Abstract
Ten spontaneous mammary gland tumors affecting guinea pigs (GP) were analyzed histologically and immunohistochemically. Histologically, 3 were benign (2 simple adenomas and 1 benign mixed tumor) and 7 were malignant (1 simple solid carcinoma and 6 simple tubulopapillary carcinomas). Immunohistochemical data revealed the glandular immunoprofile of all the tumors and suggested their ductal origin on the basis of cytokeratin 20 expression. Interestingly, cytokeratin 7 was detected in basal/myoepithelial cells. Further, all tumors were positive for type α estrogen and progesterone receptors, suggesting a role for steroid hormones in the development of these neoplasias in GP. This article describes the morphological and immunohistochemical features of the normal mammary gland and spontaneous mammary gland tumors in GP.
Keywords
Guinea pigs (GP) have recently gained popularity as pets. 14 Despite this, most studies addressing neoplasms in this species have examined spontaneous or induced tumors in animals kept under laboratory conditions. 3,10,13,19-21,41 Spontaneous tumors are relatively uncommon in GP, and although there have been isolated reports of tumors developing at 6 weeks of age, 10 they usually occur over the age of 3 years. 20 Reported incidences of tumors in GP vary greatly, with rates as high as 30% estimated for animals older than 3 years. 16 The tumors described so far are as follows: epithelial and mesenchymal tumors of the skin, 1,7,9,17,20,21,33,41 tumors of the genitourinary system, 10,13,20,33,38 endocrine tumors, 10,21,37 neoplasms originating in the blood–lymph system, 10,20,33,35 tumors of bone, 33 tumors of the alimentary tract, 33,36 and tumors of the respiratory tract. 33,36 The most common tumors of GP are bronchogenic papillary adenomas, followed by tumors of the skin and subcutis. 16
Despite this copious literature, there have been few reports of mammary tumors in GP. Retrospective reports have described the epidemiologic characteristics, gross morphology, and microscopic features of both benign and malignant mammary tumors. 2,5,20 Such tumors have been identified as papillary cystoadenomas, 4 adenomas, 10 fibroadenomas, 20 fibrocystadenomas, 10 adenocarcinomas, 20 lipofibrosarcomas, 33 and malignant mixed tumors. 10,20 Mammary tumors are generally locally invasive; they rarely metastasize; and they are often large and highly vascularized. Surgical excision of a primary mammary tumor is possible, but recurrence is likely if excision is incomplete. 2 The prevalence of mammary tumors in male GP is higher than in other species. 2,34 To the best of our knowledge, the literature lacks information on the immunohistochemical profile of mammary tumors in this species. The present study was designed to morphologically and immunohistochemically describe spontaneous mammary gland tumors in GP.
Materials and Methods
Case Selection
We examined 10 mammary gland tumor specimens from the archives obtained during mastectomies (case Nos. 1–9) and necropsy (case No. 10) that had been submitted from January 2002 to December 2007 to the Unit of Histology and Animal Pathology of the Institute for Animal Health at the University of Las Palmas de Gran Canaria, Spain. Each tumor specimen was accompanied by the corresponding case history. Tissue samples had been fixed in 10% neutral-buffered formalin for 24 hours at room temperature, processed according to standard procedures, embedded in paraffin wax, sectioned at 5 μm, and then stained with hematoxylin and eosin. In 7 cases (case Nos. 1–4, 6, 7, 10), unaffected mammary gland tissue was obtained from tumor-free surgical margins.
Immunohistochemistry
Tumor cell immunophenotypes and steroid hormone receptors were identified using cell markers and immunohistochemical techniques. An antibody panel was used comprising 11 monoclonal antibodies against cytokeratins (CKs) AE1/AE3 pancytokeratin (clone AE1 and AE3), CK 5+8 (clone RCK-102), CK 8+18 (clone NCL-5D3), CK 20 (clone Ks 20.8), CK 7 (clone OV-TL 12/30), CK 14 (clone LL002), smooth muscle actin (clone HHF35), calponin (clone CALP), vimentin (clone Vim 3B4), and type α estrogen (clone 1D5) and progesterone (clone PRA109) receptors (ERα and PR). Table 1 offers technical data on specific antibodies. 11,12,23,24 Antigen–antibody reactions were visualized by the avidin–biotin peroxidase complex (ABC) method (Vector Laboratories, Burlingame, CA). As chromogen, we used 0.5% 3,3′-diaminobenzidine tetrahydrochloride (Sigma Chemical Co, St. Louis, MO) diluted 1:10 in 0.05M Tris containing 0.3% hydrogen peroxide, which was applied to the slides for 1 to 2 minutes at room temperature. Slides were counterstained with Harris hematoxylin. Negative controls were prepared by replacing the primary antibody with normal rabbit serum diluted 1:100 in phosphate-buffered saline. In each assay, normal canine mammary gland tissue specimens were used as positive controls.
Summary of Sources, Antigen Retrieval Methods, and Dilutions of Antibodies Used a
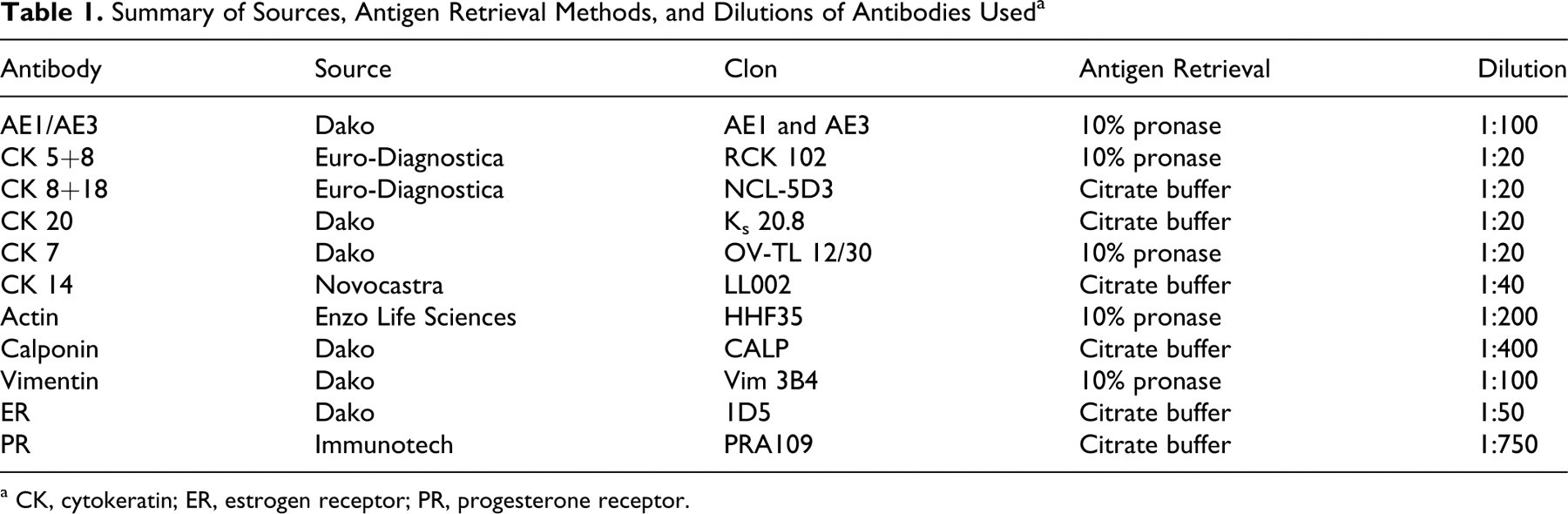
a CK, cytokeratin; ER, estrogen receptor; PR, progesterone receptor.
Histological Classification
The mammary tumors were histologically classified according to the
Results
Table 2 provides details of the results obtained for the 10 cases.
Details of the 10 Cases of Mammary Tumors in Guinea Pigs a
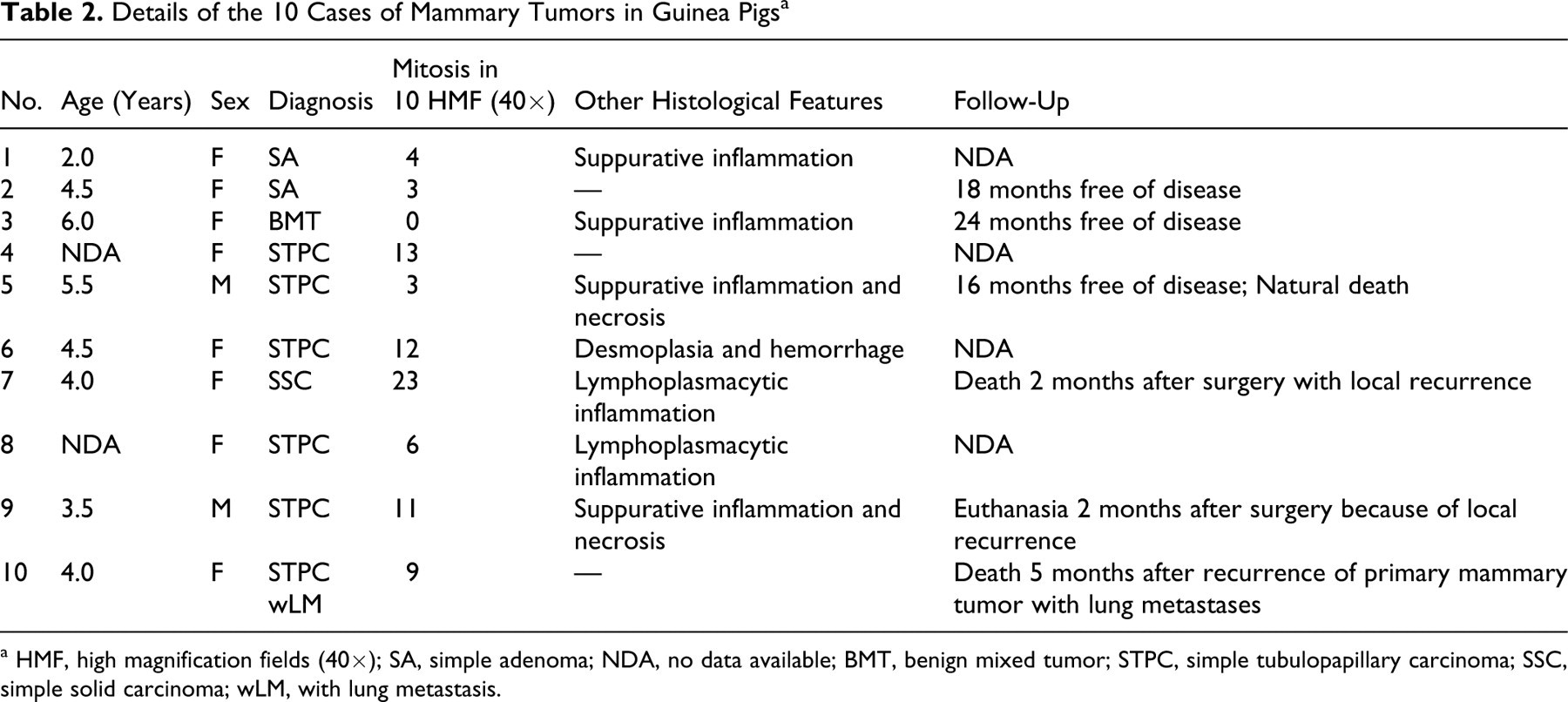
a HMF, high magnification fields (40×); SA, simple adenoma; NDA, no data available; BMT, benign mixed tumor; STPC, simple tubulopapillary carcinoma; SSC, simple solid carcinoma; wLM, with lung metastasis.
Epidemiological Findings
We had information on the ages of 8 animals, 7 of which were older than 3 years (87.5%; case Nos. 2, 3, 5–7, 9, 10). Eight tumors (case Nos. 1–4, 6–8, 10) affected female animals (80%). All animals were intact and nulliparous.
Histopathological Findings
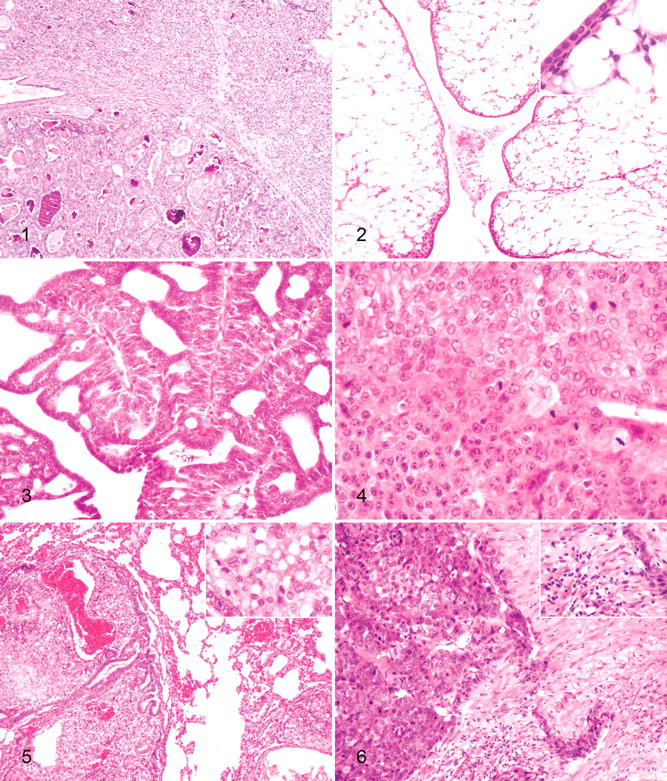
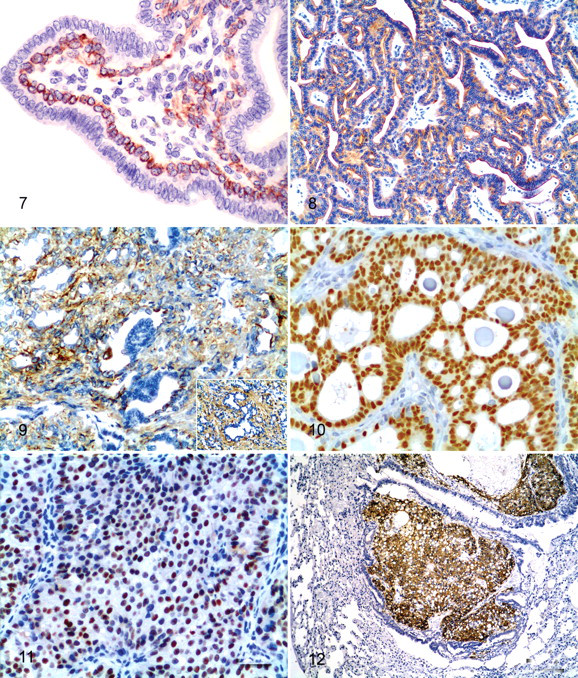
The benign tumors (case Nos. 1–3) were composed of cells morphologically consistent with epithelial cells that showed tubular or tubulopapillary patterns of growth. Case No. 1 included a cystic neoplasm comprising floating cells forming tubules and papillae intermingled with large numbers of neutrophils. Case No. 2 was a well-defined neoplasm composed of epithelial cells forming uniform, occasionally dilated, tubules, which sometimes had papillary growths. In addition, well-defined lobular hyperplasia was observed close to the tumor (Fig. 1). The third benign tumor (case No. 3) was classified as a benign mixed tumor. Histologically, the tumor mass was well delineated yet nonencapsulated, and it showed a tubular growth pattern. The tumor displayed a single layer of well-differentiated epithelial cells surrounded by a single layer of basal/myoepithelial cells and abundant, clear, vacuolated cells identified as adipocytes (Fig. 2). In some areas, this epithelial cell layer appeared thickened and showed squamous differentiation. Histologically, carcinomas differed from benign mammary tumors in at least one of the following features: atipia in the epithelial component of the tumor growth, infiltrative growth, high mitotic activity and cellularity, and/or necrosis. In general, the malignant mammary gland tumors were mostly well or moderately differentiated, and all with the exception of case No. 7 (case Nos. 4–6, 8–10) were classified as simple tubulopapillary carcinomas. These tumors appeared as tubulopapillary proliferations of the lining epithelium supported by connective tissue. There were numerous evidently mitotic cells in this type of tumor (Fig. 3). Case No. 7, classified as a simple solid carcinoma, was characterized by epithelial cells forming solid nests surrounded by areas of normal or dysplastic mammary gland tissue. This tumor was also highly vascularized and showed a high mitotic cell rate (Fig. 4). Case No. 10 was an animal whose primary mammary gland tumor had metastasized to the lung. At the primary site, no infiltration within the surrounding connective tissue or invasion of lymph and blood vessels was observed. Metastatic tissue showed the same characteristic tubulopapillary growth as the primary tumor, but most of the metastatic tissue mass was composed of large pleomorphic clear cells with a prominent vascular stroma (Fig. 5). Inflammatory cell infiltration was detected in five tumors and consisted of moderate (case Nos. 1 and 3) to severe suppurative inflammation (case Nos. 5 and 9) or a lymphocyte-plus-plasma-cell infiltrate (case Nos. 7 and 8) (Fig. 6). Furthermore, areas of desmoplasia and hemorrhage were observed in case No. 6. None of the 10 tumors showed signs of vascular invasion.
Immunohistochemical Findings
Table 3 outlines the immunohistochemical observations made in normal and neoplastic mammary gland tissue. In both normal and neoplastic mammary tissue, acinar epithelial cells of the cytoplasm were intensely labeled with both anti-AE1/AE3 and anti–CK 8+18 antibodies. An even anti–CK 5+8 antibody reaction was observed in the cytoplasm limited to the basal/myoepithelial cells (Fig. 7 ). Staining for the CK 20 antibody was intense in the normal ductal epithelial cells and all the epithelial cells composing the tumors (Fig. 8). Groups of ductal and acinar epithelial cells showed intense yet uneven anti–CK 7 antibody staining. Besides this, basal/myoepithelial cells were largely positive for CK 7 and for classic myoepithelial markers (actin, calponin, vimentin, and CK 14) in the same microscopy fields of consecutive serial sections (Fig. 9). The CK 7 was immunolocalized in the cytoplasm and in the cell membrane. Most myoepithelial cells in the normal and mammary tumor tissue were positive for CK 14, actin, calponin, and vimentin. Intense homogeneous staining for these markers was detected at the basal/myoepithelial level in normal and tumoral tissue. Finally, the hormone receptors PR and ERα were immunolocalized in the nuclei of most normal ductal epithelial cells, whereas acinar cells showed a more heterogeneous staining pattern. All the tumors were positive for PR and ERα (Fig. 10) as reflected by an intense immunostaining pattern. However, although the less-differentiated simple solid carcinoma (case No. 7) was positive for PR and ERα, the intensity of nuclear staining was more heterogeneous (Fig. 11). Myoepithelial cells were unreactive for PR and ERα. Minimal cytoplasm staining for ERα was also observed in some epithelial cells in the normal and neoplastic mammary gland (case Nos. 4, 5, 8, 10). Neoplastic cells of the lung metastasis (case No. 10) were positive for AE1/AE3, CK 20, CK 7, CK 14 (Fig. 12), calponin, vimentin, and actin. No hormone receptors were detected in the lung metastasis.
Immunohistochemical Results of Normal and Neoplastic Mammary Tumors in Guinea Pig
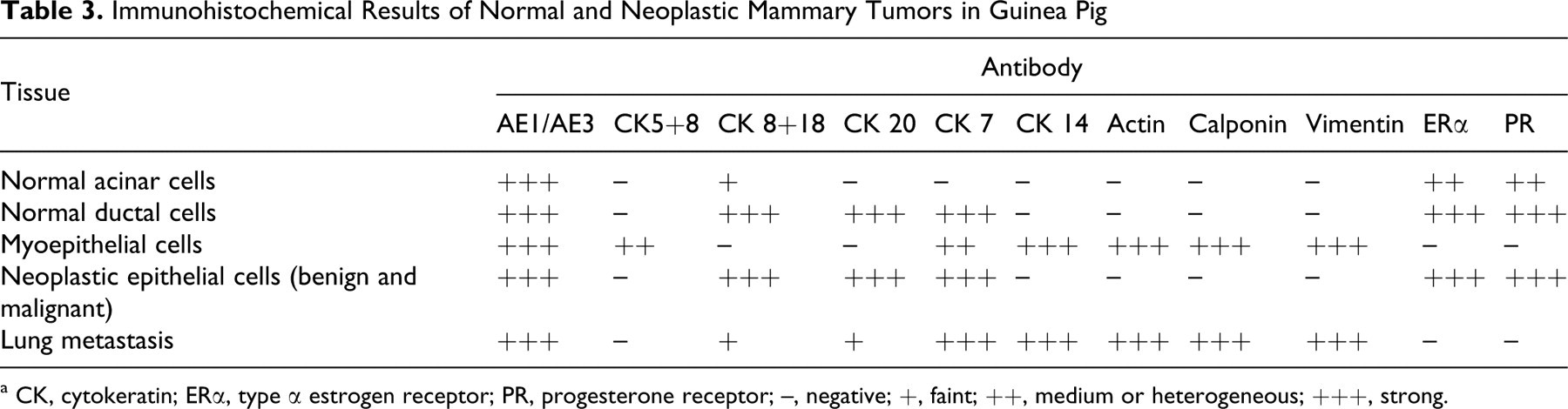
a CK, cytokeratin; ERα, type α estrogen receptor; PR, progesterone receptor; –, negative; +, faint; ++, medium or heterogeneous; +++, strong.
Discussion
Spontaneous neoplasms are generally rare in GP, especially in animals younger than 3 years of age. 20 Seven out of the eight cases (87.5%) examined here for which age data were available involved animals older than 3 years. Spontaneous tumors commonly described in this species include bronchogenic papillary adenomas, trichofolliculomas, carcinomas of the skin, ovary teratomas, and tumors of the mammary gland. 16 Mammary tumors are described in female and male GP. Of the 10 spontaneous mammary gland tumors reported here, 2 occurred in males, a finding consistent with published findings 3,16 indicating a higher prevalence of mammary tumors affecting males in GP compared with other species. 34 Despite no consensus regarding the histological classification of these neoplasms, 4 our histological findings resembled those included in the World Health Organization’s classification of mammary tumors of the dog and cat. 27 Other authors have used this classification system to classify mammary tumors in other species. 8, 22 In effect, we believe that pathologists and clinicians should routinely use this system to standardize, interpret, and accurately reproduce research results. In our study, 3 out of 10 tumors were benign, 2 were simple adenomas (case Nos. 1 and 2), and 1 was a benign mixed tumor (case No. 3). The latter was difficult to classify because it had many features of a complex adnexal tumor. 1 According to the classification system mentioned above, mixed tumors have secretory and myoepithelial components, and these myoepithelial cells later differentiate into adipocytes or other connective tissues, such as cartilage or bone. 27 Because both the epithelial and myoepithelial cells of this neoplasm had the same immunophenotype as those of the normal mammary gland and the other mammary tumors examined here, a diagnosis of a benign mixed mammary gland tumor was made. Two mammary malignant mixed tumors have been reported in GP. 10,20 These tumors were characterized by a fibrosarcomatous portion mixed with other areas exhibiting the histological features of a papillary adenocarcinoma. Features of anaplasia such as bizarre mitotic figures, an increased nuclear/cytoplasmic ratio, and hyperchromatic nuclei were not observed in our mammary benign mixed tumor. This type of neoplasm has not been reported in GP. For the malignant tumors, we describe here six simple tubulopapillary carcinomas (case Nos. 4–6, 8–10) and one simple solid carcinoma (case No. 7). The later is the first report of a simple solid carcinoma affecting the mammary gland of GP. According with the World Health Organization’s classification of mammary tumors in dogs and cats, simple carcinomas are divided into tubulopapillary, solid, and anaplastic types, reflecting increasing malignancy. 27 Our results seem to prove that GP benign mammary tumors have a long disease-free period whereas GP malignant mammary tumors show a worse biological behavior, related with local recurrence and distant metastasis.
Commercially available antibodies are widely used in humans and domestic animals, mainly dogs and cats, to characterize the immunophenotype of neoplastic cells. 32 All the commercially available antibodies employed in our study showed cross-reactivity with GP tissue. Six monoclonal antibodies (AE1/AE3, RCK 102, NCL-5D3, Ks20.8, OV-TL 12/30, and LL002) identified a variety of CKs in normal and neoplastic mammary tissue, indicating that neoplastic transformation did not alter the CK expression profile of glandular epithelial cells. The broad reactivity of the AE1/AE3 antibody precludes any conclusions regarding the expression of a given CK. The combined use of the antibodies RCK 102 and NCL-5D3—which recognize CKs 5 and 8 and CKs 8 and 18, respectively—revealed that the acinar and ductal epithelial cells of the mammary gland in the GP do not express CK 8, because only the basal/myoepithelial layer was immunostained with the RCK 102 antibody. In addition, CK 20 was clearly identified in both normal ductal and neoplastic mammary epithelial cells. In humans, CK 20 is mainly restricted to epithelia and tumors originating in the gastrointestinal tract, urothelium, and Merkel cells. 25,29,30 Despite this apparent tissue-specific distribution, ectopic CK 20 expression has been reported in sporadic cases of carcinoma derived from CK 20–negative nonaltered tissues, including breast, lung, and endometrium. 29 Feline and canine mammary tumors do not express CK 20, whereas carcinomas of the thyroid gland, intestine, lung, and kidney of cats and carcinomas of the gastrointestinal tract and ovary of dogs do express this CK. 11 Our consistent demonstration of CK 20 in normal ductal and neoplastic epithelial cells suggests the hypothesis that most mammary tumors in GP are of a ductal origin. In the current research, the pattern of reaction of CK 7 in epithelial cells of GP mammary gland and tumors is similar to those described in other species. 11,15,39,40 Mammary basal/myoepithelial cells in the present study were also positive for CK 7, whereas human, feline, canine, and mice mammary basal/myoepithelial cells do not express this CK, 11,15,39,40 although the expression of CK 7 by cells with muscle differentiation has been reported in human beings. 26,40 The expression of CK 20 and CK 7 and the lack of expression of CK 8 in GP mammary gland tissue seem to represent a significant difference with respect to the CK profiles of other mammalian species. Actin, calponin, and vimentin immunostaining was detected at the basal/myoepithelial level in agreement with the results obtained by several authors in other species. 6,12,18,23,39 However, further studies would be necessary to discuss our immunohistochemical results.
In our study, the hormone receptors (PR and ERα) were localized in the nuclei of most normal ductal epithelial cells. Both PR and ERα expression was detected in the glandular epithelial cell compartment of all the GP mammary gland tumors. Myoepithelial cells did not express hormone receptors. Both these findings, along with minimal cytoplasmic staining for ERα observed in our cases, are in line with previous immunohistochemical studies of hormone receptor expression in mammary gland tumors in humans, dogs, and cats. 24,31 The present case of lung metastasis showed no nuclear immunostaining for ERα or PR. This finding is in agreement with other studies showing lack of hormone receptors in metastatic lesions of primary positive tumors. 28 In fact, the CK pattern observed in this tissue was consistent with the mammary gland immunoprofile. The results described here confirm the fact that monoclonal antibodies against human ERα and PR cross-react with the GP ERα and PR and can be used in formalin-fixed tissues of the GP. They also show that these tumors may develop under the influence of estrogens and progesterone as in humans, dogs, and cats. 24,28,31 Nevertheless, further studies controlling the hormone status of GP must be addressed to confirm this hypothesis.
To the best of our knowledge, this is the first comprehensive study revealing both the morphological and the immunohistochemical features of the normal mammary gland of the GP and those of spontaneous mammary gland tumors.
Footnotes
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
