Abstract
Since the initial outbreak of West Nile virus (WNV) in the northeastern United States in 1999, the virus has rapidly spread westward and southward across the USA, causing high mortality in crows as well as sporadic mortality in horses, humans, and a wide variety of birds. In 2002 the epidemic widened as hundreds of equine and human cases and sporadic cases in other mammalian species were reported. This is the first report of WNV infection in three Eastern fox squirrels (
West Nile virus (WNV) was first recognized in the northeastern USA in the summer of 1999.
22
Since then, it has spread rapidly across the USA and Canada, causing high mortality in crows as well as sporadic mortality in horses, humans, and a wide variety of birds, including native and captive exotic species.
1,4,11,13,17,19
The first wild crow with naturally occurring WNV in Michigan was diagnosed on August 22, 2001. Throughout the spring and summer months (April through October) of 2001 and 2002, a multi-institutional arbovirus surveillance team composed of scientists from the Michigan Department of Community Health, the Michigan Department of Agriculture, the Michigan Department of Natural Resources, the Diagnostic Center for Population and Animal Health (DCPAH) and the Department of Microbiology and Molecular Genetics, Michigan State University (MSU) had conducted surveillance for WNV in wild birds, mammals, and mosquitoes. During 2001 all reported cases of WNV infection in Michigan involved free-ranging
In 2002 the epidemic widened as hundreds of horses and humans and sporadic cases in wildlife and exotic species were reported. Multiple anecdotal reports from the public indicated that fewer squirrels were observed in Michigan during the summer of 2002 than in previous years. Some Michigan citizens reported finding dead squirrels in their backyards and observing squirrels with neurologic signs falling from trees. Following those reports, the DCPAH conducted an investigation to determine the possible role of WNV as the cause of death of squirrels found during the summer of 2002 in Michigan. One dead adult and two moribund juvenile Eastern fox squirrels that had been submitted to the DCPAH for diagnostic work-up during August and October 2002 tested positive for WNV.
The adult squirrel had been found dead, but the juvenile squirrels exhibited various signs of neurologic disease including weakness, depression, head tilt, torticollis, lateral recumbency, uncoordinated movements, inability to right themselves when pushed over, and scratching of their foreheads with both feet and tremors. The condition of the squirrels continued to deteriorate, and the animals were humanely euthanized.
Complete necropsies were performed on all three animals. One adult and one juvenile squirrel were judged to be in fair to good body condition; the other juvenile squirrel was mildly emaciated. All animals had ingesta within their stomachs, indicating that they had eaten recently. No gross lesions were seen.
Representative samples of major organs, including brain, heart, lung, liver, kidney, spleen, gastrointestinal tract, pancreas, and gonad were collected and fixed in 10% neutral-buffered formalin for histopathologic examination. Formalin-fixed tissues were trimmed, embedded in paraffin, sectioned at 5 µm and routinely processed for microscopic examination. Immunohistochemistry (IHC) for WNV was performed on selected organs following a standard protocol using a rabbit-polyclonal anti-WNV antibody (BioReliance, Rockville, MD) at a concentration of 1 : 500. 5,20 Sections were stained on an automated stainer using a labeled streptavidin-immunoperoxidase staining procedure (Dako Corp., Carpinteria, CA). The immunoreaction was visualized with AEC (Dako Corp., Carpinteria, CA), and sections were counterstained with Mayer's hematoxylin and then dehydrated, cleared, and mounted.
For RNA extraction, paraffin-embedded formalin-fixed tissues were processed to be compatible with the RNeasy minikit (QIAGEN, Inc., Valencia, CA). WNV RNA detection was carried out using a reverse transcriptase–polymerase chain reaction (RT-PCR) assay previously developed and optimized at the Virology section of the DCPAH, MSU. 9,15 The primer pair amplifies a 155-bp region of the envelope glycoprotein E gene of WNV (forward primer: 5′ TCG GGT CAT TTG AAG TGT AGA GT 3′; reverse primer: 5′ CCA TCC GTG CCA GTG TAC TGC 3′). The RT-PCR reaction was carried out in a one-tube system using the OneStep RT-PCR kit (QIAGEN, Valencia, CA). Formalin-fixed, paraffin-embedded sections of heart and kidney from crows in which infection with WNV had been confirmed by RT-PCR served as positive controls for IHC and RT-PCR. Formalin-fixed tissues from squirrels that had been necropsied for other causes prior to 2000 served as negative controls.
All formalin-fixed tissues were examined microscopically. The only consistent lesion was observed in the kidneys. In all squirrels, there was multifocal, moderate to severe lymphoplasmacytic nephritis characterized by multifocal, randomly scattered aggregates of lymphocytes, plasma cells, and a few histiocytes that expanded the interstitium. Only the adult squirrel had severe lesions in the brain and the heart and mild lesions in the liver and lungs. All portions of the heart were affected by a severe multifocal necrotizing myocarditis characterized by pyknotic nuclei, loss of cross-striations, and hypereosinophilic sarcoplasm in affected myocytes with infiltration of lymphocytes, plasma cells, and a few histiocytes (Fig. 1). Microscopic lesions were found throughout the brain, including cerebrum, cerebellum, and brain stem, and were characterized by diffuse, moderate, nonsuppurative meningoencephalitis with lymphoplasmacytic perivascular cuffing in both the meninges and the neuropil (Fig. 2). Scattered individual necrotic neurons that were sometimes surrounded by glial cells (satellitosis) were found in the cerebrum and the hippocampus. In addition, there was focal gliosis, and a few glial nodules were scattered throughout the neuropil. Hepatic lesions consisted of moderate perivascular lymphohistiocytic inflammation with lesser numbers of plasma cells. There was moderate proliferation of the bile ducts and mild intracellular cholestasis. The lungs had mild lymphohistiocytic perivascular cuffing throughout the parenchyma. No significant microscopic lesions were found in the gastrointestinal tracts, pancreas, spleens, or gonads of the three squirrels.
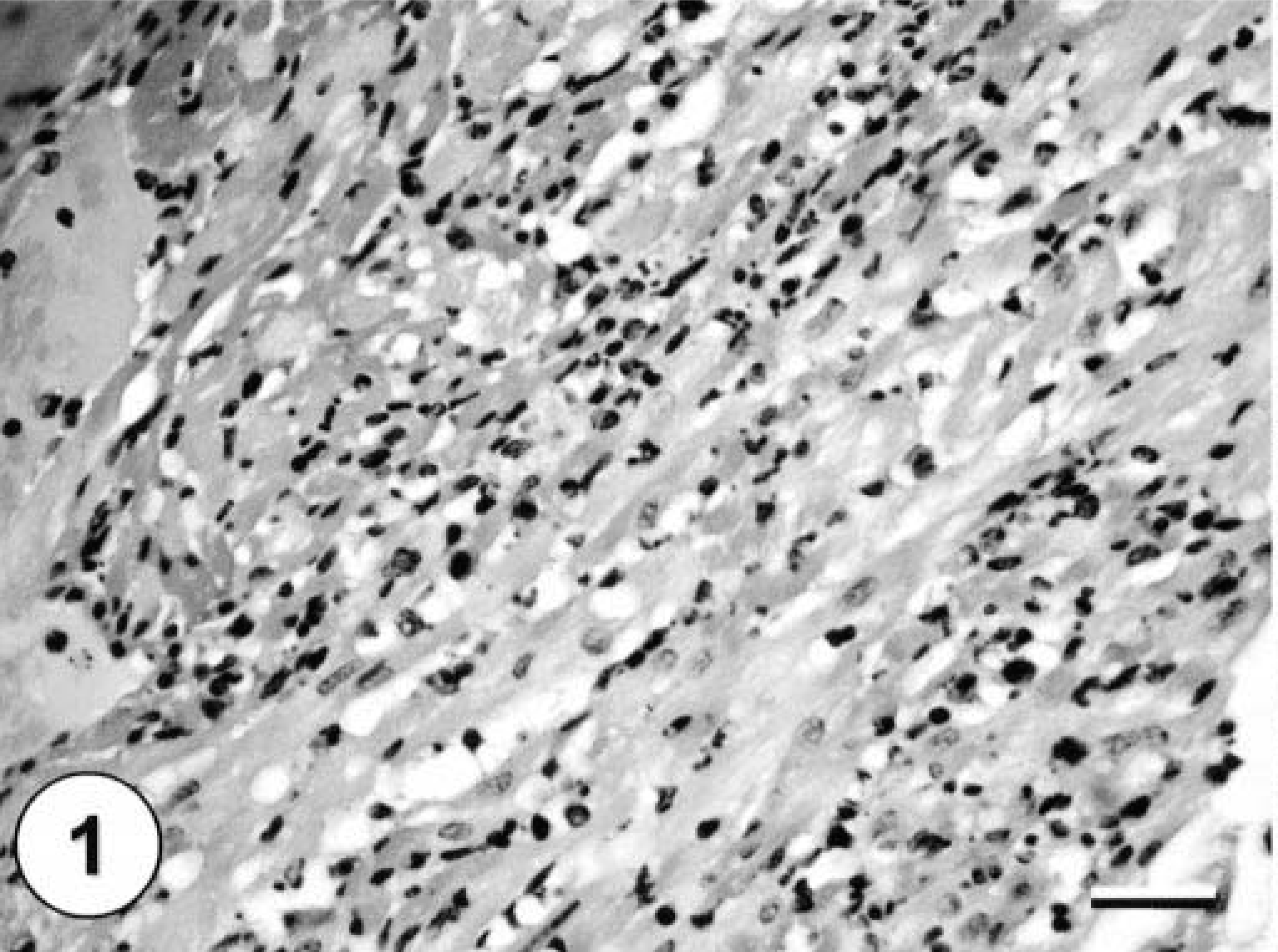
Heart from an Eastern fox squirrel with WNV with severe necrotizing myocarditis, heavy mixed mononuclear leukocyte infiltrate, and remnants of necrotic cardiomyocytes. HE staining. Bar = 60 µm.
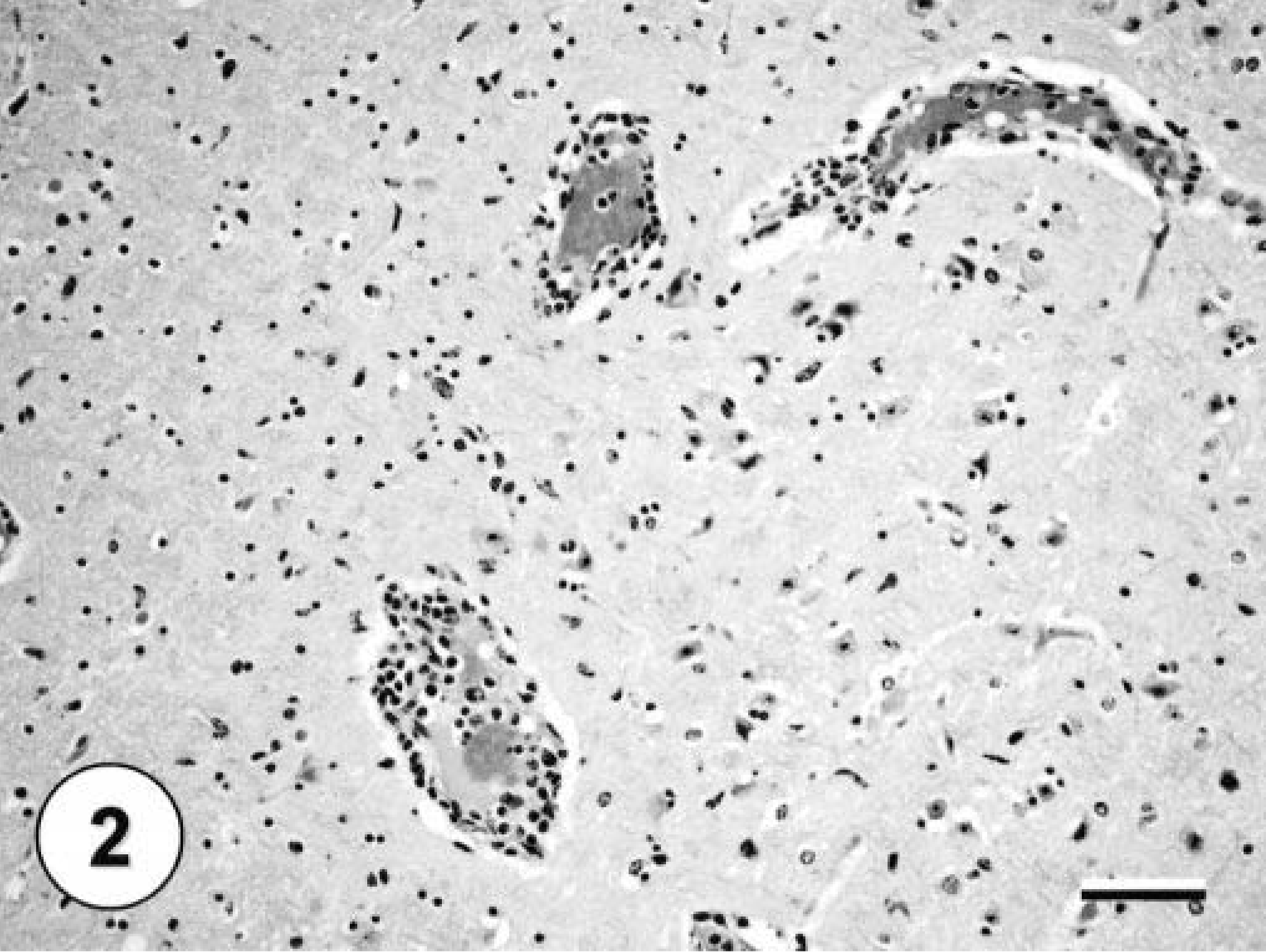
Brain from an Eastern fox squirrel with WNV with moderate perivascular cuffing of a large cerebral vessel by mononuclear inflammatory cells. HE staining. Bar = 60 µm.
All three squirrels in this report were positive for WNV by IHC staining in one or more tissues. The kidneys exhibited fairly abundant staining in all three squirrels, with both tubular epithelial cells and interstitial macrophages staining (Fig. 3). The heart, brain, liver, and lung of the adult squirrel with microscopic lesions in these organs also stained positively for WNV. Myocardial fibers and interstitial macrophages were the cells exhibiting specific staining. Staining in the brain was focal but strong. Multiple neurons and glial cells in the cerebrum and hippocampus stained positively (Fig. 4). In the liver, there were single positive macrophages. Hepatocytes and bile duct epithelial cells did not stain positively. In the lungs rare individual interstitial macrophages, especially surrounding bronchi, stained positively. No other organs stained positively. In all cell types, staining was limited to the cytoplasm.
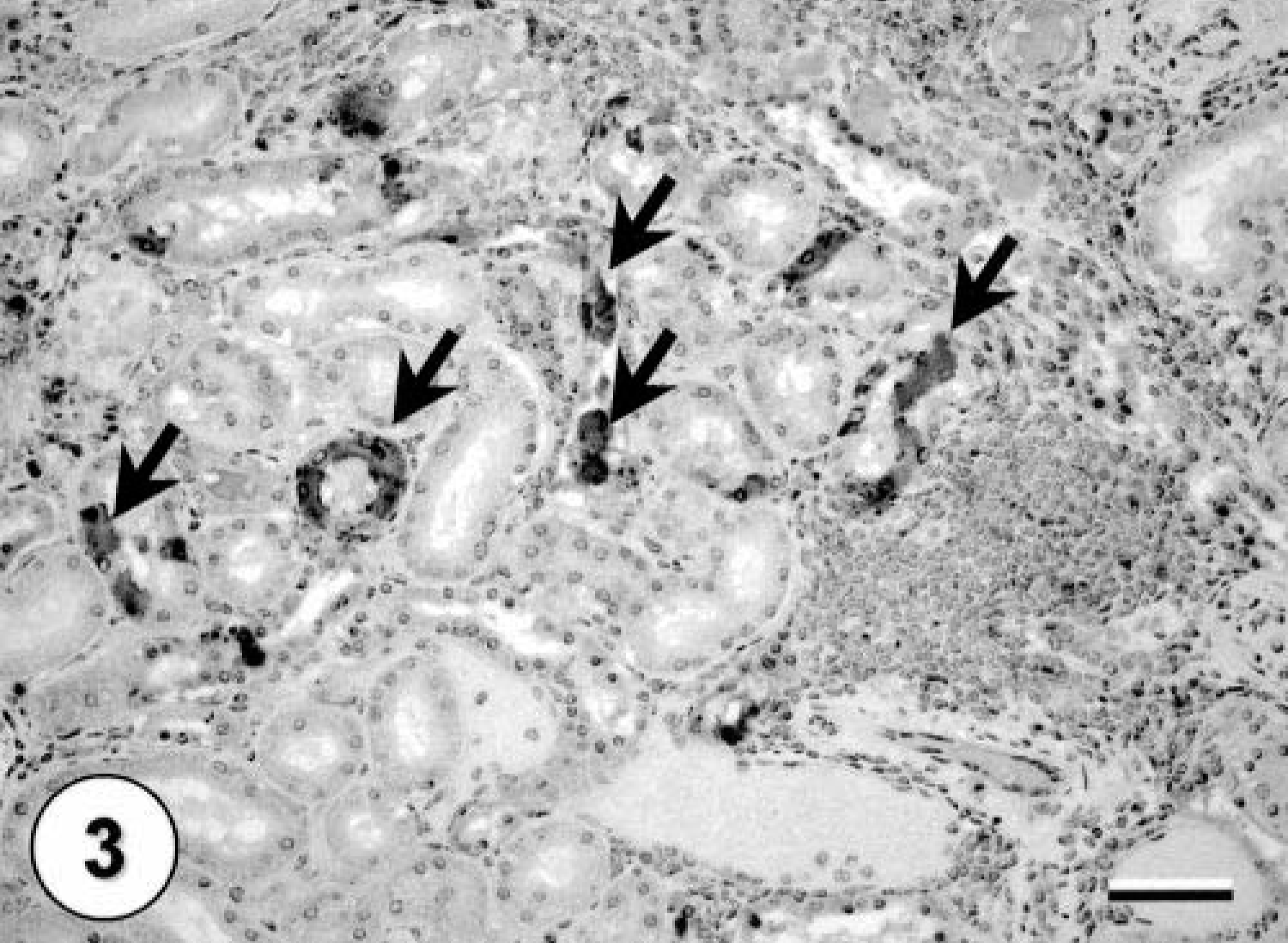
Immunohistochemical staining of a kidney from an Eastern fox squirrel with interstitial lymphoplasmacytic nephritis and multifocal tubular epithelial cells staining positive for WNV (arrows). Hematoxylin counterstaining. Bar = 60 µm.
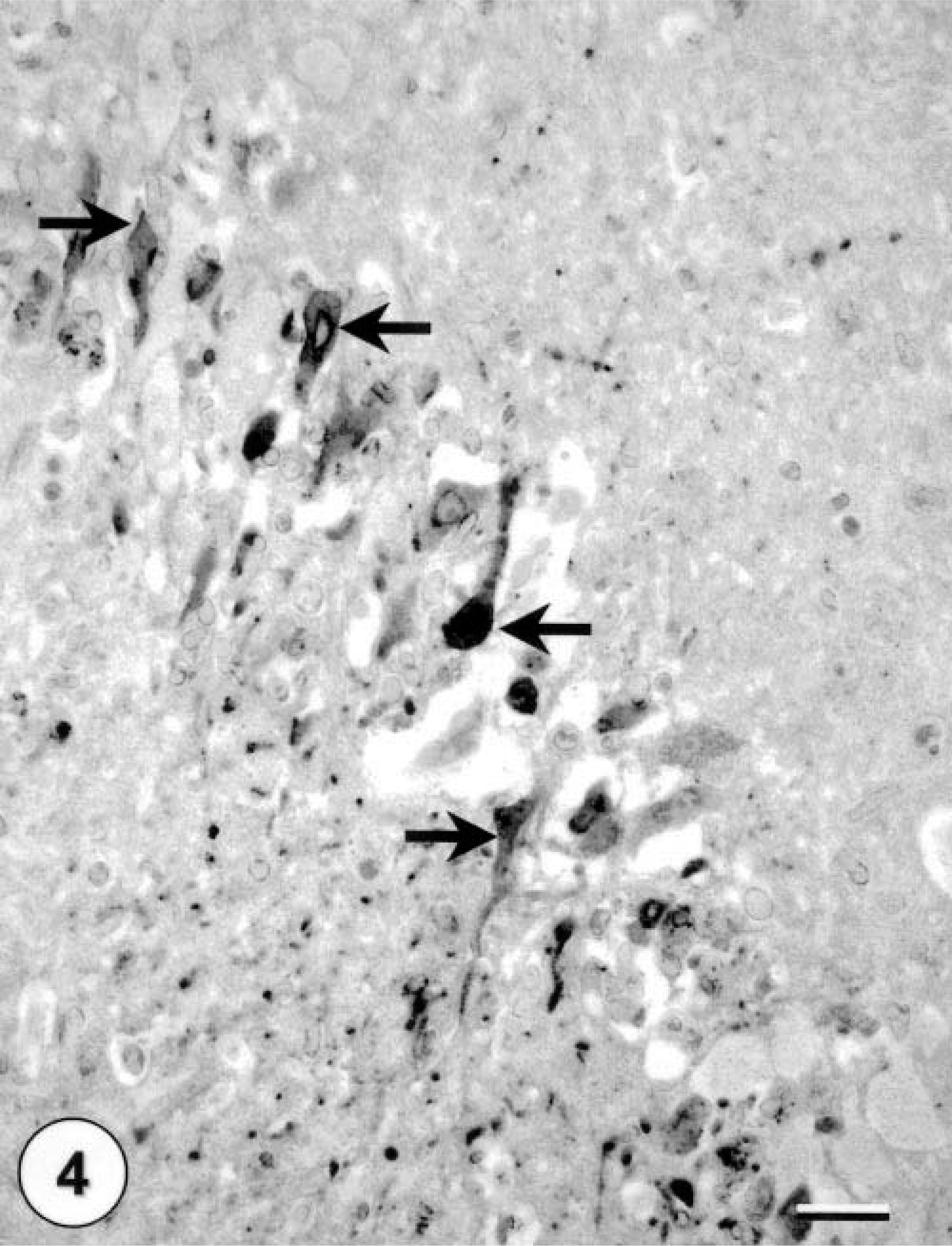
Immunohistochemical staining of a brain from an Eastern fox squirrel with multiple neurons staining positive for WNV (arrows). Hematoxylin counterstaining. Bar = 60 µm.
Formalin-fixed renal tissue from all three squirrels was tested by RT-PCR for WNV RNA, and results were positive in two cases (Fig. 5). One juvenile squirrel with immunohistochemical labeling for WNV in the kidneys was negative for WNV by RT-PCR.
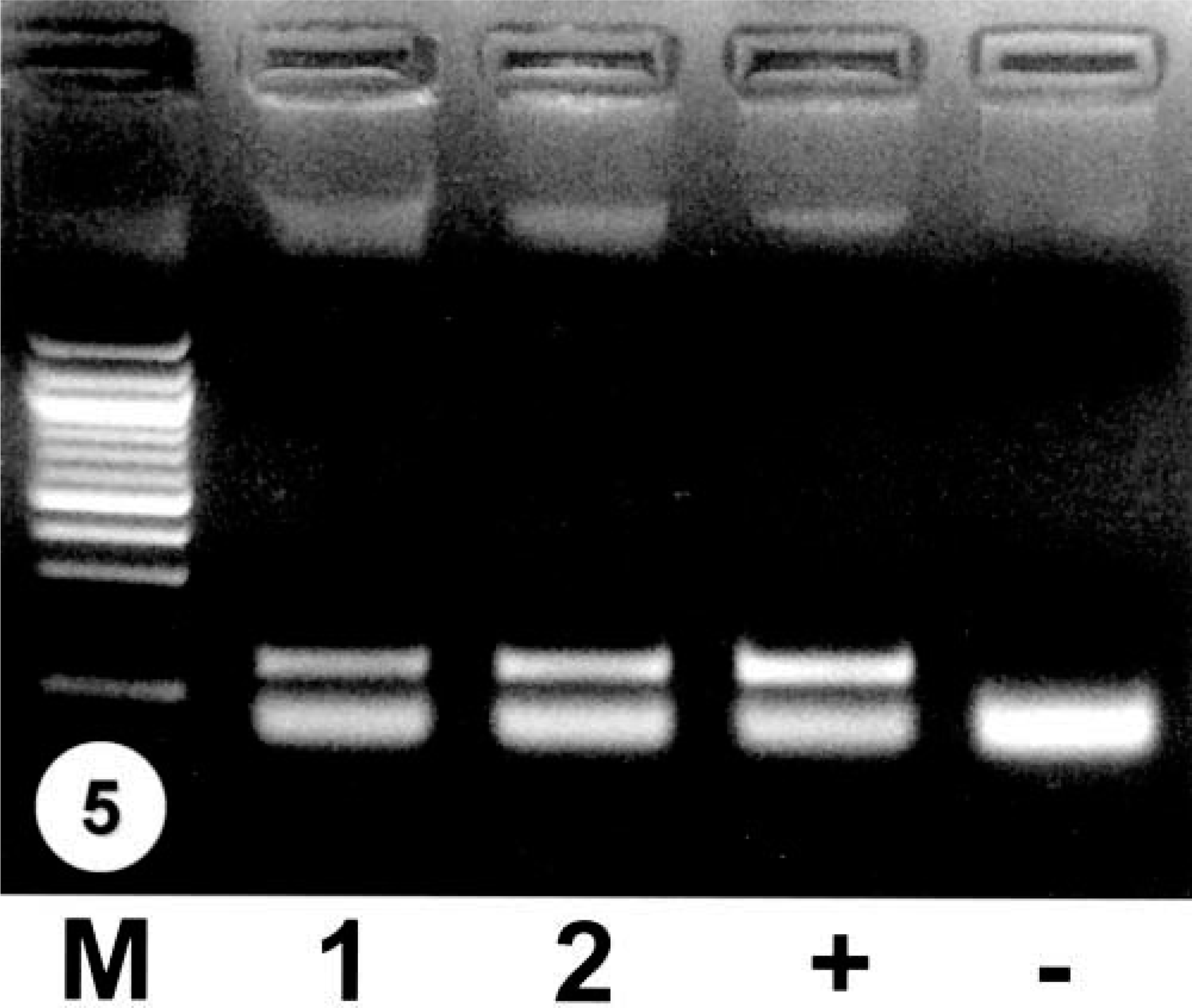
RT-PCR for WNV of squirrel tissues. M = 100 bp DNA ladder; 1 = kidney of an Eastern fox squirrel; 2 = brain of an Eastern fox squirrel; + = positive control; − = negative control.
In our experience with surveillance of hundreds of free-ranging crows with WNV infection during the last 2 years, histologic lesions tend to be mild or absent, and IHC staining confirms the presence of large amounts of viral antigen in various tissues including heart, kidney, and brain. 13,20,22 In contrast, horses tend to exhibit moderate or even severe histologic lesions in the brain and spinal cord, but very minimal WNV antigen, if any, can be detected by IHC. 5,19 Commonly nested RT-PCR is required to confirm the diagnosis. Interestingly, Eastern fox squirrels had large amounts of WNV antigen associated with moderate to severe microscopic lesions in different tissues. It is possible that squirrels are somewhat more resistant to infection than are crows but less resistant than horses, which means that infection may persist longer in squirrels than in crows and stimulate a greater inflammatory response prior to illness and death. Experimental inoculation studies with WNV in squirrels are needed to determine their relative susceptibility, compared with other mammalian species, and the pathogenesis of the infection.
Studies examining tissue tropism of WNV in corvids and noncorvids indicated that the kidney was the most consistently infected tissue in birds, regardless of the level of infection. 20,22 The brain was the next most consistently positive tissue. 20,22 As in crows, antigen was most commonly found in the kidneys of affected squirrels. Based on the limited data presented here, the optimal tissues to collect from squirrels suspected of WNV infection for routine histologic evaluation and IHC staining or RT-PCR for WNV appear to be the kidney, followed by the brain and heart.
Presently there is no information in the literature on WNV infection in squirrels. There were no reports of increased mortality associated with neurologic signs in squirrels in Michigan prior to 2002. Most likely squirrels represent a spill-over host, similar to humans and horses, none of which exhibited clinical cases of WNV during 2001 in Michigan. However, we speculate that in 2002 the prevalence and geographic distribution of both WNV-infected mosquitoes and crows had increased to some critical level that allowed significant spill-over infection into nontarget host species. 1 Alternatively, WNV may be adapting to new host species over time. Additional studies on both WNV strains and species-specific pathogenesis are needed to answer these questions.
Other mammalian species have been shown to be susceptible to experimental and naturally occurring WNV infection, including horses, mules, donkeys, sheep, dogs, and pigs. 1–4,8,13,17,19,22 Hamsters and mice have been used as a model for WNV-induced encephalitis. 13,23 Because these rodents are susceptible, perhaps squirrels are likely candidates as well. In hamsters, encephalitic signs began 6 days after intraperitoneal infection and about half the animals died between the 7th and 14th days. 23 Microscopic lesions and viral antigen were detected in the brain after 6 days of infection. 23 Experimental mouse inoculation demonstrated a classical flavivirus infection pattern. 13 A study on seroprevalence of WNV in pigs in India demonstrated WNV-specific antibodies in 3.2% of 158 pigs examined. 21 Dogs and cats have tested positive for WNV-neutralizing antibodies, but there are no reports in the literature of clinical disease or gross and microscopic lesions caused by a naturally occurring WNV infection in these species. 12
Our knowledge about disease affecting North American squirrels is limited. Parapoxvirus and poxvirus infections and larval migration of
WNV represents a new challenge for North American diagnosticians, wildlife rehabilitators, wildlife biologists, zoological staff, and others involved with wild bird and mammalian species. Although it is widely recognized that crows and other corvids are highly susceptible to this newly emergent disease, many other species are also exposed. Squirrels now appear to be among the susceptible mammals that develop both clinical signs and mortality. When presented with a squirrel with a history of neurologic disease, and microscopic nonsuppurative lesions in various tissues, particularly the heart, WNV infection should be one of the primary differential diagnoses.
Footnotes
Acknowledgements
The authors thank S. Scot Marsh and Tom Woods for their technical help.
