Abstract
Ocular lesions are common in red-tailed hawks with West Nile (WN) disease. These lesions consist of pectenitis, choroidal or retinal inflammation, or retinal necrosis, but detailed investigation of the ocular lesions is lacking. Postmortem examination of the eyes of 16 red-tailed hawks with naturally acquired WN disease and 3 red-tailed hawks without WN disease was performed using histopathology, immunohistochemistry for West Nile virus (WNV) antigen, glial fibrillary acid protein, cleaved caspase-3, and the terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling method. Retinal lesions were classified as type I or type II lesions. Type I lesions were characterized by lymphoplasmacytic infiltrates in the subjacent choroid with degeneration limited to the outer retina (type Ia lesion) or with degeneration and necrosis of the outer retina or outer and inner retina (type Ib lesion) while retinal collapse, atrophy, and scarring were hallmarks of type II lesions. Type II retinal lesions were associated with a more pronounced choroiditis. Although not statistically significant, WNV antigen tended to be present in larger quantity in type Ib lesions. Type I lesions are considered acute while type II lesions are chronic. The development of retinal lesions was associated with the presence of an inflammatory infiltrate in the choroid. A breakdown of the blood-retina barrier is suspected to be the main route of infection of the retina. Within the retina, virus appeared to spread via both neuronal and Müller cell processes.
West Nile virus (WNV) is a flavivirus of the Japanese encephalitis antigen complex that has caused significant mortality in various species of birds since its introduction to North America in 1999.
9,14,33
Mosquitoes are the primary vectors and spread the infection to birds, the natural host for the virus. Birds of prey are commonly infected with WNV.
7,11,25,36,37
Less than 1-year-old (aka hatch-year) red-tailed hawks (
Progressive visual impairment leading to blindness has been reported in various hawk species infected with WNV such as red-tailed hawks, Cooper’s hawks (
Structured in 10 layers, the avian retina is similar to the better studied mammalian retina. The components of the outer retina are the retinal pigmented epithelium (RPE) layer, photoreceptor layer, outer nuclear layer, and outer plexiform layer while the components of the inner retina are the inner nuclear layer, inner plexiform layer, ganglion cell layer, and nerve fiber layer. However, the avian retina differs from the mammalian retina in that (1) retinal capillaries are lacking in birds, (2) avian retina harbors oligodendroglia and myelinated nerve fibers in the nerve fiber layer close to the optic disc, and (3) astrocytes, strictly speaking, are absent so that Müller cells constitute the only astroglial cell type in the retina of birds. 35 The objective of this study was to further describe and classify retinal lesions present in red-tailed hawks naturally infected with WNV and to determine whether Müller cells of the retina were infected. In addition, the study addresses the question of whether apoptosis may be a mechanism of WNV-induced cell death in the retina.
This study included the ultrastructural examination of the fundus (choroid, retina, optic disc, and pecten), which will be presented in a subsequent article.
Materials and Methods
Selection Criteria
Nineteen red-tailed hawks (
Sample Collection and Preparation
Both eye globes were removed from every bird within approximately 5 minutes of euthanasia. The eyes were cut perpendicular to the visual axis behind the scleral ossicles. The fundus was macroscopically evaluated. The eyes were fixed in 10% neutral buffered formalin for histopathologic examination after tissue samples had been collected for electron microscopy. Formalin-fixed ocular tissue was trimmed and paraffin embedded approximately 24 hours after necropsy. Sections of 4 μm thickness were stained with hematoxylin and eosin (HE). Three sections, including 2 sections of the fundus and 1 section of cornea, lens, and anterior chamber, were evaluated per eye to characterize the degree of inflammation of pecten and choroid and the character of the retinal lesions. The lesions in the retina were typed based on the predominant morphology of the alteration (see the Results section). The degree of inflammation in the pecten and choroid was subjectively graded as absent, mild, moderate, or marked based on the number of infiltrating lymphocytes, plasma cells, and macrophages (Suppl. Table S2).
Immunohistochemistry
A peroxidase-based polymer system was used as previously described (Suppl. Table S3). 38 The antibody used does not have reactivity for viruses closely related to WNV such as Saint Louis encephalitis (SLE) virus and Japanese encephalitis virus. 1 The positive controls were formalin-fixed, paraffin-embedded brain tissue from known WNV positive red-tailed hawks (WNV, glial fibrillary acidic protein [GFAP], or cloacal bursa [cleaved caspase-3]). Negative controls included substitution of the primary antibody with ascites fluid from nonimmunized Balb/c mice (WNV antigen), with nonimmune irrelevant rabbit serum (GFAP), or with rabbit immunoglobulin fraction (DAKO; cleaved caspase-3, Carpinteria, CA).
The amount of antigen expression in the eyes was described as absent, mild, moderate, or marked based on the estimated number of WNV antigen-positive cells as previously described. 26,38
Terminal Deoxynucleotidyl Transferase-Mediated dUTP Nick-End Labeling
For the detection of apoptosis, the paraffin-embedded sections of eye fundus were stained by the terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) method as described previously. 17 The TUNEL assay was performed using the ApopTag Plus Peroxidase in situ apoptosis detection Kit (Chemicon, Temecula, CA) according to the manufacturer’s instructions. Formalin-fixed, paraffin-embedded cloacal bursal tissue from a red-tailed hawk hatchling was used as positive control for the TUNEL method.
Quantitative Reverse Transcription Polymerase Chain Reaction
The aqueous humor of the left and right eye and samples of pooled tissue, including heart, kidneys, and brain (cerebrum and cerebellum), were examined for WNV, SLE virus, and eastern equine encephalitis (EEE) virus using the quantitative reverse transcription polymerase chain reaction (RT-qPCR) assay as previously described. 26 Nucleic acid extraction and amplification were performed by use of the probes and protocol previously described. 21 Reactions were performed with 2 μL RNA in a 25-μL reaction. Amplification was done in a 2-stage reaction with RT 42°C for 900 seconds, denaturation at 95°C for 600 seconds, followed by 39 cycles of 95°C for 15 seconds, 50°C for 10 seconds, and 60°C for 100 seconds.
Statistical Analysis
The different lesion types were compared with the degree of inflammation of the subjacent choroid and the degree of antigen expression in the affected retinal segment (as determined by immunohistochemistry [IHC]) using a semiquantitative scale (0, absent; 1, mild; 2, moderate; 3, marked) through Kendall’s tau (τ) coefficient. Differences in the proportions of observations per lesion type with respect to the IHC scores and to the choroiditis scores were evaluated using the Fisher exact test and
Results
Two female and 1 male red-tailed hawk served as control birds. These birds included a hatch-year bird, a second-year bird, and an adult bird (Suppl. Table S1). All but 1 WNV-infected bird were hatch-year birds.
Necropsy
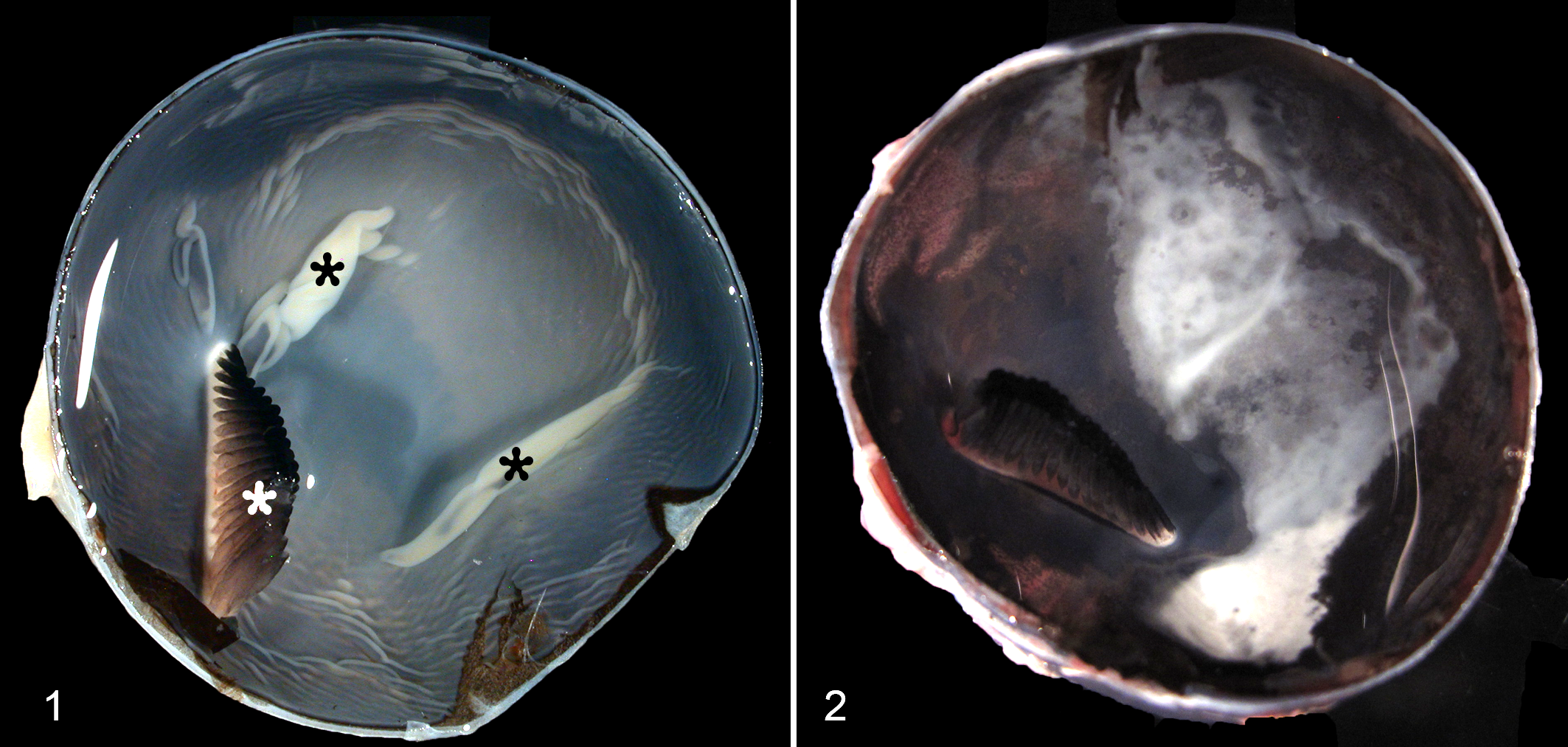
Gross lesions were not detected in any of the 6 eyes of the control birds (Fig. 1). Gross examination of the fundus of the red-tailed hawks with WN disease revealed focal geographical white to gray discoloration in 17 eyes from 12 birds (Fig. 2). These discolorations were often present in the center of the fundus in the vicinity of the pecten. The remaining 4 WNV-infected birds did not have any gross lesions in the eyes.
Histopathology
Histological lesions were absent in the 6 eyes of the 3 control birds. In 9 of 32 (28%) eyes of the 16 WNV-infected hawks, the choroid and retina were devoid of lesions (Fig. 3, Table 1), including both eyes of 2 cases (case Nos. 1 and 2). In 23 of 32 (72%) eyes of the infected birds, the choroid was segmentally infiltrated by a variable number of lymphocytes and plasma cells (Table 1). This infiltration was consistently associated with lesions of the overlying retina. Chorioretinal lesions were of 2 types (type I and type II) and were multifocal with different lesion types and normal tissue all present in neighboring chorioretinal segments.
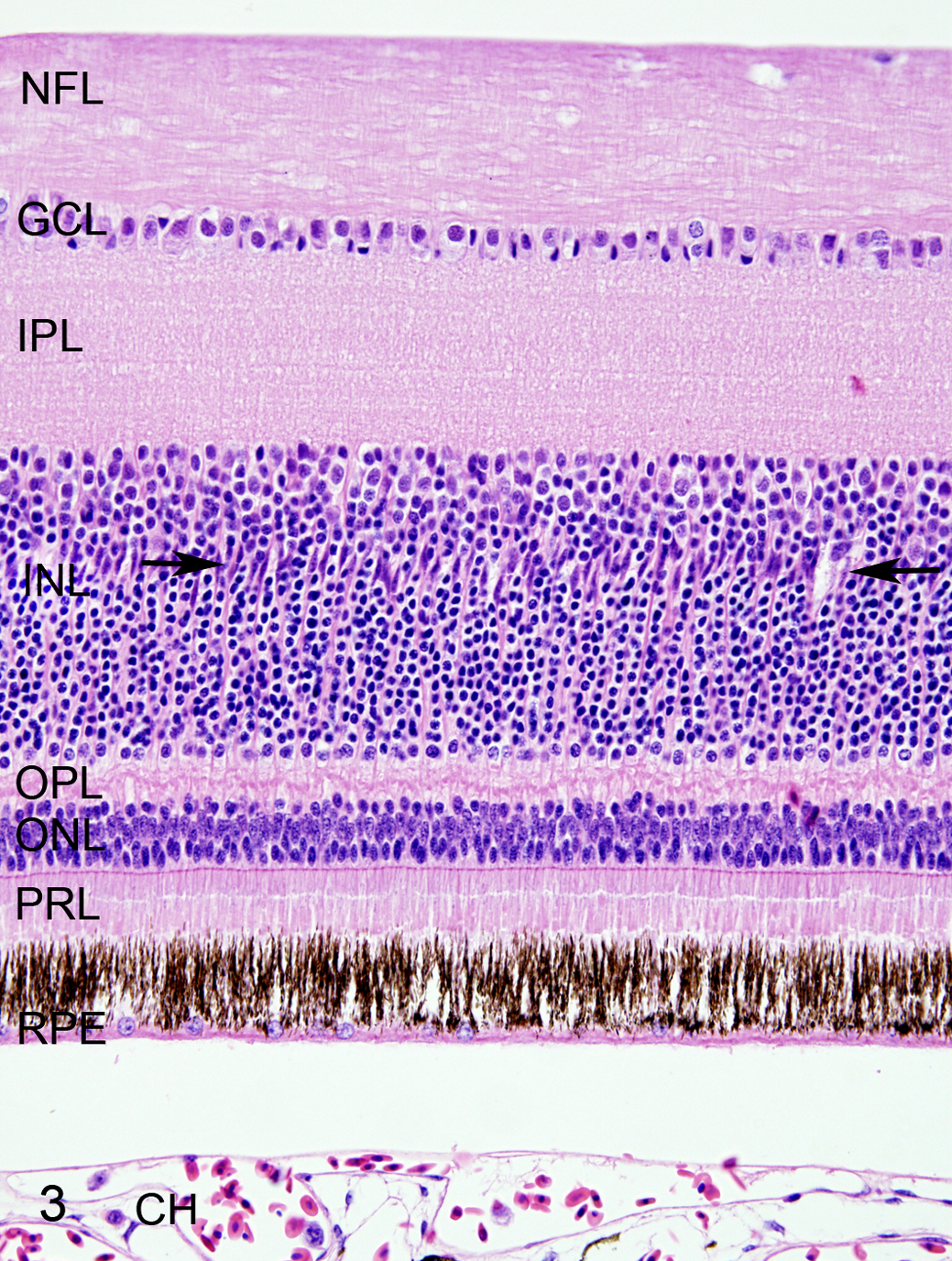
West Nile virus infection, retina, red-tailed hawk, case No. 1. The retina does not have any lesions. All layers of the retina are present, including retinal pigmented epithelium layer (RPE), photoreceptor layer (PRL), outer nuclear layer (ONL), outer plexiform layer (OPL), inner nuclear layer (INL), inner plexiform layer (IPL), ganglion cell layer (GCL), and nerve fiber layer (NFL). The RPE cells have distinct long cell processes. The Müller cell nuclei form a palisade between the upper and middle thirds of the inner nuclear layer (arrows). The space between the RPE cell layer and choroid (CH) is a processing artifact. Hematoxylin and eosin (HE).
Histologic Findings in the Eyes of 16 Red-Tailed Hawks With West Nile Disease.a
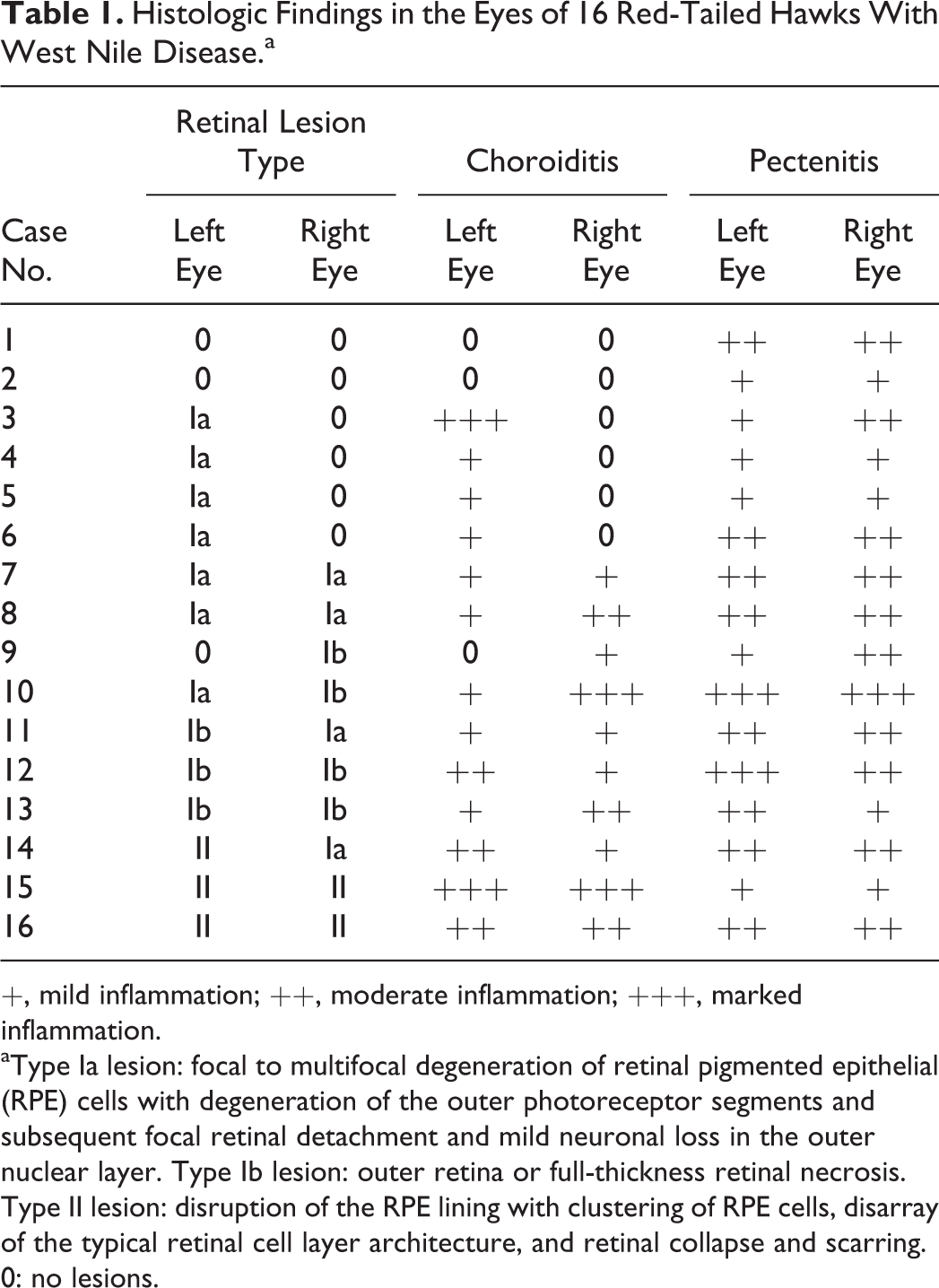
+, mild inflammation; ++, moderate inflammation; +++, marked inflammation.
aType Ia lesion: focal to multifocal degeneration of retinal pigmented epithelial (RPE) cells with degeneration of the outer photoreceptor segments and subsequent focal retinal detachment and mild neuronal loss in the outer nuclear layer. Type Ib lesion: outer retina or full-thickness retinal necrosis. Type II lesion: disruption of the RPE lining with clustering of RPE cells, disarray of the typical retinal cell layer architecture, and retinal collapse and scarring. 0: no lesions.
Type Ia lesion
The most subtle and most common retinal lesion was a focal to multifocal degeneration of RPE cells, as evidenced by the loss of their delicate apical cell processes and the assumption of a rounded cell morphology (“clumping”; Fig. 4). This lesion was detected in 11 eyes from 9 birds. The choroidal infiltrate was mild in 9 eyes with this lesion type. The RPE degeneration was associated with degeneration of the photoreceptors and subsequent focal retinal detachment followed by mild focal to multifocal loss of neurons in the outer nuclear layer. Few macrophages and lymphocytes were scattered in the nerve fiber layer close to the optic disc in 5 of these eyes.
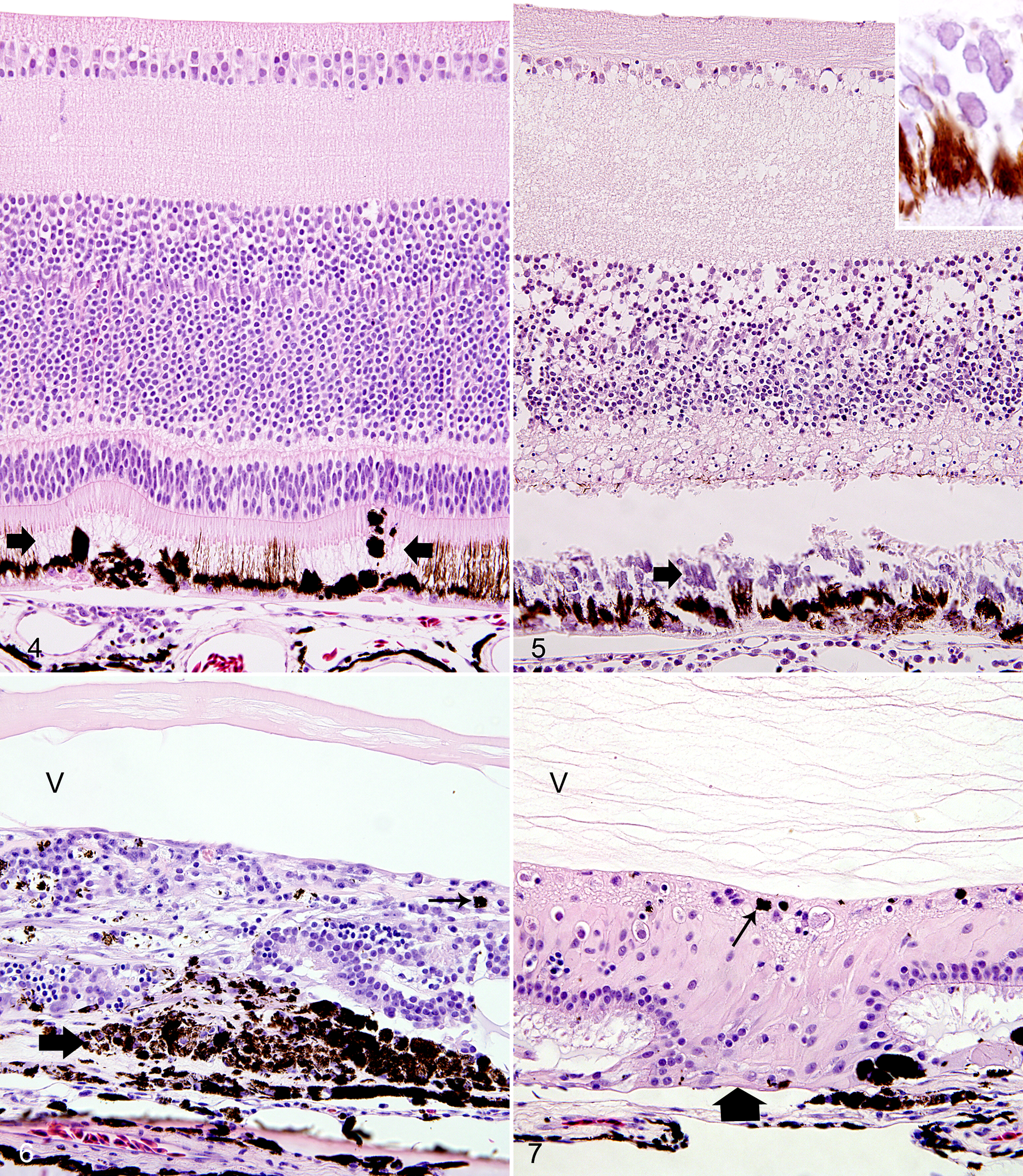
Type Ib lesion
Retinal degeneration and necrosis overlying a segment of choroidal lymphoplasmacytic infiltration were the hallmark of the type Ib lesion (Fig. 5). This lesion was detected in 7 eyes from 5 birds. The necrosis affected either the outer retina or both the outer and inner retina. The necrosis was associated with dystrophic mineralization of the outer segments of the photoreceptors in 4 eyes. Few lymphocytes and plasma cells infiltrated the nerve fiber layer close to the optic disc in 2 eyes with type Ib lesions. In 2 eyes (case Nos. 11 and 12). Type Ia and type Ib lesions occurred simultaneously, but the type Ib lesion was the predominant lesion.
Type II lesion
Lesions of retinal atrophy affected 5 eyes from 3 birds, characterized by disruptions of the RPE with clustering of RPE cells and disarray of the retinal cell layer architecture in the overlying retina (Fig. 6). Müller cell hypertrophy was a prominent feature in 2 eyes (Fig. 7). Overall, there was a marked collapse of the retina. The neurosensory retina was multifocally fused with the choroid in type II lesions of 2 birds (case Nos. 15 and 16). Occasionally, a glial scar completely replaced a retinal segment. The choroiditis subjacent to type II lesions was usually moderate to marked. In the 5 eyes with type II lesions, the nerve fiber layer close to the optic disc and just beneath the inner limiting membrane was infiltrated by a low to moderate number of plasma cells.
Compared with the control eyes (Fig. 8), the pecten was expanded by lymphocytes, plasma cells, and macrophages to a variable degree in all eyes of the birds with WN disease (Table 1). The inflammatory cells lined the luminal endothelial cell surfaces, and there were perivascular infiltrates (Fig. 9). In 2 of the 5 eyes with type II lesions, the pecten was markedly thickened due to deposition of fibrillar material within the stroma of the pecten with a minor inflammatory cell infiltration (Fig. 10).
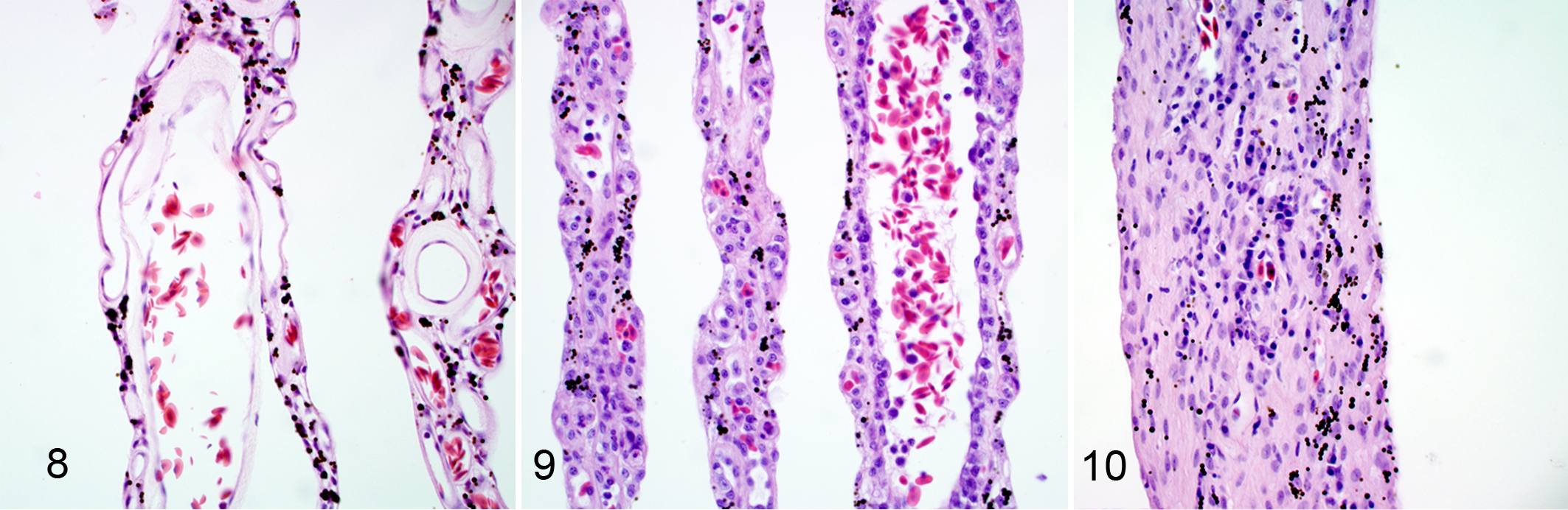
The anterior segment of 19 eyes of 11 WNV-infected birds was characterized by a subtle mononuclear iridocyclitis, with infiltration of the iris stroma particularly along the anterior border, and adherence of lymphocytes to the trabeculae of the iridocorneal meshwork.
Immunohistochemistry
WNV antigen was not detected in the eyes of the control birds. WNV antigen was detected in 25 of 32 (78%) eyes of WNV-infected birds, including at least 1 eye from all 16 animals (Table 2). The degree of WNV antigen expression varied between the eyes and birds. Of the 9 eyes that did not have any histological retinal lesions, WNV antigen was absent in 3, weakly positive in 4, and moderately abundant in 2 eyes from the same bird (case No. 1; Fig. 11). The 4 positive retinas with a low antigen score included 3 retinas in which only individual ganglion cells were positive and 1 retina in which only few cell processes in the outer and inner nuclear layer were WNV antigen positive.
Relationships Among Retinal Lesion Types and Detection of WNV by IHC and RT-qPCR.
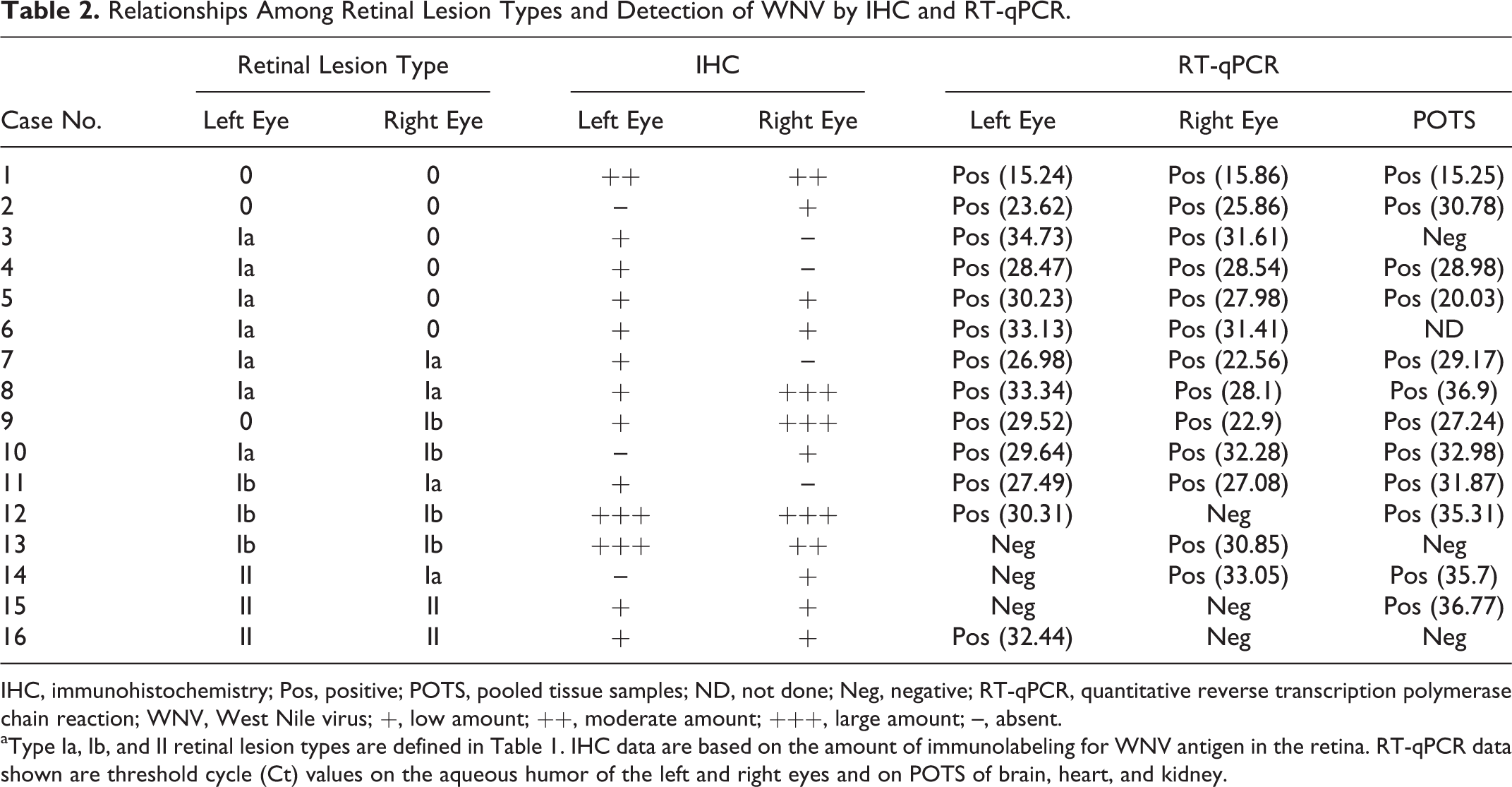
IHC, immunohistochemistry; Pos, positive; POTS, pooled tissue samples; ND, not done; Neg, negative; RT-qPCR, quantitative reverse transcription polymerase chain reaction; WNV, West Nile virus; +, low amount; ++, moderate amount; +++, large amount; –, absent.
aType Ia, Ib, and II retinal lesion types are defined in Table 1. IHC data are based on the amount of immunolabeling for WNV antigen in the retina. RT-qPCR data shown are threshold cycle (Ct) values on the aqueous humor of the left and right eyes and on POTS of brain, heart, and kidney.
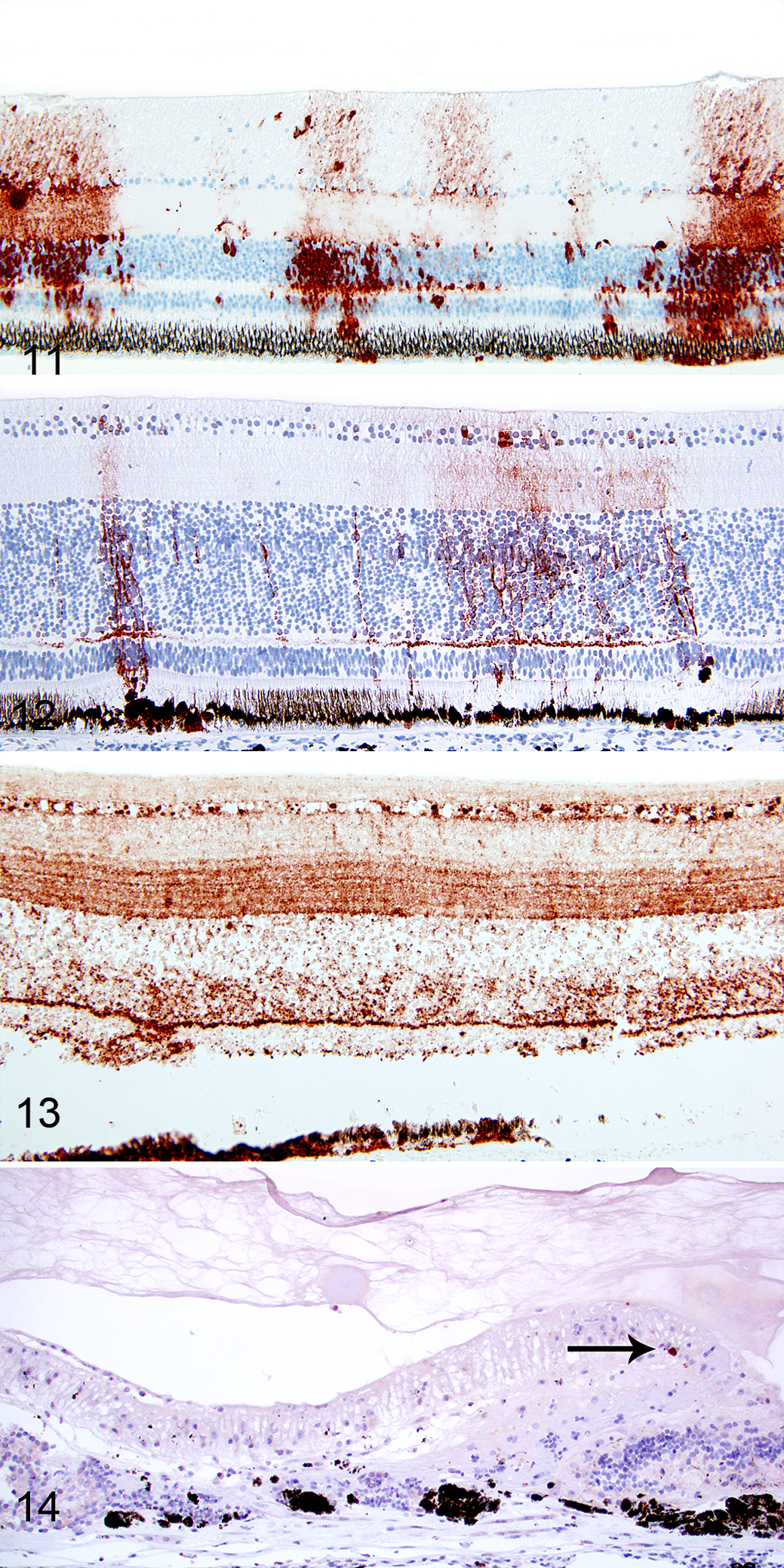
Of the 11 eyes with type Ia lesions, 2 were negative for WNV antigen, 2 had WNV antigen limited to RPE cells, and 7 had WNV antigen-positive cells in all layers of the neurosensory retina besides having positive RPE cells (Fig. 12). Of 7 eyes with overt necrosis of the neurosensory retina (type Ib lesion), 5 had a moderate to marked WNV antigen expression in all retinal layers (Fig. 13), and 2 had a mild WNV antigen expression in cells of the outer and inner retina. In 5 eyes with type II lesions, 4 had mild WNV antigen expression limited to individual cells (Fig. 14), and 1 had no detectable WNV antigen. In retinas with type I lesions, WNV antigen was present in RPE cells (Fig. 15), neurons of all layers (Fig. 15), and Müller cells (Fig. 16). The viral antigen was present in neuronal somata and horizontal and arborizing neuronal cell processes (Fig. 15) and in somata and vertical processes of Müller cells (Fig. 16) or was widespread in all layers (Fig. 17).
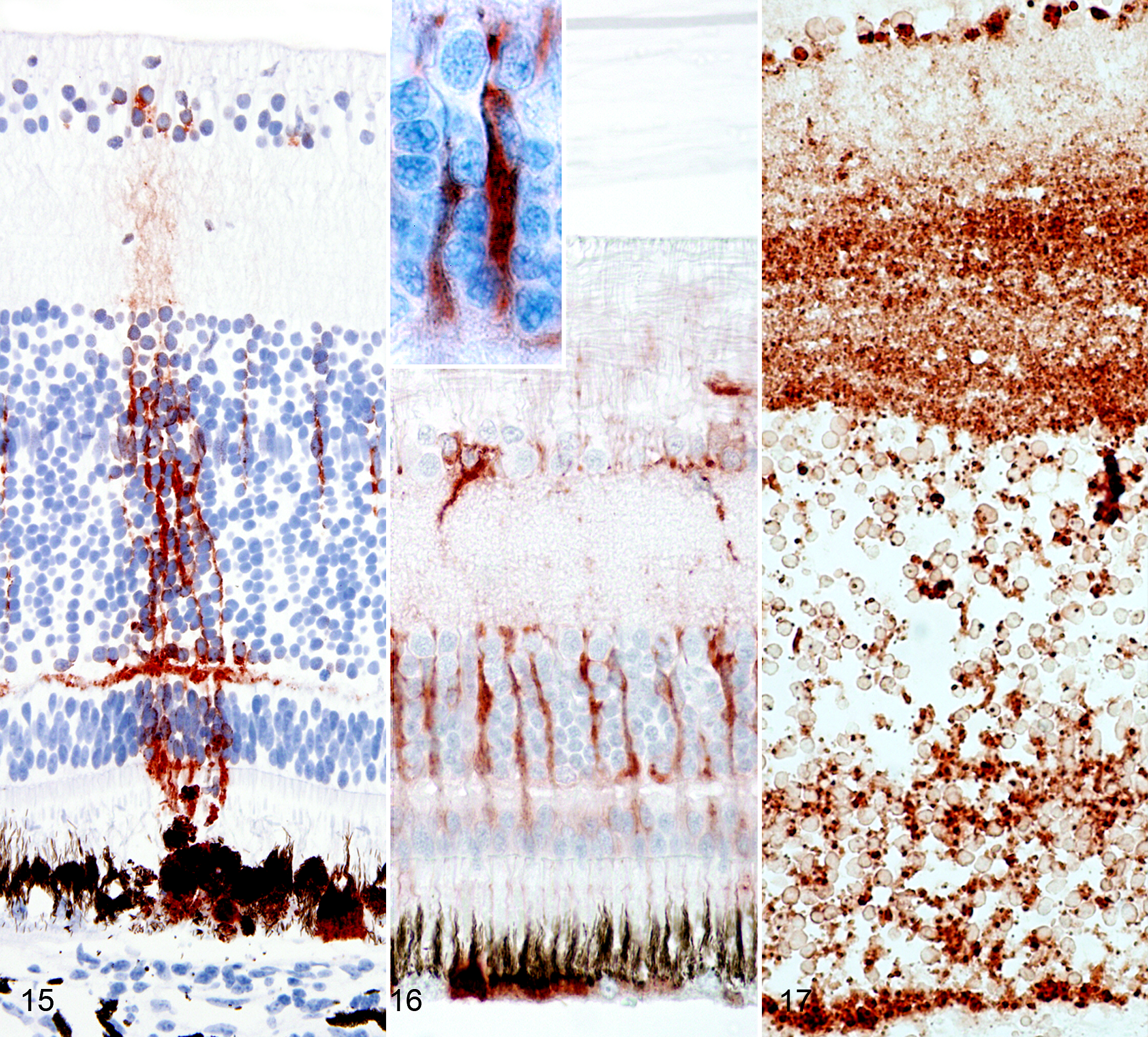
In type II lesions, WNV antigen was limited to individual ganglion cells (Fig. 14), RPE cells, and few cell processes in the inner retina. WNV antigen was detected in the anterior segment of the eye in only 1 hawk (case No. 1). In this bird, the striated muscle cells of the iris were WNV antigen positive. Despite the high prevalence of pectenitis in the examined eye globes, WNV antigen was not detected in the pecten, optic disc, or optic nerve of any eye.
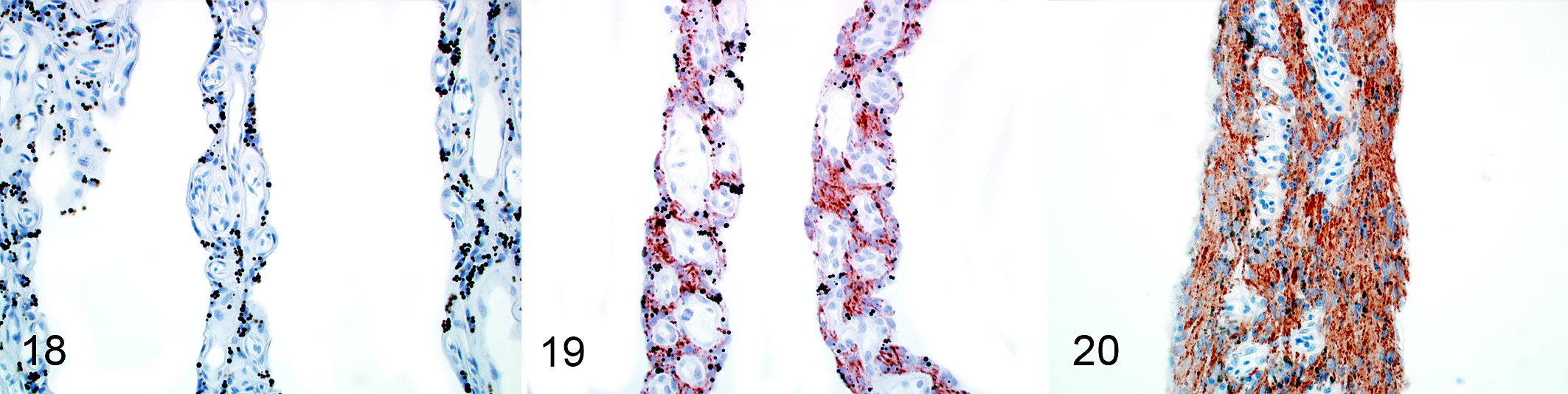
GFAP was delicately but consistently expressed in the optic nerve and optic disc of the control birds while the retina and the lamellae of the pecten were negative (Fig. 18). In contrast, GFAP was expressed in the pecten of 27 of 32 (84%) eyes and in the retina of 7 of 32 (22%) eyes of WNV-infected birds. The GFAP expression in the pecten was mild to moderate in eyes with type I lesions (Fig. 19) and moderate to marked in eyes with type II lesions (Fig. 20). In 2 of 5 (40%) eyes with chronic retinal lesions (case Nos. 15 and 16), GFAP was expressed in hypertrophied Müller cells.
Cleaved Caspase-3 and TUNEL
The retina and pecten of the control birds were devoid of TUNEL-positive or activated caspase-3–positive cells. There were rare scattered individual TUNEL-positive and activated caspase-3–positive cells among the inflammatory mononuclear infiltrate in the choroid and pecten of infected birds. The positive cells likely were lymphocytes or macrophages based on the morphology and location. However, neither TUNEL signal nor activated caspase-3 signals were detected in any of the examined retinas, including areas of histologically detected necrosis.
RT-qPCR
The tissue samples and aqueous humor samples of all control birds were negative for WNV RNA. The aqueous humor sample of at least one eye was positive for WNV RNA in 15 of 16 (93%) birds with WN disease (Table 2). In total, WNV RNA was detected in the aqueous humor of 26 of 32 (81%) eyes. The aqueous humor was positive for WNV nuclei acid in 5 of 7 (71%) eyes with type Ib lesions and 1 of 5 (20%) eyes with type II lesions. Pooled tissue was positive for WNV RNA in 13 of 16 (81%) infected birds, including the bird that was negative for WNV RNA in the aqueous humor of both eyes (case No. 15; Table 2). The pooled tissues of 3 of 16 (19%) infected birds were negative (case Nos. 3, 13, and 16) for WNV RNA. RT-qPCR for EEE and SLE yielded negative results in all cases.
Statistical Analysis
A significant correlation was observed between the lesion type and the degree of choroiditis subjacent to the lesion (τ = 0.79,
In contrast, the lesion type was not significantly correlated with the amount of viral antigen detected based on IHC scores (τ = 0.19,
Discussion
The study confirms the previously described high prevalence of ocular lesions in red-tailed hawks with WN disease. 26,36 In most infected birds, ocular lesions were grossly visible as discolorations of the fundus region in at least 1 eye upon careful examination. WNV-associated ocular macroscopic lesions have only been rarely reported in any species but are more prevalent in red-tailed hawks and related species. 37,39 Ocular manifestation of WNV infection is common in humans (eg, as funduscopically detectable chorioretinitis) but usually is clinically inapparent. 13,16 While initial work in hawks focused on the inflammatory changes in choroid and pecten, this study emphasized the histopathologic features of different types of retinal lesions. 26,36,37
As a necropsy study of naturally infected animals, the study was necessarily based on sampling at one time point of the infection only. Nevertheless, the results of this study allow speculations regarding the pathogenesis. Histologically, 2 principal retinal lesions were distinguishable herein, named type I lesions and type II lesions. Based on the morphology of the lesions but also taking into account the viral load estimated using immunohistochemistry and measured by RT-qPCR and GFAP expression, particularly in the pecten, the type I lesions represent acute lesions while the type II lesions represent chronic lesions. Type Ia lesions may be the earliest lesion of WNV-associated retinal damage.
Multiple alternative or concurrent routes of viral infection of the avian retina are possible: (1) neurogenic retrograde spread via the optic nerve, (2) hematogenous cell–associated or extracellular spread via an impaired blood-retinal barrier (BRB), and (3) hematogenous cell–associated or extracellular spread via the aqueous and vitreous humor (eg, with virus entering vitreous from the pecten and/or with virus entering the aqueous humor at the iris or ciliary body via the blood-aqueous barrier). 34 WNV has been shown to use both neurogenic and hematogenous routes to infect the central nervous system in experimental infection models. 8 In experimental infection of red-legged partridges, the findings suggested that the retina became infected via the hematogenous route since viral antigen was detected in the retina several days prior to infection of the brain. 10
In our study, the infection of RPE cells in the majority of red-tailed hawks with retinal type I lesions supports the hypothesis that a sustained infection of the retina occurs via the BRB. The virus appeared to spread toward the inner retina via both neuronal processes and Müller cell processes. By comparison, in patients with human immunodeficiency virus–associated immunosuppression, human cytomegalovirus infects the retina via the BRB, although in mammalians, the intraretinal vasculature serves as a portal of entry. The avian retina is avascular, and hence the BRB is restricted to the choriocapillaris-RPE interface. 29
One of the infected birds was apparently euthanatized during a very early stage of infection (case No. 1). This bird carried an unusually large WNV load, as evidenced by the low Ct values in the aqueous humor and a pooled tissue sample. Furthermore, in contrast to the other infected animals, this animal was in a good nutritional state and had massive myocardial necrosis (data not shown). This was the only bird that had any nonretinal WNV antigen-positive cells in the eye (ie, striated muscle cells of the iris). In addition, this bird had abundant viral antigen in the retina without any histologic chorioretinal degenerative or inflammatory lesions, although the pecten was infiltrated by mononuclear inflammatory cells. The immunopositivity of the striated muscle cells of the iris for WNV antigen suggests that WNV may pass the blood-aqueous barrier in the early phase of the infection at least transiently.
WNV antigen was not detected in the optic nerve in any of the examined sections, indicating that this is not a likely route of viral spread. Retrograde transport of virus from the brain to the eye via the optic nerve is the route of infection by equine Borna disease virus. 20 The neurogenic route of infection of equine Borna disease virus is also reflected in its antigen distribution in the retina, in that the antigen is detected first in the nerve fiber layer and ganglion cell layer before it spreads to the outer retina in experimentally infected rats. 32
In our study, retinal lesions of any type were invariably associated with infiltration of the subjacent choroid with mononuclear inflammatory cells. Furthermore, the presence of choroiditis was usually associated with viral antigen in the overlying retinal segment. Hence, we speculate that choroiditis may constitute a prerequisite for the development of retinal lesions. Immunopathologic consequences of the antiviral immune response appear to contribute to the pathogenesis of neuroinfection in several animal models of WNV. 8 Degeneration and necrosis of photoreceptor cells are associated with the RPE cell damage and may then progress to necrosis of the outer nuclear layer of the retina and ultimately full-thickness retinal necrosis with or without calcification of the outer retinal segment (type Ib lesion). Retinal scarring with loss of neurons, disarray of layers, upregulation of GFAP expression in Müller cells, and fibrillary astrocytosis in the pecten may be a late stage of the retinal injury. In the proposed pathogenesis of the retinal lesion, partial or complete elimination of virus may explain the scarcity or absence of WNV antigen in type II lesions.
Infiltration of the retina with inflammatory cells was not a prominent feature, although few scattered lymphocytes and macrophages were present in the nerve fiber layer of some eyes with type Ia and Ib lesions, and plasma cells were present along the inner limiting membrane of the nerve fiber layer in few eyes with type II lesions.
With respect to the cell tropism, this study provided evidence that both glial cells and neurons are targeted by WNV. WNV antigen was present in RPE cells and neurons of all layers (ie, ganglion cells, photoreceptor cells, and neurons of the inner nuclear layer). It is well established that neurons are the major target of WNV in the central nervous system. 3,12 Human RPE cells are susceptible to WNV infection in vitro and produce interferon β in response to the infection. 5 Experimentally, WNV-infected human RPE cells upregulate proinflammatory cytokine gene expression, including chemokines that have the potential to attract mononuclear cells and neutrophils. 24 The combined effects of WNV on RPE cells likely impair their function as part of the blood-retinal barrier, allowing for the infection of the neurosensory retina. 24 In addition to neurons, retinal Müller cells were also infected by WNV in this study. Müller cells are critical support cells, regulating ion and water homeostasis of the retina and providing neurons with trophic substances and removing waste products. 2
In recent years, the role of glial cells, particularly astrocytes, in the pathogenesis of WNV-associated brain disease has received increased attention. In vitro studies suggest that astrocytes are permissive to infection with both pathogenic and nonpathogenic WNV strains and do not undergo rapid cell death but instead may allow viral replication and assembly of infectious particles for a long time. 6,15,23 Therefore, astrocytes are thought to play a role in the spread of WNV in the central nervous system. One case report also suggests that glial cells may have a role in maintaining WNV infection in immunocompromised WNV-infected humans. 27 In our study, hypertrophic Müller cells in type II lesions did not show evidence of WNV infection based on IHC.
Viral infection may trigger caspase-3–dependent and caspase-3–independent apoptotic pathways in neurons. 31 Apoptosis, particularly caspase-3–dependent apoptosis, did not appear to be a significant mechanism of cell death in the retina in the examined hawks based on the lack of labeling of cells by the TUNEL method and lack of immunoreactivity for activated (cleaved) caspase-3. Caspase-3–dependent cell death has been shown to play an important role in the pathogenesis of experimentally induced WN disease in mice and hamsters, in neuron cell culture, and in Vero cell culture. 4,18,19,30,40 WNV is capable of causing necrosis and inducing apoptosis in Vero cell cultures dependent on the degree of the infection. Low-dose infection was associated with apoptosis while high-dose infection resulted in necrosis of the infected Vero cells. 4 Human RPE cells infected with WNV upregulate gene expression of a proapoptotic mediator and downregulate the expression of an antiapoptotic mediator. 24
In conclusion, WNV infection is commonly associated with gross and histologic retinal lesions in red-tailed hawks. Retinal changes are classified as acute type I lesions and chronic type II lesions. The findings of our study support the hypothesis that WNV infects the retina hematogenously after breakdown of the blood-retina barrier and spreads toward the nerve fiber layer via neuronal and Müller cell processes. WNV antigen tends to be present in larger quantity in type I lesions partly in the absence of retinal damage, while WNV antigen quantity tends to be scant or absent in type II lesions. The development of retinal lesions is associated with the presence of an inflammatory infiltrate in the choroid. Apoptosis, and more specifically caspase-3–dependent apoptosis, does not appear to be a significant mechanism in retinal damage in red-tailed hawks.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Morris Animal Foundation (D07ZO-303).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
