Abstract
This study describes the clinicopathologic findings in naturally occurring West Nile virus (WNV) infection in horses. WNV was diagnosed in a foal by immunohistochemical and in situ hybridization methods, and the presence of WNV antibodies was detected in 5 other horses with clinical signs suggestive of WNV infection. At necropsy of the foal, lymph nodes were edematous and enlarged, and the intestines showed diffuse congestion and focal hemorrhages. The most significant histologic lesions in this case were nonsuppurative meningoencephalomyelitis, particularly in the brainstem and spinal cord. Identification of viral RNA by in situ hybridization and viral antigen by immunohistochemistry was concentrated primarily in nerve fibers, glial cells, and their processes in brainstem and spinal cord and, to a lesser extent, within the cerebral hemispheres and cerebellum.
West Nile virus (WNV)—an enveloped positive-sense single-stranded RNA virus of the genus Flavivirus, family Flaviviridae—causes epidemics and/or epizootics of encephalomyelitis and mortality in certain domestic and wild birds. 6 During peak infection periods, as governed by favorable climatic conditions, adult female mosquitoes carrying WNV can transmit the virus to humans, horses, and other mammalian species, which are all considered to be dead-end hosts. 8 Clinically, WNV disease in horses is characterized by neurologic signs such as ataxia, weakness, hyperesthesia, muscle fasciculations, and paralysis. 1 Histopathologically, lesions of the central nervous system (CNS) are characterized by nonsuppurative meningoencephalomyelitis and perivascular hemorrhages, localized mostly in the brainstem and spinal cord. 1,6
The first serologic evidence of WNV infection in Turkey was reported in the 1970s based on the detection antibodies, while the first human cases with neurologic signs associated with WNV were reported in 2009 from Aegean Region and subsequently from other regions in Turkey. 2 A seroepidemiologic study in domestic animals from 8 representative provinces of Turkey has shown the positivity for WNV-neutralizing antibodies in 1 of 40 (2.5%) donkeys and mules, 4 of 100 (4%) cattle, 43 of 114 (37.7%) dogs, 35 of 259 (13.5%) horses, 18 of 88 (20.4%) humans, and 1 of 100 (1%) sheep. 4 Recently, WNV RNA was detected in sick horses (buffy coat) and in humans (cerebrospinal fluid), and the partial nucleotide sequences of the E-gene coding region were closely related to viruses of lineage 1, clade 1a. 3 This study reports the detection of WNV-infected horses with neurologic signs in Turkey, using immunohistochemistry (IHC), in situ hybridization (ISH), and ELISA techniques.
A 2-month-old female Rahvan foal from the Aegean Region of Turkey was presented for postmortem evaluation in July 2011 with a 1-week history of stagnation and posterior paralysis prior to death. Necropsy was performed the same day, and collected tissue samples included sections of the brain, intestines, lymph nodes, spleen, liver, lungs, and kidneys, which were fixed in 10% buffered formalin and embedded in paraffin. For histopathologic examination, sections were cut at 5-μm thickness and stained with hematoxylin and eosin. Replicate unstained sections were used for IHC and ISH.
For serologic and laboratory examinations, blood samples were drawn from 5 adult horses (ranging in age from 5 to 8 years), all of which had clinical signs consisted with WNV infection. The serum samples were tested for equine viral arteritis virus (by virus neutralization test), equine infectious anemia virus (by agar gel immunodiffusion test), and WNV infections (by antibody ELISA) in Etlik Veterinary Control and Research Institute.
For immunolabeling of the WNV antigen, the fluorescent antibody method was used according to Toplu et al. 9 In that method, mouse anti-WNV polyclonal antibody (kindly provided by Dr A. Özkul, Turkey) was used as primary antibody. The secondary antibody was goat anti-mouse IgG-FITC conjugate (Santa Cruz Biotechnology).
For the labeling of viral RNAs, ISH was performed on each brain section with the digoxigenin (DIG)–labeled probe using the ISH detection system core kit-DD130-60 K (Biogenex Technology, USA). Positive-control WNV RNA was kindly provided by Dr Nicholas Johnson (Veterinary Laboratories Agency, Weybridge, UK) and used for preparation of the DIG-labeled probe. Initially, the NS5 and 3′NTR of the WNV genome from reference strain were amplified using primers (EMF1/VD8) 5 and a 789–base pair WNV-specific amplicon was obtained by reverse transcription polymerase chain reaction (PCR), the product of which was purified using a commercial kit (Combokit, Qiagen, Germany). This purified PCR product was used as template DNA and labeled with the PCR DIG Probe Synthesis Kit (Cat No. 11 636 090 910, Roche, Germany). After labeling, the probe was analyzed in 1% agarose-ethidium bromide gels in Tris-acetate buffer and visualized on an ultraviolet transilluminator.
Serum samples were analyzed for the detection of WNV antibodies by a commercial ELISA test (anti-pr-E antibodies in multiple species, ID Innovative Diagnostic VET, ID Screen West Nile Competition, France) according to the manufacturer’s instructions. The resulting coloration depended on the quantity of specific antibodies present in the sample tested. Photometric measurement at a wave length of 450 nm was done according to the manufacturer’s instructions.
In addition to the case that was autopsied, 4 other foals (2 to 3 months of age) died with similar clinical signs of paralysis during the summer months of 2010 and 2011. In addition, deaths of foal with neurologic signs were also anecdotally reported in neighboring farms. It was epidemiologically important that the case clusters were most prevalent in farms next to the Büyük Menderes River, where high humidity (about 90%) and high temperature (about 35°C to 40°C), coincide with mosquito abundance during the summer months.
Clinical signs compatible with a neurologic disorder and consistent with WNV were reported in the 5 adult horses that were sampled for diagnosis. This clinical presentation included signs such as fever, ataxia, muscle/limb weakness, and hyperesthesia. Other relevant signs noted in these animals with decreasing frequency included paralyzed lips, recumbency, depression, twitching, teeth grinding, and muscle fasciculations (Table 1). Interestingly, all 5 horses were markedly hyperesthetic. Additionally, the autopsied foal had a history of severe neurologic signs involving many of the symptoms described but also posterior paralysis. No concurrent etiologies compatible with any other vector-transmitted diseases were reported in any of the 6 evaluated cases.
Clinical Findings in Horses Infected With West Nile Virus (N = 5).
Serum samples of all 5 horses were positive for WNV with antibody ELISA test with anti-pr-E, negative for equine viral arteritis with virus neutralization, and negative for equine infectious anemia with AGID (EIAV Ab Test Kit, IDEXX, Maine, USA).
At necropsy, lymph nodes of the foal were edematous and enlarged. In small and large intestines, there was diffuse congestion and focal hemorrhagic lesions on the serosal surfaces. Splenic lesions consisted of petechial hemorrhages. The lungs were observed to have leakage of serous exudate from cut surfaces. No additional macroscopic findings were noted.
Histopathologically, severe microscopic changes consisted of a nonsuppurative meningoencephalomyelitis, localized particularly in the brainstem (medulla oblongata and pons and, to a lesser extent, the thalamus and mesencephalon) as well as the spinal cord. Congested vessels showed mild to severe perivascular infiltrations with lymphocytes and few macrophages and plasma cells. Perivascular hemorrhages were also marked in these areas (Fig. 1). Focal or scattered gliosis and occasional neuronophagia were noted. Spheroids (swollen axons) and vacuoles were observed in the spinal cord and medulla oblongata. Additionally, neuronal degeneration characterized by central chromatolysis and cell shrinkage was conspicuous, particularly in the motor neurons of the brainstem and spinal cord.
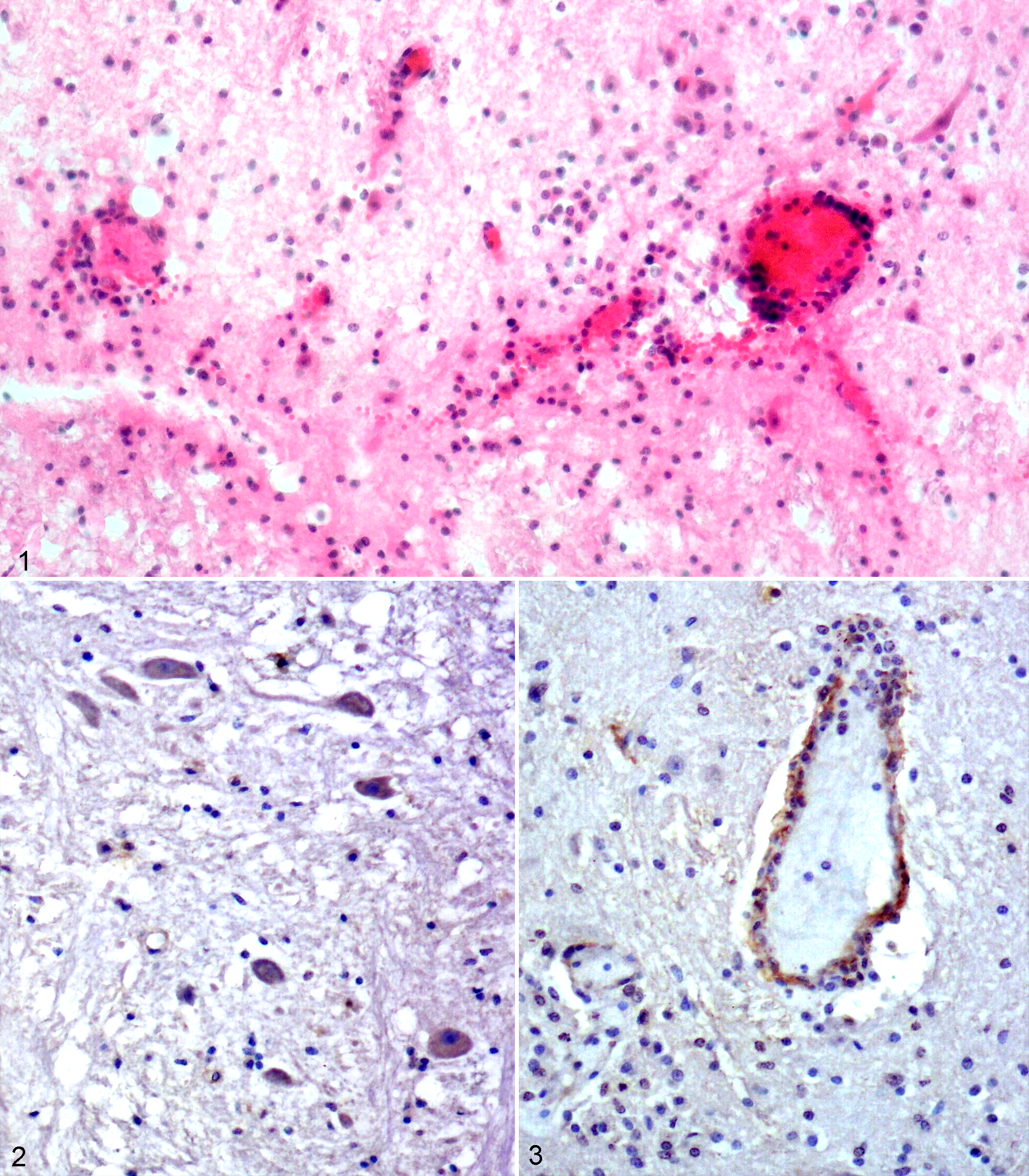
West Nile virus (WNV) infection, brainstem, foal.
In the intestines, lesions consisted of diffuse edema of the submucosa and infiltration of the lamina propria mucosae with mononuclear cells and a few eosinophils. Many nematode sections were observed among the villi. In the spleen, lymph nodes, and tonsils, there were marked follicular hyperplasia with germinal center formations and destruction of lymphoid follicles in cortical and paracortical regions, characterized by lymphoid depletion and karyorrhexis. Sinusoids were dilated and filled with macrophages and plasma cells. Coagulative necrosis was seen predominantly in midzonal areas of the liver. The lungs showed mainly edema in bronchi, bronchioles, and alveoli and interstitial pneumonia.
ISH and IHC revealed that tissue localization of WNV nucleic acid and antigens were associated with the histopathologic findings. Results of ISH with both DIG-labeled probes and IHC with WNV antibody showed that there was a good correlation between the intensity and distribution of both the viral nucleic acid with that of the viral antigen. Labeling of viral RNA and antigen was mainly detected in the nerve fibers, spheroids, and glial cells of the brainstem and spinal cord, and to a lesser extent within cerebrum and cerebellum (Fig. 2). Additionally, occasional labeling of viral RNA and antigen were also observed within endothelial cells (Fig. 3) particularly in brainstem. Conversely, only slight labeling of neuronal cell bodies was seen with ISH and IHC.
Previous reports state that histologic lesions of the CNS in horses infected with WNV, similar to those seen in the present case, are characterized by polioencephalomyelitis, with typical involvement of the brainstem and ventral and lateral horns of the spinal cord, and are associated with moderate to severe hemorrhage within the CNS. 1 Neuronal degeneration characterized by central chromatolysis and cell shrinkage as noted in the present case has also been described. 1 The mechanism of neuronal degeneration is controversial. In a hamster model of WNV infection, many degenerating neurons were theorized to be undergoing apoptosis, but there did not appear to be an association with inflammatory cells, and the authors suggested that the cellular damage was caused by the direct effect of WNV infection. 10 In contrast, the lack of viral nucleic acid and antigen in degenerated motor neurons in the present case indicates that WNV might not be associated with direct neuronal damage. Cantile et al suggested that neuronal damage in natural WNV infection of horses might have an immunopathologic component, since inflammatory changes were present despite the absence of significant detectable WNV antigen. 1 The presence of significant lesions in spinal cord, cerebral hemispheres, and cerebellum, associated with less antigenic staining and fewer viral transcripts than in the brainstem (pons, thalamus, medulla oblongata) in our case supports the idea of an immunopathologic component in the pathogenesis of WNV infection.
Pathogenesis studies indicated that WNV, after viremia, might spread rapidly throughout the neuropil and replicate in particularly sensitive areas, such as hippocampus in mice and thalamus, substantia nigra, and cerebellum in monkeys and humans. 7 The strong positive reactions of WNV antigen in the neuropil and in glial cells indicate that the animal of this study had a high viral load and died during the period of acute infection. In the present case, abundant WNV was localized in nerve fibers, and glial cells, but ISH and IHC showed scant labeling in the neurons and neuronal processes. Similar observations within neurons and necrotic areas, mainly of the brainstem and spinal cord, have been demonstrated by IHC in an investigation of human WNV encephalitis. The disassociation of virion density from lesion severity suggests an immunopathologic mechanism in the pathogenesis of WNV disease in horses. 1
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
