Abstract
Fifty feline sarcomas associated with vaccine-site injection were evaluated to determine the immunohistochemical expression of p53 protein, basic fibroblast growth factor (FGF-b), and transforming growth factor-α (TGF-α). Forty-one tumors (82%) were fibrosarcomas (FS), eight (16%) were malignant fibrous histiocytomas (MFH), and one (2%) was a chondrosarcoma (CS). Overexpression of p53 protein was observed in the nuclei of tumor cells in 28 (56%) sarcomas; FGF-b expression was found in the cytoplasm of tumor cells in 40 (80%) feline sarcomas, but the staining was more intense in the spindle-shaped cells of FS than in polygonal or round cells of MFH. The single CS faintly expressed FGF-b. The majority of feline vaccine-associated sarcomas (43 of 50, 86%) expressed moderate or intense staining for TGF-α in the cytoplasm of tumor cells. Heterogeneous immunolabeling for p53, FGF-b, and TGF-α was present in neoplastic, multinucleated giant cells. Intense expression of FGF-b was statistically associated with younger cats (P < 0.01) and with tumors with nodular growth patterns (P = 0.02). In addition, sarcomas negative for p53 protein expressed FGF-b more frequently than did p53-positive tumors (P = 0.04). The frequency of FGF-b immunostaining was significantly higher in sarcomas with intense expression of TGF-α (P = 0.05). Immunohistochemical detection of p53 protein, FGF-b, and TGF-α suggests that these growth-regulating proteins may play different roles in the development of sarcomas associated with vaccine sites.
Vaccine-associated sarcoma in cats has been investigated since the early 1990s by numerous authors. 4,8,15,16,19,24 However, despite an increasing incidence of this disease, the mechanism of tumorigenesis has not been determined nor has a relationship emerged between the occurrence and specific manufacturers of adjuvant or vaccine product. 14,15,25
Transition between inflammation or wound healing and tumors has been frequently observed in different animal models of virus- or oncogene-induced tumors, in which inflammatory compounds appear to play a role in carcinogenesis. 20,42 In cats, early vaccination sites show persistent inflammatory or foreign-body reactions characterized by areas of necrosis, aggregates of lymphocytes and plasma cells, and granulation tissue formation. 4,6,15,19 This reaction is thought to predispose fibroblasts or myofibroblasts to proliferate, leading to neoplastic transformation through different mechanisms including activation of oncogenes and inactivation of tumor suppressor genes. 15 On the other hand, agents that promote inflammation such as acidic fibroblast growth factor (FGF-a) and basic FGF (FGF-b) create a favorable environment for expression of oncogenes and subsequent development of tumors. 27
The p53 tumor suppressor gene is a transcription factor that regulates the expression of genes involved in cell-cycle control, apoptosis in cells with defective deoxyribonucleic acid (DNA), cellular differentiation, and genetic instability. 13,37 Mutations in the p53 gene have been associated with human and animal neoplasms. 13,26,37,46 To investigate the role of the p53 gene in oncogenesis in cats, molecular cloning and chromosomal mapping of the feline p53 tumor suppressor gene were carried out. 35 p53 genetic aberrations have been observed in codons 180 and 248 from exons 5–7 in two of 10 feline fibrosarcomas (FS). 29 Of 60 tumors investigated, missense mutations were also detected in two FS, one malignant fibrous histiocytoma (MFH), one undifferentiated carcinoma of the skin, and one mammary carcinoma. The problematic histopathologic overlap between FS and MFH was also identified by Mayr et al. 28 Recently, a single missense mutation in the exons 5 through 8 and intron 5 was found in 5 of the 40 feline vaccine site–associated sarcomas. 31
The wild-type p53 protein is not detected by immunohistochemistry because of the short half-life of about 15–20 minutes. 37 Missense mutations leading to amino acid substitutions may induce p53 protein stabilization, resulting in accumulations of nuclear p53 proteins that are detectable by immunohistochemistry. 17,37 p53 protein was detected using immunohistochemistry in various feline cancer types, including carcinomas and sarcomas. 32,33
Recently, different authors have confirmed that growth factors (GFs) not only promote proliferation but also induce malignant transformation and regulation of angiogenesis. 10,18,38,40 Overexpression of GFs has been found in different human tumors and is considered to be one of the causes of carcinogenesis. 11,45
FGF-b is a prototype member of FGF family that comprises 20 members, with pleiotropic effects in different cells and organ systems. 1,12 FGF-b is involved in inflammation and wound healing 2,27,36 and in promoting nerve survival and regeneration after central nervous system injury. 23 It is also known that FGF-b can activate DNA synthesis in mesenchymal cells such as fibroblasts and smooth muscles, stimulating growth of tumors derived from these cells. 2,12 In addition, numerous studies have shown that FGF-b can stimulate division and migration of vascular endothelial cells, essential for sustaining tumor growth and enabling metastasis. 10,18
Transforming growth factor-α (TGF-α) is a protein of 50 amino acids belonging to the epidermal growth factor (EGF) family that was initially called sarcoma growth factor because of its profound effects on the morphology of rat fibroblasts. 7 TGF-α binds the EGF receptor (EGF-R), triggering a cascade of events that leads to regulation of epithelial and mesenchymal cell growth. 21,38 EGF- or TGF-α–induced mutation in the p53 gene with overexpression of the mutant p53 product causes enhanced signaling in vulvar squamous carcinoma cell line. 5 Expression of TGF-α can be induced by several viral and cellular oncogenes and causes a mitogenic effect in a variety of cells. 34 An increase in TGF-α expression has been detected in several malignant neoplasms in humans, including MFH. 11,40,44
The aims of this study were to establish the immunoexpression patterns of p53 protein in feline vaccine-associated sarcomas and to investigate its relationship to the presence of FGF-b and TGF-α. Both GFs participate actively in malignant progression of tumors derived from mesenchymal cells. In addition, p53, FGF-b, and TGF-α staining patterns were correlated with clinical and morphologic features of these tumors in an attempt to shed light on their pathogenesis.
Materials and Methods
Fifty primary sarcomas located in areas routinely used by veterinarians for vaccination (dorsal neck or interscapular region, dorsolateral thorax, hind limbs, dorsal lumbar region) were included in this study. Tumors were obtained from animals surgically treated in the Veterinary Teaching Hospital of the Veterinary School of Madrid and from biopsies received by the Pathology Service of the hospital. History and clinical information were obtained from the owners or veterinarians and included age; breed; sex; vaccination histories of the cats; and gross descriptions of the tumors including size, adhesion to surrounding tissues, and ulceration of overlying skin. Follow-up data (recurrence, metastases, and time and cause of death) were also recorded when available.
Tumor samples were fixed in 10% buffered formalin, embedded in paraffin, cut into 4-µm sections, and stained with hematoxylin and eosin. The histologic diagnosis was made following the criteria previously described. 15,25 The histologic grade (I–III) was established by scoring cellular differentiation, presence and extension of necrosis, and mitotic rate according to the grading system previously described. 6 The criteria of Couto et al. 6 were used to classify the multinucleated giant cells as neoplastic, reactive or of undetermined origin.
Immunohistochemistry
Immunolabeling was performed on formalin-fixed, paraffin-embedded samples using the streptavidin–biotin–peroxidase complex method. Two distinct high-temperature pretreatments were used as antigen unmasking protocols. For p53 and TGF-α, slides were placed in a pressure cooker containing 2 liters of 10 mM sodium citrate buffer (pH 6.0) and heated for 2 minutes. The slides were cooled in distilled water and washed in Tris-buffered saline (TBS) (0.1 M Tris base, 0.9% NaCl, pH 7.4). For FGF-b detection, the slides were placed in plastic jars and boiled in a microwave oven at maximum power with sodium citrate buffer (3 times for 5 minutes each) and cooled at room temperature (RT) in the buffer for 20 minutes. After the pretreatment, sections were incubated for 15 minutes with 1% hydrogen peroxide to block endogenous peroxidase activity. After a 5-minute wash in distilled water, the sections were incubated for 10 minutes with 0.05% saponin (Sigma Chemical Co., S-2149, St. Louis, MO) and then with protein-blocking sera (10% normal goat and rabbit serum) to reduce nonspecific reactions.
The primary antibodies used were polyclonal anti-human p53 (Novocastra NCL p53-CM1, at a dilution of 1 : 200, incubated overnight at 4 C), polyclonal anti-FGF-b (Oncogene, 5 µg/ml, overnight, 4 C), and monoclonal anti-TGF-α (Oncogene clone 213-4.1, 1 : 100, overnight, 4 C). The specificity of these antibodies was previously confirmed in feline vaccine-associated sarcomas 32,33 and human tumors. 11,45 The slides were then incubated with anti-mouse biotinylated secondary antibody (Dako EC 4233, 1 : 200, 30 minutes at RT) for anti-TGF-α monoclonal primary antibody or with goat anti-rabbit biotinylated secondary antibody (Vector Laboratories BA 1000, 1 : 400, 30 minutes at RT) for p53 and FGF-b polyclonal primary antibodies. The slides were next incubated with streptavidin–biotin–peroxidase complex (Zymed P50242, 1 : 400, 30 minutes at RT). All washes (5 minutes, three times between each incubation step) and dilutions were made in TBS. Immunoreactivity was observed with 3-3′ diaminobenzidine tetrachloride (Sigma Chemical Co., D5050) and H2O2 (0.01%) in TBS. After washing in distilled water for 10 minutes, slides were counterstained for 2 minutes with Gill's hematoxylin, washed in tap water, dehydrated, and mounted. Negative control slides were prepared by substituting the primary antibody with TBS or with normal mouse or rabbit serum. In addition, to avoid the possible defects in antibody specificity or fixation protocols, normal fibroblasts of the skin adjacent to sarcomas were taken as negative control in every slide. Feline mammary carcinomas positive for p53 protein (presence of p53 gene mutation demonstrated by polymerase chain reaction, data not shown) and feline sarcomas previously stained and positive for p53, FGF-b, and TGF-α (following the same protocol described before) were used as positive controls for every immunolabeling.
Assessment of staining for p53 was performed using a computer-assisted image analyzer (Olympus Microimage™ image analysis, software version 4.0 for Windows) and recorded as the percentage of brown nuclei in the tumor cells in 10 representative high-power fields (minimum of 1,000 cells). Following the criteria previously described, 46 sarcomas were considered positive when more than 10% of nuclei immunolabeled for p53. Evaluation of the FGF-b and TGF-α immunoreactivity was graded according to the number and the intensity of staining in the tumor cells as follows: grade 0, negative; grade 1, tumors with less than 25% of cells weakly stained; grade 2, tumors with 25–50% of cells moderately positive labeled; grade 3, tumors with more than 50% of cells intensely positive for FGF-b or TGF-α.
Statistical analysis
The association among p53, TGF-α, and FGF-b immunoexpression and clinical variables studied was established using the Biomedical Data Processing Software. To avoid data dispersion, age was categorized into three groups: young (1–5 years), medium (6–10 years), and old (11–15 years). For the same reason, FGF-b and TGF-α expression was considered negative for grades 0 and 1 and positive for grades 2 and 3.
Analysis of variance was used to study differences between the groups of categorical variables (F-test, pooled t-test if variances are assumed to be equal, and Welch test or separate t-test if variances are not equal). Values of P ≤ 0.05 were considered significant.
Results
Of the 50 primary vaccine-associated sarcomas, 29 were from male and 21 from female cats (age range: 3–13 years). These tumors were classified as FS, MFH, or chondrosarcoma (CS) according to their histologic features.
A diagnosis of FS was made in 41 of 50 (82%) sarcomas when cells were arranged in fascicles and consisted predominantly of stellate or spindle-shaped cells with minimal cytoplasm. Neoplastic, multinucleated giant cells were observed in 12 of 41 (29.3%) of these FS. Eight of 50 (16%) tumors were classified as MFH and had a preponderance of atypical histiocytoid cells characterized by round to polygonal shape, abundant cytoplasm, large nuclei, and prominent nucleoli.
Neoplastic giant cells were observed in 4 of 8 (50%) of MFH. Neoplastic, multinucleated giant cells in FS and MFH had similar morphologic features: pleomorphic appearance with irregularly shaped nuclei and prominent nucleoli. A single tumor was diagnosed as CS (1 of 50; 2%) and consisted of sheets of polygonal cells embedded in lacunae surrounded by a basophilic cartilaginous matrix. This tumor had no neoplastic giant cells.
The determination of histologic grade revealed that 12 of 50 (24%) were of grade I, 20 of 50 (40%) were of grade II, and 18 of 50 (16%) were of grade III. Regardless of the histologic classification, 23 of 50 tumors (46%) showed a nodular growth pattern with well-demarcated limits, and some of these were partially encapsulated by connective tissue. In contrast, 27 of 50 sarcomas (54%) presented with an infiltrative pattern, where the cellular growth invaded the surrounding tissue.
The majority of sarcomas (46 of 50; 92%) showed an inflammatory infiltrate of lymphocytes and macrophages. Necrotic areas were also a common finding in our study (39 of 50; 78%) in all the histologic types of sarcomas. Central necrosis was observed not only in well-demarcated sarcomas but also in tumors with an infiltrative growth pattern.
Immunohistochemistry
Immunostaining of p53 was demonstrated in the nuclei of tumor cells in 28 tumors: 20 of 41 (48.7%) FS, 7 of 8 (87.5%) MFH, and 1 of 1 (100%) CS. Only intensely staining nuclei were considered positive (Fig. 1). Two different groups of sarcomas could be observed: in the first group, more than 80% of nuclei stained, and in the second group, the percentage was not higher than 5%, and these samples were considered negative for p53 protein. In some sarcomas, a pale immunolabeling was observed in lymphocytes and adjacent nonneoplastic tissue but was considered nonspecific staining. Neoplastic, multinucleated giant cells uniformly failed to demonstrate p53 protein (Fig. 1). Staining was not seen in any of the negative controls.
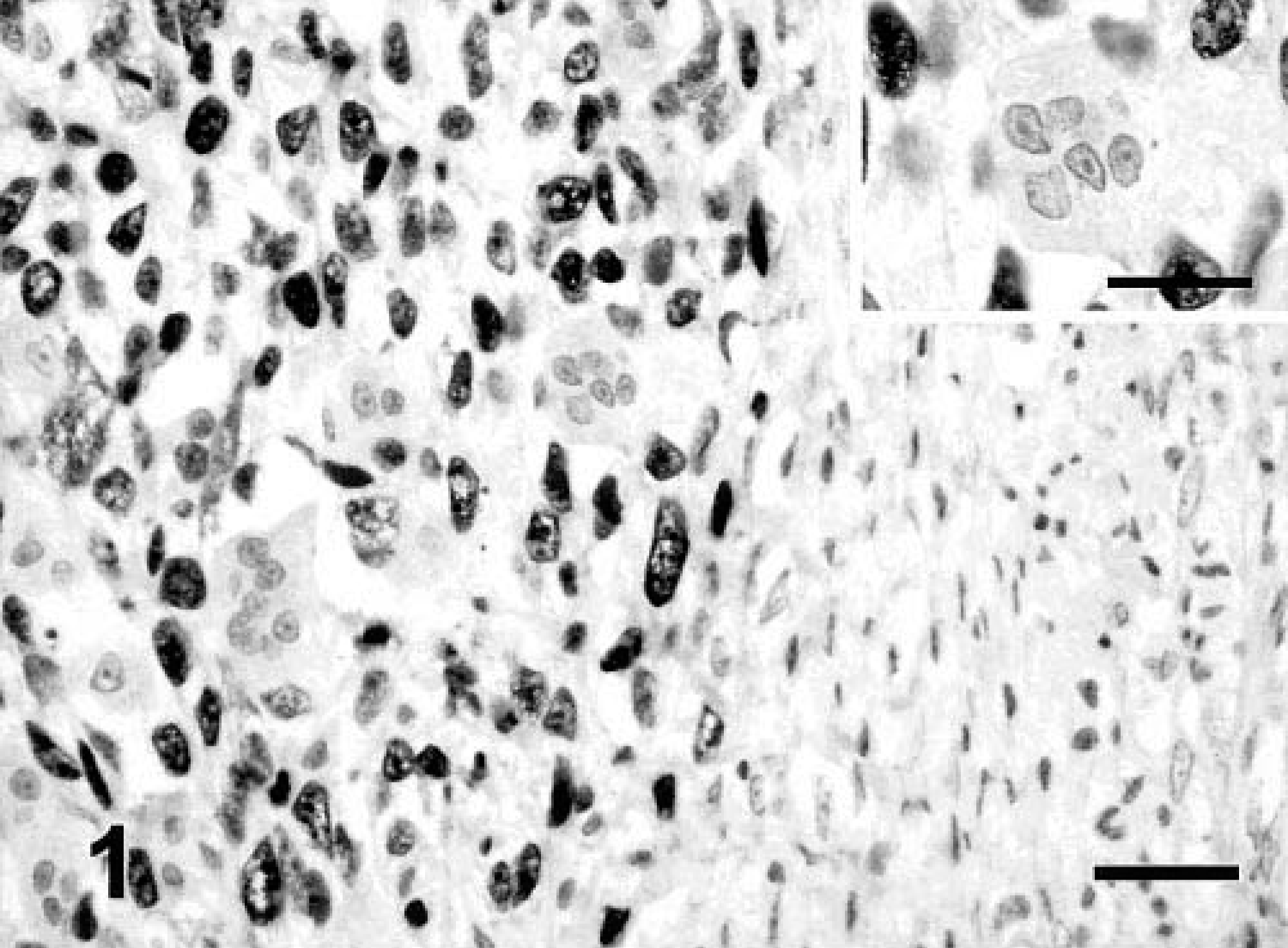
Vaccine-associated sarcoma; cat No. 35. MFH variant. Nuclear p53 immunostaining in a high percentage of neoplastic cells with nonreactive, multinucleated giant and stromal cells. Streptavidin–biotin complex method, polyclonal antibody anti-p53 CM-1, Gill's hematoxylin counterstain. Bar = 60 µm. Inset: Detail of a multinucleated giant cell with no p53 nuclear staining. Neoplastic cells show intense nuclear staining. Bar = 36 µm.
Immunolabeling to FGF-b was located in the cytoplasm of tumor cells in 32 of 41 (78%) FS and 5 of 8 (62.5%) MFH. The CS was negative for FGF-b. In the majority of the sarcomas, the intensity of staining showed marked heterogeneity (Fig. 2). Fibroblasts of tumor stroma and lymphocytes were negative. Neoplastic, multinucleated giant cells showed an intense immunostaining for this GF (Fig. 3).
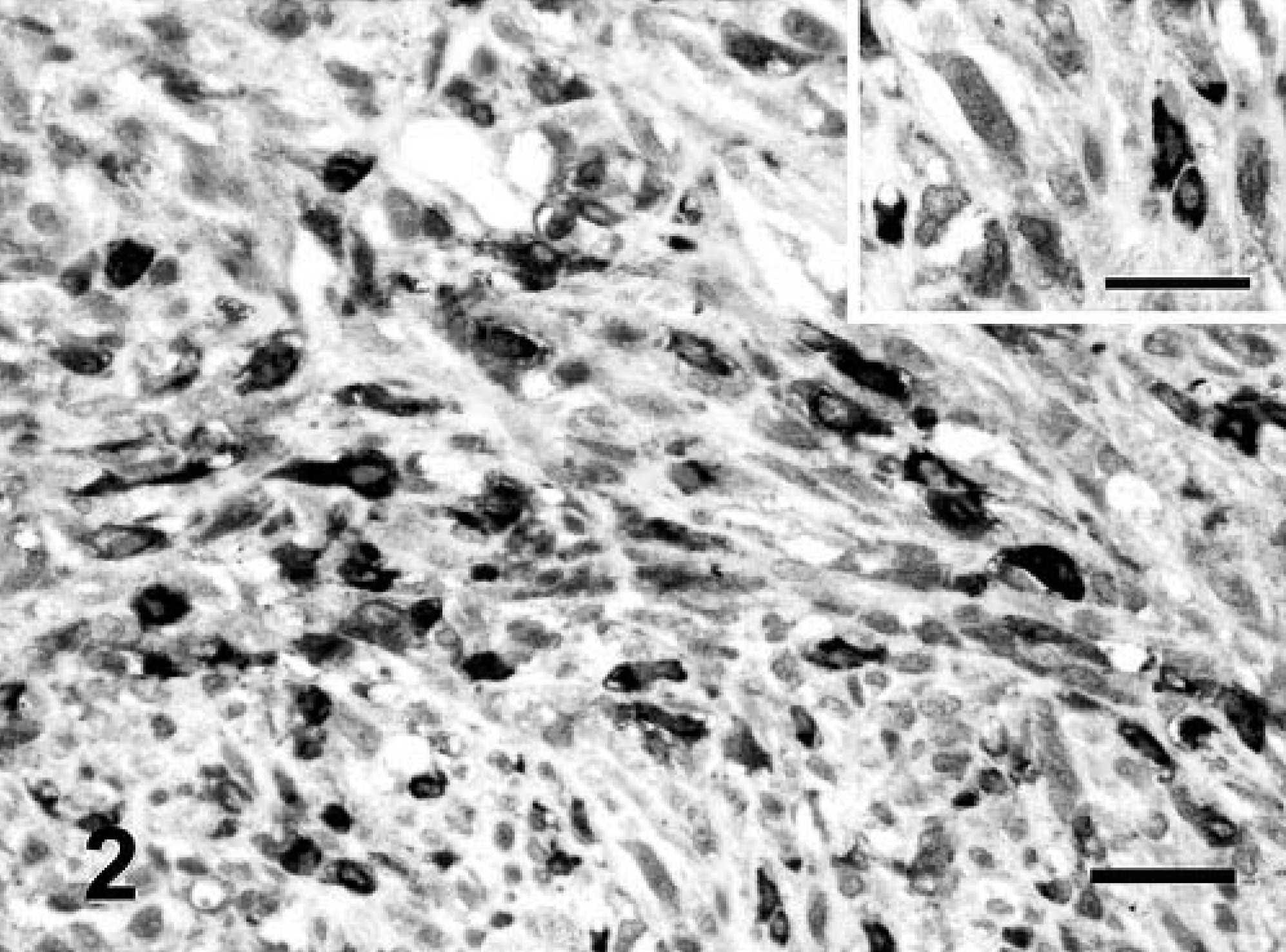
Vaccine-associated sarcoma; cat No. 12. FS variant. Heterogeneous staining of the cytoplasm in neoplastic cells for FGF-b. Streptavidin–biotin complex method, polyclonal antibody anti-FGF-b, Gill's hematoxylin counterstain. Bar = 60 µm. Inset: Detail of cytoplasmic staining. Bar = 36 µm.
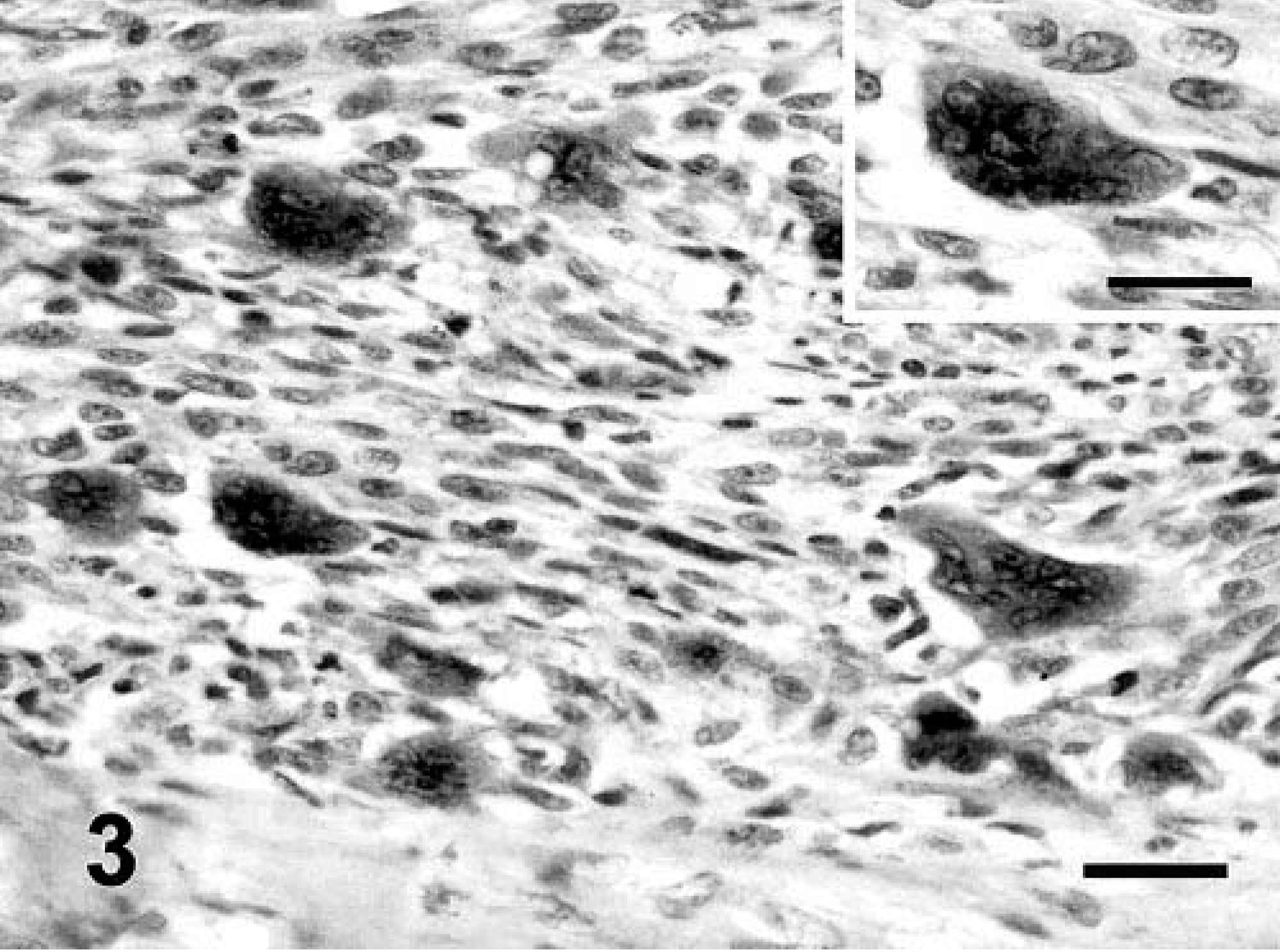
Vaccine-associated sarcoma; cat No. 27. FS variant. Moderate number of neoplastic cells show cytoplasmic immunostaining for FGF-b. The cytoplasm of the neoplastic, multinucleated giant cells is intensely positive. Streptavidin–biotin complex method, polyclonal antibody anti-FGF-b, Gill's hematoxylin counterstain. Bar = 60 µm. Inset: Detail of cytoplasmic staining in the neoplastic, multinucleated giant cells. Bar = 36 µm.
The majority of feline vaccine-associated sarcomas expressed a moderate (grade II) (17 of 50; 34%) or intense (grade III) (26 of 50; 52%) immunoexpression of TGF-α. The staining was located in the cytoplasm, and the intensity was not related to the cellular morphology (Fig. 4). Stromal cells were weakly positive, and lymphocytes were negative for TGF-α. Neoplastic, multinucleated giant cells presented a heterogeneous response to TGF-α: in some tumors, the cytoplasm of these cells showed intense expression (Fig. 5), and in others no staining was detected.
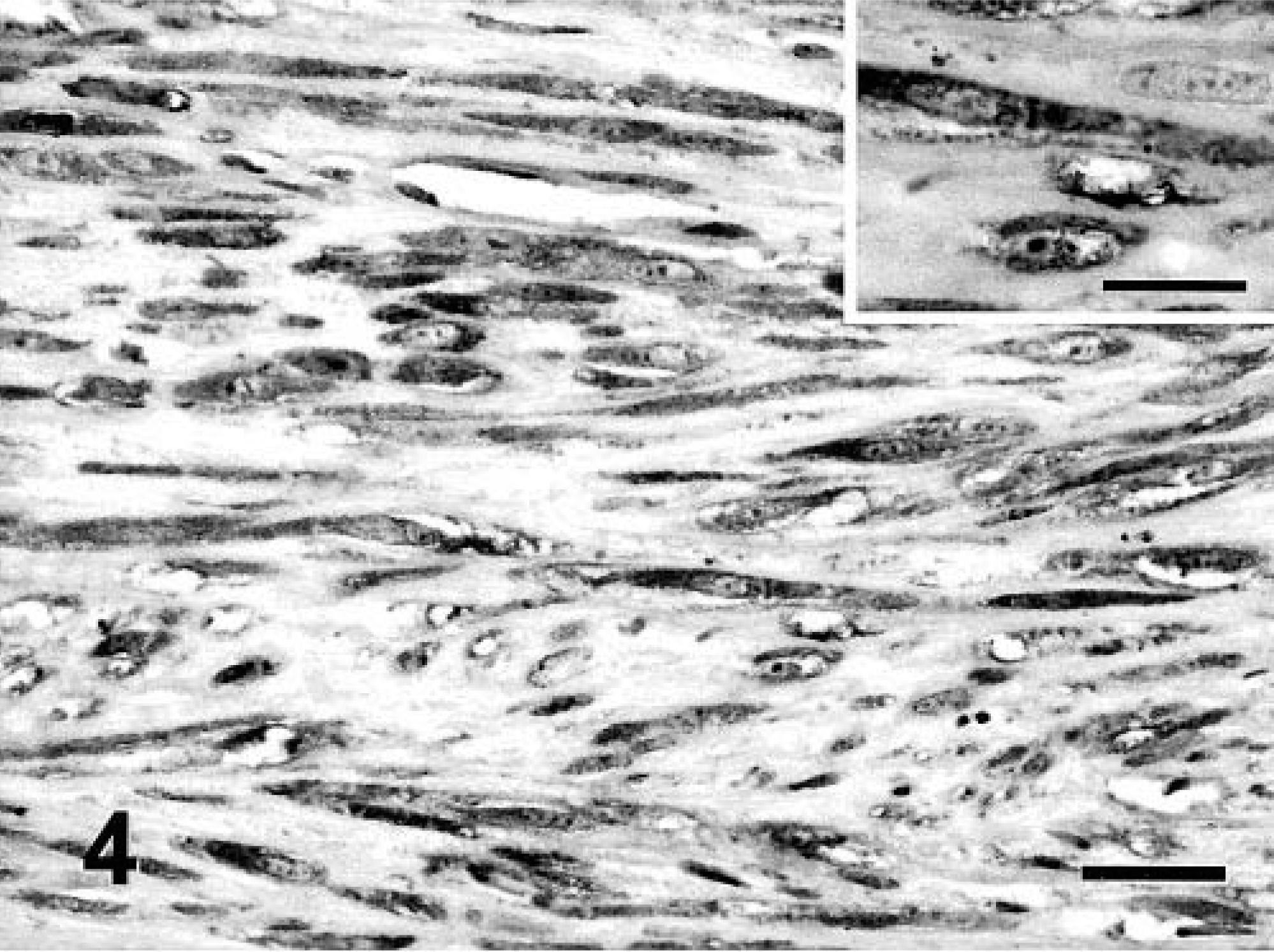
Vaccine-associated sarcoma; cat No. 43. FS variant. Intense TGF-α cytoplasmic immunostaining in spindle neoplastic cells. Streptavidin–biotin complex method, monoclonal antibody anti-TGF-α, Gill's hematoxylin counterstain. Bar = 60 µm. Inset: Detail of cytoplasmic staining. Bar = 36 µm.
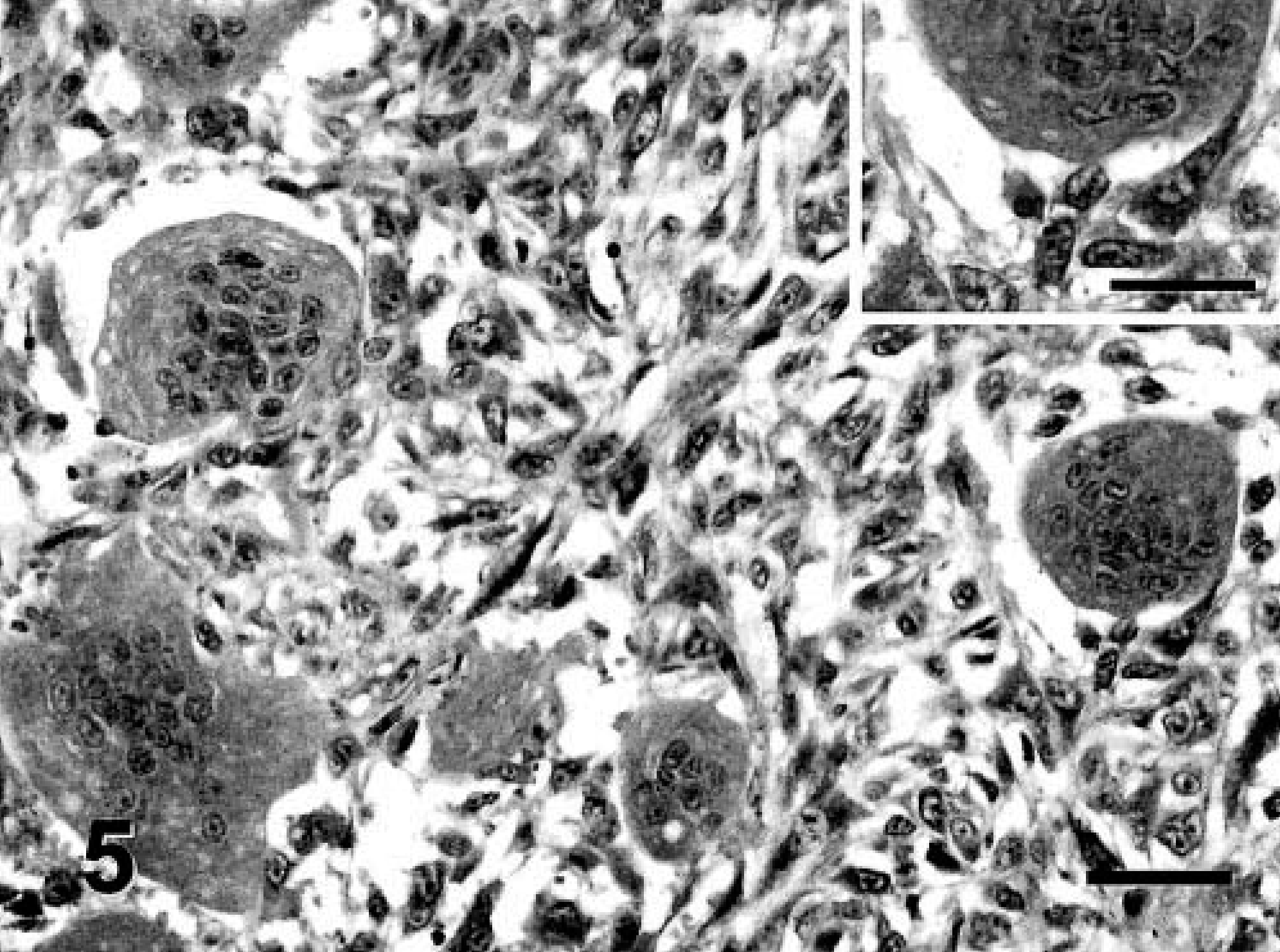
Vaccine-associated sarcoma; cat No. 16. MFH variant. A high percentage of neoplastic cells show intense cytoplasmic expression of TGF-α. The neoplastic, multinucleated giant cells are positive. Streptavidin–biotin complex method, monoclonal antibody anti-TGF-α, Gill's hematoxylin counterstain. Bar = 60 µm. Inset: Detail of the cytoplasmic staining of neoplastic, multinucleated giant cells. Bar = 36 µm.
Statistical analysis
A significant association was observed between FGF-b immunolabeling and age (P < 0.01): tumors from young cats (< 5 years) expressed FGF-b more frequently than did those from old animals (> 11 years). In addition, tumors with a nodular growth pattern also expressed FGF-b more frequently than did infiltrative sarcomas (P = 0.02). An association was observed between FGF-b immunoexpression and the presence of necrosis, but this was not statistically significant (P = 0.06).
No significant association was found between p53 protein nuclear overexpression and clinical or pathologic variables. However, nuclear staining for p53 protein tended to be more frequent in MFH (P = 0.06) and in sarcomas with multinucleated giant cells (P = 0.08).
No significant association was found between a high expression of TGF-α and age of cats (P = 0.11), presence of necrosis (P = 0.07), or absence of giant cells (P = 0.10).
Sarcomas negative for p53 protein showed a significantly higher incidence of FGF-b expression than tumors positive for p53 protein (P = 0.04). FGF-b immunostaining was significantly higher in sarcomas with an intense expression of TGF-α (P = 0.05). However, no relationship was observed between overexpression of p53 protein and TGF-α immunostaining.
Discussion
Feline vaccine-associated sarcoma has been described by numerous authors during the past decade, but the pathogenesis remains elusive. Microscopically, one of the characteristics that differentiates these tumors from sarcomas unassociated with injection is the presence of a infiltrate of lymphocytes and macrophages at the tumor periphery. 4,6,15,19 The hypothesis commonly accepted is that a persistent inflammatory response to an adjuvant residue or an immunologic reaction, or both induces a fibrous connective tissue response and in some cases, neoplasia. 4,8,14–16,19,24,25 Interestingly, studies using the Rous sarcoma virus indicate that it is possible to prevent tumor growth if the inflammation can be suppressed. 27 Activated T lymphocytes in Kaposi's sarcoma (a sarcoma in humans characterized by an inflammatory cell infiltrate) have been shown to release mediators such as TNF-α, interleukin-1, and Interferon-γ, which probably cause the tumor cells to secrete FGF-b. 41 Similar mechanisms may play a role in feline vaccine-associated sarcomas because there is a significant inflammatory cell component observed in both these tumors.
In addition to the inflammatory cell component surrounding the tumors, FS consisted predominantly of spindle cells arranged in fascicles. These cells have been described immunohistochemically and ultrastructurally as fibroblasts and myofibroblasts. 15,25 These cell types have also been implicated in inflammation and wound healing and can participate in a variety of pathologic conditions such as hypertrophic scars, fibromatosis, and desmoplastic response. 30,39 Although the origin of myofibroblasts remains controversial, some authors have proposed that they represent a differentiation of fibroblasts or macrophages, which occurs during the wound healing. 9 In addition, neoplastic transformation of these myofibroblasts had been proposed as the origin of human MFH. 3 and neoplastic myofibroblasts are found in feline vaccine-associated FS. 6,15,25
Immunoexpression of p53 protein was analyzed to examine the role of p53 gene mutation in fibroblastic and myofibroblastic cell transformation and its relationship with the expression of GFs, FGF-b and TGF-α. FGF-b is a GF closely related to the inflammation process, angiogenesis, 2,18,23,36 and mesenchymal tumor development, 1,2,10,12 and TGF-α has a potent mitogenic effect in sarcomas. 11,34
Approximately 90% of the feline p53 gene has been cloned and sequenced and is 82.9% homologous to the human p53 gene. 35 Because mutations of the p53 gene were demonstrated in a large variety of cancers, 13,26,37 it is possible that p53 plays an important role in feline tumor progression. Several studies have demonstrated missense mutations in feline neoplasms, including FS and feline vaccine site–associated sarcomas. 28,29,31 Immunohistochemical detection of p53 protein in tumors has several advantages over the nucleotide sequencing because sequencing is restricted to genetic evaluation of specific exons most frequently implicated in disease. 7 However, two reports showed conflicting results concerning p53 protein immunoexpression in feline sarcomas: one study failed to demonstrate p53 protein immunolabeling in three feline sarcomas, 33 whereas the other found that 42.5% of 40 tumors expressed p53 protein. 32 In our study, p53 protein immunoexpression was found in 56% of sarcomas analyzed, especially in MFH where 87.5% of tumors were positive. These results could be attributed to a predominant role of p53 gene in the development of MFH. However, no staining was observed in 44% of feline vaccine-associated sarcomas. In these cases, the mechanism of carcinogenesis may be something other than p53 gene mutation.
In addition to participating in inflammation and tissue repair, GFs also stimulated cellular proliferation. 34,43 However, no previous studies were found that looked at the expression of FGF-b or TGF-α in feline vaccine-associated sarcomas. The only reference to GFs is a preliminary study, in which cells of sarcomas associated with vaccine-site injection showed a strong positive reaction to platelet-derived growth factor, whereas this same GF was not detected in non–vaccine-associated sarcomas. 14
In this study, the majority of sarcomas express FGF-b, indicating a role of this GF in the development of feline vaccine-associated sarcomas. As was noted previously, the inflammatory mediators may be responsible for release of FGF-b by tumor cells, leading to increased tumor growth. 41,42 In addition, some authors have suggested that FGF-b has a mitogenic action in the short term and over the long term may block differentiation of myoblasts, producing undifferentiated cells that lead to tumor development. 22 Because myofibroblasts are one of the predominant cells implicated in feline vaccine site–associated sarcomas, FGF-b might work similarly in these tumors.
EGF-R activation was due to continued expression of TGF-α that acts as a potent mitogenic stimulus for epithelial and fibroblastic cells. 7,21,43,44 Coexpression of TGF-α and its receptor was investigated in different tumors (including human soft-tissue tumors), demonstrating an autoregulation and autostimulation of growth more intense in the malignant tumors. 11,38,40,45 Elevated levels of TGF-α in feline vaccine-associated sarcomas could signify a role of this GF in the development of these tumors. There are no significant differences in TGF-α expression among the types of sarcomas; almost all MFH (87.5%) expressed TGF-α as did the majority of the FS (85.3%).
Neoplastic, multinucleated giant cells are another common cell type in feline vaccine-associated sarcomas and are associated with tumors of a high histologic grade. 6 Immunohistochemically, neoplastic giant cells stained positive for FGF-b, but p53 protein was not detected. Staining for TGF-α was heterogeneous. Considering these results, neoplastic, multinucleated giant cells may be an important source of FGF-b (and probably TGF-α) that could increase the growth of sarcomas. These results, together with the pleomorphic cellular appearance, indicate a poorly differentiated tumor, in accordance with previous reports. 6
Univariate analysis demonstrated an association between FGF-b and age. FGF-b could play a more important role in young cats than in older cats, where a prolonged contact with other environment carcinogens could also be implicated in tumorigenesis. FGF-b expression was also related to nodular growth, an early event in the development of feline vaccine-associated sarcomas and closely related with the inflammatory process after the injection. In addition, nodular types of growth frequently present a central necrotic area. The presence of necrosis was associated with an increase in FGF-b expression, although this correlation was not statistically significant.
Statistical analysis of immunolabeling results demonstrated a relationship between p53 protein expression and FGF-b: tumors with a positive immunolabeling to p53 protein showed only faint staining for FGF-b. Contrary to our findings, other authors have found that mutant p53 enhances the FGF-b expression. 2 Further studies are necessary to know the significance of this finding. Regarding FGF-b and TGF-α immunostaining, there was a significant association between tumors positive for FGF-b and tumors that express TGF-α. This may suggest that both GFs participate actively in the development of feline vaccine-associated sarcomas. Although some authors found an increase in mutant p53 product after EGF-R stimulation by TGF-α, 5 we failed to demonstrate an association between p53 protein and TGF-α immunoexpression.
Feline vaccine-associated sarcomas immunoexpress p53 protein, FGF-b, and TGF-α, indicating their participation in the pathogenesis of these tumors. In the case of FGF-b, its role can be related to inflammatory compounds and mitogenic effect on fibroblasts and myofibroblasts present in the sarcomas. TGF-α could act via the EGF-R to initiate growth and division of mesenchymal cells. Currently, the authors are conducting studies further to explore the association among immunoexpression of p53 protein, FGF-b, and TGF-α and prognostic variables, such as disease-free survival and overall survival.
Footnotes
Acknowledgements
This study was supported by the Complutense research Project PR 48/01-9895. We thank Dr. Pedro Cuesta for this statistical work and Pedro Aranda, Mario Hernando, and Tamara Panadero for their technical assistance. We also appreciate the assistance of Mr. David Bruhn and Deborah Gillette in preparing the English manuscript.
