Abstract
Ovine pulmonary adenocarcinoma (OPA) is a naturally occurring cancer in sheep that is caused by the Jaagsiekte sheep retrovirus (JSRV). Because the pathologic and epidemiologic features of OPA are similar to those of bronchoalveolar carcinoma in humans, OPA is considered a useful animal model for pulmonary carcinogenesis. In this study, 3,512 lungs from various breeds of sheep were collected and macroscopically examined. OPA was identified in 30 sheep, and samples of these animals were further examined by histologic, immunohistochemical (p53 protein, surfactant protein A [SP-A], proliferating cell nuclear antigen [PCNA], JSRV matrix protein [MA]), and PCR methods. Papillary or acinar adenocarcinomas were detected microscopically in the affected areas. Immunoreactivity for p53 PAb240 was detected in 13 sheep, whereas p53 DO-1 was not detected in any of the OPA animals. PCNA immunoreactivity was recorded in 27 animals. SP-A and JSRV MA protein was immunopositive in all 30. JSRV proviral DNA was detected by PCR analysis in all of the lung samples collected from OPA animals. In addition, the pulmonary SP-A levels were increased in tumor cells. The results of this study suggest that PCNA and p53 protein expression may be useful indicators in monitoring malignancy of pulmonary tumors.
Introduction
Ovine pulmonary adenomatosis (OPA, also known as Jaagsiekte, pulmonary adenocarcinoma) is a contagious lung tumor of sheep, and rarely of goats. This malignancy is caused by Jaagsiekte sheep retrovirus (JSRV; family Retroviridae, subfamily Orthoretrovirinae, genus Betaretrovirus).6,22 This tumor, which can develop naturally or be experimentally induced, originates from alveolar type II pneumocytes and Clara cells.8,26 The disease occurs in sheep-breeding areas worldwide, but has been eradicated in Iceland. The economic ramifications of OPA are significant, reportedly leading to losses of up to 80% in herds following first exposure to the virus. The disease is very difficult to eradicate because effective treatments or vaccination are not available (Spickler AR, 2009, Ovine pulmonary adenocarcinoma. Emerging and exotic diseases of animals. Ames, IA: Iowa State University. Available at: http://goo.gl/hSTLv3).
The incubation period of OPA is long. The lung is the most highly affected tissue, and the tumor rarely metastasizes to the intra- and/or extrathoracic lymph nodes.9,19 Tumor-bearing lungs become consolidated, with gray, firm foci of various sizes in the apical, middle, and caudal lung lobes. These tumors result in respiratory distress, and a watery nasal discharge is typical in infected animals. 14 The morphological, pathological, and epidemiological features of OPA and human bronchoalveolar carcinoma are similar. Because JSRV has been detected in tumors of both humans and sheep, animals affected by OPA have been suggested as an appropriate model for human pulmonary adenocarcinomas.15,23 Oncogenic retroviruses lead to tumorigenesis by stimulating cell proliferation via oncogenes or encoded proteins. JSRV encodes the classical retroviral genes gag, pro, pol, and env. The gag gene encodes the structural internal virion proteins that comprise the matrix (MA), capsid (CA), and nucleocapsid (NC). MA proteins are produced as N-terminal parts of Gag polyproteins and play a critical role during viral replication.14,24 The MA domain has either leading or supportive roles in genome selection and encapsidation, depending on the virus. The expression of p53 protein increases in response to various stressors, including DNA damage, hypoxia, viral infections, and tumors. The most common genetic abnormalities in lung cancer are mutations of the p53 gene and the overexpression of the p53 protein.13,18 The p53 antibodies have many different epitopes, which can be classified into 3 groups. BP53, DO-1, and DO-7 in the first group adhere to the N terminal of p53. 2 PAb240 in the second group adheres to the body of p53. The p53-positivity ratio has been determined in human and animal cancer. This ratio shows marked differences according to the antibody used and the tumor.10,11,18
Proliferating cell nuclear antigen (PCNA) is a nuclear nonhistone protein that plays a role in the initiation of cell proliferation by increasing the expression of DNA polymerase. The level of PCNA expression correlates with the grade and mitotic activity of the tumor. At least 50% of cells in malignant tumors are generally PCNA positive, and the number of PCNA-positive cells negatively correlates with prognosis.11,25 It was reported that p53 overexpression was associated with increased mean PCNA.27,28
Pulmonary surfactants are lipoprotein complexes that cover the alveolar surface of the lung and prevent alveolar collapse by reducing surface tension. The complex consists of ~90% lipids and 10% proteins. Four surfactant proteins (SPs) whose structures and functions differ have been identified. These 4 SPs (SP-A, SP-B, SP-C, SP-D) play an important role in innate lung defense, enhance the opsonization of microbes, and limit lung inflammatory responses. 4 All SPs are synthesized from alveolar type II pneumocytes. SP-A, SP-B, and SP-D are also synthesized from respiratory tract cells, such as Clara cells and submucosal cells. SP-A immunostaining is used to characterize lung tumor cells of sheep with OPA. The SP-A gene is highly expressed in alveolar epithelial type II (ATII) cells and Clara cells, and its protein is a major component of pulmonary surfactant, which is produced in abundance in sheep with OPA.3,26 Smoking is considered to play a role in the etiology of human lung cancer, but researchers have also studied other etiologic factors, because the incidence of tumors in nonsmokers, especially women, is increasing.12,17 Studies have focused on retroviruses, specifically JSRV, because retroviruses have been detected in some human lung tumors.5,15 Human pulmonary adenocarcinoma and OPA are similar to each other both morphologically and histologically. Therefore, some researchers suggest that OPA caused by JSRV may serve as a good model for human lung cancer.5,16 Useful diagnostic markers include p53, PCNA for many carcinomas,11,28 and SP-A1,7 for pulmonary carcinoma. The aim of this study was to investigate the lungs of sheep with pulmonary adenomatosis by gross, histological, immunohistochemical (JSRV MA, SPA, p53, PCNA), and PCR methods.
Materials and methods
Samples
Lungs (n = 3,512) from various breeds of sheep, 3–4 years of age, were examined at Van slaughterhouse in Van in East Anatolia, Turkey, and 30 lungs suspected to be natural cases of OPA by macroscopic evaluation were further examined. Tissue samples were collected for histologic and PCR investigation. Tissues were stored frozen at −80°C until tested by PCR. Samples of normal lungs were obtained from 5 healthy sheep for immunohistochemistry (IHC).
Histology and immunohistochemistry
Tissue samples were fixed in 10% formalin, processed routinely, embedded in paraffin, and sections stained with hematoxylin and eosin for histologic examination. Immunohistochemical staining was performed using the avidin–biotin immunoperoxidase complex method. For IHC, sections on adhesive slides were deparaffinized and dehydrated. Peroxidase activity was blocked in 3% hydrogen peroxide–methanol solution. The sections were washed with phosphate buffered saline (PBS), and nonspecific proteins were blocked by incubating protein-blocking a sera for 5 min. The sections were incubated with primary antibodies (p53/DO1, b Pab 240, c PCNA, d SPA, e JSRV MA protein f ) for 1 h at room temperature (Table 1). After washing with PBS, sections were incubated for 20 min with biotinylated goat anti-rabbit antibodies at room temperature. After another PBS rinse, sections were treated with streptavidin–horseradish peroxidase complex for 20 min. After washing in PBS, 9-ethylcarbazol-3-amine (AEC) was used as chromogen, and Mayer hematoxylin was used for counterstaining. All sections were treated with PBS after each step. As controls, PBS was substituted for the primary antibody in each slide run; the rest of the procedure was identical. Slides were examined on a light microscope and photographed. Ten fields were examined on each slide at high magnification. The intensity of staining was scored as no staining (–), low (+), moderate (++), and marked (+++). The extent of staining was scored as – (0%), + (1–10%), ++ (1–50%), and +++ (≥51%) based on the percentage of positively stained cells. Each field was graded according to the score and then the total score was divided by 10. Thus, the average score was calculated for each slide.
Characteristics of antibodies used for immunohistochemistry.*

PCNA = proliferating cell nuclear antigen; SP-A = surfactant protein A; JSRV MA = Jaagsiekte sheep retrovirus matrix protein.
Heat-induced antigen retrieval was performed using citrate buffer.
PCR
All 30 OPA samples were also analyzed by PCR. The samples were homogenized with a pestle and mortar and reconstituted with sterile PBS. DNA was extracted with a DNA extraction kit g according to the manufacturer’s instructions.
The presence of viral genomic DNA was detected with heminested PCR as described previously.20,21 In the first stage of PCR, PI (TGGGAGCTCTTTGGCAAAGCC) 21 and APO2R (CTCCCACCTTCTTCCATGTCTC) 20 primers were used. In the second stage, 5 µL of PCR product was used as template, and the PI and APO5R (CGAGCTCGAGCCCCACGTTG) primers were used to amplify DNA. The PCR reactions were conducted in a total volume of 50 µL consisting of 2.5 mM MgCl2, 10 mM Tris (pH 8.3), 50 mM KCl, 200 μM deoxyribonucleotide triphosphate, 0.125 μM of each primer, 1.25 U Taq DNA polymerase, h and 5 μL (100 ng/μL) of extracted DNA template. The DNA was amplified for 40 cycles consisting of 94°C for 1 min, 61°C for 1 min, and 72°C for 1 min with an initial denaturation of 10 min at 95°C and a final extension of 5 min at 72°C. Distilled water was used as a negative control. The PCR products were separated on a 1% agarose gel stained with ethidium bromide and visualized with ultraviolet excitation.
Results
In 30 of the 3,512 examined lungs, diffuse or limited neoplastic masses were often localized on the ventral portion of the caudal lobe and/or cranial and middle lobes (Fig. 1A, 1B). In 8 cases, lesions were located only in the cranial and middle lobes, whereas they were only located in the caudal lobes in 16 cases. Lesions were found in both the caudal and cranial lobes in 6 cases. In 24 cases, the masses were of various sizes and gray-white. Their edges were irregular, and they were somewhat shiny in appearance with a stiff nodular structure. These nodules were present either as areas of multiple nodules ranging from 2–3 mm to 2 cm or as solitary nodules of 2–6 cm diameter. In 6 cases, neoplastic foci were diffusely distributed throughout all lung lobes. These lungs were firm and gray-white. In 3 of these 6 cases, the pleura were very thick. Pleural adhesions between lobes were observed in 9 cases. The lungs had not collapsed in proportion to the severity of the lesions. An atelectatic red border was observed around the large nodular foci. The cut surface of the foci located in the cranial lobe was wet, whereas the cut surface of foci located in the caudal lobe was dry. Necrosis and abscesses were observed in 5 cases. In 3 cases, parasitic foci and neoplastic foci were evident in the caudal lobes.
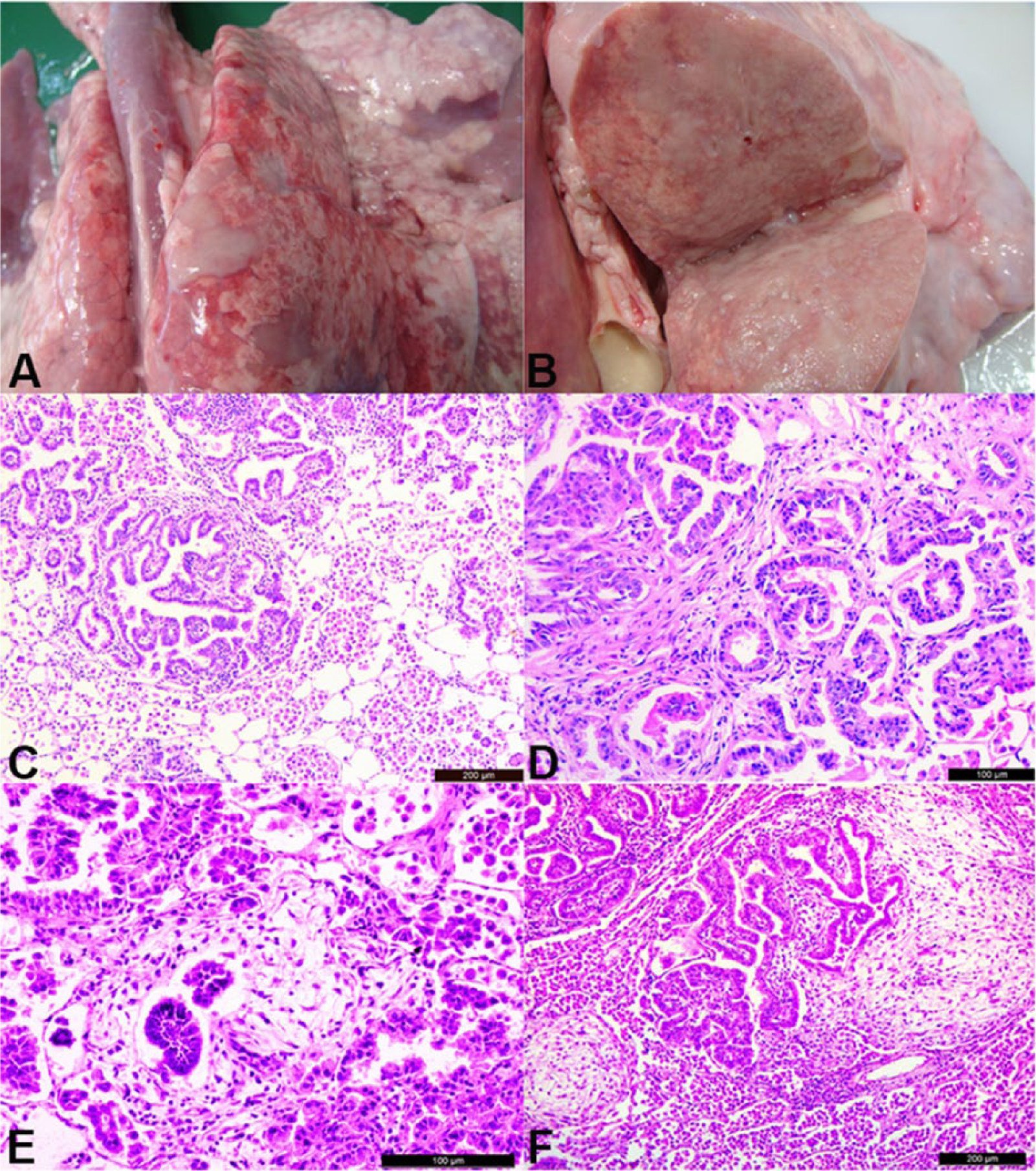
Macroscopic and histologic appearance of ovine pulmonary adenocarcinoma.
The detailed histological results from the OPA lungs are shown in Table 2. Unencapsulated neoplastic foci consisted of cuboidal to columnar cells whose cytoplasm was occasionally vacuolar, forming papillary to acinar patterns (Fig. 1C, 1D). The stroma consisted of lymphoplasmacytic infiltrates and fibrous connective tissue. In most cases, stromal tissue surrounded and divided the neoplastic foci (Fig. 1D). In 20 cases, follicular lymphoid proliferation was observed around the bronchi, bronchioles, and vessels. In 2 cases, extensive myxoid foci (Fig. 1E, 1F) were observed. In all 30 cases, moderate and marked accumulations of alveolar macrophages with vacuolated cytoplasm were observed in the alveolar lumens around the neoplastic foci (Fig. 1C). The neoplastic foci compressed the adjacent alveoli, causing atelectasis demarcated by condensed stroma. In some sections, pale eosinophilic, amorphous masses were observed in the alveoli. Neutrophil and leukocyte-rich exudate was observed in the lumen of the neoplastic foci and in some alveoli. In 3 cases, different developmental stages of parasites and neoplastic foci in the alveolar lumen were observed. The pattern of neoplastic foci was cystic in 3 cases, and this pattern was severe in 1 case.
Histologic and immunohistochemical findings of naturally occurring ovine pulmonary adenomatosis.
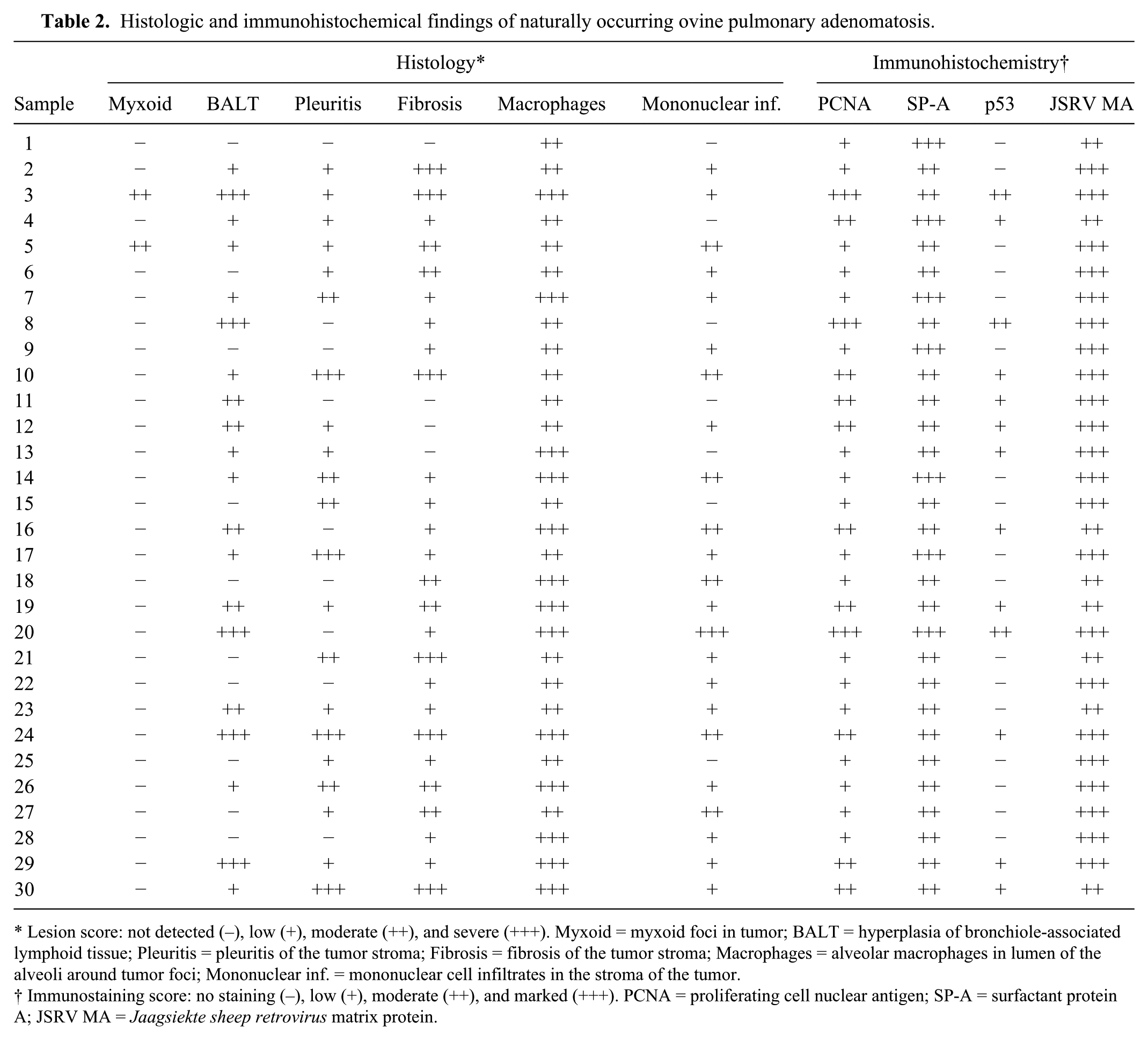
Lesion score: not detected (–), low (+), moderate (++), and severe (+++). Myxoid = myxoid foci in tumor; BALT = hyperplasia of bronchiole-associated lymphoid tissue; Pleuritis = pleuritis of the tumor stroma; Fibrosis = fibrosis of the tumor stroma; Macrophages = alveolar macrophages in lumen of the alveoli around tumor foci; Mononuclear inf. = mononuclear cell infiltrates in the stroma of the tumor.
Immunostaining score: no staining (–), low (+), moderate (++), and marked (+++). PCNA = proliferating cell nuclear antigen; SP-A = surfactant protein A; JSRV MA = Jaagsiekte sheep retrovirus matrix protein.
Jaagsiekte sheep retrovirus MA protein expression was intense in the tumors of 22 of 30 lungs and moderate in 8 of 30 lungs (Fig. 2A, 2B), but was not detected in the lung tissue of normal sheep. SP-A staining was moderate or intense in the cytoplasm of neoplastic cells in all 30 tumors examined (Fig. 2C). In all cases, positive staining was identified by diffuse labeling in the cytoplasm and the cell membrane of the neoplastic cell. SP-A staining was also immunopositive in the normal sheep lungs, but was much less intense than in the neoplastic cells of sheep with OPA (Fig. 2D). PCNA expression was also not detected in the lung tissues of normal sheep. The neoplastic cells showed rare and weak nuclear staining in 18 of 30 lungs, moderate staining in 9, and intense staining in 3 (Fig. 2E). p53 (PAb240) was not expressed in the normal lung tissue but was mildly or moderately expressed in the neoplastic epithelial cells of 13 of 30 lungs (Fig. 2F). Positive staining for p53 (PAb240) was observed in both the nuclei and the cytoplasm in all positive cases. However, p53 (DO-1) staining was not observed in the healthy lung tissue and neoplastic cells. Thirteen cases were positive for both PCNA and p53 (PAb240). These findings were confirmed in all samples by heminested PCR, and a 395-bp virus-specific PCR amplicon was visualized by electrophoresis (Fig. 3). No band was detected in the negative control samples.
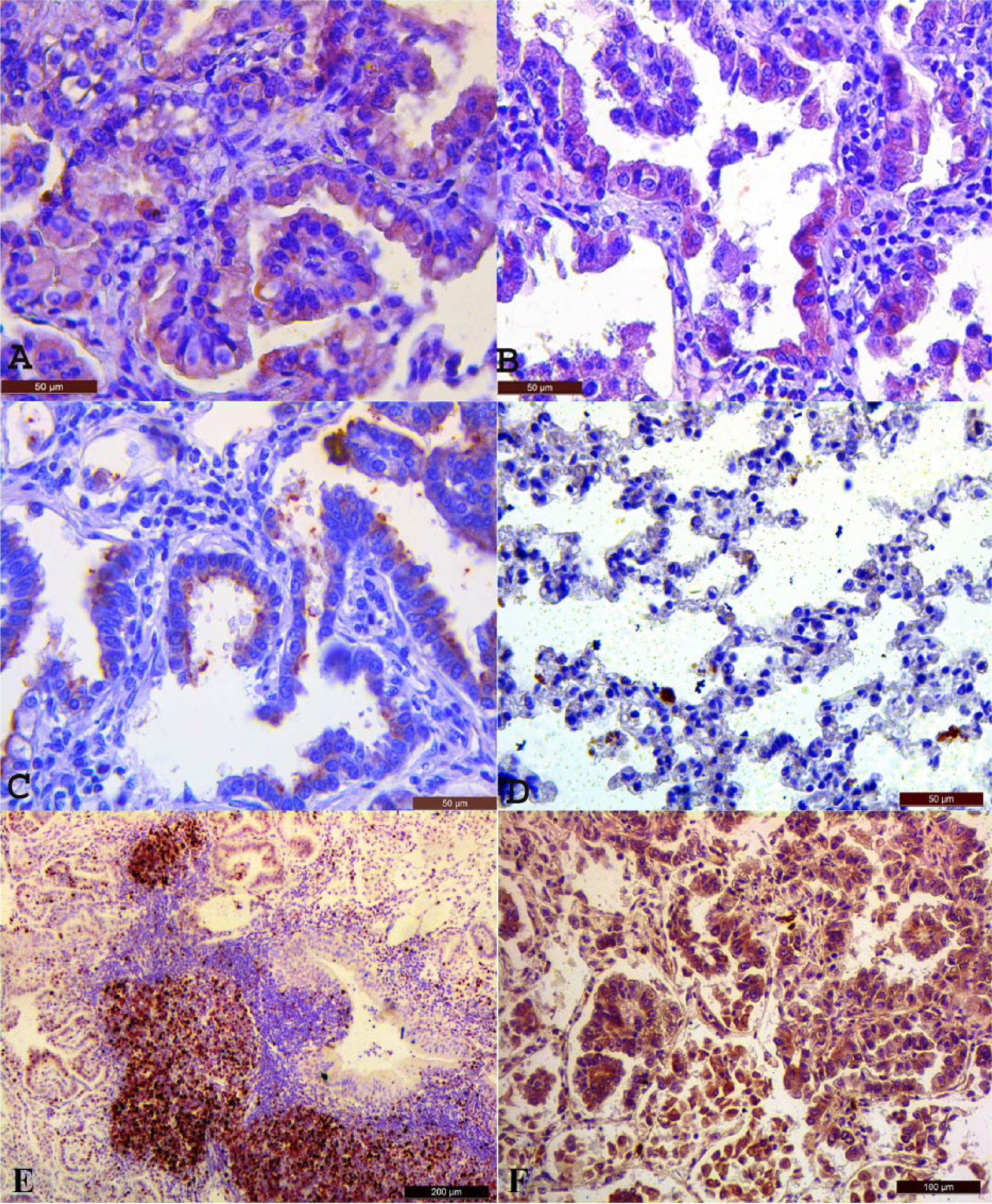
İmmunohistochemistry (IHC) of ovine pulmonary adenocarcinoma.
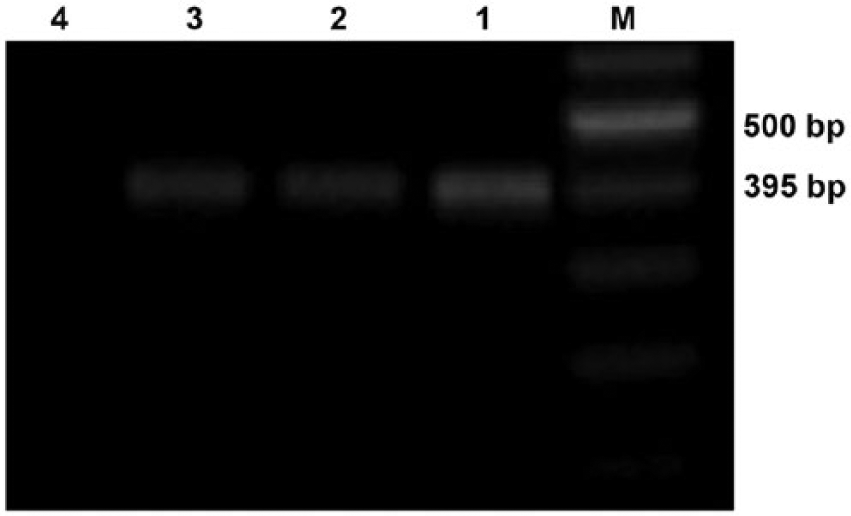
Detection of ovine pulmonary adenocarcinoma proviral DNA by agarose gel electrophoretic analysis of PCR amplicons. Lanes 1–3: 395-bp PCR amplicon of positive samples; lane 4: negative control; lane M: marker.
Discussion
Ovine pulmonary adenocarcinoma is a disease generally seen in sheep aged between 2 and 4 years8,14 and, in the current study, it was recorded in 3–4-year-old animals. Two forms of OPA, classical and atypical, are known.6,8 Classical lesions are nodular or diffuse, firm, and are seen as gray-white foci that form by the fusion of nodules, particularly in the cranioventral portions of the lobes. The cut surfaces of these foci are wet, and large amounts of frothy exudate are evident in the bronchi and bronchial areas. Atypical OPA lesions are either solitary or occur in groups; their structure is demarcated, and they often arise in the caudal lobe. Cut surface foci are pearl white, firm, and dry. In the current study, more macroscopic lesions were found in the caudal lobes, which is consistent with the atypical form of the disease. The stroma consists of mononuclear cells and fibrous connective tissue that divides and surrounds neoplastic foci. Lymphoid hyperplasia is a common feature of OPA, and it was detected in 20 of 30 cases in the present study. In our study, macroscopic analysis revealed eosinophilic amorphous material in the alveoli that manifested as a frothy exudate, a significant portion of which consisted of SP-A. 26 The myxomatous foci consisted of stellate and fusiform cells, which were similar to foci identified in a limited number of previously published studies.8,19
In our study, the JSRV MA protein was detected in the cytoplasm of neoplastic epithelial cells in all of the affected lung samples. A small amount of extracellular staining was also observed in some cases. All lungs were positive for JSRV MA protein and lesions specific to OPA. IHC and PCR assays are known to effectively detect JSRV antigens (http://goo.gl/wlBVT4). 8 All PCR-positive lungs were also positive for JSRV MA protein and showed characteristic features of OPA both grossly and microscopically. In the current study, PCNA expression was observed in an increased number of alveolar type II cells and in fewer Clara cells. It is also showed that high PCNA expression in tumors indicates histologic malignancy.27,28 The PCNA staining intensity appeared to be positively correlated with malignancy. Twenty-seven of 30 cases were positive for PCNA, suggesting that this protein could be a useful marker of tumor malignancy.
The detection of p53 protein by IHC reportedly correlates with p53 gene mutations, which are present in many human and animal cancers and play an important role in determining malignancy.2,10 The p53 protein can be used to estimate malignancy in various lung tumors as well. However, as was the case in our study, different p53 antibodies can give completely different results. Therefore, different p53 antibodies targeting different epitopes (DO-1, DO-7, PAb240) might be required for proper diagnostic value. For example, it was reported that p53 (DO-7) was positive 71% 13 and 20.4% 18 in human pulmonary adenocarcinoma. These differences in positive staining have been suggested to arise from the quantity of material being small, all material examined being malignant, and/or the difference in antibodies. In our study, the presence of 2 different types of p53 protein was investigated in detail in the tumor foci of OPA using the DO-1 and PAb240 monoclonal antibodies to p53. Whereas DO-1 did not result in staining, PAb240 produced stains in 13 cases. Positive reactions of p53 antibodies were correlated with p53 overexpression in the corresponding tumor, suggesting that the positive reaction of these antibodies may be related to the accumulation of p53 protein in the primary tumors. At the same time, these positive cases were also positive for PCNA. A similar direct correlation between PCNA and p53 expression has been described previously.11,28 SP-A is an alveolar type II cell–specific marker. Alveolar type II cells originating in OPA lung express JSRV as detected by PCR and hybridization. 1 Small amounts of SP-A are also reportedly present in alveolar and bronchiolar epithelial cells of healthy lungs, but its expression is more intense in the cytoplasm of neoplastic cells in OPA.3,26 In the current study, SP-A was detected in both type II pneumocyte cytoplasm and surface. We utilized the tumor markers p53 protein and PCNA, and the IHC staining of related markers, to determine the malignancy of OPA. The IHC staining of SP-A in tumor cells confirmed the correlation between the increase in SP-A expression and the Jaagsiekte viral infections in OPA.
Footnotes
Authors’ contributions
F İlhan contributed to conception and design of the study; contributed to acquisition and interpretation of data; drafted the manuscript; and critically revised the manuscript. SA Vural contributed to conception of the study; contributed to acquisition of data; and critically revised the manuscript. S Yıldırım contributed to design of the study and contributed to acquisition and analysis of data. İ Sözdutmaz and ME Alcigir contributed to analysis of data and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Dako North America Inc., Carpinteria, CA.
b.
Leica Biosystems, Newcastle Upon Tyne, United Kingdom.
c.
Thermo Fisher Scientific Inc., Waltham, MA.
d.
Leica Biosystems, Newcastle Upon Tyne, United Kingdom.
e.
EMD Millipore, Billerica, MA.
f.
JSRV MA kindly provided by Drs. Palmarini and Golder, MRC-University of Glasgow Centre for Virus Research, Glasgow, Scotland.
g.
Qiagen GmbH, Hilden, Germany.
h.
Fermentas Inc., Burlington, Ontario, Canada.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Research Fund of the Yüzüncü Yıl University (2010-VF-B002).
