Abstract
Forty-four primary feline vaccine-associated fibrosarcomas and 16 recurrences were examined histologically for detailed morphologic characterization with emphasis on tumor grade, presence of neoplastic multinucleated giant cells, presence and proportion of T and B lymphocytes within the tumor, and thin and intermediate filament contents of neoplastic and stromal cells. The microvascularity and proliferation rates of central and peripheral areas of the tumors were also quantified by computerized image analysis. For primary fibrosarcomas, 11 of 44 (25%) were grade I, 21 of 44 (47.7%) were grade II, and 12 of 44 (27.3%) were grade III. The recurrences followed a similar pattern: 4 of 16 (25%) were grade I, 8 of 16 (50%) were grade II, and 4 of 16 (25%) were grade III. A positive correlation was found between the presence of neoplastic multinucleated giant cells and tumor grade. These cells were present in 9 of 12 (75%) of grade III and none of the grade I tumors. Prominent peritumoral lymphoid aggregates or follicles were present in 59% of the tumors, and many contained high proportions of T lymphocytes, varying from 19 to 87%. All fibrosarcomas were immunoreactive for vimentin and 28 of 44 (64%) were reactive for α-smooth muscle actin. The actin-positive cells were either part of the tumor or formed a capsule around tumor nodules. The peripheral vascularity was significantly higher than the central vascular density but no difference was found in tumor cell proliferation rates between the two areas. Centrally located, fluid-filled micro- or macrocavitations were frequently observed in the large vaccine sarcomas and probably formed secondary to rapid tumor growth and central necrosis.
Keywords
A unique tumor of cats was described by Hendrick and Goldschmidt in 1991. 10 The appearance of feline vaccine-associated sarcomas coincided with increased rabies vaccination frequency and introduction of its subcutaneous route of administration. Aluminum vaccine adjuvants, common in killed virus vaccines, were suggested to play a role in the etiopathogenesis of these sarcomas. 11 An epidemiologic study published in 1993 revealed a causal relationship between feline leukemia virus and rabies virus vaccinations and sarcoma development. In that study, risk for sarcomas after vaccination was associated with both adjuvanted (with or without aluminum) and nonadjuvanted vaccines. 17
Most vaccine-associated sarcomas are categorized as fibrosarcomas but other histologic variants include rhabdomyosarcoma, myxosarcoma, chondrosarcoma, undifferentiated sarcoma, and malignant fibrous histiocytoma. 5,9,19,20 Many of these sarcomas are associated with inflammation. The inflammatory reaction is characterized by follicular aggregates of lymphocytes and smaller numbers of plasma cells that are usually located perivascularly at the tumor periphery. 5 Large round macrophages with blue-gray cytoplasm, presumably associated with phagocytosed adjuvant material, are commonly found within, around, or adjacent to these sarcomas. 9,11–13,28 Tumors are often contiguous with granulation tissue that surrounds areas of necrosis at the vaccine site. Because of these histologic findings, chronic or exacerbated inflammation in response to adjuvants or other vaccine components has been implicated in the pathogenesis of the vaccine-associated sarcomas. 5,11,12,20
Various histologic features of soft-tissue sarcomas have undergone scrutiny in both human and veterinary medicine. These include histologic grade, the presence of intratumoral neoplastic giant cells, intratumoral microvessel density, tumor proliferation index, and the presence of myofibroblasts. These morphologic attributes were examined in a subset of feline vaccine-associated sarcomas in the present study in an effort to separate tumors into categories that might offer clues to tumor biology, or be tested as prognostic criteria for cats with sarcomas with or without treatment. The peritumoral lymphocytic infiltrates were also phenotyped to provide additional information that might be useful when considering future immune therapy protocols.
Materials and Methods
Morphology
Paraffin blocks containing fibrosarcomas collected from cats between 1991 and 1999 were retrieved from the pathology service of the Veterinary Medical Teaching Hospital at the University of California, Davis. Sarcomas arising in cats with appropriate vaccination histories at subcutaneous sites commonly used for vaccination (interscapular, lateral cervical, and thoracic regions, and caudal aspect of the thighs) with characteristic inflammatory responses were included within the category of presumed vaccine-associated sarcomas. A total of 44 primary tumors (first occurrences) and 16 recurrences were examined. Ten fibrosarcomas arising at unrelated sites (nonvaccination sarcomas) were examined similarly for comparison. For each specimen, 5-µm, hematoxylin and eosin–stained sections were made and examined microscopically. Tumors were graded using a scheme previously adapted to the dog based on cellular differentiation, presence and extension of necrosis within the neoplasm, and mitotic rate. 25 All sarcomas were scored 1–3 for overall differentiation (1 = tumor cells closely resembled the mature differentiated type; 2 = tumors that had a defined histologic phenotype; and 3 = poorly differentiated tumors), mitotic rates (1 = 1–9 mitotic figures per ten 400× fields; 2 = 10–19 figures per ten 400× fields; and 3 = 20 or more figure per ten 400× fields), and necrosis (1 = no necrosis; 2 = necrosis of <50% of the total area of the specimen; and 3 = necrosis of >50% of the total area). Final scores of 3 or 4 were designated grade I; scores of 5 or 6 were designated grade II; and scores of 7, 8, or 9 were designated grade III. When present, multinucleated giant cells were classified as of neoplastic, reactive, or of undetermined origin, according to previously described criteria. 15,27 Briefly, multinucleated giant cells within tumors were considered to be neoplastic based on immunoreactivity for proliferation marker Ki-67 and lack of reactivity for macrophage marker CD18.
When present, the inflammatory infiltrate within and surrounding the tumors was examined and characterized as to its intensity, distribution, and cell types. Inflammatory cells were identified as lymphocytes, plasma cells, macrophages, and neutrophils. To determine the intensity of inflammation, we focused on the peritumoral lymphoid aggregates. The specimens were examined and scored subjectively based on the presence of lymphoid cells within all peritumoral fields in low magnification (40×). When lymphoid aggregates or follicles were seen in all fields surrounding the tumor, the infiltrate was considered to be abundant (score 3). When lymphoid cells, diffusely distributed or forming smaller follicles, were present in less than 50% of the peritumoral fields, the infiltrate was considered to be moderate (score 2). When a careful search was required to determine the presence of small numbers of lymphoid cells with minimal or no follicular formation, the infiltrate was considered to be mild (score 1). When no inflammation was seen, the tumor was given a score of 0.
Immunohistochemistry
Immunohistochemistry was used to examine tumor-associated inflammatory infiltrates, tumor cellular proliferation, intratumoral vessel density, and tumor cytoskeletal filaments in primary fibrosarcomas. Ten nonvaccine sarcomas were examined only for thin and intermediate filament content (desmin, α-smooth muscle actin [α-SMA], and vimentin).
Leukocyte subpopulations
Twenty vaccine-associated sarcomas that contained a prominent inflammatory reaction (inflammation score 3) were selected from the 44 primary tumors. Tumors selected consisted of appropriate-sized biopsy specimens from cats with spindle cell sarcomas morphologically consistent with fibrosarcomas, preceding any form of treatment. Needle or punch biopsy specimens were omitted from examination. For each tumor, 5-µm sections were immunostained to allow localization and differentiation between T lymphocytes, B lymphocytes, and macrophages. Tissue sections were immunostained using primary antibody, biotinylated secondary antibody, streptavidin–peroxidase as third reagent, 3-amino-9-ethycarbazol as chromagen, and hematoxylin as counterstain. Monoclonal antibodies were anti-CD3 for T lymphocytes (clone CD3–12, Vector Laboratories, Burlingame, CA), anti-CD79a (clone HM57, Dako Corp., Carpinteria, CA) for B lymphocytes, and anti-CD18 (clone Fe3.9F2, courtesy of P. F. Moore, University of California, Davis) for histiocytic cells. Three lymphoid aggregates or follicles per specimen were randomly selected for quantification of B and T lymphocytes. Sections stained for CD3 were selected for image analysis because of the clarity and specificity of staining. Immunostains for CD79a included nonspecific (background) stain and, therefore, were considered to be inappropriate for computer imaging. The CD79a-stained sections were used as reference for identifying unstained cells within the lymphoid follicles marked for CD3.
Computerized image analysis
Entire lymphoid follicles immunostained for CD3 were digitally captured (100×) in tagged image file format with no compression (748 × 580 pixels) using a digital camera (ProgRes 3008, Kontron Elektronik, München, Germany) mounted on a microscope (Zeiss Axioskop, Carl Zeiss Inc., Thornwood, NY) and connected to a computer. The images were analyzed on a Power Macintosh 8500/120-mhz computer using Photoshop 4.0 and NIH Image 1.61 (National Institutes of Health, Bethesda, MD) to quantitate the proportion of T cells per follicle. Such calculation was based on area with positive staining for CD3 with a color filter that selected areas with immunoreactivity (red) using L∗a∗b color mode (L = luminance; a = color components green to red; b = color components blue to yellow). The entire follicle was selected and copied to a white background to be used as a base image from which quantification of T lymphocytes was done. Areas of immunoreactivity (T cells) within the follicle were selected using the filter, converted to black pixels, and copied to a white background (image A). The same procedure was used to determine areas representing non–T lymphocytes (image B) with a filter that selected light blue (hematoxylin-counterstained cells), assumed to be mostly B lymphocytes based on the correlation of the CD79a-stained sections. NIH Image 1.61 software was used to count the black and white pixels within each image. The proportion of black pixels in image A represented total T cells (based on relative area), and the proportion of black pixels in image B represented the area of the follicle occupied by non–T (mostly B) lymphocytes. The blue filter was preferred over a total follicular area to exclude the white spaces (i.e., acellular areas and vascular lumens) and minimize the error caused by variation in cellular density among the follicles. The percentage of T cells per follicle was determined to be the area of black pixels in images A, divided by the sum of the area of black pixels in image A and B (T cells/total number of cells). The total estimated proportion of T lymphocytes per sarcoma was defined as the average area occupied by T cells among the three follicles counted per tumor.
Tumoral vessel density
Tissue sections of sarcomas were examined for tumoral vessel density both along the periphery of the tumor (growth fronts or hot spots) and within the central areas of the mass. 1,33 Ten randomly selected fields of each region (center and periphery) of 20 primary tumors were evaluated. Vascular endothelium was identified with an immunolabel for factor VIII–related antigen in 10 randomly selected fields within both regions of each tumor. Tissues sections were stained using polyclonal rabbit anti-human factor VIII–related antigen (FVIII-rag, DAKO Corp., Santa Barbara, CA) as the primary antibody, followed by biotinylated, anti-rabbit secondary antibody, and alkaline phosphatase–labeled streptavidin as a third reagent (StrAviGen Super Sensitive Kit, BioGenex, San Ramon, CA). The reaction was developed with a red chromagen (Fast Red, BioGenex) and counterstained with hematoxylin. Quantitation of tumoral vessels was done by digital imaging with methods similar to those described above but specifically adapted for factor VIII-rag in canine tissues. 7 Total tumor vessel density was calculated to be the average of the immunostained endothelium from three images analyzed (200×) from each specimen or tumor.
Cell proliferation index
Proliferative fractions of 20 primary vaccine sarcomas were determined by means of immunohistochemistry for Ki-67 antigen. Ki-67 was detected on 5–µm-thick tissue sections with a primary antibody, biotinylated alkaline phosphatase–labeled streptavidin as secondary reagent, red chromagen (Fast Red, BioGenex), and hematoxylin counterstain. The primary antibody was murine anti-human Ki-67 (MIB-1, Oncogene Science Inc., Cambridge, MA). Positive and negative controls consisted of normal feline small intestines; intestinal crypts are considered sites of proliferation. The number of proliferating cells (positive cells for MIB-1 antibody) was manually counted with the assistance of computer imaging. Images of six nonoverlapping, 400× magnification fields along the periphery (growth fronts or hot spots) of the tumor were captured as previously described for leukocyte counts. Positive and negative nuclei were counted manually with the NIH 1.61 image analysis program, and the total numbers of labeled (red) and unlabeled (blue) cells were stored on a database. The number of fields was calculated so that a minimum of 1,000 cells was counted per specimen. The total number of cells counted per field varied depending on the cellular density of each tumor. Values obtained for positive and negative cells were summed and the proliferative fraction of peripheral regions of each tumor was determined to be the number of cells immunolabeled with MIB-1 (red) divided by the total number of cells counted per tumor. For comparison, the same procedure was applied to six nonoverlapping, 400× fields within the central region of each tumor.
Cytoskeletal filaments
Immunohistochemistry was used to identify thin and intermediate filaments including α-SMA, desmin, and vimentin in 44 primary fibrosarcomas. Tissue sections were stained with primary antibody, biotinylated secondary antibody, streptavidin–peroxidase as third reagent, 3-amino-9-ethycarbazol as chromagen, and hematoxylin as counterstain. Monoclonal antibodies were used to identify α-SMA (BioGenex), vimentin (Dako Corp., Carpinteria, CA), and desmin (BioGenex). Specimens were scored as positive or negative for vimentin and desmin, whereas specimens stained for α-SMA were scored on the basis of proportion of positive cells within the sarcoma: score 0 = no immunolabeled tumor cells; 1 = less than 50% of tumor cells immunolabeled; 2 = approximately 50% of tumor cells immunolabeled; and 3 = majority of tumor cells immunolabeled.
Statistical analysis
A computer program was used for all analyses (SPSS 9.0 for Windows, Microsoft, Redmond, WA). The chi-square test was used to assess associations between tumor grade and presence of α-SMA, degree of inflammation, and presence of giant cells. Fisher's exact test was used to assess the association between α-SMA and degree of inflammation among all 44 tumors.
Comparisons between tumor categories with respect to age, average vascularization, and central and peripheral proliferation rates were performed using the Kruskal-Wallis nonparametric analysis of variance. Sperman's correlation was used to assess correlation between proliferation within the periphery and central regions of the tumor, and between peripheral and central vascularity. A Wilcoxon signed rank test was used to assess the differences in the mean peripheral and central proliferation rates. A level of significance of 0.05 (P < 0.05) was used for all tests.
Results
The average age for cats at the time of first tumor diagnosis was 10.3 years (median = 9.5; range 3–14 years). The 44 cats with first occurrence sarcomas included 21 females (47.7%) and 23 males (52.3%). Fibrosarcoma was the most frequent histologic variant; however, marked cellular pleomorphism occurred within an individual mass and among different tumors. Forty-four primary fibrosarcomas, 16 recurrent tumors, and 10 nonvaccine sarcomas were evaluated microscopically for determination of histologic grade, presence of giant cells, intensity and type of inflammation, and presence of foreign material within macrophages (putative adjuvant). For primary fibrosarcomas, 11 of 44 (25%) were grade 1, 21 of 44 (47.7%) were grade II, and 12 of 44 (27.3%) were grade III. The histologic grade distribution was similar for 16 recurrent tumors: 4 (25%) were grade I, 8 (50%) were grade II, and 4 (25%) were grade III. Neoplastic multinucleated giant cells were present in 22 of 44 (50%) primary tumors and in 6 of 16 (37.5%) recurrent tumors (Fig. 1). Putative adjuvant material was seen in 10 of 44 (22.7%) primary tumors (22.7%), and 4 of 16 (25%) recurrent tumors. Six of 10 (60%) nonvaccine sarcomas were grade I, 2 of 10 (20%) were grade II, and 2 of 10 (20%) were grade III.
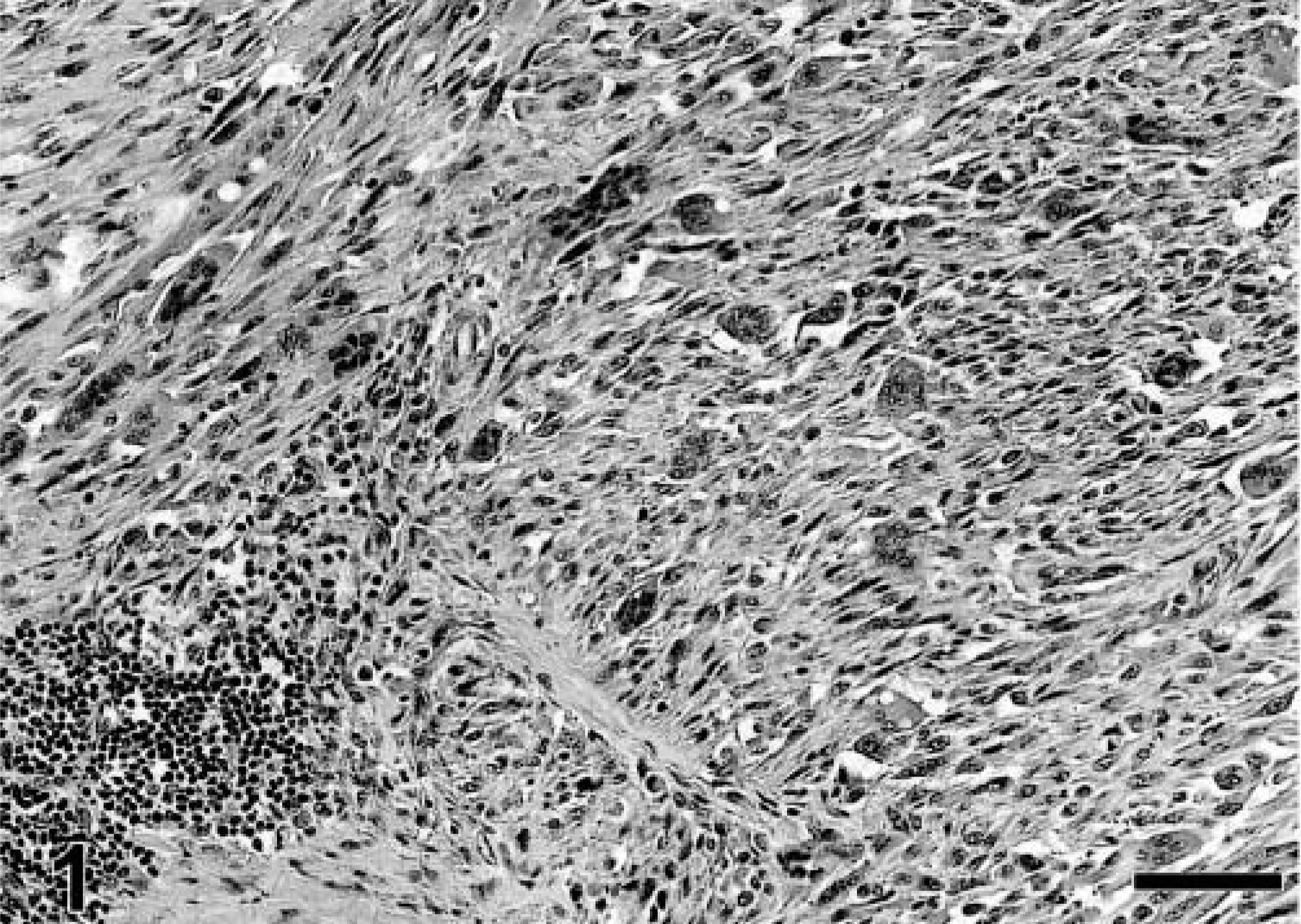
Vaccine-associated sarcoma; cat. High-grade fibrosarcoma with numerous neoplastic multinucleated giant cells and a small focus of lymphocytic inflammation (bottom left). HE. Bar = 72 µm.
In the vaccine-associated sarcomas examined herein, neutrophils were usually present within areas of necrosis, and small numbers of macrophages were admixed with lymphoplasmacytic infiltrates. Large, round macrophages containing blue-gray material (presumed adjuvant) within their cytoplasm were seen in 10 out of 44 (22.7%) primary tumors and in 4 out of 16 (25%) of recurrent tumors. The blue-gray macrophages formed clusters that were either entrapped within the periphery of the neoplasm or were distant from the tumor mass (Fig. 2). All sarcomas (primary and recurrent tumors) contained prominent lymphocytic inflammation. Prominent or abundant inflammation was seen in 59.1% of the primary tumors and 56.3% of recurrent tumors. Neoplastic multinucleated giant cells were present in 9 out of 12 (75%) grade III sarcomas, and in 13 of 21 (61.9%) grade II sarcomas, but none were seen in grade I sarcomas. A positive correlation was found between the presence of multinucleated giant cells and histologic grade. The proportion of giant cells increased as histologic grade progressed from 1 to 3 (P < 0.001). No association was found between the presence or intensity of inflammation and histologic grade (P = 0.305). None of the nonvaccine sarcomas had associated lymphofollicular inflammation, multinucleated giant cells, or blue-gray macrophages.
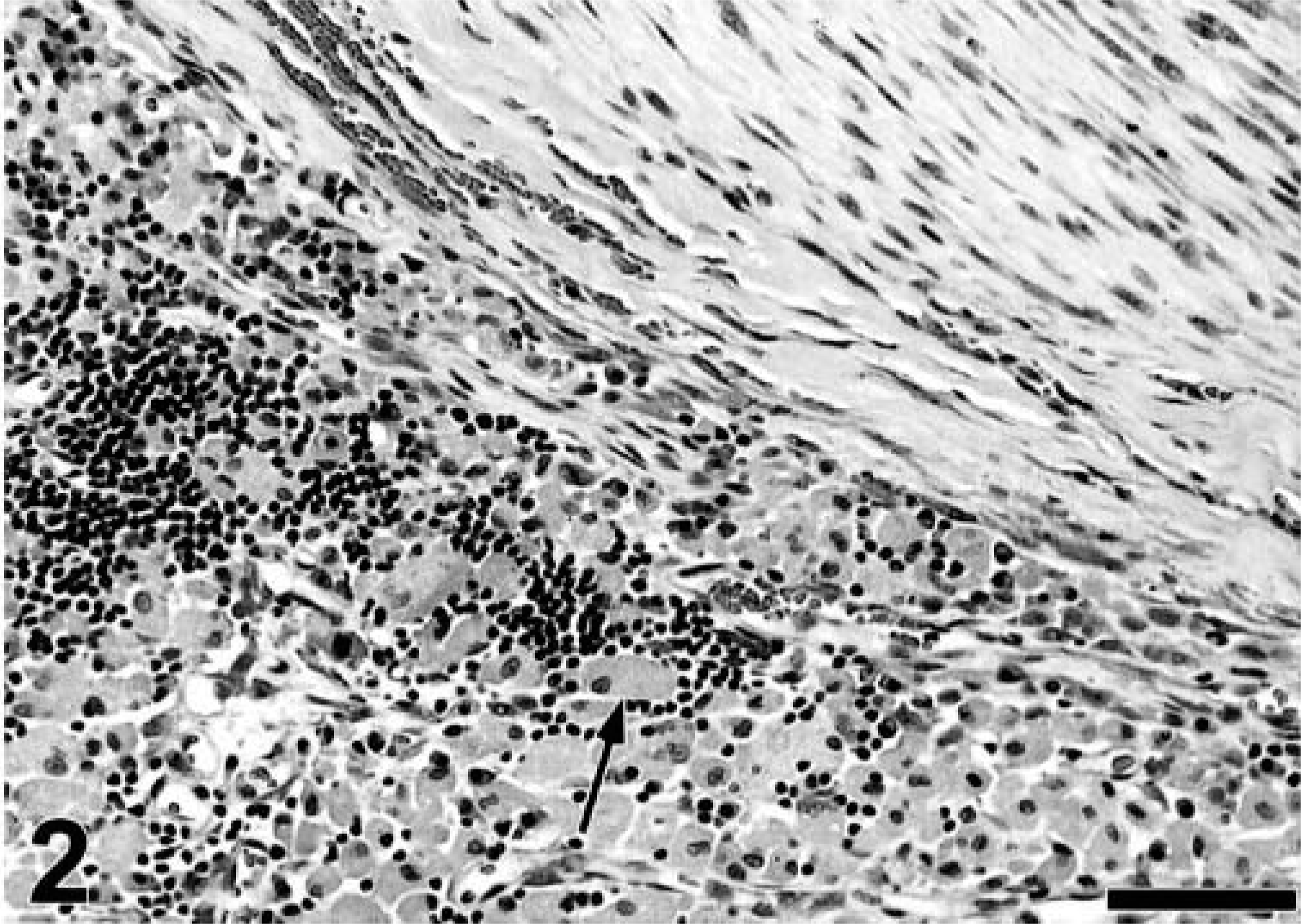
Vaccine-associated sarcoma; cat. Large aggregates of blue-gray macrophages containing presumed vaccine adjuvant material (arrow) are interspersed with lymphocytes along the periphery of a tumor nodule (upper right). HE. Bar = 104 µm.
A frequent macroscopic and occasionally microscopic finding among the feline vaccine-associated sarcomas was the presence of cavitations within central areas of tumors. Extensive, clear, fluid-filled cavities, often clinically detected as a fluctuating area similar to a seroma, were found in the larger tumor masses (Fig. 3). Histologically, the larger cavities were surrounded by a rim of necrotic tissue. Smaller microscopic cavities were lined by viable tumor cells that resembled reactive granulation tissue.
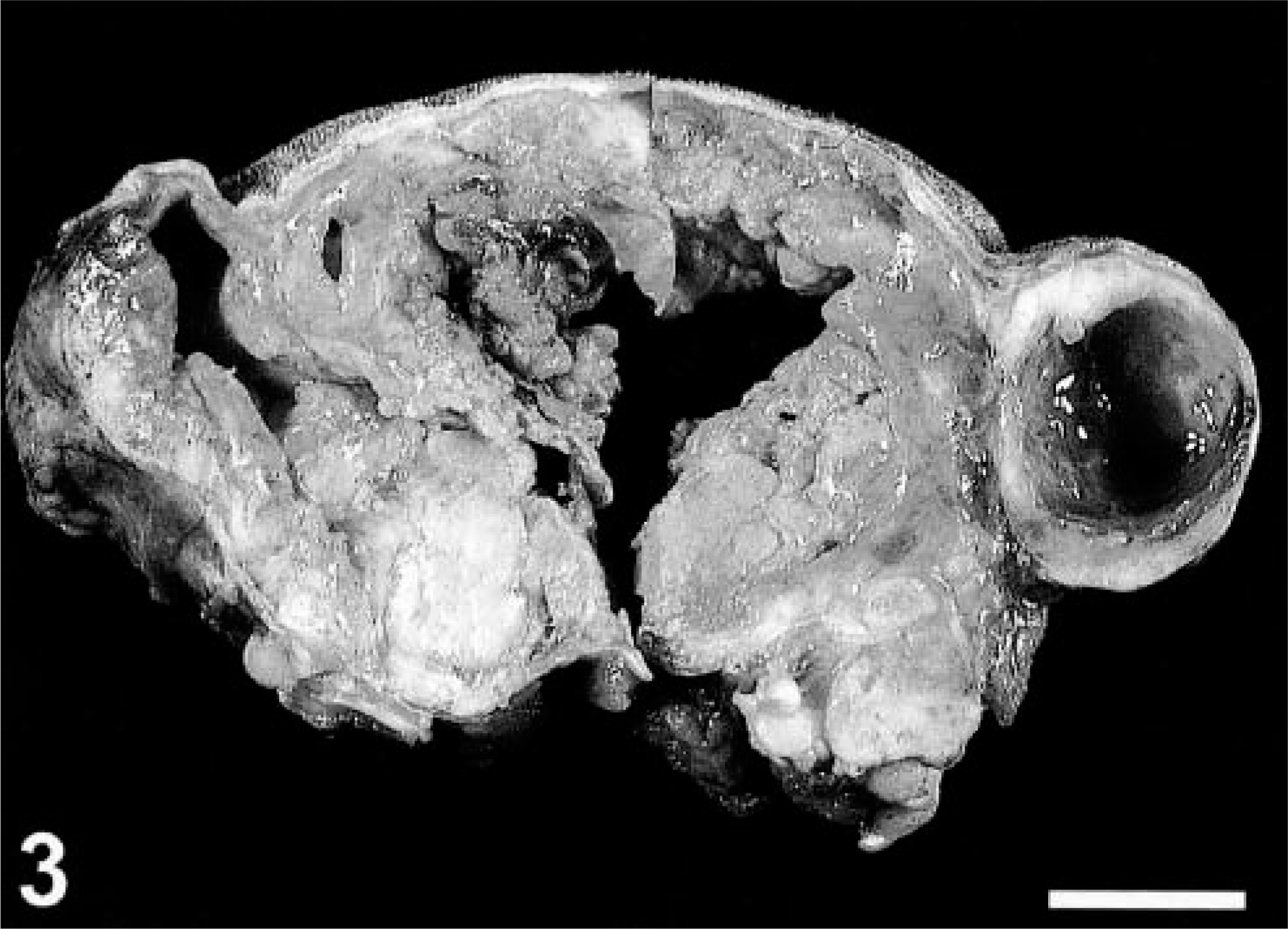
Vaccine-associated sarcoma; cat. Central area of necrosis and cavitation within a large tumor. Note smaller, well-defined cavities. Surgical specimen. Bar = 2 cm.
The proportion of T lymphocytes was quantified in 20 primary sarcomas that had a prominent inflammatory infiltrate (inflammation score 3). Marked variation occurred in the T lymphocyte content between lymphoid follicles within the same tumor and among different tumors; the average T cell proportion of individual tumors ranged from as low as 19.3% to as high as 87.7%. The average T cell proportion within lymphoid follicles of all tumors was 46.9% with a standard deviation of ± 18.8%. No significant difference was found in the average proportion of T cells among tumors of the three different histologic grades (P = 0.8630).
All 44 fibrosarcomas were strongly positive for vimentin and negative for desmin intermediate filaments. Twenty-eight of 44 (64%) tumors were immunoreactive for α-SMA but the immunolabel was heterogenous in intensity and distribution throughout the tumors. Of 28 actin-positive tumors, 28.6% had abundant immunolabeled cells (score 3), 32.1% had approximately 50% immunolabeled cells (score 2), and 39.3% had few immunolabeled cells (score 1). Immunoreactive cells were usually plump, polygonal or elongated, and contained abundant pink cytoplasm and large, often lobular nuclei. Tumor cells that were nonreactive for actin were generally smaller elongated spindle cells containing scant cytoplasm and fusiform nuclei—a more typical fibroblastic morphology. Multinucleated giant cells were either positive or negative for actin in different tumors.
No significant associations were found between tumor histologic grade and α-SMA expression (P = 0.173), or α-SMA immunoreactivity and degree of inflammation (P = 0.508). Two of 10 nonvaccine-associated sarcomas showed weak reactivity for α-SMA, one located on the pinna and another from the mammary gland.
Tumoral vascular density at the periphery or growth fronts of the tumor was examined in 44 primary sarcomas of different histologic grades. The average tumoral vascular density varied from 0.30% to 4.84% among the different tumors. The average vascular density was 1.152% for grade I tumors, 1.766% for grade II tumors, and 1.556% for grade III tumors. No significant associations were found between tumoral vascularity and histologic grade. The tumoral vascular densities and cell proliferation indexes of central (presumably quiescent) and peripheral areas (growth fronts) of 20 randomly selected primary tumors were compared. The average tumoral vascular density of the central areas from all tumors was 0.36% and the average proliferation index for the same region was 19.53%. The average vascular density from the periphery of all tumors (1.67%) was significantly higher than the central vascular density (P < 0.001). No significant difference was found between the proliferation index of the periphery (22.15%) and central areas of the tumors (P = 0.086). No significant differences were found among the average peripheral proliferation rates (P = 0.703) or vascular densities (P = 0.512) among tumors of different histologic grades.
Discussion
Feline vaccine-associated fibrosarcomas were examined for morphologic distinctions. A grading scheme for soft-tissue sarcomas from human medicine that has been previously used to categorize canine soft-tissue sarcomas allowed separation of feline fibrosarcomas into three groups based on light microscopic attributes—cellular differentiation, presence or absence of necrosis, and mitotic rate. 2,3,25 Histologic grading is currently the most important prognostic factor for human adult soft-tissue sarcomas with regard to probability for distant metastasis and overall survival rate. 16,22,34 Whether this grading scheme has prognostic merit for the cat awaits clinical trial. Although the majority of cats from which tumors were derived in this study received treatment, inconsistencies with regard to tumor sizes, surgical methods, and postsurgery chemotherapy or radiotherapy preclude comparisons of tumor-free intervals and survival times between histologic grades. In cases in which more than one tumor from the same animal was available, the histologic grade of the recurrent tumor remained the same as that of the primary tumor (three out of seven), was lower than that of the primary tumor (two out of seven), or was higher than that of the primary tumor (two out of seven). Although these observations are limited, recurrent tumors do not seem to assume a more anaplastic phenotype when compared to the original (primary) tumor.
Multinucleated giant cells are a common finding in feline vaccine-associated fibrosarcoma. 5 Analyses of the data suggested that the presence of intratumoral neoplastic giant cells may indicate a more poorly differentiated phenotype. In human patients with soft-tissue sarcomas, the presence of intratumoral neoplastic giant cells correlates with an aggressive, invasive tumor phenotype and is used as part of the paradigm to estimate prognosis. 15,27
Because immunotherapy or biological methods of therapy might be used as adjuvants after surgical treatment of feline vaccine-associated sarcomas, objective methods for quantifying immune responses are required. Image analysis was used to quantitate B and T lymphocytes associated with peritumoral lymphoid follicles. Although considerable differences were found in lymphoid cell distribution among the fibrosarcomas studied, a high proportion of T lymphocytes occurred within tumor-associated lymphoid follicles in many specimens, in spite of the fact that follicular formations are primary B cell areas of lymphoid structures (Fig. 4). 4,32 The role of the lymphocyte in tumorigenesis or host response to feline vaccine-associated sarcomas is unknown.

Vaccine-associated sarcoma; cat. High proportion of T lymphocytes (arrow) within a lymphofollicular aggregate along the tumor nodule (T). Scattered T lymphocytes occur throughout the mass. CD3, avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 144 µm.
Myofibroblasts, or cells with myofibroblast features (myofibroblast-like), have been described previously in feline vaccine-associated sarcomas based both on immunohistochemical and ultrastructural observations. 21 Myofibroblasts are mesenchymal spindle cells that have fibroblastic phenotype and produce collagen, but are distinguished by their capacity for contraction, the presence of cytoplasmic α-SMA, and the presence of subplasmalemmal plaques of cytoskeletal filaments on ultrastructural examination. 6,9,29 These cells are important in normal and abnormal wound healing, and are pivotal elements in reactive proliferative conditions and within the stroma of certain neoplasms. Myofibroblasts are recognized in a variety of pathologic conditions well characterized in humans, including hypertrophic scars, fibromatoses, nodular fasciitis, and desmoplastic response to lung and mammary ductal carcinomas. 23,26,30
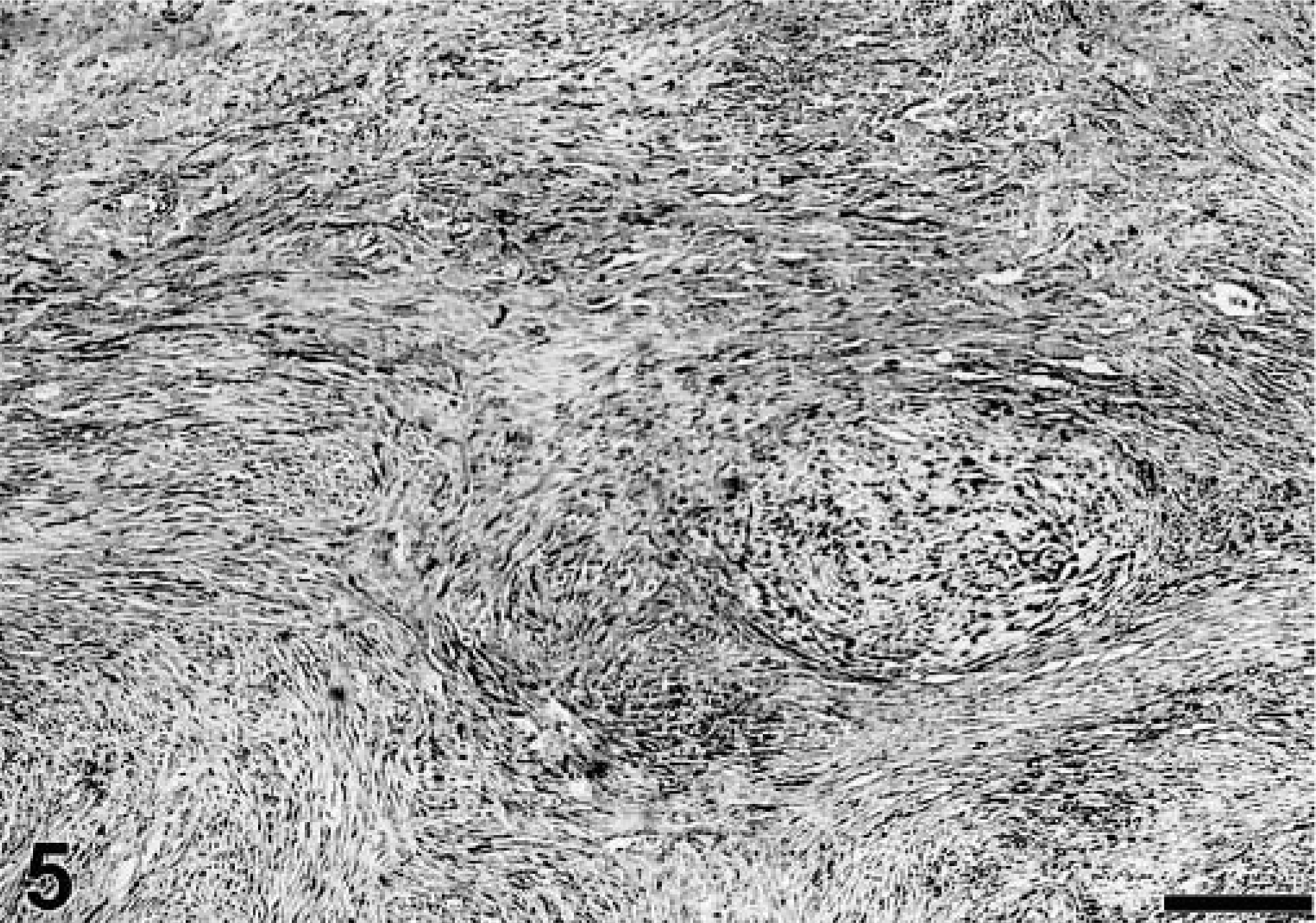
Immunolabeling of tumors herein is consistent with those previous observations, and suggests a pervasive myofibroblast-like phenotype within the histologic spectrum of feline vaccine-associated fibrosarcoma—more than 60% of tumors studied contained cells labeled for α-SMA. In some specimens, the majority of the tumor cells were immunoreactive for α-SMA. Immunolabeled cells were clearly neoplastic and consisted of a differentiated cell population within the tumors (Fig. 5), or, in less frequent cases, formed a capsule surrounding the neoplasm adjacent to areas of intense peritumoral inflammation (Fig. 6). Based on morphology and location, the cells along the periphery of the tumors (capsular pattern) seemed to be reactive rather than neoplastic. In one recent study in a rat colorectal tumor model, tumor-activated myofibroblasts formed a capsule that enveloped tumor nodules, mechanically preventing penetration of T lymphocytes and macrophages into the tumor. 18 If this mechanical action of myofibroblasts is true for vaccine-associated sarcomas, it may account for the presence of abundant lymphocytes along the periphery of the tumors. T lymphocytes that would normally penetrate the tumor mass conceivably are blocked at the periphery of the nodule by the presence of the myofibroblastic capsule. This might explain the high proportion of T cells in peritumoral follicular aggregates.
Vaccine-associated sarcoma; cat. Spindle neoplastic cell population (myofibroblast-like cells) positive for α-smooth muscle actin (α-SMA; black-stained cells) interspersed with nonreactive cells within the tumor. α-SMA, avidin–biotin–peroxidase complex, Mayer's hematoxylin counterstain. Bar = 144 µm.

Vaccine-associated sarcoma; cat. α-smooth muscle actin (α-SMA)–positive cells (myofibroblast-like) forming a capsule around tumor nodule (T = tumor). α-SMA, avidin–biotin–peroxidase complex, Mayer's hematoxylin counterstain. Bar = 72 µm.
The function and biological implications of myofibroblasts in tumor growth are controversial. The proposal has been made that when present in the tumor stroma, myofribroblasts prevent invasion by stabilizing the extracellular matrix through the secretion of lysyl oxidase. 18 Alternatively, adenocarcinomas transplanted into myofibroblast-rich granulation tissue assumed a more invasive and aggressive phenotype. 18 The origin of the myofibroblast is controversial and any mesenchymal cell is a candidate. Whatever the cell of origin, differentiation into the myofibroblastic phenotype is mainly modulated by cytokines. Myofibroblastic differentiation is induced by transforming growth factor-β and granulocyte-macrophage colony-stimulating factor (GM-CSF), produced by activated macrophages. However, lymphocytes also play a key role in the activation of macrophages, and therefore, indirectly influence the modulation of these cells. 29 True tumors derived from myofibroblasts are rare and not very well characterized in the cat. 8 The myofibroblastic differentiation of the vaccine sarcomas and the prominent associated inflammation seem to be unique morphologic characteristics of this category of tumors that may be somehow related. Although we found no significant association between the presence of myofibroblast-like cells (α-SMA-positive spindle cells) and inflammation, further investigation of the cytokines that form the microenvironment of vaccine-reaction granulomas and vaccine sarcomas might elucidate some of the mechanisms involved. The therapeutic implications of GM-CSF, interferon-γ, or other immune regulatory compounds are currently speculative but could prove valuable for the treatment of vaccine sarcomas. In humans, myofibroblasts are more common in tumorlike proliferations and low-grade soft-tissue sarcomas, and, therefore, the presence of myofibroblasts has been associated with a favorable prognosis. 31 This does not seem to be true in the case of feline vaccine-associated sarcomas, because these tumors are locally invasive.
A close relationship between intratumoral microvascular density and tumor proliferation rate has been described recently. 33 Mitotic activity quantitated within fast-growing areas along the periphery of a tumor (hot spots) also is known to be a more useful predictor of prognosis than mitotic activity quantitated from random tumor fields. 1,14 Tumor cell proliferation depends, in part, on adequate blood supply provided by neovascularization. Besides providing oxygen and nutrients, neoformed vessels influence tumor growth and spread by means of several growth factors and other important substances, including collagenases, urokinases, and plaminogen activator, secreted by activated endothelial cells. These activated endothelial cells are believed to be located at newly formed capillaries within the growth fronts at the periphery of the tumor. 1,33 High tumor microvessel density has been correlated with increased risk for metastasis and decreased survival time in human patients and with decreased postsurgical disease-free interval in dogs with mammary tumors. 7,33 In the vaccine-associated sarcomas studied herein, although significantly increased tumor microvessel density occurred at the tumor periphery compared to other tumor areas, no significant difference was found between cellular proliferation rates when the two sites were compared. This could represent a biological characteristic of the feline vaccine-sarcomas and would be a possible explanation to the commonly observed central areas of cavitation within the larger masses. 5 If the proliferation rate of the tumor is equivalent throughout the mass but the vascularity is higher along the peripheral areas, the tumor cells located within the central areas may eventually outgrow the vascular supply, undergoing necrosis and subsequently forming areas of cavitation. Lung carcinomas are another example of tumors that commonly present a similar central cavitary portion secondary to necrosis. 24 However, the cavities lined by granulation tissue may form secondary to a vaccine-induced necrotizing reaction with seroma formation from which the tumor subsequently arises.
In summary, in the 44 fibrosarcomas examined, wide variations occurred in cellular differentiation, mitotic index, and necrosis that allowed separation of the tumors into three histologic grades. Independent of the grade, the fibrosarcomas shared several morphologic characteristics, including the presence of peritumoral T cell–predominant lymphoid inflammation, accentuated peripheral vascularity, variably distributed myofibroblast-like cells, and neoplastic multinucleated giant cells. Macrophages containing presumed adjuvant material were not always seen in the tumors. Central cavitations, possibly resulting from previous vaccine-induced necrotizing panniculitis, were often observed in gross specimens. The neoplastic multinucleated giant cells only occurred in high histologic grade tumors and may indicate a more poorly differentiated tumor phenotype. Whether the presence of these giant cells, or other morphologic attributes of feline vaccine-associated fibrosarcoma, may provide prognostic information requires further investigation. Fibrosarcoma was the most frequent histologic type; however, because of the marked cellular pleomorphism within each tumor, we agree with the current and more accurate classification of these tumors as simply vaccine-associated sarcomas.
Footnotes
Acknowledgements
We wish to thank Dr. Alexandre Basso for assistance with initial data collection, Diane Naydan for immunohistochemistry, Dr. Peter F. Moore for valuable help with photomicrography, and Dr. Frank Verstraete for the image analysis equipment. This study was supported by the Dubee Fund, the Sacramento County Veterinary Medical Association, and the Center for Companion Animal Health, School of Veterinary Medicine, University of California.
