Abstract
A 24-year-old female rhesus macaque (Macaca mulatta) presented with a clinical history of chronic heart disease and prolonged recovery from sedation and anesthesia. At necropsy, the heart was markedly enlarged, with thinned ventricular walls, dilated chambers, and severe left atrioventricular valvular insufficiency. The ventricular walls contained numerous narrow, pale, often coalescing streaks that often extended along vessels into the deeper myocardium. Histologically, bands of interstitial fibrosis in the heart were associated with areas of myofiber atrophy, myofibril degeneration and loss, and inflammation. The left adrenal gland contained a 2 × 1 × 0.5 cm tumor with an unusual angiomatous pattern consisting largely of blood-filled sinusoids lined by one to four layers of low cuboidal to polyhedral tumor cells. In most sinusoids, the tumor cells appeared to be in direct contact with blood, although endothelial cells lined some sinusoids. Ultrastructurally, the tumor cells contained abundant electron-dense cytoplasmic granules. These granules were argyrophilic by Sevier-Munger staining and intensely immunoreactive for synaptophysin, chromogranin A, neuron-specific enolase, and S-100. These findings helped confirm the diagnosis of angiomatous pheochromocytoma. The heart lesions match those reported for catecholamine cardiomyopathy in other species.
Pheochromocytomas are neuroendocrine tumors arising from the catecholamine-secreting chromaffin cells of the adrenal medulla and are the most common adrenal medullary tumors of animals. They have been reported most frequently in cattle and dogs and are common in rats. 2 Here we report the microscopic, ultrastructural, and immunohistologic characteristics of a functional angiomatous pheochromocytoma and an associated cardiomyopathy in a rhesus monkey.
An adrenal mass was discovered at necropsy in a 24-year-old female rhesus macaque with a long-term clinical history of heart disease and delayed recovery from sedation and anesthesia. This monkey had developed a grade IV–V holosystolic heart murmur and usually required several hours to recover after receiving low doses of Telazol® (Fort Dodge Animal Health, Overland Park, KS) (normal recovery time is less than 30 minutes). The medical history showed that Ketaset®-associated (Fort Dodge Animal Health) arrhythmias were detected when the monkey was 11 years old and a systolic murmur was detected at 12 years. The heart was approximately twice the normal size, with thinned ventricular walls, dilated chambers, and severe left atrioventricular valvular insufficiency. The outer left and right ventricular walls were streaked by numerous narrow coalescing bands of myocardial pallor that often extended along larger vessels into deeper myocardium. The left adrenal gland was distorted by a 2 × 1 × 0.5 cm dark red mass that originated from and replaced the medulla and almost completely replaced the cortex except for a discontinuous compressed thin margin of cortical tissue.
Tissues were fixed in 10% neutral-buffered formalin and embedded in paraffin. Paraffin-embedded sections of the adrenal tumor were cut at 5 µm and stained with hematoxylin and eosin (HE) and also by the Sevier-Munger argyrophil stain method. Serial sections of the tumor were also immunohistochemically stained using polyclonal antibodies for synaptophysisn (Ventana Medical Systems, Tucson, AZ), neuron-specific enolase (NSE), neurofilament-200MW (NFP), glial fibrillary acidic protein (GFAP), S-100 protein, and mouse monoclonal antibodies for chromogranin A (CGA) and vimentin (Dako Corp., Carpinteria, CA) according to the manufacturers' instructions. For electron microscopy, a 2-mm cube of tumor tissue was cut from the paraffin block with a hot blade, deparaffinized, and then processed routinely for transmission electron microscopy (TEM) and examined in a Philips CM100 TEM (Mahwah, NJ).
Microscopic examination of HE-stained sections of the tumor showed that the expansile mass was completely encapsulated, had effaced normal adrenal medullary tissues, and was only partially surrounded by a thin compressed margin of cortical adrenal cells. Mitoses were very rare, and invasion of blood vessels and surrounding tissues was not evident. The tumor was characterized by a highly vascular angiomatous component, consisting largely of myriad blood-filled sinusoids that appeared to be lined by one to four layers of low cuboidal to polyhedral tumor cells having moderate amounts of finely granular, pale eosinophilic cytoplasm, ovoid nucleus and inconspicuous nucleolus (Fig. 1). However, electron microscopy showed that an endothelial cell layer actually lined some sinusoids. Most tumor cells contained abundant electron-dense cytoplasmic granules characterized by a dense homogeneous internal core and narrow clear submembranous space, which are ultrastructural features consistent with epinephrine granules.
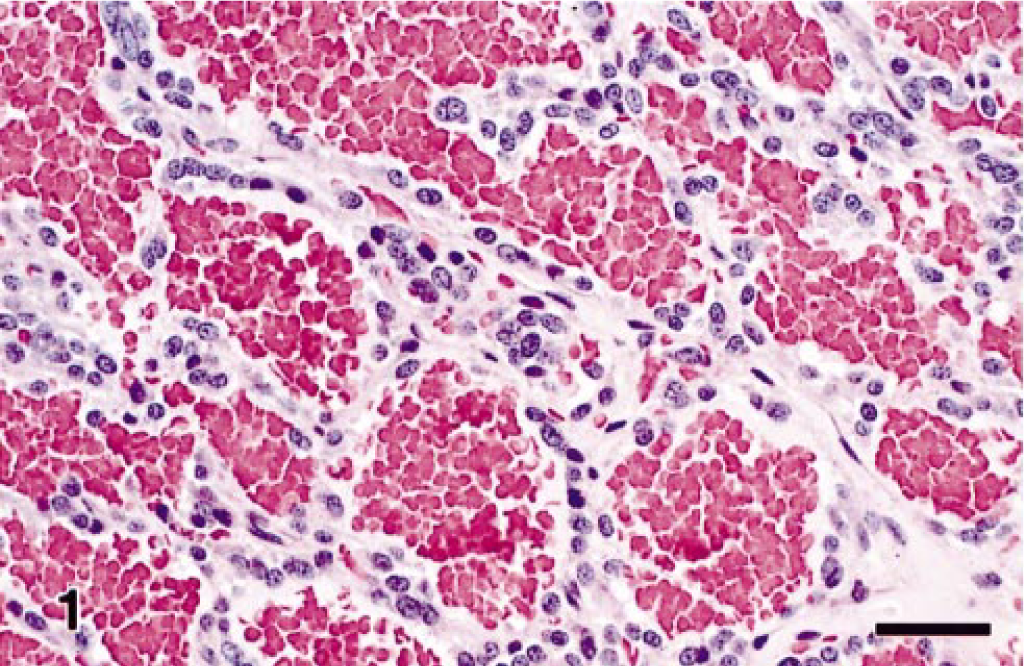
Angiomatous pheochromocytoma; rhesus macaque. Cuboidal to polygonal neoplastic cells line blood-filled sinusoids and are supported by a fine fibrovascular stroma. HE. Bar = 30 µm.
Pheochromocytomas with angiomatous features have been reported rarely. 14 Because pheochromocytomas can vary markedly in their histologic appearance, a definitive diagnosis may depend on application of specific histochemistry, immunohistochemistry, and ultrastructure techniques. Poorly differentiated adrenocortical carcinomas and pheochromocytomas may be differentiated by the combined use of argyrophilic staining and electron microscopy. However, both these methods depend on the detection of chromaffin granules, which can be scanty in some tumors. Thus, antibodies to several different neuroendocrine markers have proven useful in identifying atypical pheochromocytomas by histochemistry. Antibodies to NSE, synaptophysin, and CGA have proven useful in the widest range of species. 3 , 5 , 13 In this case, Sevier-Munger argyrophil–stained sections showed numerous dense brown to black cytoplasmic granules in the tumor cells (Fig. 2). By immunohistochemistry, the tumor cells were intensely immunoreactive for synaptophysin, CGA, NSE, and S-100 and were negative for GFAP, NFP, and vimentin (Fig. 3). These findings helped confirm the diagnosis of pheochromocytoma, which is a well-known, albeit rare cause of cardiomyopathy in humans.
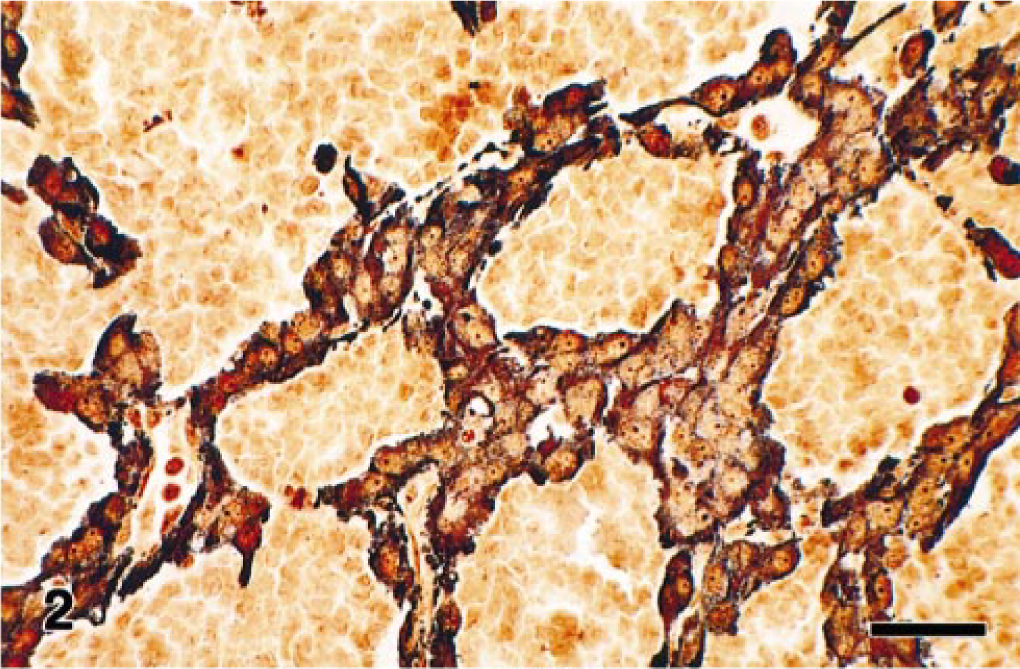
Angiomatous pheochromocytoma; rhesus macaque. The cytoplasm of tumor cells contains argyrophilic granules. Seiver-Munger method. Bar = 25 µm.
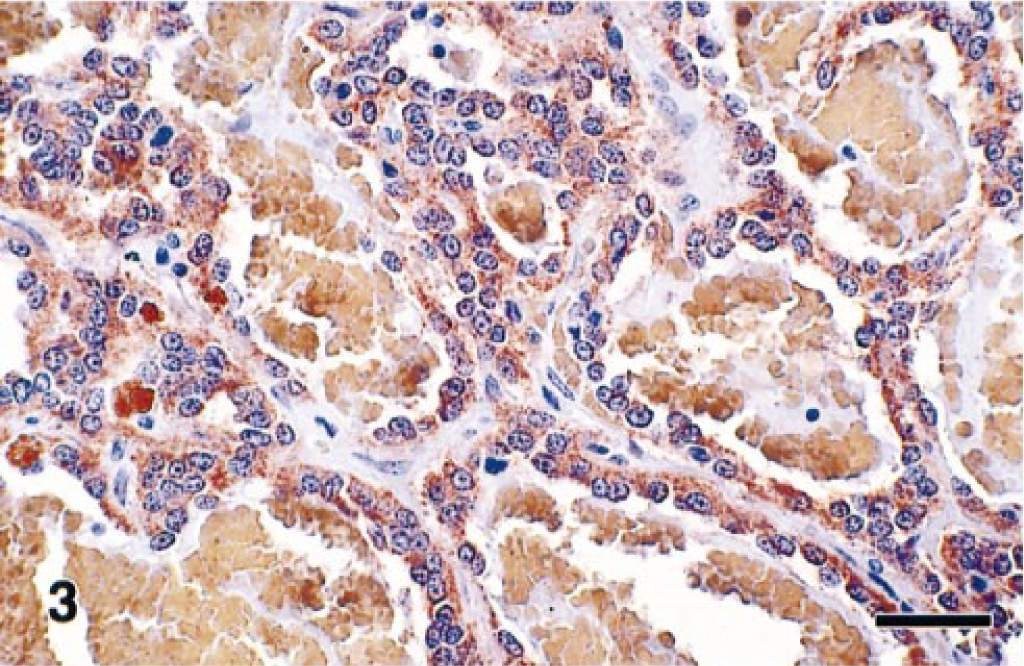
Angiomatous pheochromocytoma; rhesus macaque. Representative immunohistochemical staining of tumor cells immunoreactive for neuron-specific enolase. Immunoperoxidase technique. Bar = 30 µm.
Microscopic examination of this monkey's heart revealed that the bands of replacement and interstitial fibrosis were invariably associated with areas of myofiber atrophy, myofibril degeneration and loss, and inflammation. The multifocal to coalescing fibrotic lesions had a perivascular orientation and were most common in the outer layers of myocardium, although some extended into the subendocardium. Interstitial fibrosis frequently extended from these areas of scarring into the adjacent more normal myocardium (Fig. 4). In affected areas, irregular interwoven bands of fibrous connective tissue disrupted the normal orientation of variably sized myofibers, which ranged from thin remnants of atrophic and degenerating myofibers to very large hypertrophic myofibers. Most of the atrophic myofibers contained small amounts of pale disorganized granular sarcoplasm, whereas the hypertrophic myofibers had abundant hyperchromatic sarcoplasm and pleomorphic karyomegalic nuclei (Fig. 5). A few degenerating myofibers contained contraction bands (Fig. 6). Scattered amongst the degenerating and atrophic muscle fibers were necrotic myofibers with pyknotic or absent nuclei and scanty amounts of pale staining, globular, or highly vacuolated sarcoplasm. Macrophages, lymphocytes, and eosinophils were often present around or within the necrotic and disrupted myofibers (Fig. 7). Catecholamine-induced vascular lesions were not observed in other tissues. By TEM, early myofibrillar degeneration was characterized by out of register, nonparallel, or smeared Z lines (Fig. 8). By TEM, marked myocyte degeneration was characterized by myofibrillar loss and condensation of z-bands to form contraction bands (Fig. 9).
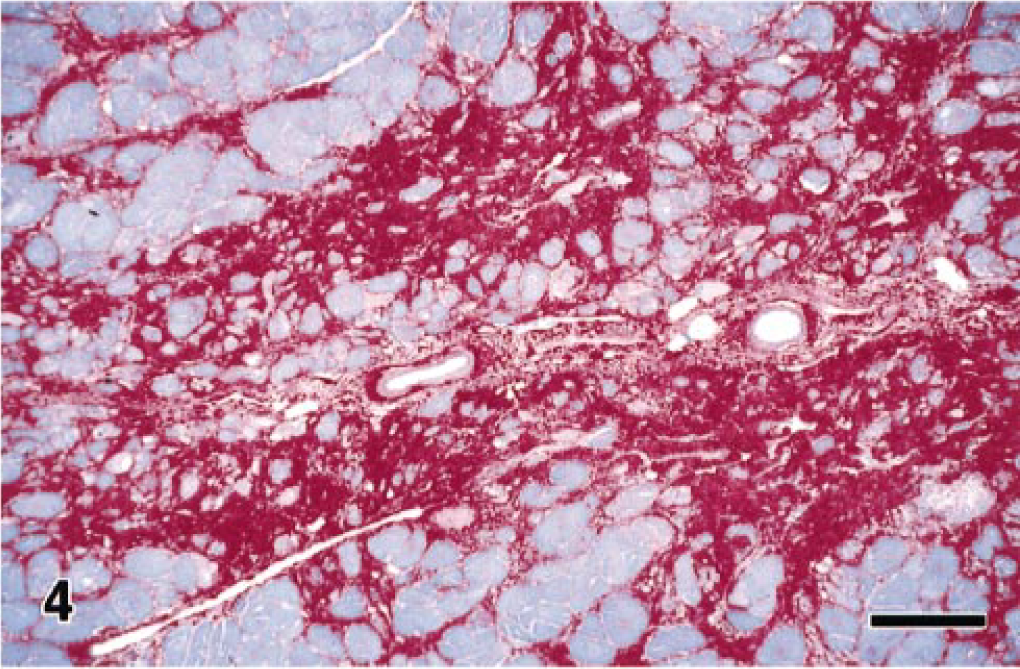
Heart; rhesus macaque. There is a perivascular distribution of replacement fibrosis, with extension of interstitial fibrosis into more normal surrounding myocardium. PTAH. Bar = 125 µm.
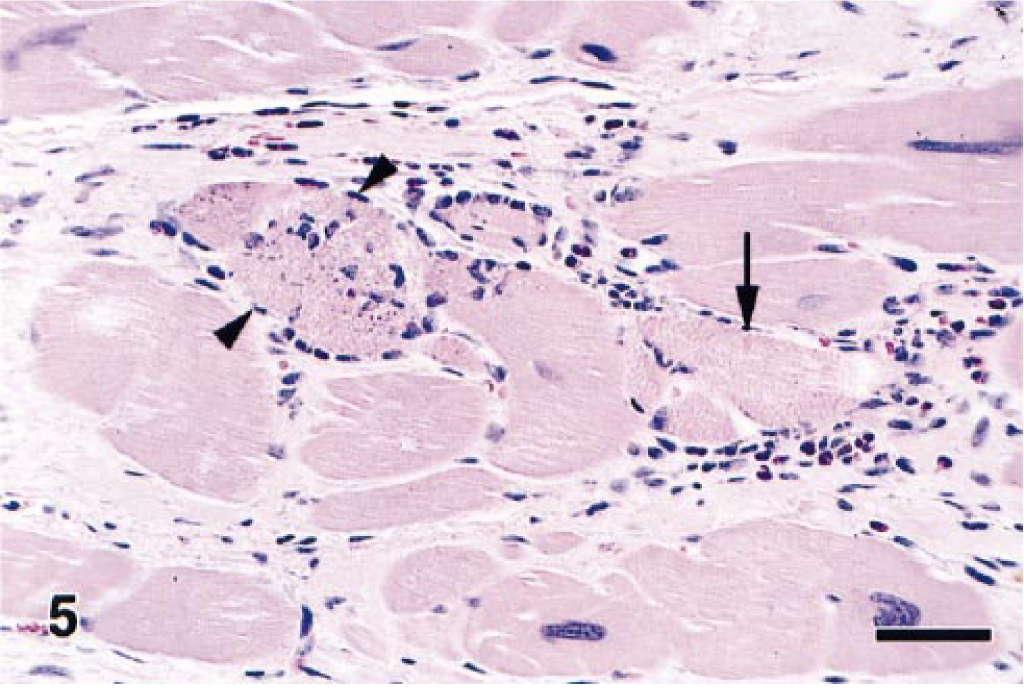
Heart; rhesus macaque. Increased fibrous connective tissue surrounds the variably sized myofibers. There are atrophic myofibers containing small amounts of pale granular sarcoplasm, hypertrophic myofibers containing pleomorphic karyomegalic nuclei, and necrotic (arrowheads) and degenerating myofibers having prominent contraction bands (arrow). HE. Bar = 60 µm.
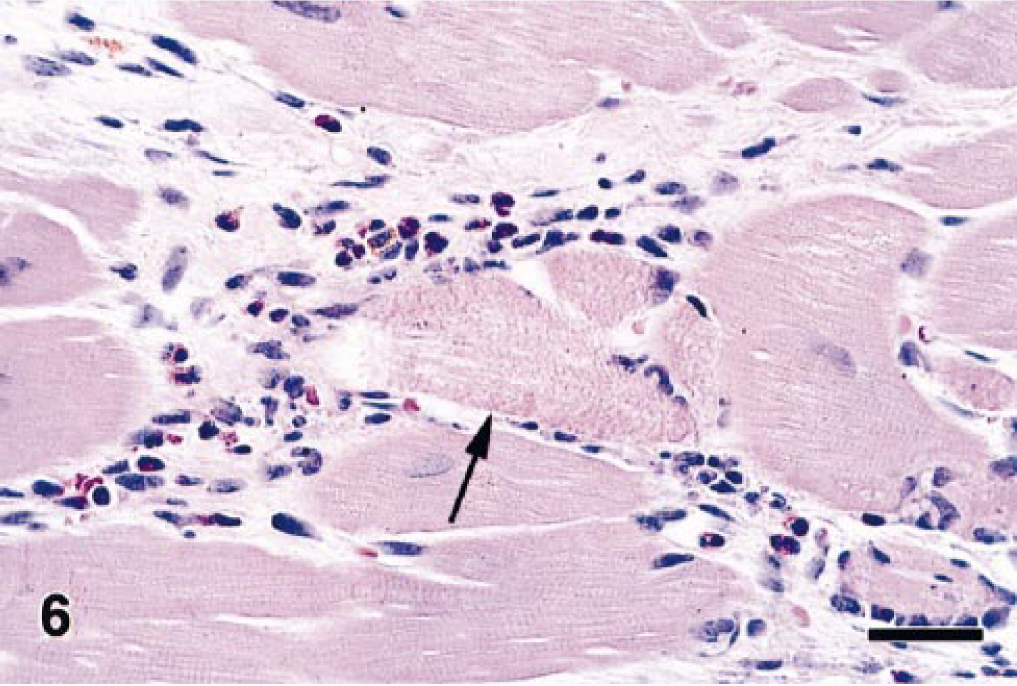
Heart; rhesus macaque. Higher magnification of Fig. 5 showing inflammation consisting of macrophages, lymphocytes, and eosinophils associated with a myofiber with prominent contraction bands (arrow). HE. Bar = 30 µm
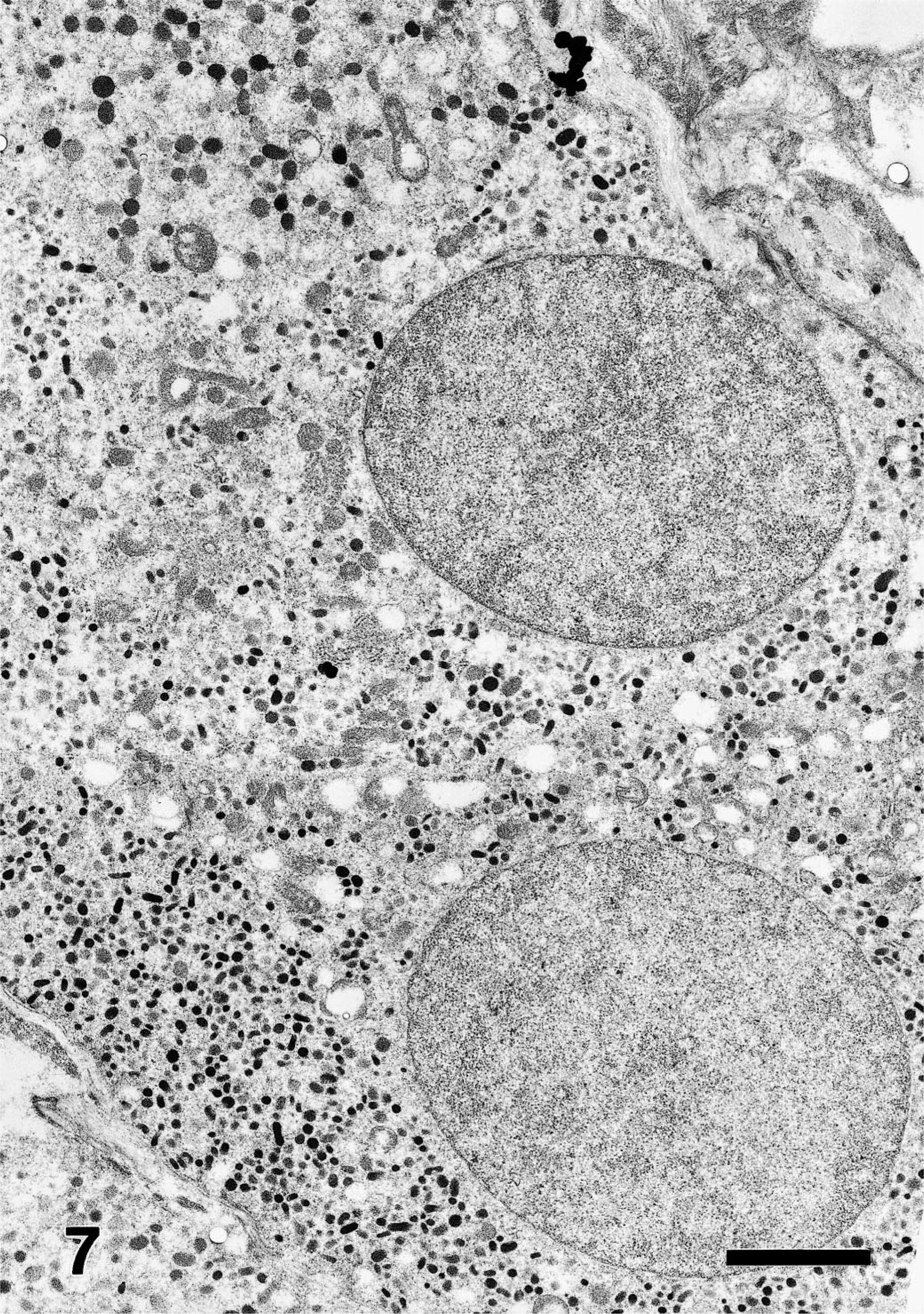
Angiomatous pheochromocytoma; rhesus macaque. Transmission electron micrograph showing tumor cells containing numerous cytoplasmic granules that have a coarsely granular internal core surrounded by a narrow clear submembranous space. Lead citrate, uranyl acetate poststain. Bar = 2.5 µm
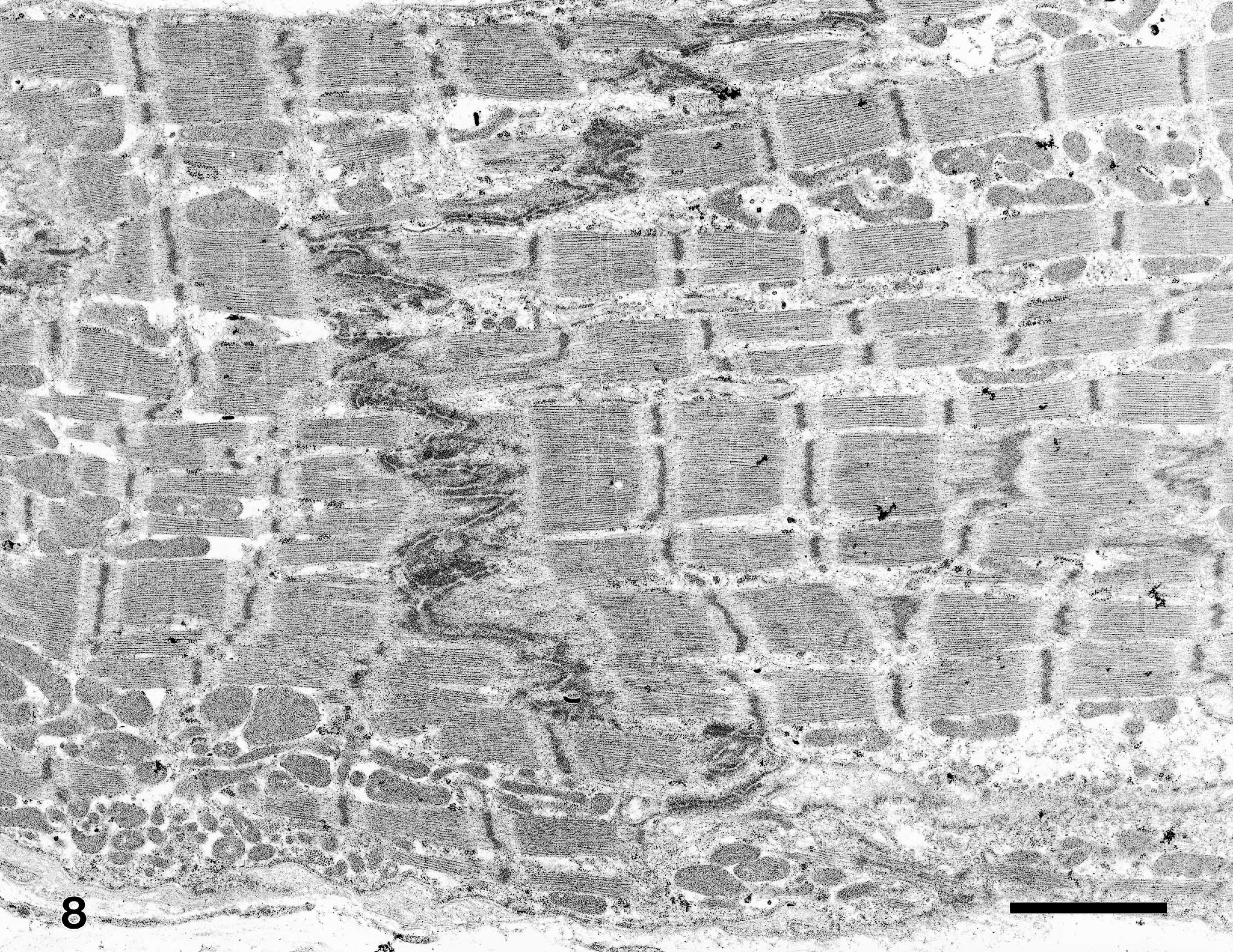
Myocardium; rhesus macaque. Transmission electron micrograph of mild myofibrillar degeneration. On both sides of the intercalated disk, note that z-lines are frequently out of register, are occasionally nonparallel, and rarely smeared. Lead citrate, uranyl acetate poststain. Bar = 2.5 µm

Myocardium; rhesus macaque. Transmission electron micrograph of marked myofibrillar degeneration. Note areas of myofibrillar loss with condensation of z-lines to form contraction bands. Lead citrate, uranyl acetate poststain. Bar = 5 µm
In humans, functional pheochromocytomas are known to induce hypertension, pulmonary edema, myocardial infarction, ventricular fibrillation, cerebral hemorrhage, and congestive heart failure. In dogs, pheochromocytomas are frequently incidental findings at necropsy or surgery; however, hypertension and vascular lesions are commonly observed in dogs with functional pheochromocytomas that are diagnosed antemortem. 16 Abnormal clinical findings in dogs are highly variable, but in those cases where blood pressure was measured, most affected dogs are hypertensive (systolic range, 164–325 mm Hg; diastolic range, 110–198 mm Hg). 7 Experimentally, both hypertension and cardiomyopathy are induced in rats implanted with transplantable pheochromocytomas. 8 These rats develop multifocal lesions of interstitial and replacement cardiac fibrosis, increased granularity of the myocyte cytoplasm and contraction band necrosis, and mixed inflammatory infiltrates. 12
An experimental model of catecholamine cardiotoxicity has been induced in the rhesus macaque. 11 In this rhesus monkey model, norepinephrine infusions (20 µg/kg body wt/minute) were given for 2 hours daily on three consecutive days. Myofibril degeneration, myocytolysis, and vacuolization with aggregation of lymphocytes and monocytes were seen at 2 hours after the last infusion. At later time points, the proliferation of fibroblasts became more marked, and by day 21 after infusion there was fibrosis in the affected areas. 11 The lesions of catecholamine cardiomyopathy are most likely due to the ischemia-induced reactive oxygen species (free radicals) that are released after catecholamine-induced vasomotor constriction of arterioles. 10 However, calcium overload due to the enhanced permeability of the myofiber membranes and even toxic oxidation products of norepinephrine (adrenochromes) may be involved. 10
In our rhesus macaque, an apparently functional angiomatous pheochromocytoma presented with clinical signs indistinguishable from those of idiopathic dilated cardiomyopathy. Immunohistochemistry and electron microscopy were invaluable in confirming the diagnosis of pheochromocytoma in this atypical angiomatous tumor. Although the presence of this tumor was not suspected antemortem, and blood levels of metanephrines were therefore not measured, the heart lesions observed in this rhesus macaque match those reported for pheochromocytoma-associated catecholamine cardiomyopathy in humans. 9 Historically, many pheochromocytomas in human patients are unsuspected clinically and are discovered either at autopsy or during surgery for unrelated conditions. 4 , 15 However, the antemortem diagnosis of pheochromocytoma as the proximate cause of otherwise “idiopathic” cardiomyopathy is important because effective medical or surgical treatment of the tumor has been shown to rapidly normalize catecholamine levels and blood pressure, resolve congestive heart failure symptoms, and gradually reverse the cardiomyopathy. 6 , 9 Increased awareness of the syndrome and increased use of assays for catecholamines and chromogranin should improve the antemortem diagnosis of pheochromocytomas both in humans and in veterinary medicine. In humans, it can be expected that the more widespread use of these assays will increase the proportion of occult pheochromocytomas that are diagnosed before, rather than after, cardiac transplantation.
Footnotes
Acknowledgements
We thank J. Brubaker and K. Kuehl for their expert technical assistance and Drs. K. Steele and D. Scott for immunohistochemistry support. We also thank Drs. Kelly Davis, Mark Martinez, Nate Powell, and Ms. Katheryn Kenyon for their critical review of this manuscript. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the US Army. Research was conducted in compliance with the Animal Welfare Act and other Federal statutes and regulations relating to animals and experiments involving animals and adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, 1996. The facility where this research was conducted is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
