Abstract
A spontaneous complex pheochromocytoma was diagnosed in the adrenal gland of an aged female Fischer 344 rat. This expansile neoplasm consisted of pheochromocytoma and areas of ganglioneuroma. The supporting stroma of both neoplastic components contained spindle-shaped cells, which also formed large fascicles. Immunohistochemically, pheochromocytoma cells stained for synaptophysin and chromogranin, scattered ganglioneuroma cells stained for neurofilament protein, and the spindle-shaped stromal cells were positive for S-100 protein. Special stains demonstrated Nissl substance in the ganglioneuroma cells and nerve fibers in the fascicles.
Complex pheochromocytoma, a rarely reported adrenal medullary tumor, is characterized by a pheochromocytoma component and a coexisting, variably differentiated neural component composed of ganglioneuroma and/or neuroblastoma, nerve fibers, and Schwann cells. Here, we describe the characteristics of a complex pheochromocytoma of the adrenal medulla in an aged female Fischer 344 (F344) rat that was routinely necropsied after 518 days in a sentinel program.
Fresh tissues were routinely fixed and processed. Paraffin-embedded sections were stained with hematoxylin and eosin (HE) or were submitted for special staining procedures such as the cresyl violet (Vogt's) method for Nissl substance, the Seiver–Munger method for argyrophilic granules, Bodian's method for nerve fibers and nerve endings, and the Bielschowsky method for neurofibrils. Additional sections were mounted on positively charged slides (Fisher Laboratory Products, Pittsburgh, PA) for immunoperoxidase detection of synaptophysin (Ventana Medical Systems, Tucson, AZ), and chromogranin, S-100 protein, glial fibrillary acid protein, and neurofilament-200MW (Dako Corp., Carpinteria, CA) according to the manufacturers' instructions.
An oval, 2.5- × 4-mm, expansile, unencapsulated mass replaced the adrenal medulla and compressed the cortex. Polygonal pheochromocytes 12–45 µm in diameter were arranged in packets and cords and were separated by a fine fibrovascular stroma containing spindle-shaped cells (Fig. 1). There were scattered mitoses and occasional extension into the compressed cortex. Scattered pheochromocytes stained for the neuroendocrine markers synaptophysin (Fig. 2) or chromogranin. Staining of argyrophilic granules was not evident with the Seiver–Munger method.
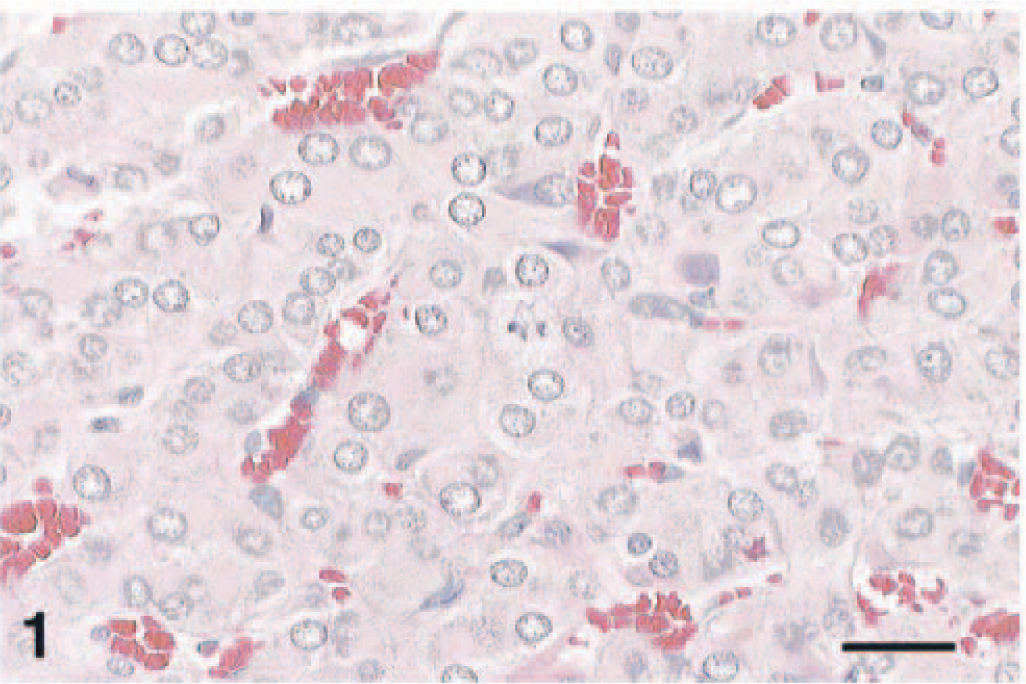
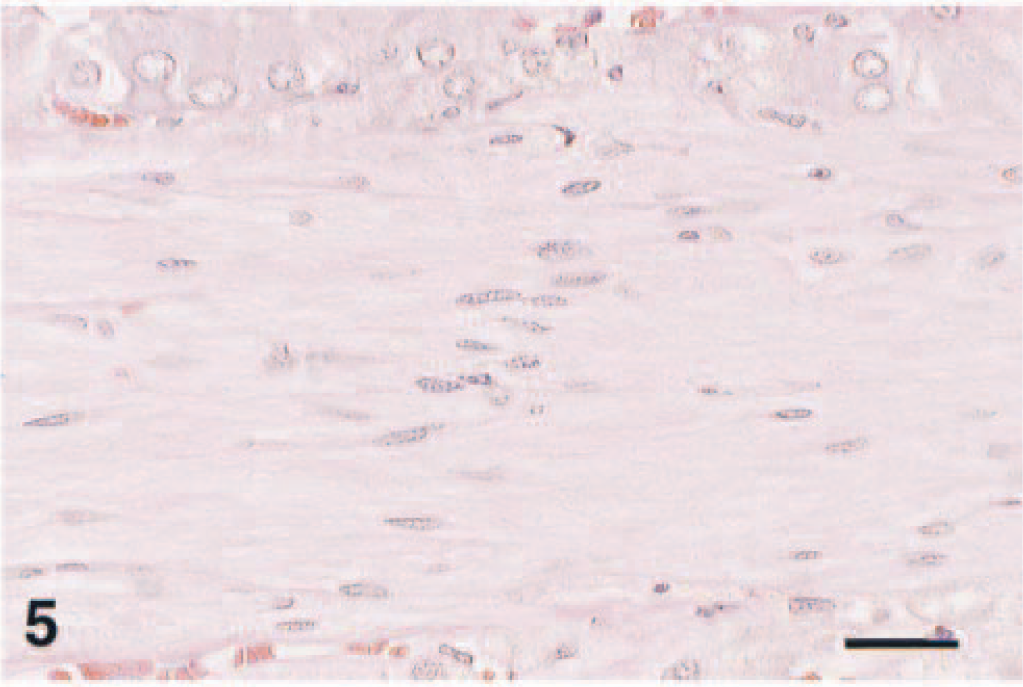
Complex pheochromocytoma; F344 rat. Neoplastic chromaffin cells are arranged in packets, nests, and cords. HE. Bar = 40 µm.
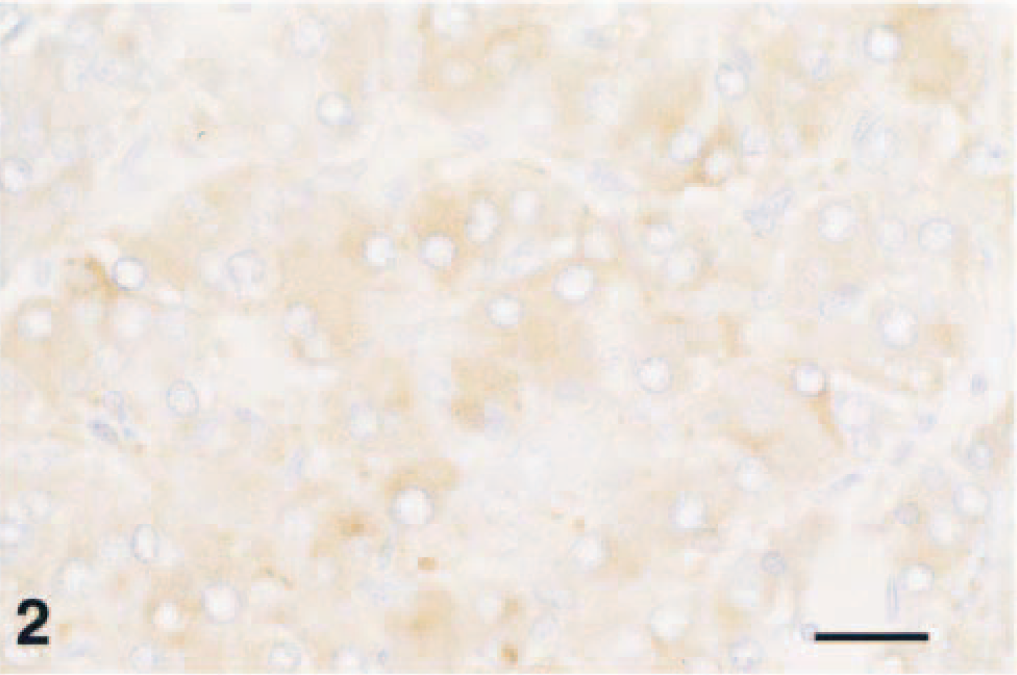
Complex pheochromocytoma; F344 rat. Many neoplastic chromaffin cells stain for synaptophysin. Avidin–biotin–peroxidase complex, Richard–Allen hematoxylin counterstain. Bar = 40 µm.
Less cellular areas contained individual or clustered ovoid to stellate ganglioneuroma cells supported by a loosely arranged fibrillar stroma containing spindle-shaped cells (Fig. 3). No mitoses were seen. The largest ganglioneuroma cells had elongated, tapered ends, reached 50 µm in diameter, and contained amphophilic to basophilic peripheral cytoplasm, which stained with the cresyl violet staining method for Nissl substance. Staining for neurofilament protein was occasionally seen; however, staining of the largest cells was not evident (Fig. 4). Dendritelike processes in the fascicles also stained for neurofilament protein. In some areas, ganglioneuroma and pheochromocytoma cells were intermixed.
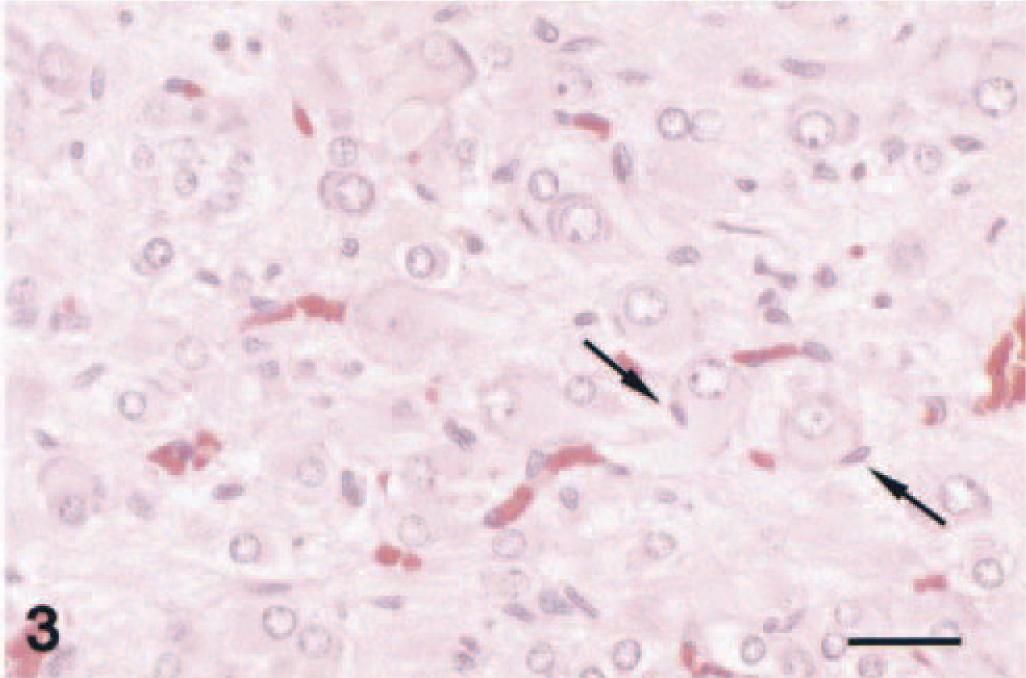
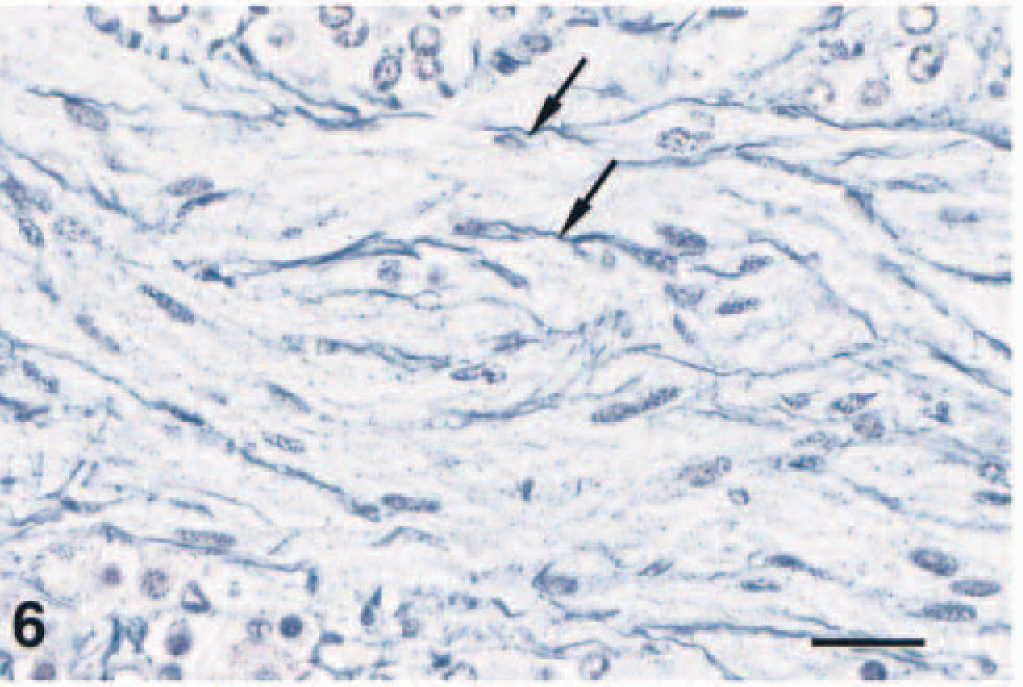
Complex pheochromocytoma; F344 rat. The ganglioneuroma component of this neoplasm is characterized by neoplastic ganglion cells, which are often closely supported by spindle-shaped cells (arrows). Note the supporting fibrillar stroma. HE. Bar = 40 µm.
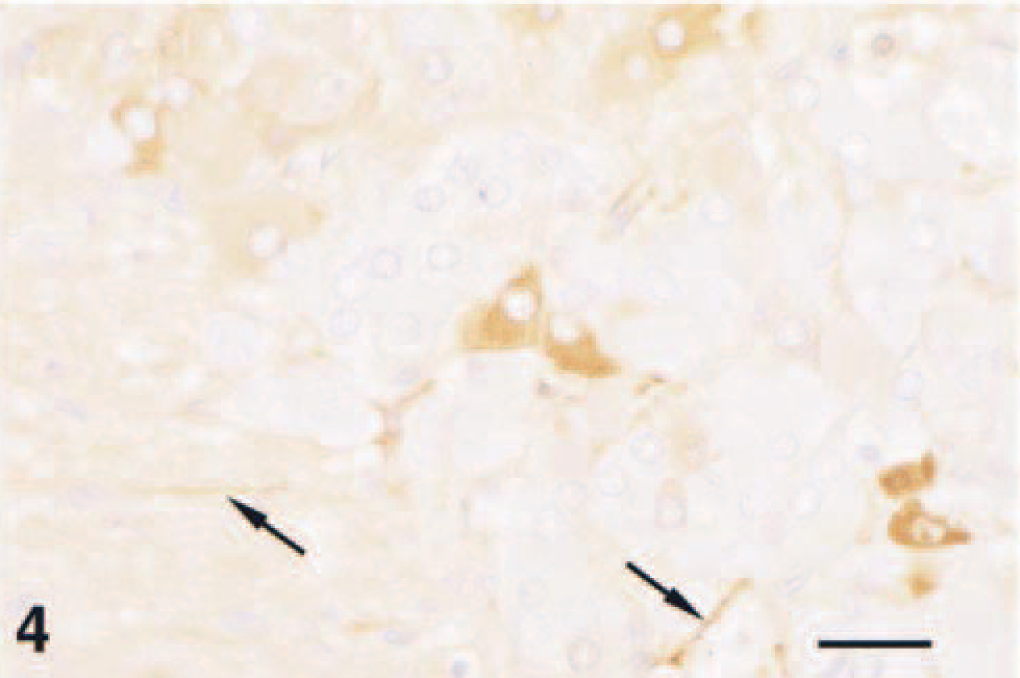
Complex pheochromocytoma; F344 rat. Neoplastic cells showing neuronal differentiation occasionally stain for neurofilaments, as did structures interpreted as nerve fibers (arrows). Avidin–biotin–peroxidase complex, Richard–Allen hematoxylin counterstain. Bar = 40 µm.
Ganglioneuroma stromal cells were interpreted as Schwann cells and multifocally were arranged linearly in branching fascicles 50–300 µm wide (Fig. 5). Similar cells separating pheochromocytes were interpreted as sustentacular or satellite cells. Regardless of pattern, both cells stained for S-100 protein. Bodian's method revealed many small, 3–6-µm-thick, undulating nerve fibers in the fascicles and extending into the pheochromocytoma (Fig. 6). The Bielschowsky method highlighted nerve fibers in the fascicles and few ganglioneuroma cells. Neither tumor nor stromal cells stained for glial fibrillary acidic protein. Hyperplasia of the pars distalis was also seen.
Complex pheochromocytoma; F344 rat. Fascicles containing spindle-shaped cells occasionally course through the pheochromocytoma component. Note nuclear palisading in center. HE. Bar = 40 µm.
Complex pheochromocytoma; F344 rat. Fascicles containing spindle-shaped cells also contain many nerve fibers (arrows). Bodian's method for nerve fibers and nerve endings. Bar = 40 µm.
The term complex pheochromocytoma is often used when the neural component is <80% of the neoplasm. 4 The features of the tumor reported here are similar to those found in F344 rats 7,8 and in Alderley Park Wistar-derived rats. 3 The presence of Schwann cells and sustentacular or satellite cells is in agreement with findings by others, and these cells most likely are normal cells growing into the tumor in response to microenvironmental factors. 9 The S-100 protein positivity of these cell types is similar to that in rats and humans. 1,5 Maekawa et al. observed ganglioneuromas in adrenal medullae of aged female F344 rats and neuroblastoma-like structures in some malignant pheochromocytomas. 6 Features of malignancy were not evident in the pheochromocytoma component of the tumor reported here, and as commonly seen in rats and humans, the ganglioneuroma component also appeared benign.
Spontaneous ganglioneuroma is rare in the adrenal medullae of F344 rats, may reach 10 mm in diameter, and is unilaterally and closely associated with pheochromocytoma. 2,7,8 It occurs in the adrenal medulla, autonomic ganglia, cranial nerve ganglia, and neuraxis and has been reported in several species. A similar association between ganglioneuroma and pheochromocytoma exists in humans, and areas of intermediate cytomorphology suggest a common stem cell for both components. 2 Reports of ganglioneuroma associated with thyroid C-cell proliferation in Sprague-Dawley rats support the belief that both C cells and autonomic ganglion cells arise from neuroectoderm. 2 Neural crest–derived cells are involved in familial multiple endocrine neoplasia (MEN) type 2b in humans, in which there are associations among pheochromocytoma, C-cell carcinoma, and multiple mucosal neuromas. 2
Pheochromocytoma frequently occurs in aging F344 rats, males more so than females, and may be solitary or multiple and unilateral or bilateral. It occurs in a wide range of species, commonly in cattle and dogs, and is the most common endocrine neoplasm in New World primates. In bulls, it sometimes develops concomitantly with functional thyroid C-cell tumors, reminiscent of familial MEN. In these cases, there often is adrenal medullary hyperplasia, but its role in the development of pheochromocytoma (with thyroid C-cell tumors) is not yet understood. In rats, pheochromocytoma and other proliferative adrenal medullary lesions may develop concomitantly with proliferative lesions of the endocrine pancreas, thyroid gland C cells, or as in this case, the pituitary gland. 4 In many strains of rats, including the F344, focal medullary hyperplasia and pheochromocytoma may represent a morphologic continuum; the increased incidence of either lesion is age dependent. 4
Primary adrenal medullary neoplasms such as pheochromocytoma, ganglioneuroma, and neuroblastoma are believed to have a common histogenesis. Although the adrenal cortex arises from primitive mesothelial cells from the urogenital crest, the adrenal medulla originates from neuroectodermal cells of neural crest origin. These neuroectoderm cells give rise to sympathogonia cells, which differentiate both to cells of the sympathetic nervous system and to secretory cells of the adrenal medulla. Normally, rare ganglion cells or myelinated nerve fibers are seen in the rat adrenal medulla. 4
A common stem cell suggests that coexisting pheochromocytoma and ganglioneuroma do not represent two neoplasms developing independently but, rather, divergent differentiation of existing cells. 2,4,8 Whether divergence results from differentiation of the ganglioneuroma or the pheochromocytoma or transformation of undifferentiated stem cells is uncertain. 4,7,9 Pheochromocytes may differentiate to neural cells, and rat pheochromocytes and immature rat or adult guinea pig chromaffin cells retain the capacity to differentiate to neural cells. Although this example of differentiation occurs under varied in vitro conditions, including the presence of nerve growth factor or glucocorticoids, the precise mechanism of histogenesis for complex pheochromocytoma remains uncertain. 4,9,10
Footnotes
Acknowledgements
We thank J. Brubaker and L. Ostby (US Army Medical Research Institute of Infectious Diseases) and the Division of Immunopathology and Department of Scientific Laboratories (Armed Forces Institute of Pathology) for their excellent technical assistance and Drs. D. Fritz, T. Larsen, and D. Scott for their critical review.
