Abstract
Several immunohistochemical markers have been used to demonstrate the presence of myoepithelial cells in order to determine their role in the histogenesis of mammary tumors. p63, a recently characterized p53 homologue, is consistently expressed in myoepithelial cells of the human breast; however, no assessment of its immunoreactivity has been reported so far in canine mammary tissues. We investigated p63 immunohistochemical expression, as a novel myoepithelial cell nuclear marker, in 81 samples of normal (n = 2), hyperplastic (n = 11), and neoplastic (n = 68) canine mammary tissues. Myoepithelial phenotype was confirmed by using complementary monoclonal antibodies: alpha-smooth muscle actin, cytokeratin 14, cytokeratin AE1/AE3, and vimentin. p63 expression was observed in 91.4% (74/81) of the samples evaluated. Normal mammary glands, mammary hyperplasias, and benign tumors showed 100% immunoreactivity, with p63 expression restricted to myoepithelial cell nuclei. In general, benign mixed tumors showed a basal cell compartment immunoreactive to p63, with a gradual decrease of its expression during myoepithelial transformation. p63 expression was found in 72% of malignant tumors, allowing myoepithelial or basal cell identification in spindle-cell carcinomas (2/2), tubulopapillary carcinomas (8/9), solid carcinomas (7/10), and carcinosarcomas (1/3). The osteosarcoma analyzed was p63 negative. In our series, stromal components were consistently nonreactive to p63. In conclusion, the present study reveals p63 as a sensitive and highly specific marker of myoepithelial cells in canine mammary tissues, and the authors suggest p63 as an additional marker for defining myoepithelial histogenesis.
Mammary gland tumors are the most commonly occurring neoplasm in the female dog and are known for their biologic and histomorphologic heterogeneity. 6 , 19 , 20
The classification of canine mammary neoplasms is based mainly on standard histopathology (descriptive morphology) and only to a lesser extent on histogenesis. Histogenetic classification is hampered by the difficulty in determining a specific cell type's origin in certain mammary tumors and also by the conflicting ideas concerning the role of myoepithelium in the genesis of complex and mixed tumors, 18 the most frequent canine benign neoplasms. 12 , 19
The high frequency of tumors showing myoepithelial or basal cell proliferation is one of the unique features of canine mammary tumors. 19 In contrast, similar lesions are considered rare in human breast tumors. Canine complex adenomas and mixed tumors are characterized by the presence of morphologically different types of myoepithelial cells as well as cartilage, bone, or fat in addition to proliferated luminal epithelial cells. 3 , 9 , 19 , 32
Because the identification of myoepithelial cells, which constitute the basal cell layer of normal mammary epithelia, may be difficult in routinely stained sections, several immunohistochemical markers have been used to demonstrate their presence in order to discriminate their role in the histogenesis of mammary tumors. In particular, as a result of the combined epithelial and contractile phenotype of these cells, the immunohistochemical markers available are based on their cytoskeletal components and on the presence of specific actin subtypes. 1 , 23 Monoclonal antibodies directed against alpha-smooth muscle actin (ASMA) and cytokeratin 14 (CK14) are known to immunoreact with myoepithelial cells, both in human breast tissue 7 , 11 , 15 , 26 , 31 and in the canine mammary gland. 3 , 32–34
p63, a recently characterized p53 homologue, 35 is necessary to maintain a stem cell epithelial population, and it is consistently expressed in basal cells of several types of multilayered epithelia 14 , 16 , 21 , 36 and in myoepithelial cells of the human breast 1 and is considered a possible marker of stem or reserve cells. 27 p63 germline inactivation suggests that p63 is necessary for the development of certain tissues, such as the mammary gland. 2 , 17 , 37
The aim of the present study was the evaluation of p63 immunoreactivity as a novel myoepithelial cell nuclear marker in canine mammary tissues and to provide some information concerning the histogenesis of these tumors. So far and to the best of our knowledge, no assessment of p63 immunoexpression in canine mammary tissues has been reported.
Materials and Methods
Tissue samples
We evaluated immunohistochemically eighty-one 10% neutral formalin-fixed, paraffin-embedded canine mammary tissues collected at the time of surgical excision or necropsy, obtained from the histopathology files of the Institute of Biomedical Science of the University of Porto and the University of Trás os Montes e Alto Douro, Vila Real, Portugal. Three-micrometer paraffin sections were cut and routinely stained with hematoxylin and eosin (HE) for histologic examination.
Histologic examination
Histologic diagnosis was conducted by three pathologists on HE-stained sections according to the World Health Organization's diagnostic criteria for tumors of the mammary gland of the dog. 18 All tumors were reevaluated using the same guidelines to ensure consistency of diagnosis. The tissues examined included specimens of normal canine mammary glands (n = 2) and hyperplastic (n = 11) and neoplastic (n = 68) samples (Table 1).
Classification and number (n) of the different mammary tissues evaluated for the expression of p63.



Antibodies
p63 expression was evaluated on additional sections of each specimen by immunohistochemistry, using a specific mouse monoclonal antibody (clone 4A4, Neomarkers, Freemont, CA, USA), which recognizes the human p63 protein, diluted 1 : 150 in phosphate-buffered saline (PBS), pH 7.4, according to the streptavidin–biotin–peroxidase complex method. To further assess the epithelial or myoepithelial phenotype of the cells, complementary monoclonal antibodies were used on consecutive serial sections of selected samples, using the same immunohistochemical procedure. The additional markers used were ASMA (1 : 50; NCL-SMA, Novocastra, UK), CK14 (1 : 20; NCL-LL 002, Novocastra, UK), cytokeratin AE1/AE3 (CK AE1/AE3) (1 : 50; DAKO Corp., Carpinteria, CA, USA), and vimentin (1 : 100; NCL-V9, Novocastra, UK).
Immunohistochemistry
Briefly, the tissue sections were deparaffinized and rehydrated, and antigen retrieval was carried out by microwave treatment 3 times for 5 minutes apiece (p63) or once for 5 minutes (CK14, ASMA, CK AE1/AE3, and vimentin) at 750 W (Míele M 734) in a 10 mM citrate buffer, pH 6.0. After cooling (20 minutes at room temperature), the sections were immersed in 3% hydrogen peroxide (H2O2) and distillated water for 30 minutes to block endogenous peroxidase activity. Nonspecific staining was eliminated by 20-minute incubation with normal rabbit serum (DAKO Corp.). Excess normal serum was removed and replaced by the primary antibody and incubated for 2 hours in a humid chamber at room temperature. After washing the slides, the sections were incubated with a 1 : 300 dilution of biotin-labeled anti-mouse secondary antibody followed by avidin–biotin complex (ABC) for 30 minutes. These reagents were purchased from DAKO Corp. Subsequently, the color was developed with 3,3′-diaminobenzidine tetrahydrochloride with H2O2 in PBS buffer for 10 minutes. Slides were counterstained with Gill's hematoxylin, dehydrated, and mounted. Primary antibodies, biotinylated secondary antibodies, and ABC reagents were diluted in PBS.
Adjacent normal mammary tissues were used as internal positive controls. Negative controls were carried out by replacing the primary antibody with PBS.
Evaluation of immunohistochemical data
Positivity was indicated by the presence of distinct brown nuclear (p63) or cytoplasmic (CK14, ASMA, CK AE1/AE3, and vimentin) staining. The number of immunoreactive cells was assessed semiquantitatively: − = no stained cells; ± = less than 10% positive cells; + = 10–50% positive cells; and ++ = more than 50% positive cells.
Results
The immunohistochemistry results for p63 are summarized in Tables 2, 3 and discussed below using the diagnostic categories listed in Table 1. The immunoreactivity for CK AE1/AE3, CK14, ASMA, and vimentin of normal mammary gland and selected cases of mammary lesions is indicated in Tables 2 and 4, respectively.
Immunoreactivity of different tissue components of the canine normal mammary gland for cytokeratins (CK14 and AE1/AE3), alpha-smooth muscle actin (ASMA), vimentin, and p63 antibodies.∗
∗ - = no stained cells; ++ = more than 50% positive cells.
Immunoreactivity of canine mammary hyperplasias and tumors for p63 antibody.
∗ LE = luminal epithelial cells; RME = resting myoepithelial cells; PME = proliferating myoepithelial cells; SP = spindle-shaped interstitial cells; ST = stellate-shaped interstitial cells; B = basaloid epithelial cells; CH = chondrocytes; OS = osteoblasts.
† - = no stained cells; ± = less than 10% positive cells; + = 10–50% positive cells; ++ = more than 50% positive cells.
Immunoreactivity of selected mammary hyperplasias and tumors for cytokeratins (CK14 and AE1/AE3), alpha-smooth muscle actin (ASMA), and vimentin.
∗ LE = luminal epithelial cells; RME = resting myoepithelial cells; PME = proliferating myoepithelial cells; SP = spindle-shaped interstitial cells; ST = stellate-shaped interstitial cells; B = basaloid epithelial cells; CH = chondrocytes; OS = osteoblasts.
† - = no stained cells; ± = less than 10% positive cells; + = 10–50% positive cells; ++ = more than 50% positive cells.
Normal mammary gland (n = 2)
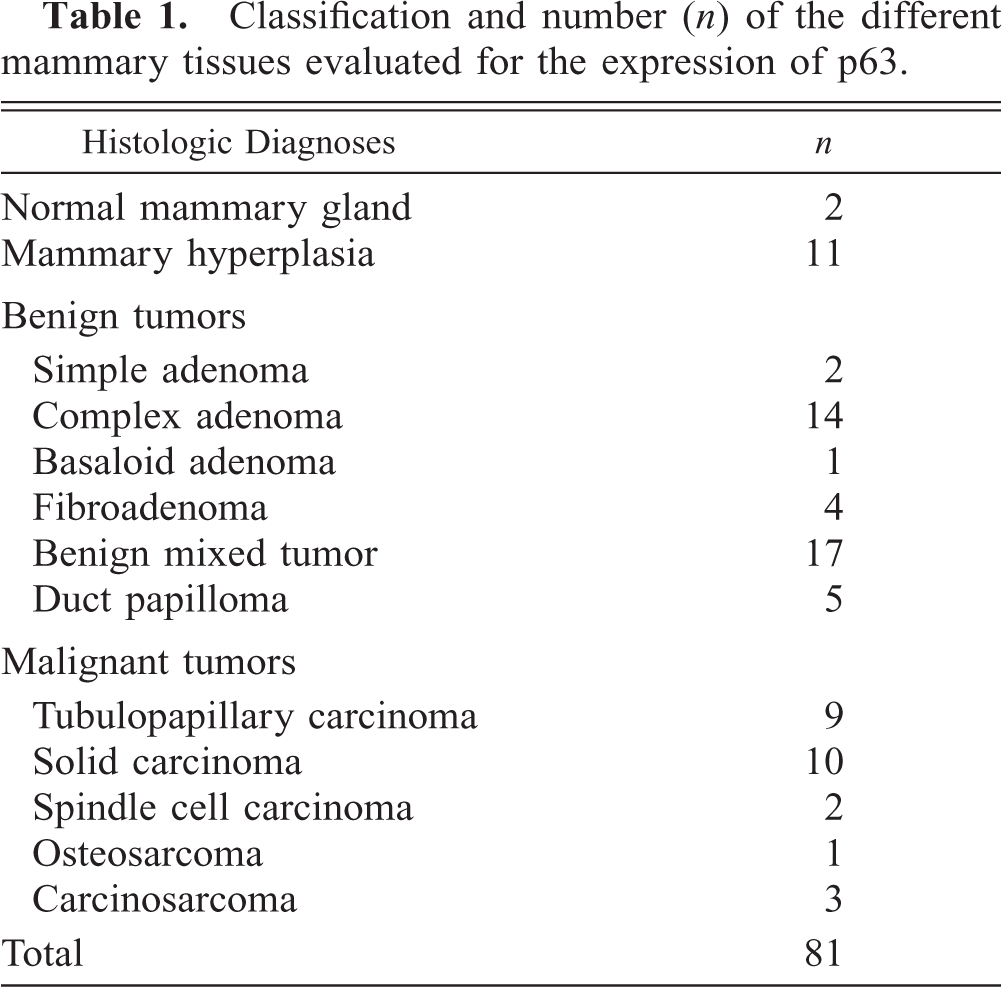
In the normal mammary tissues, strong p63 immunoreactivity (++) highlighted the myoepithelial cell nuclei evenly distributed along the ductal and alveolar structures (Fig. 1). Myoepithelial phenotype was confirmed by using the classic myoepithelial cell markers. Mammary secretory cells and stromal components were consistently nonreactive to p63, whereas ASMA immunoreacted with muscle stromal cells, and the pan-epithelial CK AE1/AE3 also stained the luminal epithelium. Both myoepithelial and mesenchymal cells were vimentin positive, in accordance with previous studies. 13 , 29 Whenever skin was present, p63 antibody also stained basal epidermal cell layers, as in human skin, 25 allowing the use of epidermis as an additional internal control.
Normal mammary gland; dog. Alveoli with strong nuclear p63 immunoreactivity of the myoepithelial cell layer. Streptavidin–biotin complex method. Gill's hematoxylin counterstain. Bar = 37µm.
Mammary hyperplasia (n = 11)
Hyperplastic mammary tissues showed a similar p63-labeling pattern when compared with normal mammary gland, with its expression confined to the resting myoepithelial cells.
Similar immunoexpression patterns to normal mammary tissue also were observed for the additional antibodies used.
Benign mammary tumors (n = 43)
Simple adenoma ( n = 2)
These tumors were composed of simple proliferations of well-differentiated luminal epithelial cells. The labeling patterns in simple adenomas correspond largely to those observed in normal mammary tissue, with p63-immunoreactive cells exclusive to myoepithelium and resembling normal tissues; all p63-positive cells consistently coexpressed cytoplasmic myoepithelial cell markers.
Complex adenoma (n = 14)
A mixed proliferation of secretory epithelial cells and cells resembling myoepithelial cells in a mucinous stroma characterized this type of tumor. As in previous studies, 30 we identified distinct myoepithelial cell morphologies: resting and proliferative suprabasal cells and spindle- and stellate-shaped interstitial cells. Suprabasal cells were located between the basement membrane and the luminal epithelium and exhibited flattened spindle (resting cells) or polygonal morphologies (proliferative cells). Interstitial cells were frequently arranged in solid nests apposed to epithelial elements or isolated in the interstitium. Suprabasal cells showed strong p63 immunoreactivity, whereas most interstitial cells showed a fainter positivity. No p63 expression was observed in luminal cells. All myoepithelial cell types were vimentin positive, whereas there was a reduction of immunoreactivity in both cytokeratins and ASMA from resting to stellate myoepithelial cells.
Basaloid adenoma (n = 1)
This tumor was characterized by the cluster proliferation of monomorphic, basaloid epithelial cells. The peripheral basal epithelial cells showed strong p63 immunostaining, whereas the centrally located were p63 negative.
Fibroadenoma (n = 4)
These tumors consisted of a mixture of epithelial (luminal and myoepithelial cells) and stromal elements. p63 expression was confined to myoepithelial cells lining p63-negative luminal epithelial components.
Benign mixed tumor (n = 17)
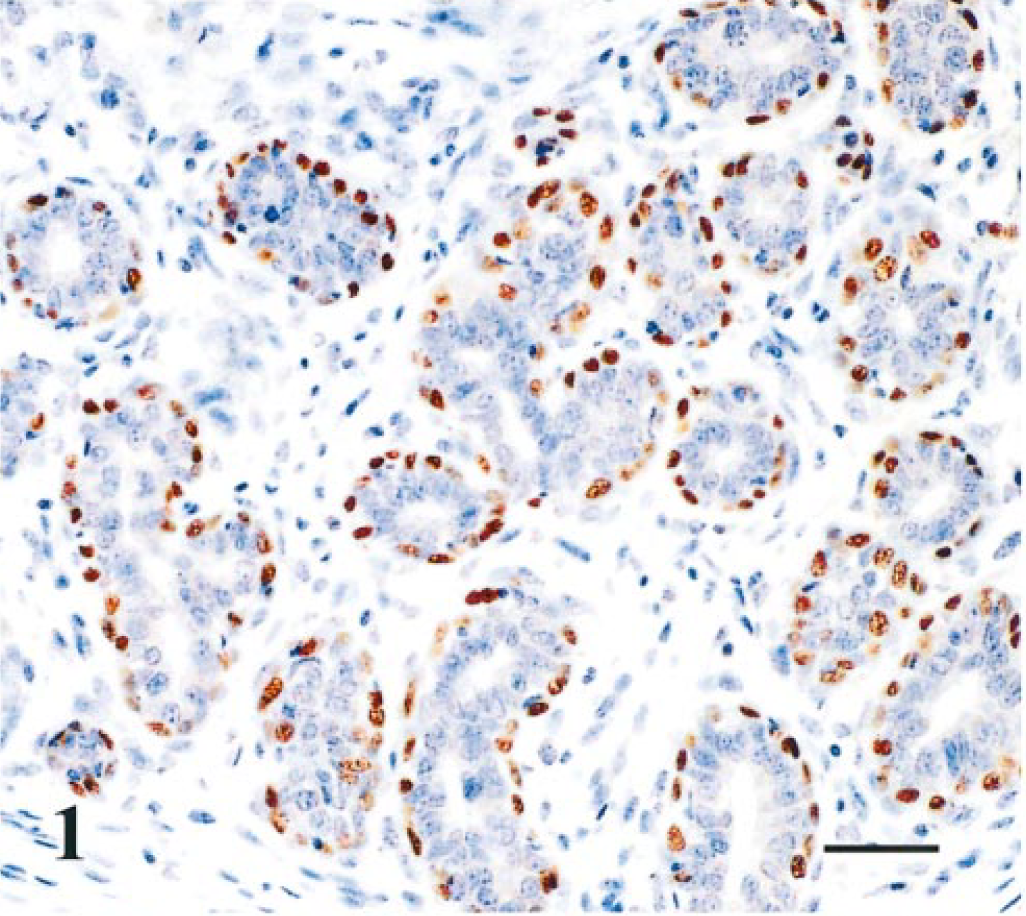
These complex tumors were characterized by the proliferation of both glandular (luminal and myoepithelial) and mesenchymal elements like cartilage (n = 17) or cartilage and bone (n = 4). Distinct myoepithelial cell morphologies, as in complex adenomas, were observed. The basal compartment of myoepithelial cells was strongly p63 immunoreactive and a decrease of p63, cytokeratins, and ASMA expression was observed from resting to stellate cell morphologies. Mesenchymal elements like cartilage or bone were p63 negative (Fig. 2). Vimentin strongly immunostained all myoepithelial cell types, chondroblasts, and osteoblasts.
Mammary gland; benign mixed tumor; dog. Luminal epithelial cells p63 negative and myoepithelial cells p63 immunoreactive (on the left) and mesenchymal components (cartilage) p63 negative (on the right). p63-Immunoreactive cells include suprabasal myoepithelial cells (arrow heads) and some interstitial cells (black arrows) near the cartilage. Streptavidin–biotin complex method. Gill's hematoxylin counterstain. Bar = 37µm.
Duct papilloma (n = 5)
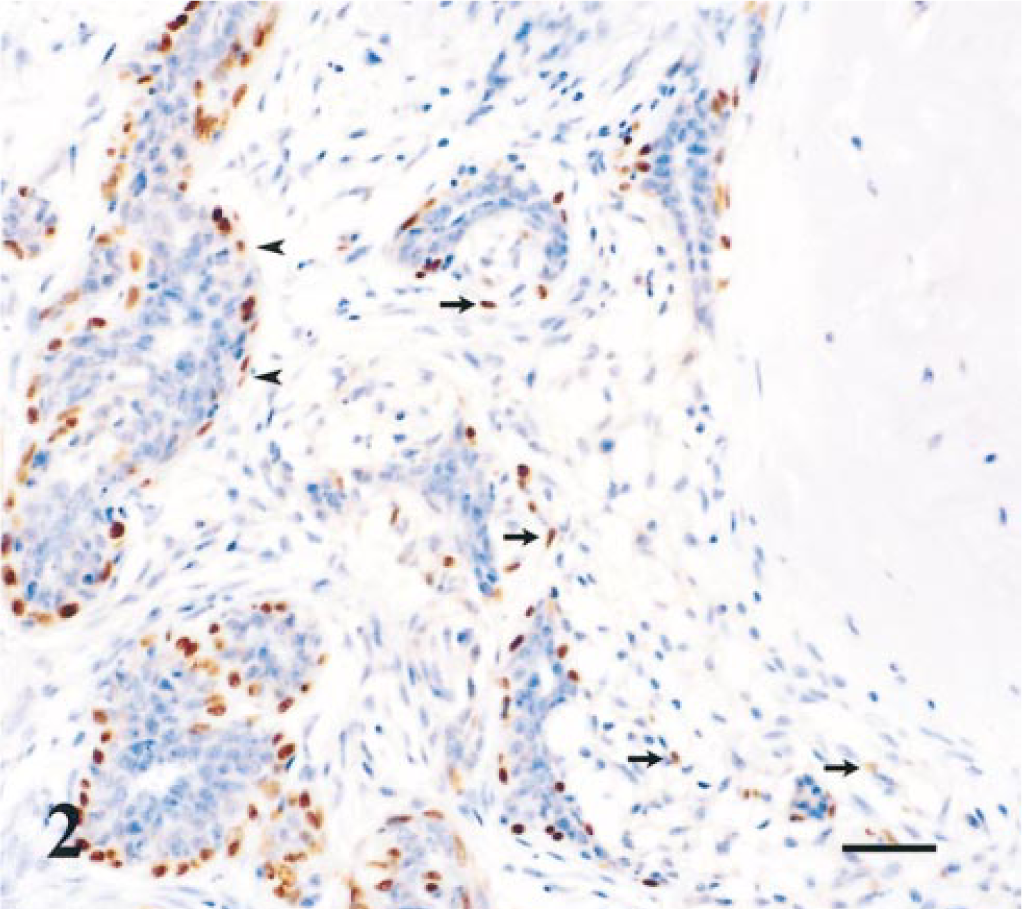
These tumors consisted of papillomatous projections in distended ducts lined by epithelial and resting myoepithelial cells and supported by stalks of vascularized connective tissue. In two cases, we also identified interstitial, spindle myoepithelial cells. All the myoepithelial cells observed showed strong p63 immunoreactivity (Fig. 3).
Mammary gland; duct papilloma; dog. Nuclear immunoreactivity of resting and proliferating myoepithelial cells for p63. Streptavidin–biotin complex method. Gill's hematoxylin counterstain. Bar = 37µm.
Malignant mammary tumors (n = 25)
Tubulopapillary carcinoma (n = 9)
These carcinomas were composed of a proliferation of cells resembling luminal epithelial cells and showing a tubular or papillary arrangement. A strong p63-immunoreactive myoepithelial resting cell layer was observed, with the exception of an invasive carcinoma, which was p63 negative. In addition, five of the nine tubulopapillary carcinomas were characterized by the presence of interstitial myoepithelial cells that were p63 immunoreactive (complex carcinomas). In selected cases subjected to additional immunohistochemistry, p63-positive cells also were stained for both cytokeratins, ASMA and vimentin. The luminal epithelium was p63 and ASMA negative and was stained for cytokeratins in all cases and for vimentin in half the cases.
Solid carcinoma (n = 10)
These carcinomas were characterized by the arrangement of luminal epithelial tumor cells, usually uniform in size and shape, in solid sheets, cords, or nests. In one case, we observed intraductal proliferations with central necrosis (comedocarcinoma).
Three solid carcinomas were characterized by tumor cells that were p63 negative; however, the comedocarcinoma showed a thin, discontinuous layer of flattened cells over the basal lamina, with p63 immunoexpression similar to that in normal mammary gland. These p63-positive cells also were identified by CK14 and ASMA, but to a lesser extent. p63-Negative cells stained for both cytokeratins and vimentin and were ASMA negative.
The other seven solid carcinomas presented p63 expression in a diffuse (n = 1) or peripheral (n = 6) manner. One carcinoma exhibited a simple proliferation of p63-positive cells (Fig. 4), and the remaining six were characterized by a dual cellular proliferation: p63-immunoreactive cells were generally located peripherally, and p63-negative cells were located centrally in the solid nests or cords. p63-positive cells also stained with CK14, ASMA (except in two cases), and vimentin. ASMA and vimentin decorated stromal myofibroblasts in all cases.
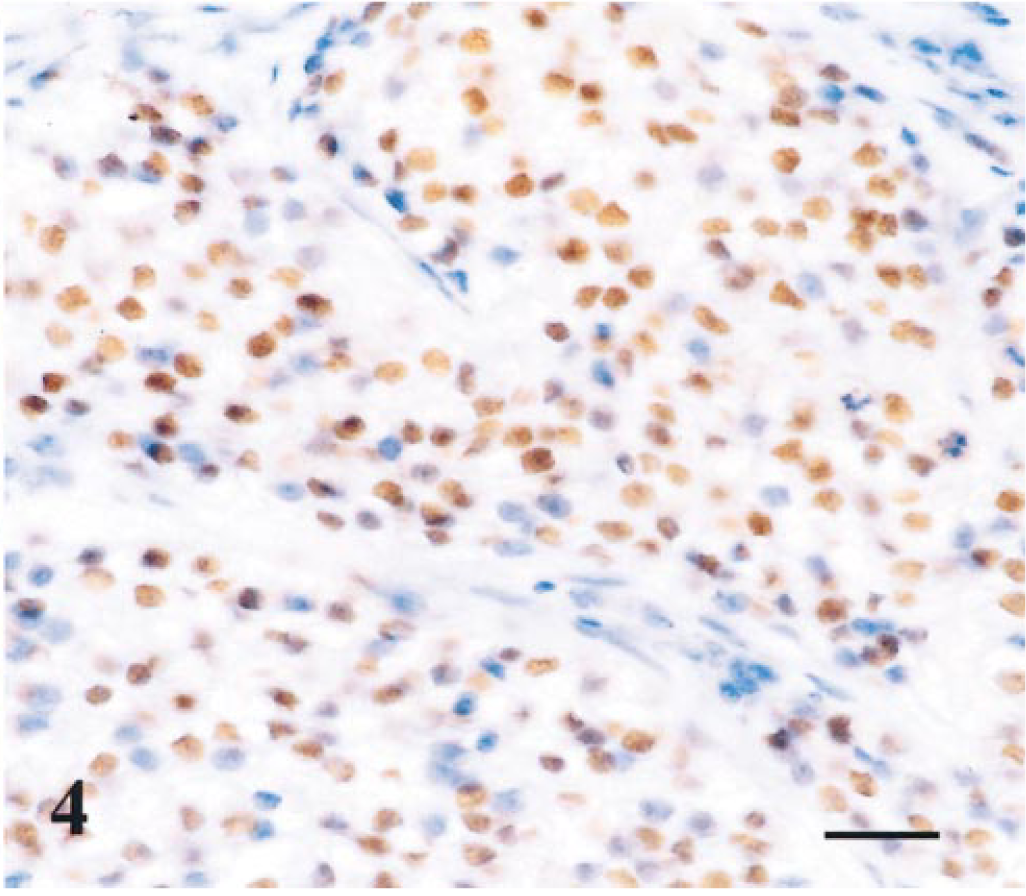
Mammary gland; simple solid carcinoma; dog. Neoplastic epithelial cells showing strong nuclear p63 immunoreactivity. Streptavidin–biotin complex method. Gill's hematoxylin counterstain. Bar = 18.5 µm
Spindle-cell carcinoma (n = 2)
These tumors were composed of spindle cells that were usually arranged in solid epithelial patterns, with the formation of bundles and nests.
Spindle cells showed a strong nuclear p63 immunoreactivity (++) and also stained for ASMA and vimentin. These cells also expressed both cytokeratins, but to a lesser extent.
Osteosarcoma (n = 1)
This tumor was characterized by the presence of polyhedral and pleomorphic neoplastic cells that were p63 negative. Tumor cells were positive only for vimentin.
Carcinosarcoma (n = 3)
These tumors were composed of both epithelial and mesenchymal malignant components. Sarcomatous components were consistently p63 negative. Two carcinosarcomas presented carcinomatous areas that were p63 negative; however, some p63-positive myoepithelial cell remnants were identified around the carcinomatous area. These cells were also positive for CK14 and ASMA, but to a lesser extent. The other carcinosarcoma was characterized by a squamous cell carcinoma, which showed strong p63 immunoreactivity (++) of basal and parabasal cells. The sarcomatous component was always vimentin positive and cytokeratin negative. The ASMA antibody immunoreacted with one of the three sarcomatous areas and also decorated myofibroblasts. The carcinomatous areas were ASMA negative and CK AE1/AE3 positive, and two out of three showed strong CK14 and moderate vimentin immunoreactivity.
Discussion
The p63 gene is expressed in the basal cells of several epithelial organs, and it has been suggested that the p63 gene plays a major role in the maintenance of the stem cell compartment in these organs. The role of p63 as a tumor suppressor protein, as in its p53 homologue, is still uncertain, but p63 germline mutations are associated with severe mammary gland developmental defects. 17 , 35 , 37 Barbareschi et al. (2001) reported that in human breast tissue, p63 is preferentially expressed by myoepithelial cells and proposed a close link between myoepithelial and stem or breast progenitor cells, supporting the hypothesis that the myoepithelial cell compartment includes stem cells.
To our knowledge, this is the first study evaluating p63 expression in canine mammary tissues, and our results demonstrated specificity of p63 monoclonal antibody for basal or myoepithelial cells in the normal and neoplastic canine mammary gland. p63 expression was found in 91.4% (74/81) of the samples evaluated. Normal mammary glands, mammary hyperplasias, and benign tumors showed 100% immunoreactivity, with p63 expression restricted to myoepithelial cell nuclei. Malignant tumors showed 72% of p63 expression: 100% of spindle-cell carcinomas, 89% of tubulopapillary, 70% of solid carcinomas, and 33% of carcinosarcomas immunoreacted with p63; osteosarcoma was not immunoreactive.
Myoepithelial cells are contractile elements showing a combined epithelial and smooth muscle phenotype. Among the several immunohistochemical markers available to detect myoepithelial cells, ASMA and CK14 are frequently used. Recently, calponin, a novel smooth muscle protein, was used in human and canine mammary tissues to identify myoepithelium. 4 , 5 , 7 The disadvantage of the muscle actin immunohistochemical markers is the additional staining of stromal myofibroblasts, which exhibit smooth muscle phenotypic properties. 5 , 7
In our series, the selected cases subjected to immunostaining on consecutive serial sections showed a comparable number of myoepithelial cells with similar staining intensity decorated by p63 and cytoplasmic myoepithelial cell markers: ASMA and CK14. However, p63 immunoreactivity was most specific, being restricted to myoepithelial cells only, whereas antibody to ASMA also stained stromal myofibroblasts and smooth muscle cells, which confirmed the previously reported results. 13
In the normal canine mammary gland and mammary hyperplasia, the p63 antibody reacted exclusively with myoepithelial cell nuclei, and the labeling patterns within the benign mammary tumors were to a large extent in accordance with those seen in the normal tissues.
In benign mixed tumors, the histogenesis of mesenchymal elements, such as bone or cartilage, remains controversial. Three hypotheses have been proposed: metaplasia from epithelial cells, metaplasia from stromal connective tissue, and metaplasia from basal epithelium (myoepithelium). 3 , 5 , 8 , 10 , 18 , 28–30 , 32 As in previous reports, 30 in this study, myoepithelial cells were classified into different morphologic types: suprabasal resting, proliferative myoepithelial, and spindle- and stellate-shaped myoepithelial cells located in the interstitial area. Immunohistochemistry for cytokeratins, ASMA, and vimentin revealed that suprabasal myoepithelial cells maintain their normal immunohistochemical characteristics, whereas interstitial myoepithelial cells tend to gradually decrease their expression of cytokeratins and ASMA, as already reported by other authors. 3 , 32 Our results reinforce the putative role of myoepithelium in mesenchymal metaplasia, which is supported by our observations of p63 immunoreactivity of some interstitial, modified, myoepithelial cells of complex and mixed tumors. Myoepithelial cells, by mechanisms not fully understood, can undergo metaplastic transformation to cartilage or bone, which is accompanied by molecular alterations that include the loss of expression of cytokeratins, ASMA, and p63 (as shown here) and increased vimentin expression, acquiring a mesenchymal phenotype (fibroblastic-like), which is in accordance with previous electron microscopy and immunohistochemistry studies. 3 , 9 , 29 An emerging concept is that the differentiated state of specialized cells exhibits a remarkable plasticity, and the idea of epithelial to mesenchymal transition of tumor cells has gradually emerged. An increasing availability of markers for the myoepithelial lineage will help to distinguish specific cell types in the microenvironment of tumors, unraveling some differentiation pathways. 15 , 22
In malignant mammary tumors, p63 immunoexpression revealed a myoepithelial phenotype not always identified by routine histopathology, as in previous reports using different myoepithelial markers. 3 , 10 In tubulopapillary carcinomas, p63 antibody allowed the identification of interstitial myoepithelial cells occupying the papillary core area. In addition, in solid carcinomas, p63 immunoreactivity revealed a major component of myoepithelial cells in 7 of 10 carcinomas: one of seven was a simple carcinoma, and the remaining six were reclassified as complex carcinomas, characterized by a dual immunoreactivity with p63-negative cell nests bordered by p63-positive cells.
Spindle-cell carcinomas showed nuclear p63 immunoreactivity, which confirms the myoepithelial nature of these neoplasms. In addition, these tumors were also positive for ASMA and vimentin. CK14 was not a reliable marker, showing faint immunoreactivity in these carcinomas. On the other hand, the osteosarcoma reacted exclusively with vimentin, an observation consistent with its mesenchymal origin.
As expected, sarcomatous components of carcinosarcomas were devoid of p63 and cytokeratin immunostaining and were strongly positive to vimentin. The carcinomatous areas presented differences due to the different tumor types involved: the squamous cell carcinoma showed strong p63 immunoreactivity with a basal cell distribution, as in results obtained in human squamous cell carcinomas, where p63 seems to be a valuable diagnostic marker of anaplasia of keratinocyte tumors. 25 p63 also identified myoepithelial cell remnants, which were not clearly identified by classic myoepithelial markers: CK14 also stained luminal epithelium and ASMA immunoreacted with myofibroblasts.
In the current series, p63 identified myoepithelial cells without any concurrent staining of stromal myofibroblasts, which is especially useful in those cases where it is difficult to distinguish true myoepithelial cells from stromal myofibroblasts lying just beneath the basement membrane. In addition to its possible diagnostic value as a myoepithelial cell marker, in view of the postulated functional role of p63 in normal breast development and maintenance of epithelial stem cell population, 17 , 24 , 37 the presence of p63 protein that is exclusive to canine myoepithelium might support a close link between myoepithelial and stem cells. However, further studies are needed to assess this hypothesis and the biologic relevance of p63 protein in canine mammary tissues.
In conclusion, our study reveals that p63 is a sensitive and highly specific marker of basal or myoepithelial cells in canine mammary tissues, being a reliable nuclear marker of these cells in normal and neoplastic mammary lesions. A better identification of cell-type origins is essential to clarify the classification of canine mammary tumors, and the authors propose p63 as an additional marker in immunohistochemical panels defining a myoepithelial histogenesis in these tumors.
Footnotes
Acknowledgements
We thank Mrs. Lígia Bento for expert technical assistance. This work was supported by the Institute for Agrarians and Agro-Food Sciences and Technologies, University of Trás os Montes e Alto Douro, Vila Real, Portugal.
