Abstract
The aim of this retrospective study (2004–2011) was to examine mammary tumors and tumor-like lesions in 24 pet rabbits by histopathology and immunohistochemistry. Rabbits were aged 2 to 8 years. Seventeen were female and 7 female-spayed. Diagnosed tumor-like lesions were lobular hyperplasia (2 rabbits) and multiple cysts (10 rabbits). Tumors included cystadenoma (7 tumors; 3 rabbits), intraductal papilloma (2 tumors; 1 rabbit), intraductal papillary carcinoma (1 tumor), adenocarcinoma (14 tumors; 13 rabbits), adenosquamous carcinoma (2 tumors; 2 rabbits), and matrix-producing carcinoma (1 tumor). The most frequently diagnosed lesion was invasive carcinoma (n = 17). Ten rabbits had several lesions. Immunohistochemistry for calponin and p63 showed that the diagnosed tumor-like lesions, benign tumors, and noninvasive carcinoma had a peripheral myoepithelial layer that was lacking in the invasive carcinomas. In 13 of 14 (93%) of the invasive carcinomas, however, there were variable numbers of calponin- and/or p63-immunopositive cells ranging from 0.1% to 40% with morphological features of either retained nonneoplastic myoepithelial cells or neoplastic epithelial cells with a myoepithelial differentiation. Tumor recurrence was reported in the rabbit with the matrix-producing carcinoma and in 3 rabbits with mammary adenocarcinomas displaying ≥20 mitotic figures in 10 high-power fields and high numbers of neoplastic cells with a myoepithelial differentiation (19%–39%). The rabbit with the matrix-producing mammary carcinoma developed cutaneous metastases confirmed by histopathology. This study shows that different types of mammary tumor-like lesions and tumors can occur in pet rabbits.
The most common tumor of the rabbit (Oryctolagus cuniculus) is the uterine adenocarcinoma, 20 whereas mammary tumors and tumor-like lesions are rarely reported in pet and laboratory rabbits. In a study of uterine disorders in 59 rabbits, the concurrent presence of a uterine and a mammary gland adenocarcinoma was observed in 4 rabbits, 2 of which had endometrial hyperplasia as well. 28 In another investigation of 47 rabbits with uterine disorders, mammary gland disease including cystic lesions was diagnosed in 15 of these rabbits by physical examination. 24 Histopathological features of neoplastic and nonneoplastic proliferative lesions of the mammary gland of rabbits are described in only a few publications that relate to laboratory rabbits. 11,26
In a breeding colony of rabbits, most invasive and noninvasive neoplasms developed in mammary glands with preexisting cystic disease. 11 A cystic mammary adenocarcinoma was diagnosed in a 44-month-old female nulliparous New Zealand White laboratory rabbit with concurrent mammary hyperplasia and dysplasia and elevated prolactin levels due to a pituitary adenoma. 26 To the authors’ knowledge, a detailed histopathological and immunohistochemical characterization of a case series of rabbit mammary tumors and tumor-like lesions has not been reported so far. The aim of this retrospective study was to characterize mammary tumors and tumor-like lesions of 24 pet rabbits by histopathology and immunohistochemistry.
Materials and Methods
Materials
This retrospective study encompasses all mammary tumors and tumor-like lesions diagnosed in pet rabbits (n = 24) between July 2004 and September 2011 in the Institute of Pathology, Faculty of Veterinary Medicine, University of Leipzig, Germany. From these excisional biopsy submissions, the reported clinical histories, hematoxylin-eosin (HE)–stained slides, and formalin-fixed, paraffin-embedded (FFPE) tissues were available.
Histochemical Stains
Applied histochemical stains were a periodic acid–Schiff (PAS) reaction (Nos. 1–24), an Alcian blue stain (No. 5), a Safranin O stain (No. 5), and a Papanicolaou stain (Nos. 10, 11).
Characterization of Mammary Gland Carcinomas
Carcinomas with different growth patterns and without significant squamous differentiation were classified as adenocarcinomas, 2,11 whereas carcinomas with squamous differentiation in more than 25% of the tumor area were consistent with adenosquamous carcinomas. 13,16 Tissue invasion was scored as – (lack of invasion) and +, ++, or +++ (mild, moderate, or marked tissue invasion, respectively). Noninvasive carcinomas were characterized by the presence of a continuous peripheral myoepithelial layer that was lacking in invasive carcinomas. 25,29 Mild (+) or moderate (++) invasion was defined as the presence of nodular tumor masses and infiltration of neoplastic cells in the immediately adjacent (mildly invasive) or the more distant (moderately invasive) peritumoral soft tissue, respectively. Marked invasion (+++) was defined as diffuse infiltration of the mammary gland and adjacent soft tissue by neoplastic cells. Moreover, it was evaluated if tumor cell emboli were absent (–) or were observed in vessels (V) or lymphatics (L). Secretory activity and intratumoral necrosis were evaluated according to their distribution within the tumor (multifocal or focal) and according to the size of these areas in relation to the entire tumor mass examined. The following semiquantitative evaluation was applied: –, when necrosis or secretory activity was absent, and (+), +, ++, or +++, when necrosis or secretory activity was minimal (involving <10% of the tumor area), mild (encompassing 10%–39% of the tumor area), moderate (involving 40%–69% of the tumor area), or marked (encompassing ≥70% of the tumor area), respectively. The number of mitotic figures in 10 dry high-power fields (HPFs) (40× magnification) was counted by the use of an Olympus BX 41 standard laboratory microscope (Olympus, Hamburg, Germany) in randomly selected tumor areas without significant necrosis. According to the modified method of Elston and Ellis, 7 invasive mammary tumors were graded in the following histological grades: grade I, well differentiated; grade II, moderately differentiated; and grade III, poorly differentiated. This semiquantitative method analyzes the following 3 histomorphological features by the use of a scoring system (scores 1–3): the tumor area with a tubular growth pattern (>75%, score 1; 10%–75%, score 2; and <10%, score 3), the degree of nuclear pleomorphism (mild, score 1; moderate, score 2; and marked, score 3), and the number of mitotic figures in 10 HPFs counted under consideration of the field diameter of the microscope (scores 1–3). 6,7 For each tumor, 3 individual scores were obtained and added to a total score that determined the histological grade and indicated the tumor differentiation: scores 3 to 5, grade I; scores 6 to 7, grade II; and scores 8 to 9, grade III. 7
Immunohistochemistry
The performed immunohistochemistry is summarized in Table 1. Sections of FFPE tissues were dewaxed, rehydrated, and treated with 3% H2O2 in methanol for 30 minutes at room temperature (RT). For antigen retrieval, sections were heated in 10 mM citrate buffer (pH 6) at 95°C for 25 minutes and subsequently remained in the citrate buffer for 17.5 minutes. The following primary antibodies were used: anti–cytokeratin AE1/AE3, antivimentin, anticalponin, anti–smooth muscle actin, and anti-p63. Sections were incubated with the primary antibodies overnight at RT and rinsed thoroughly with Tris-buffered saline (TBS). Antibodies and the PAP-complex were diluted in TBS containing 1% bovine serum albumin (BSA). For the peroxidase-antiperoxidase (PAP) method, sections were incubated with rat anti–mouse immunoglobulin G (1:100; Dianova, Hamburg, Germany) for 30 minutes at RT and rinsed thoroughly with TBS prior to the application of mouse-PAP-complex (1:100; Dianova) for 30 minutes at RT. For the Histofine method, sections were incubated for 30 minutes at RT with Histofine (Nichirei Biosiences, Tokyo, Japan) followed by a washing step. The chromogen 3,3′-diaminobenzidine tetrahydrochloride (DAB) was used. Slides were counterstained with a Papanicolaou solution (Merck, Darmstadt, Germany). For the negative controls, a nonrelated monoclonal mouse immunoglobulin G antibody 12 was applied in the same dilution as the primary antibody. Internal positive controls were the epidermis and adnexal structures for cytokeratin, the fibrous connective tissue for vimentin, the epidermis and hair follicles for p63, and the smooth muscles surrounding the mammary gland ducts and the vascular tunica media for calponin. With regard to the 2 different methods (PAP method and Histofine) used for the detection of p63, no major differences in the numbers of immunolabeled cells were observed in most cases; only in 2 cases (No. 8: ductal papillomas; No. 17: intraductal papillary carcinoma) did the Histofine method show a higher sensitivity. Therefore, for the latter 2 tumors, the results obtained by the Histofine method are described in the Results section.
Immunohistochemistry Performed on Rabbit Mammary Tissue.
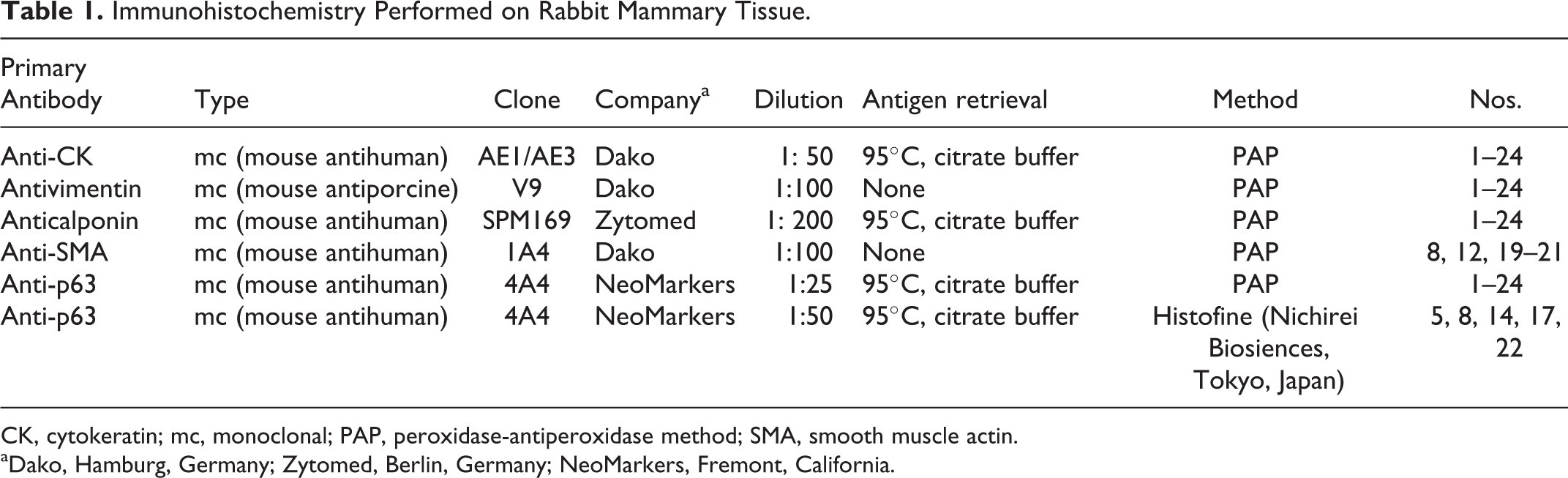
CK, cytokeratin; mc, monoclonal; PAP, peroxidase-antiperoxidase method; SMA, smooth muscle actin.
aDako, Hamburg, Germany; Zytomed, Berlin, Germany; NeoMarkers, Fremont, California.
Evaluation of the Immunohistochemical Signal
In normal and hyperplastic mammary glands as well as in benign tumors and the intraductal carcinoma, the immunostaining of luminal and basal epithelial cells was evaluated semiquantitatively (ie, <10%, 10%–49%, 50%–89%, and ≥90%). Only in cysts and cystadenomas was the immunostaining of the myoepithelium (ME) evaluated according to the immunolabeled proportion of the circumference of the cystic structures (ie, <10%, 10%–49%, 50%–89%, ≥90%) since the cell borders of the attenuated myoepithelial cells (MECs) were difficult to distinguish. In invasive carcinomas, the immunostaining of the neoplastic cells was investigated quantitatively by the use of image analysis. Depending on the size and the density of the neoplastic cells, digital images were taken from 5 or more representative areas of a tumor (S Plan Apo 40× objective, Olympus BH2 microscope) using an Olympus DP12 camera and the Soft Imaging System 5 (Olympus). For each applied antibody, the immunopositive and immunonegative tumor cells were counted with Adobe Photoshop, CS4 Extended (Adobe, San Jose, CA) by the use of the count tool analyzing at least 600 cells, and the percentage of immunopositive cells was calculated. Depending on the size and the density of the neoplastic cells within the digitized tumor areas, the number of counted cells varied between 603 and 997 cells. According to their histomorphological features, the calponin, p63, and SMA (only tumors Nos. 20a, 21) immunopositive cells were further divided into the 2 subcategories: retained nonneoplastic MECs and neoplastic cells with an MEC differentiation.
Questionnaire Study
In September 2011, a questionnaire was submitted to the referring veterinarians. With regard to the rabbits of this study, information about gravidities, the application of a long-term medication, the diagnosis of a uterine carcinoma, recurrence of the mammary lesion(s), and the development of metastasis was requested. In cases when the animal had died, the cause of spontaneous death or reason for euthanasia was asked.
Results
Animals
All rabbits of this study were pet rabbits (O. cuniculus domesticus). Seventeen rabbits were female and 7 (Nos. 4–7, 9, 15, 18) female-spayed. The age, known for 20 rabbits, ranged from 2 to 8 years. The mean age was 4.9 years. The breed of 7 rabbits was reported: 3 were dwarf rabbits (Nos. 13, 21, 24), 2 were lionhead rabbits (Nos. 3, 6), 1 was a dwarf lop rabbit (No. 18), and 1 was a lop rabbit (No. 11).
Tumor-like Lesions and Tumors of the Mammary Gland
The histopathological diagnoses are listed in Table 2 and include lobular hyperplasia (Nos. 5, 15), multiple cysts (Nos. 1, 4, 8, 13–17, 22, 24), 2 ductal papillomas (No. 8), 7 cystadenomas (Nos. 13, 16, 22), 1 intraductal carcinoma (No. 17), 14 adenocarcinomas (Nos. 2–4, 6, 7, 9, 12, 18–21, 23, 24), 2 adenosquamous carcinomas (Nos. 10, 11), and 1 matrix-producing carcinoma (No. 5). Besides the intraductal carcinoma, all carcinomas were invasive (17 of 18; 94%). In 10 rabbits (42%), the concurrent presence of 2 or more mammary lesions was observed, including cysts and lobular hyperplasia (No. 15), 2 to 3 cystadenomas and cysts (Nos. 13, 16, 22), 2 ductal papillomas and cysts (No. 8), 1 carcinoma and cysts (No. 4, 17, 24), 1 carcinoma and lobular hyperplasia (No. 5), and 2 carcinomas (No. 20). Secretory activity was observed in cysts (7 of 10 rabbits); lobular hyperplasia (2 of 2 rabbits), cystadenomas (2 of 3 rabbits), carcinomas (11 of 17 rabbits), and the normal mammary gland tissue (18 of 20 rabbits) were characterized by intraluminal proteinaceous material. Within secretory areas, the lining epithelial cells often contained intracytoplasmic clear vacuoles that had well-defined margins and stained PAS negative, indicative of lipid vacuoles. With regard to the mammary carcinomas with secretory activity (n = 11; Table 3), the numbers of vacuolated cells ranged between 2% and 50% of the tumor cells (semiquantitative evaluation).
Histopathological Diagnoses of Mammary Lesions in 24 Pet Rabbits.a
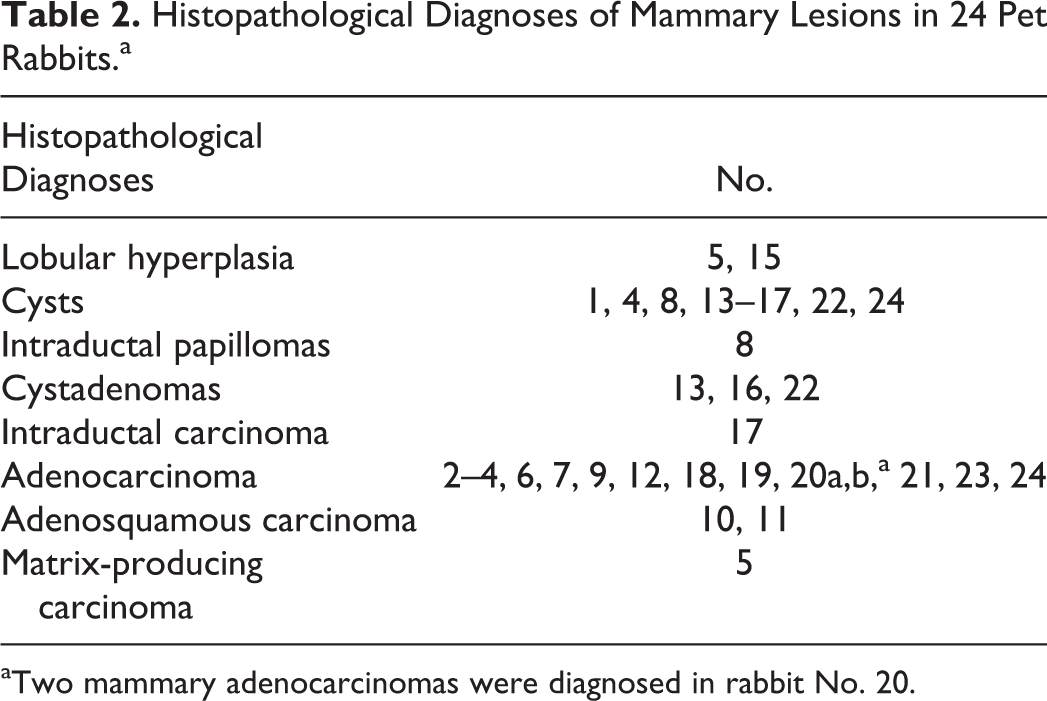
aTwo mammary adenocarcinomas were diagnosed in rabbit No. 20.
Microscopic Features of the Mammary Gland Carcinomas in Pet Rabbits.
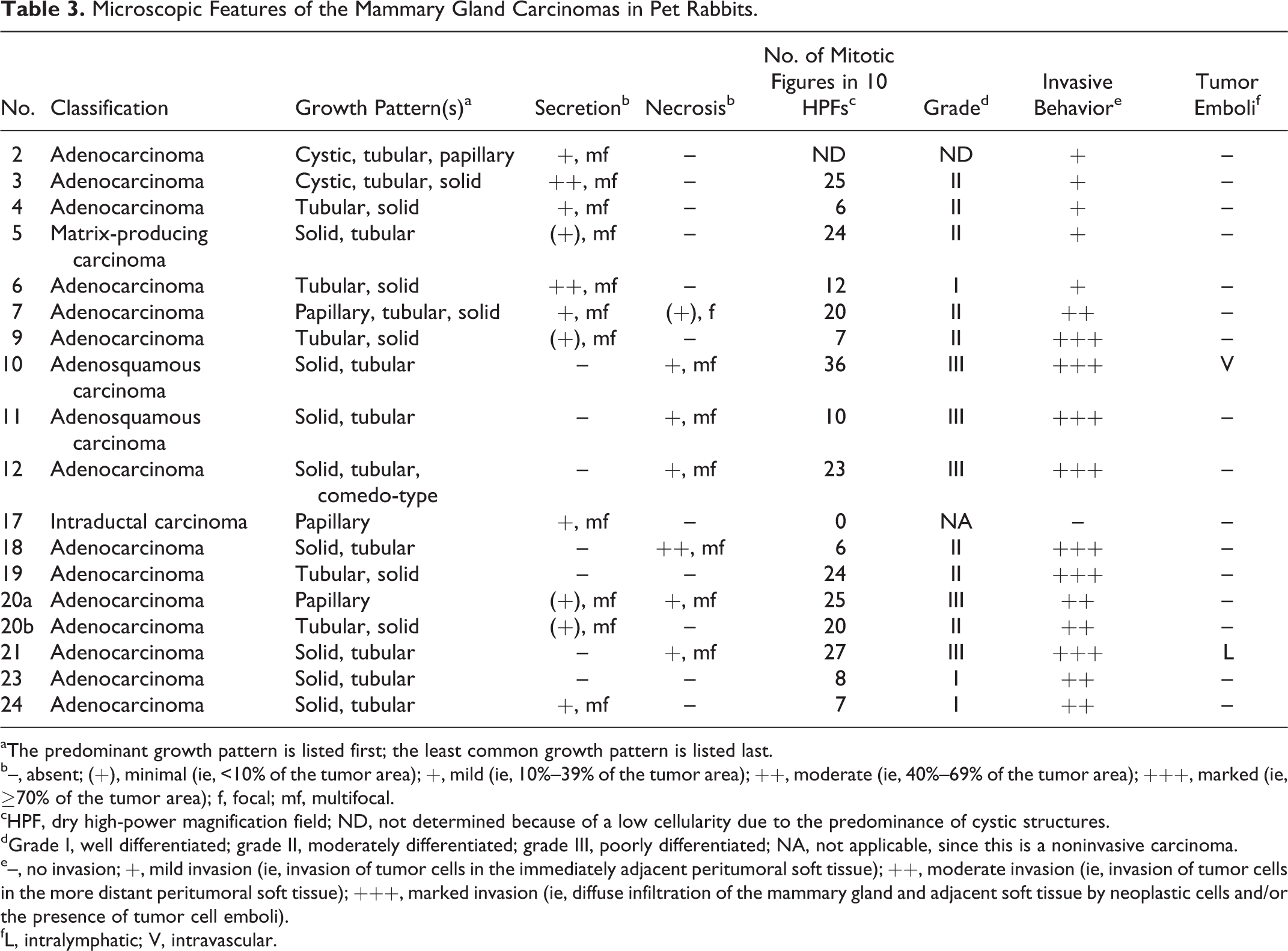
aThe predominant growth pattern is listed first; the least common growth pattern is listed last.
b–, absent; (+), minimal (ie, <10% of the tumor area); +, mild (ie, 10%–39% of the tumor area); ++, moderate (ie, 40%–69% of the tumor area); +++, marked (ie, ≥70% of the tumor area); f, focal; mf, multifocal.
cHPF, dry high-power magnification field; ND, not determined because of a low cellularity due to the predominance of cystic structures.
dGrade I, well differentiated; grade II, moderately differentiated; grade III, poorly differentiated; NA, not applicable, since this is a noninvasive carcinoma.
e–, no invasion; +, mild invasion (ie, invasion of tumor cells in the immediately adjacent peritumoral soft tissue); ++, moderate invasion (ie, invasion of tumor cells in the more distant peritumoral soft tissue); +++, marked invasion (ie, diffuse infiltration of the mammary gland and adjacent soft tissue by neoplastic cells and/or the presence of tumor cell emboli).
fL, intralymphatic; V, intravascular.
Immunohistochemical Characterization of the Normal Mammary Gland Tissue
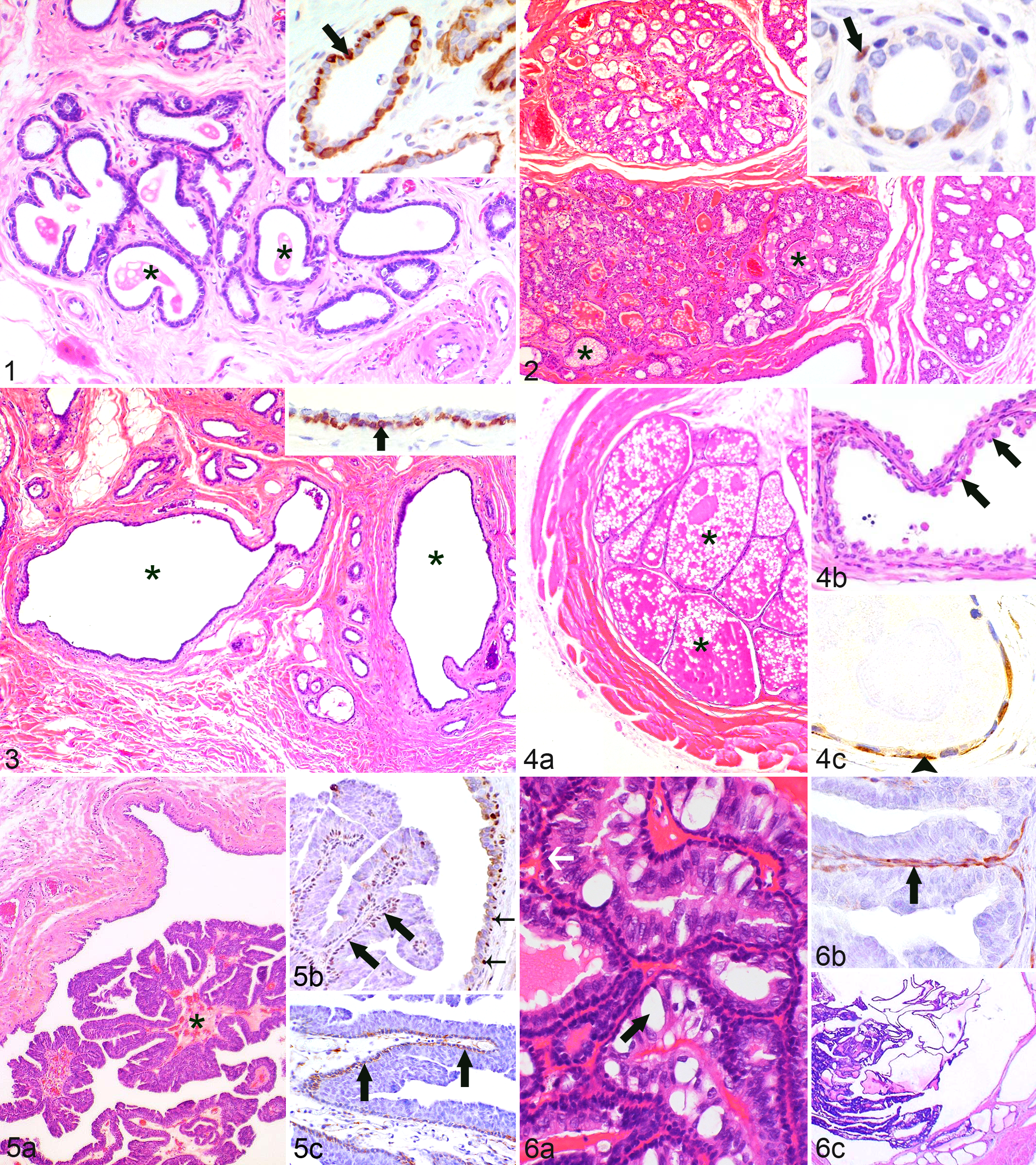
Within the terminal ductal lobular unit (TDLU) of the normal mammary tissue (observed in sections of Nos. 1–2, 4–8, 10–14, 16–18, 20–24), most luminal secretory and basal MECs stained diffusely immunopositive for cytokeratin AE1/AE3 (CK), whereas only the MEC layer was labeled with antibodies detecting calponin (Fig. 1), p63, vimentin, and SMA. Most MECs were immunopositive for calponin and in Nos. 8, 12, 20, and 21 also for SMA. In contrast, the number of p63 and vimentin immunolabeled MECs was variable, ranging from ≥90% (p63) and 50% to 89% (vimentin) to a lack of immunostaining.
Histopathological and Immunohistochemical Features of Tumor-like Lesions
The mammary tissue with lobular hyperplasia (Nos. 5, 15; Fig. 2) showed a similar immunostaining pattern for CK and calponin as the nonaltered mammary tissue of the TDLU. Immunostaining for p63 (No. 15) was observed in 10% to 49% of the MECs (Fig. 2) and could not be determined in No. 5, since hyperplastic areas were not present in the examined slide. Vimentin labeling was detected in 10% to 49% of the MECs in No. 5 and was negative in No. 15.
Cysts (Nos. 1, 4, 8, 13–17, 22, 24) were formed by the cystic dilation of elements of the TDLU and were lined by a double-layered epithelium (Fig. 3) that stained diffusely CK immunopositive. The basal epithelial layer was identified as an MEC layer. Within this layer, calponin immunostaining encompassed nearly the entire circumference (Fig. 3), and immunostaining for p63 or vimentin was variable, ranging from 50% to 89% (p63) or 10% to 49% (vimentin) of the circumference to a lack of immunostaining.
Histopathological and Immunohistochemical Features of Benign Tumors
All adenomas (Nos. 13, 16, 22) were cystadenomas. The variably sized cystic spaces were lined by a double-layered epithelium consisting of cuboidal to attenuated luminal cells with segmental apocrine metaplasia and attenuated basal MECs (Fig. 4). Both epithelial layers stained diffusely CK immunopositive and were vimentin negative; basally located cells were positive for calponin (Nos. 13, 16, 22; Fig. 4) and p63 (No. 13 and 1 tumor of No. 22). Calponin immunostaining encompassed 50%–89% to <10% of the circumference, whereas p63 immunostaining ranged from 10% to 49% of the circumference to a lack of immunostaining.
The 2 ductal papillomas (No. 8) were composed of intraluminal papillary proliferations that were lined by a double layer of well-differentiated epithelial cells (Fig. 5) and showed focal contiguity to the surrounding cystic dilated duct. Epithelial cells of the papilloma and the duct expressed CK in the majority of cells, whereas the basally located cells were identified as a myoepithelial cell layer. These were labeled by p63 (papilloma: ≥90%, duct: 10%–49%; Fig. 5), SMA (papilloma and duct: 50%–89%, Fig. 5), and calponin and vimentin (papilloma and duct: 10%–49%).
Histopathological and Immunohistochemical Features of Carcinomas
The histopathological features are listed in Table 3, and the results of the immunohistochemistry of the invasive carcinomas are in Table 4.
Immunhistochemical Evaluation of the Invasive Mammary Carcinomas in Pet Rabbits.a
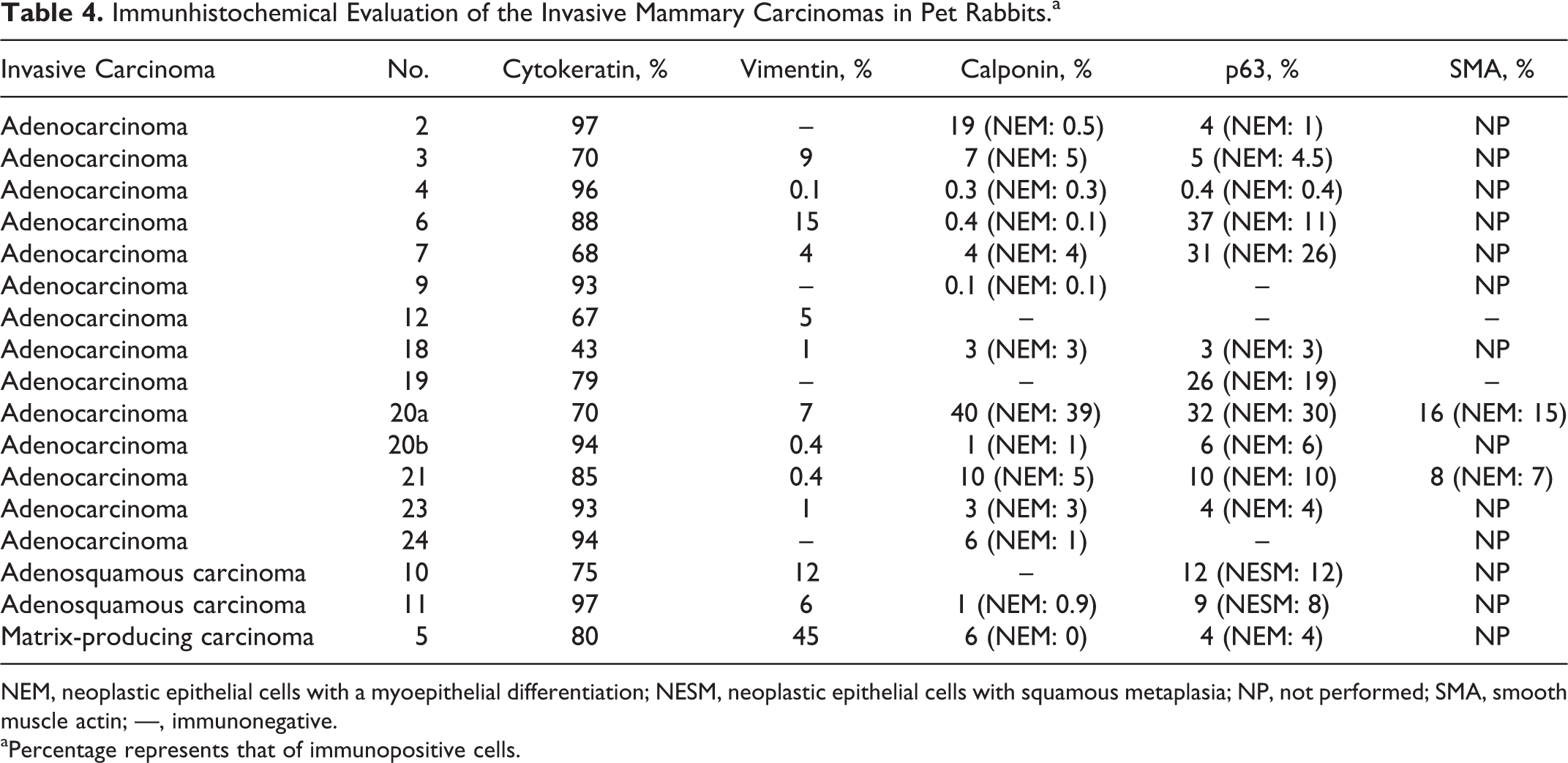
NEM, neoplastic epithelial cells with a myoepithelial differentiation; NESM, neoplastic epithelial cells with squamous metaplasia; NP, not performed; SMA, smooth muscle actin; —, immunonegative. aPercentage represents that of immunopositive cells.
The intraductal carcinoma (No. 17) showed a papillary growth pattern. It was a noninvasive (in situ) carcinoma contained within the lumen of a dilated duct and its branches and had multifocal contiguity to the epithelium of the surrounding duct. Its papillary formations were composed of a central thin fibrous connective tissue stalk and were lined by a basal layer of attenuated cells and 1 to 3 layers of cuboidal to columnar neoplastic epithelial cells with mild anisocytosis and anisokaryosis and multifocal mild secretory activity (Fig. 6). No mitotic figures were observed within either cell population in 10 dry HPFs. Calponin and p63 immunostaining in ≥90% of the basal epithelial cells was indicative of the presence of an intact peripheral myoepithelial layer (Fig. 6). Based on semiquantitative evaluation, 80% of the epithelial cells (encompassing luminal and myoepithelial cells) were CK and 10% vimentin immunopositive. The surrounding duct was lined by a bilayered epithelium, and ≥90% of the MECs were p63 and calponin immunopositive, indicative of an intact peripheral myoepithelial layer.
Adenocarcinomas
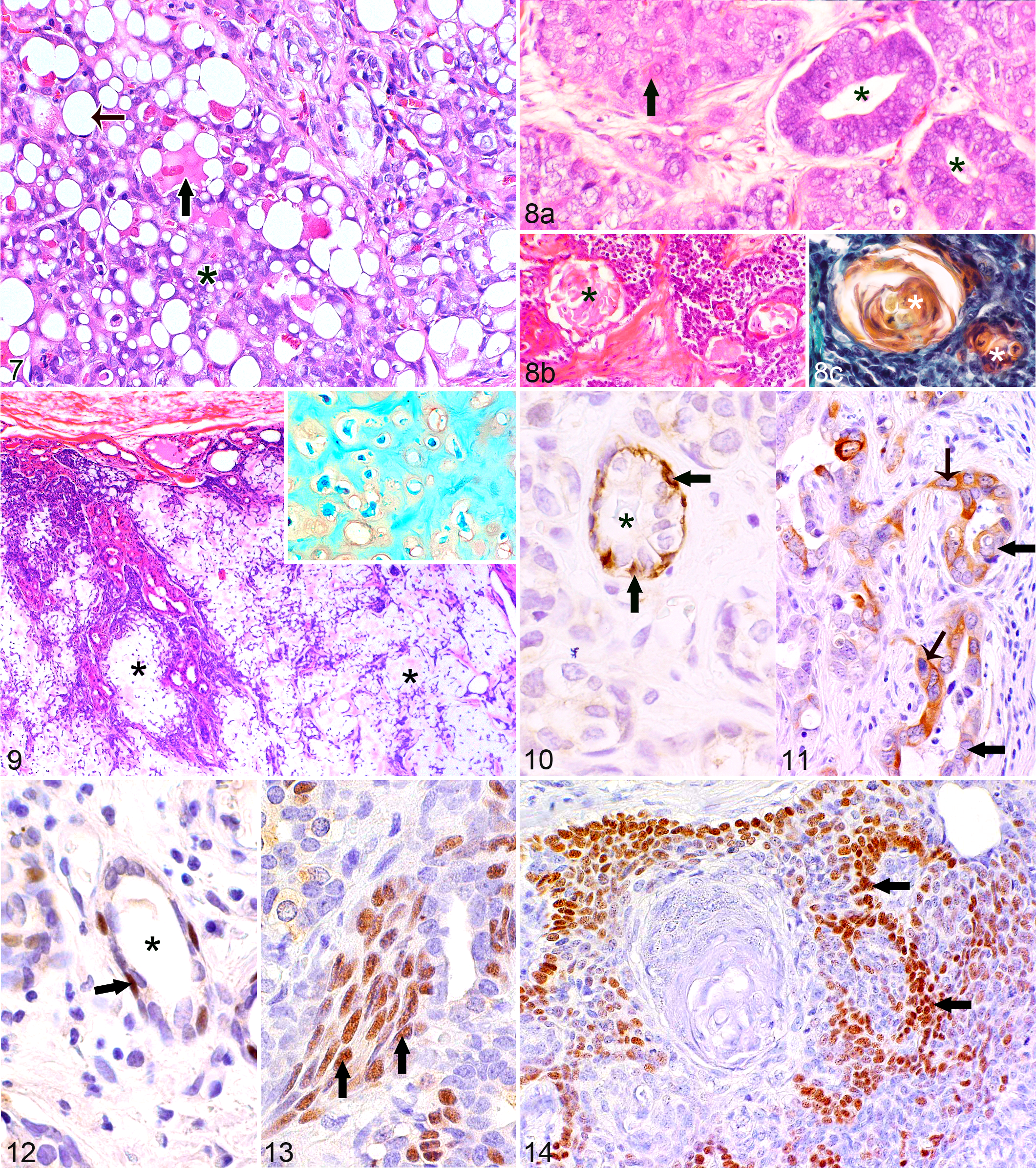
The observed growth patterns were tubular, solid, cystic, papillary, and comedo-type. Tumors with the combined presence of tubular and solid areas were most common (n = 9; 64%). In 4 tumors, 3 growth patterns were present. The adenocarcinoma with solely 1 growth pattern displayed a papillary morphology (No. 20a). The tubular growth pattern was characterized by the presence of tubular structures that were lined by 1 to 2 cell layers of neoplastic cells, had well-defined lumina, and often contained secretory material. Solid areas were composed of variably sized cellular aggregates. The cystic growth pattern was formed by the presence of variably sized cystic spaces lined by 1 to 4 layers of attenuated to cuboidal neoplastic epithelial cells. Some cystic spaces contained proteinaceous material. The papillary growth pattern was characterized by the presence of papillary proliferations that were lined by 2 to 6 layers of cuboidal to columnar neoplastic epithelial cells and were supported by small amounts of fibrovascular stroma. Comedo-type areas were composed of aggregates of tumor cells with central necrosis. Mild squamous differentiation in less than 5% of the tumor area was present in 3 adenocarcinomas (Nos. 2, 7, 20a). Secretory activity was observed in 64% of the tumors (n = 9; Fig. 7). Within secretory areas, these tumors contained variable numbers of vacuolated neoplastic cells ranging from 2% to 50% of the tumor cells (Fig. 7). The number of mitotic figures in 10 HPFs ranged from 6 to 27 mitoses per 10 dry HPFs. Moderately differentiated tumors (grade II) were most frequently observed (50%, n = 7). Tissue invasion was mild in 4, moderate in 5, and marked in 5 adenocarcinomas. Intralymphatic tumor cell emboli were present in 1 adenocarcinoma (No. 21). Most tumors (13 of 14; 93%) expressed CK in the majority of neoplastic cells (67%–97%); in 1 tumor (No. 18), 43% of the tumor cells were labeled. Vimentin immunostaining ranged from 0% to 15% of the neoplastic cells. The majority of carcinomas (10 of 14; 71%) contained minimal to moderate numbers of calponin-immunopositive cells (0.1%–40%) and p63-immunopositive cells (0.4%–37%). Three adenocarcinomas (Nos. 9, 19, 24) were immunopositive for only 1 marker: No. 19 for p63 (26% stained cells) and Nos. 9 and 24 for calponin (0.1% and 6% labeled cells, respectively). Lack of staining for calponin and p63 was observed in 1 tumor (No. 12). According to their histomorphological features, the p63 or calponin-immunopositive cells belonged to 2 different cell populations: retained nonneoplastic MECs (Figs. 10, 12) and neoplastic cells with a myoepithelial differentiation (Figs. 11, 13). The retained nonneoplastic MECs were located at the periphery of tubular structures and solid nests, were spindle shaped to ovoid, and had small ovoid nuclei (Figs. 10, 12). The neoplastic cells with a myoepithelial differentiation were polygonal with vesicular nuclei and showed moderate anisocytosis and anisokaryosis. They were intermingled with neoplastic epithelial cells within tubular and cystic structures, solid nests, and papillary formations; some tubular structures were formed entirely by neoplastic cells with a myoepithelial differentiation. In addition, they were often located at the infiltrative tumor margin (Figs. 11, 13). Immunostaining for SMA was observed in epithelial cells (No. 20: 16%, No. 21: 8%) resembling either retained nonneoplastic MECs or neoplastic cells. p63-immunopositive cells were also observed in the small areas with squamous differentiation (Nos. 7, 20a).
Adenosquamous carcinomas
These 2 tumors (Nos. 10, 11) were formed by poorly differentiated (grade III) nonsecretory mammary adenocarcinomas with solid and tubular areas and squamous differentiation in 40% (No. 10) and 30% (No. 11) of the tumor area, respectively (Fig. 8). Squamous differentiation was characterized by keratinization of individual cells or groups of cells, the formation of horn pearls, and the presence of cystic structures with intraluminal keratinized debris (squamous cysts) that was highlighted by a Papanicolaou stain (Fig. 8). Vascular tumor cell emboli were observed in tumor No. 10. The majority of neoplastic cells (No. 10: 75%, No. 11: 97%) were CK immunopositive, whereas vimentin immunolabeling was observed in 12% (No. 10) and 6% (No. 11) of the tumor cells. respectively. Both tumors contained p63-immunopositive cells (No. 10: 12%; No. 11: 9%) and 1 tumor (No. 11) minimal numbers of calponin-positive cells (1%). p63-positive cells were mainly located in areas with squamous differentiation (Fig. 14). Rare p63-labeled cells and the calponin-positive cells of tumor No. 11 showed histological features of either retained nonneoplastic MECs or tumor cells with a myoepithelial differentiation.
Matrix-producing carcinoma
This tumor (No. 5) was segmentally surrounded by compressed fibrous connective tissue and showed moderate tissue infiltration in the remaining areas. Neoplastic cells were polygonal or ovoid; arranged in nests, cords, and tubules; and displayed moderate anisocytosis and anisokaryosis. Peripheral areas of the tumor were highly cellular. Toward the central tumor area, there was a gradual increase in intercellular Alcian blue– and PAS-positive myxoid matrix associated with a decreased cellularity as well as an increased number of individualized neoplastic cells (Fig. 9). The myxoid matrix contained foci of cartilaginous metaplasia (Safranin O stain; Fig. 9). This grade II tumor displayed minimal secretory activity and 24 mitotic figures within 10 HPFs (counted in peripheral areas). CK was expressed in 80% and vimentin in 45% of the neoplastic cells. Small numbers of cells were calponin (6%) and p63 (4%) immunoreactive. These showed morphological features of retained nonneoplastic MECs or neoplastic cells with a myoepithelial differentiation and were mainly located at the tumor periphery.
Questionnaire Study
In 9 rabbits (Nos. 5, 6, 12–17, 19), a previous gravidity could be excluded with certainty, and 21 rabbits (1, 3–22) had definitively not received a long-term medication; for the remaining rabbits, this information was not available. The presence of a uterine carcinoma was diagnosed in 7 rabbits, excluded in 4 rabbits (Nos. 10, 21–23), and unknown for the remaining rabbits. Rabbits with a reported uterine carcinoma had a mammary adenocarcinoma and cysts (No. 4), mammary adenocarcinomas (Nos. 6, 9, 18), a matrix-producing mammary carcinoma and lobular hyperplasia (No. 5), ductal papillomas and cysts (No. 8), and mammary cysts and lobular hyperplasia (No. 15). Recurrence of a mammary gland carcinoma was reported in 4 rabbits (Nos. 5, 7, 19, 20). Notably, in 3 rabbits (Nos. 7, 19, 20) with reported recurrences, adenocarcinomas with a relatively high mitotic count (≥20 mitotic figures in 10 HPFs) and the highest numbers of neoplastic epithelial cells with a myoepithelial differentiation (19%–39%) were observed. The remaining rabbit (No. 5) with reported tumor recurrence had a matrix-producing mammary carcinoma. Tumor metastases were observed in 3 rabbits (Nos. 5, 8, 16). The rabbit with the matrix-producing mammary carcinoma (No. 5) was euthanized 10 months after mammary surgery due to tumor metastases. Reportedly, it had developed 30 to 40 cutaneous nodules on the back; histopathological examination of 2 of these lesions showed the presence of dermal and subcutaneous neoplasms with similar histomorphological features as the mammary tumor. Therefore, these tumors were regarded as metastases of the matrix-producing mammary carcinoma (No. 5). In addition, radiographs showed the presence of pulmonary metastases (No. 5) that may have developed from the mammary carcinoma or the uterine carcinoma. Rabbit No. 8 was euthanized 11 months after mammary surgery because pulmonary metastases were observed by radiography. Since in this rabbit, mammary ductal papillomas and cysts were diagnosed, the reported pulmonary metastases likely developed from the uterine carcinoma. Examined mammary tissue of rabbit No. 16 contained cystadenomas and cysts. This rabbit was euthanized 5 days after mammary surgery due to pulmonary metastases diagnosed by radiography; the metastases may have originated from the reported additional mammary lesions that were not investigated by histopathology; the presence of a uterine carcinoma was unknown. The following survival times were reported for the remaining rabbits with tumor-like lesions and/or benign tumors (Nos. 1, 13–15, 22): rabbit No. 1, which had mammary cysts, survived 4.5 years; the cause of death was unknown. The remaining rabbits were still alive at the end of this study; the time between mammary surgery and the end of this study ranged from 4 months and 10 days to 1 year and 5 months. The rabbit with the intraductal carcinoma and cysts (No. 17) was still alive at the end of the study; the time between mammary surgery and the end of this study was 9 months and 3 weeks. With regard to the rabbits with a mammary adenocarcinoma, information about the survival time and the cause of death was known for 8 rabbits (Nos. 3, 4, 6, 9, 18–20, 23). With regard to the 4 rabbits with a mammary adenocarcinoma and a uterine carcinoma (Nos. 4, 6, 9, 18), 2 died immediately after mammary surgery (Nos. 6, 9) and 2 were euthanized 6 and 7 days after mammary surgery due to a poor body condition (No. 4) or poor wound healing and paralysis (No. 18), respectively. The rabbit with the concurrent presence of 2 mammary adenocarcinomas was euthanized 6 months after mammary surgery due to infection with Encephalitozoon cuniculi. Rabbit No. 23 was still alive at the end of this study, 4 months after mammary surgery. Rabbit No. 11 had a mammary adenosquamous carcinoma and was euthanized 2 years and 2.5 months after mammary surgery due to a urinary bladder neoplasm. No follow-up information was available for rabbits Nos. 10 (adenosquamous carcinoma) and 21 (adenocarcinoma) that had grade III mammary carcinomas with the highest numbers of mitotic figures in 10 HPFs and intravascular or intralymphatic tumor cell emboli.
Discussion
This study shows the occurrence of different types of mammary tumor-like lesions as well as benign and malignant tumors in pet rabbits. In accordance with previously reported mammary tumors in laboratory rabbits, 11,20,26 all observed tumors were epithelial neoplasms. To the best of our knowledge, mesenchymal mammary tumors have not been published in rabbits so far.
Notably, the most frequently diagnosed mammary lesion was invasive carcinoma. Moreover, 10 rabbits (42%) showed the concurrent presence of several identical or different mammary tumor-like lesions and/or tumors, suggesting a predisposition of pet rabbits to develop multiple mammary lesions, a finding also frequently observed in dogs. 18 In laboratory rabbits, Greene 11 observed a progression of nonneoplastic mammary cysts into intracystic benign and malignant papillary epithelial tumors as well as invasive mammary adenocarcinomas. 11 A transition from cysts to neoplasia may occur in pet rabbits as well; mammary cysts were diagnosed in 38% of the rabbits in this study (n = 9) and occurred in 5, together with benign or malignant tumors.
All tumor-like lesions (lobular hyperplasia, cysts) and benign tumors (ductal papilloma, adenoma) as well as the vast majority of malignant tumors (intraductal carcinoma, adenocarcinoma, adenosquamous carcinoma) displayed histopathological features similar or identical to those described in the World Health Organization (WHO) classification of canine and feline mammary tumors 19 and/or the proposed classification of canine mammary tumors by Goldschmidt et al. 10 Only 1 carcinoma differed from the carcinoma subtypes reported in dogs, 18,19 cats, 18,19 laboratory rats, 13,16 or laboratory mice, 2,16 but showed histopathological features diagnostic for a matrix-producing breast carcinoma of human beings. 5 The characteristic feature of a matrix-producing breast carcinoma is the immediate transition between an invasive carcinoma and chrondromyxoid or osseous matrix and the lack of an interjacent population of spindle cells. 5 This carcinoma is distinguished from the carcinoma mixed-type described in dogs due to the lack of a spindle cell population embedded in myxomatous stroma indicative of MECs. 10 All but 1 mammary adenocarcinoma showed 2 to 3 different growth patterns; notably the combined presence of solid and tubular areas was observed most frequently (64% of the invasive carcinomas). In the WHO classification, 19 feline and canine (simple) mammary carcinomas are subclassified into tubulopapillary, solid, anaplastic (dogs and cats), and cribriform (only cats) carcinomas; in dogs, the recognition of these subtypes has prognostic significance. 19 For mammary carcinomas that display a heterogeneous histomorphology, Misdorp et al 19 recommend combined diagnoses, in which the prevailing growth pattern is listed first. In this study, the diagnosis of “adenocarcinoma” without further subclassification was used due to the predominance of a heterogeneous histomorphology and the lack of a confirmed prognostic relevance of (a) particular growth pattern(s) in pet rabbits. The combined presence of several growth patterns is also described in mammary carcinomas of mice 2 and laboratory rabbits, 11 which were diagnosed as adenocarcinomas without subclassification. Invasive carcinomas were graded according to the method of Elston and Ellis. 7 This method is mainly applied to human breast cancer 27 but has also been used for canine 10,17 and feline 3,17 mammary carcinomas. It is also considered valid for pet rabbits, since the assessed histomorphological features appear to be species-independent indicators for the degree of differentiation of a mammary carcinoma. In humans, this grading system has prognostic significance. 6,7 The prognostic significance of the predominance of poorly and moderately differentiated mammary carcinomas in pet rabbits has to be examined in further studies, since in this investigation, the follow-up information was often incomplete, the case numbers were relatively small, and 5 rabbits with invasive mammary carcinomas also had a uterine carcinoma.
The neoplastic and nonneoplastic lesions of the mammary gland in pet rabbits were further examined by immunohistochemistry. The retained normal mammary tissue of the TDLU displayed an identical staining pattern as observed in the human 23 and canine mammary gland 8,9 (ie, CK AE1/AE3 stained luminal secretory epithelial cells as well as basal MECs, whereas the basal MECs were immunopositive for calponin, p63, SMA, and vimentin). This confirms that—also in rabbits—calponin, p63, and SMA can be used as MEC markers. Often, more MECs were detected by immunostaining for calponin than by p63 immunolabeling, suggesting that calponin has a higher sensitivity for the detection of MECs in rabbit mammary tissue. Since archived tissue samples were investigated, however, it cannot be ruled out that differences in the sample preservation and/or the length of formalin fixation influenced the results of the immunostaining. By the use of several MEC markers, differences between the numbers of immunostained MECs have also been observed in human breast tissue. 29 Thus, the simultaneous application of a cytoplasmic marker (eg, calponin) and the nuclear marker p63 is recommended. 29 As reported for duct papillomas of dogs 4,10 and human beings, 6,27 the intraluminal papillary proliferations of these rabbit tumors were lined by a double-layered epithelium consisting of a luminal secretory epithelium and basal MECs. A similar double-layered epithelium surrounded the cystic structures in rabbit mammary adenomas. Likewise, tubular formations of canine (simple) mammary adenomas 4 and human tubular breast adenomas 27 are lined by such a double-layered epithelium. In humans and dogs, MEC markers are used to distinguish noninvasive (in situ) and invasive mammary carcinomas. 1,25,29 The detection of a peripheral MEC layer in the noninvasive carcinoma (intraductal papillary carcinoma) but not in the invasive carcinomas suggests that for this purpose, MEC markers could also be applied in pet rabbits.
Based on the evaluation of routine stained (HE-stained) sections, all invasive adenocarcinomas resembled simple carcinomas, 10,19 but immunohistochemically, most contained minimal to moderate numbers of cells immunopositive for the applied MEC markers. Whereas some of these cells displayed morphological features of retained nonneoplastic MECs, others were consistent with neoplastic epithelial cells showing an MEC differentiation. Similarly, in canine and feline mammary carcinomas with histomorphological features of simple carcinomas, retained nonneoplastic MECs and tumor cells with an MEC differentiation were detected by immunohistochemistry. 17 Furthermore, these MEC populations are also described in human breast carcinomas. 1,15,17,21 The simultaneous presence of neoplastic epithelial cells and MECs is also observed in the canine complex (type) mammary carcinomas 10,18,19 and the canine mammary carcinoma and malignant myoepithelioma. 10 According to Goldschmidt et al, 10 a complex-type carcinoma is composed of malignant epithelial cells and benign MECs that can be recognized as bundles of interstitial spindle-shaped cells surrounded by a myxoid matrix. 10 Therefore, the rabbit carcinomas with MECs were not diagnosed as complex carcinomas, since their histopathological features resembled (simple) carcinomas, they lacked proliferated interstitial spindle cells embedded in a myxomatous matrix, the MEC component was detected solely by immunohistochemistry, and varying percentages of intratumoral MECs displayed features of malignancy. The carcinoma and malignant myoepithelioma is a rare canine mammary tumor composed of malignant epithelial cells and malignant MECs; the 2 cell populations are located separately from each other and show different histomorphological features (ie, the malignant epithelial cells form tubules and solid aggregates, whereas the malignant MECs are located in the interstitium embedded in a mild amount of myxoid matrix). 10 This tumor differs from the rabbit mammary adenocarcinomas with MECs of this study since in the latter, malignant epithelial cells and MECs were often intermingled with each other in tubules, solid nests, and papillary formations; some tubular structures were entirely composed of malignant MECs; the simultaneous presence of retained and malignant MECs was a common finding; and MECs were not surrounded by a myxoid matrix.
p63 is expressed in not only MECs but also areas of squamous epithelial cell differentiation, 14 a finding observed in the 2 rabbit mammary adenosquamous carcinomas.
This questionnaire study showed that pet rabbits with mammary lesions should be examined for the presence of uterine carcinoma, a tumor diagnosed in 29% of the rabbits of this study. There was no association between a long-term medication and the development of mammary tumors or tumor-like lesions in pet rabbits. Laboratory rabbits, which received a long-term application of cyclosporine A, developed mammary hyperplasia and hyperprolactinemia. 22 In this study, a prognostic evaluation was limited by the small numbers of particular mammary lesions, the often incomplete follow-up information, and the different time intervals between the mammary surgery and the questionnaire study for individual rabbits. Notably, mammary adenocarcinomas in the 3 rabbits with reported recurrence displayed the highest numbers of neoplastic cells with an MEC differentiation counted in this study (19%–39%). In human breast carcinomas, MEC differentiation may reflect a shift to a more aggressive behavior, 1 and the aforementioned rabbit tumors had a high mitotic activity, were grade 2 or 3, and showed moderate to marked tissue invasion. This observation, however, has to be treated with caution due to the restricted case numbers and thus has to be investigated further in future studies that have collected higher case numbers.
In conclusion, this study identified types of mammary tumors and tumor-like lesions that can occur in pet rabbits and described their histopathological and immunohistochemical features.
Footnotes
Acknowledgements
We thank Ms M. Wipplinger for performing the immunohistochemistry; the histology laboratory, Institute of Pathology, Faculty of Veterinary Medicine, University of Leipzig for excellent technical support; and the referring veterinarians for providing the clinical information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
