Abstract
A 12-year-old intact female dog was submitted to surgery to remove a well-circumscribed mass located near the left inguinal mammary gland. At histological examination, the mass was unencapsulated and composed by lobules of fat cells and scattered isles of cartilaginous tissue. Chondroblasts and chondrocytes showed moderate signs of atypia and often were located singularly or in small clusters within the stroma of the neoplasm. Immunohistochemical analysis revealed that cells were vimentin and S-100 positive, whereas no immunoreactivity was showed for cytokeratin, cytokeratin 5/6, cytokeratin 14, and P63. A diagnosis of chondrolipoma was made based on microscopic findings.
Tumors of the adipose tissue are among some of the most common soft tissue tumors encountered by pathologists. Most of these entities are lipomas that pose no diagnostic difficulty; however, the diagnosis of some tumor variants could be more challenging. Lipomatous lesions can show a broad morphological spectrum, and a 2011 study focusing on these tumors led to the delineation of new tumor variants often resembling each other (mainly chondroid lipoma and myxoid liposarcoma) as well as the introduction of new concepts regarding the interactions of molecular genetics and pathology. 3 In the World Health Organization (WHO) classification of human tumors of soft tissue and bone, there are 14 types of benign tumors of adipose tissue, including specific variants such as fibrolipoma, myxolipoma, angiolipoma, hibernoma, chondroid lipoma, and chondrolipoma. 8 Some cases of chondrolipoma with breast location have been reported in human patients.1,2,4,5 In dogs, only 2 previous cases of chondrolipoma have been reported: the first tumor was located between the peritoneum and abdominal muscles, 12 and the second tumor was located in the intra-pelvic space. 9 In the current report, a case of canine chondrolipoma with mammary location is described.
A 12-year-old intact female Golden Retriever dog was presented for clinical examination of a well-circumscribed mass located near the left inguinal mammary gland. The mass measured 20 mm × 20 mm × 15 mm, was firm and elastic at palpation, and did not appear to be fixed to the underlying tissues. Due to the mammary location of the neoplasm, mastectomy of the whole left chain was performed by the practitioners.
On gross examination, the mass was white-yellow and gritty on cut surface. The surgical samples were fixed in 10% neutral buffered formalin and processed by routine methods for histological investigation. Four µm–thick sections were cut and stained with hematoxylin and eosin for microscopic examination. Further tissue sections were prepared for histochemical (Masson trichrome stain, Gomori, stain for reticular fibers, periodic acid–Schiff ) and immunohistochemical staining. Immunohistochemistry was performed by the biotin–streptavidin–peroxidase method with primary antibodies against human cytokeratin (CK), CK 5/6, CK 14, vimentin, p63, and S-100 protein. The chromogen was 3,3’-diaminobenzidine a for all antibodies (10 min at room temperature). Canine skin samples were used as control tissues. Immunohistochemical protocols and results are described in Table 1.
Primary antibodies for immunohistochemistry and reactivity of a mammary chondrolipoma.*

CK = cytokeratin; MM = mouse monoclonal; RP = rabbit polyclonal; MW = microwave; samples were microwaved in 0.01 M citrate buffer (pH 6.0) at 650 W for 30 min and at 350 W for 15 min.
Santa Cruz Biotechnology Inc., Santa Cruz, CA; Dako North America Inc., Carpinteria, CA; Thermo Fisher Scientific Inc., Waltham, MA; Novocastra Laboratories Ltd., Newcastle upon Tyne, UK.
Histological examination revealed a discrete, unencapsulated, nodular mass. The mass was composed by lobules of adipose tissue separated and surrounded by connective tissue septa. Adipocytes showed anisocytosis. Mature adipocytes and lipoblasts showed no cellular atypia; nuclei were uniform and hyperchromatic. Variably sized foci of chondrification within a wide basophilic matrix were also present within the adipose tissue (Fig. 1). These foci were composed of chondrocytes and chondroblasts showing pleomorphic features such as anisocytosis, anisokaryosis, and nuclear atypia. Nucleoli were sometimes large and prominent with angular borders (Fig. 1 inset). Within foci of chondrification, scattered vacuolated cells were identifiable. Multivacuolated lipoblasts, the hallmark of chondroid lipoma, were absent. Solitary or small groups of chondrocytes were also identifiable among the connective stroma near groups of lipoblasts (Fig. 2). Due to these microscopic features, a diagnosis of chondrolipoma was made. The histological examination did not reveal any mammary tissue around the lesion.
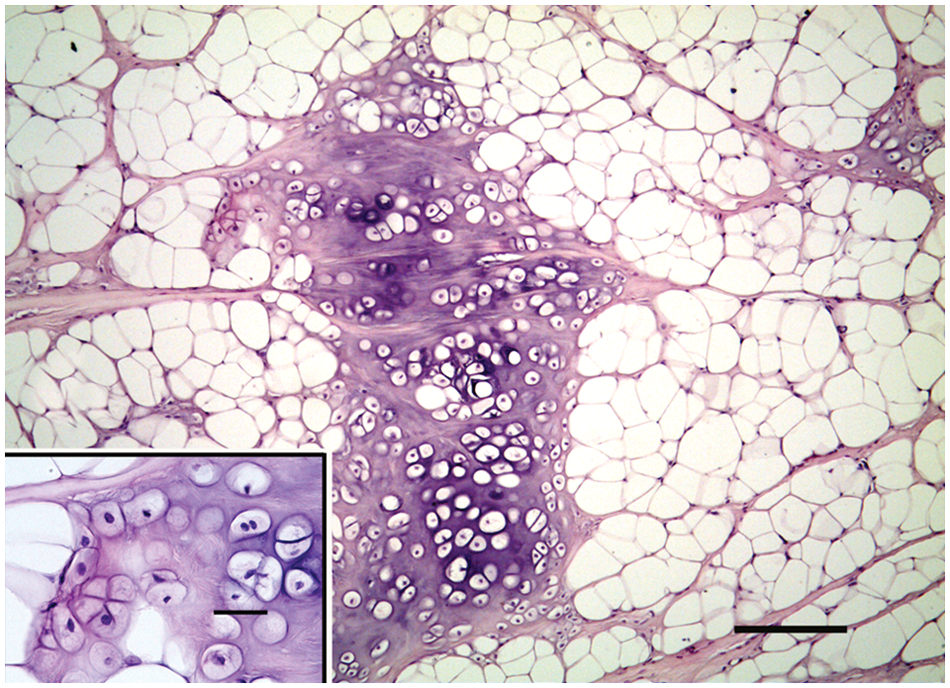
Mammary gland; dog; chondrolipoma. A large island of cartilage within lobules of neoplastic adipose tissue. Hematoxylin and eosin (HE). Bar = 200 µm. Inset: higher magnification of nuclear atypia of cartilaginous cells. HE. Bar = 20 µm.
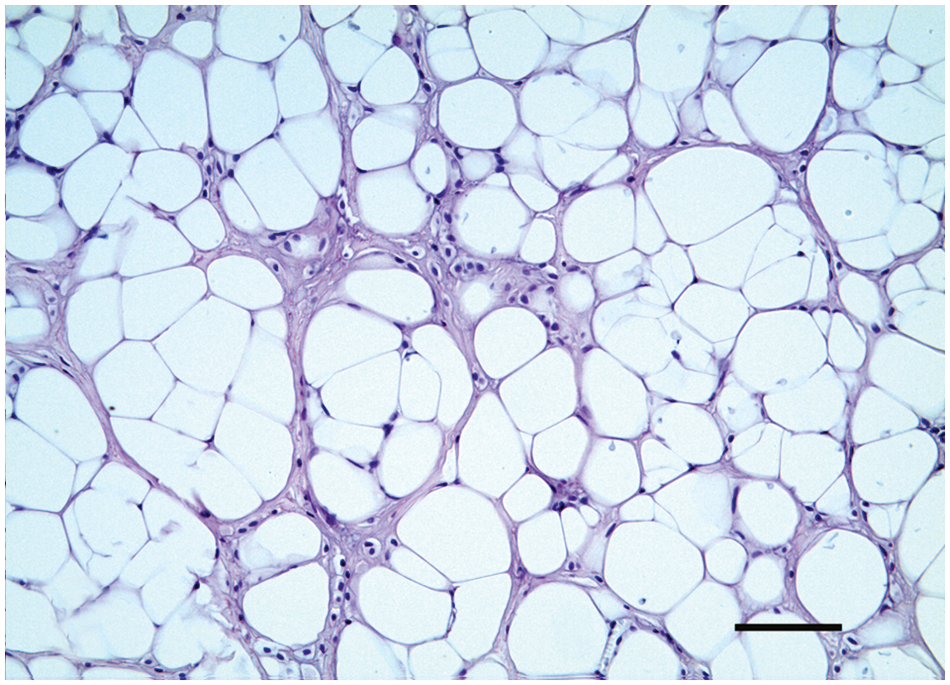
Mammary gland; dog; chondrolipoma. Single or small groups of chondroblasts located within the tumor connective tissue stroma. Hematoxylin and eosin. Bar = 100 µm.
Immunohistochemistry showed a strong cytoplasmic reaction of fat cells, chondrocytes, and chondroblasts to vimentin antibody. These same cells failed to stain for CK, CK 5/6, CK 14, and p63. The S-100 protein was located only in the cytoplasm and nucleus of cartilaginous cells (Fig. 4). Collagen fibers located in the connective stroma were strongly positive to the Masson trichrome stain, while cartilage matrix was mildly positive (Fig. 3). Periodic acid–Schiff stain showed glycogen in the cartilaginous cells. Gomori and reticular fiber stains showed numerous reticular fibers of varying thickness supporting the adipocytes.
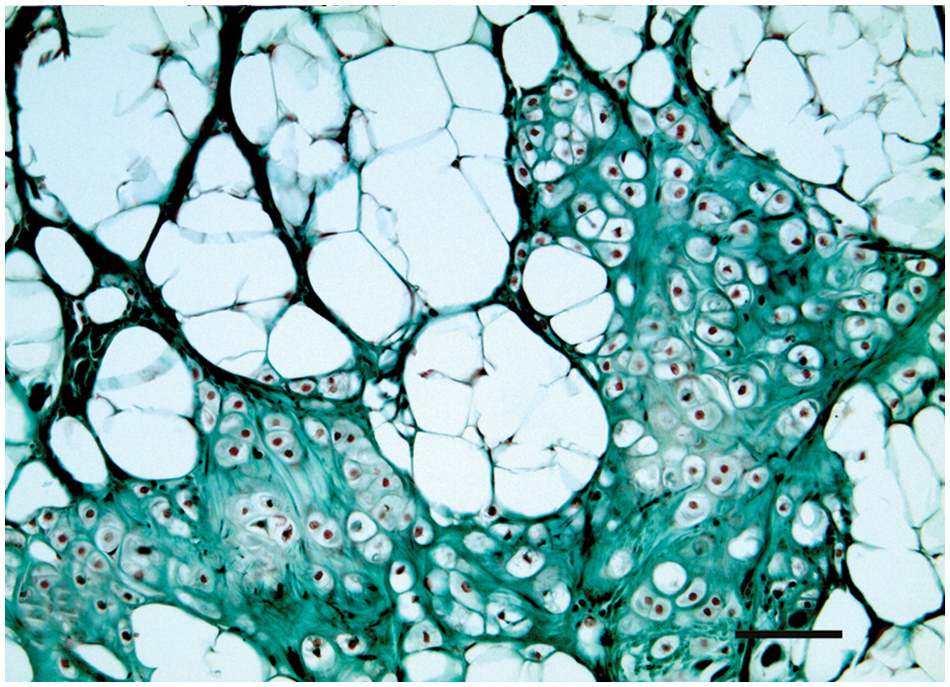
Mammary gland; dog; chondrolipoma. Strong positivity of connective tissue septa and moderate positivity of cartilage matrix to Masson trichrome stain. Bar 100 = µm.
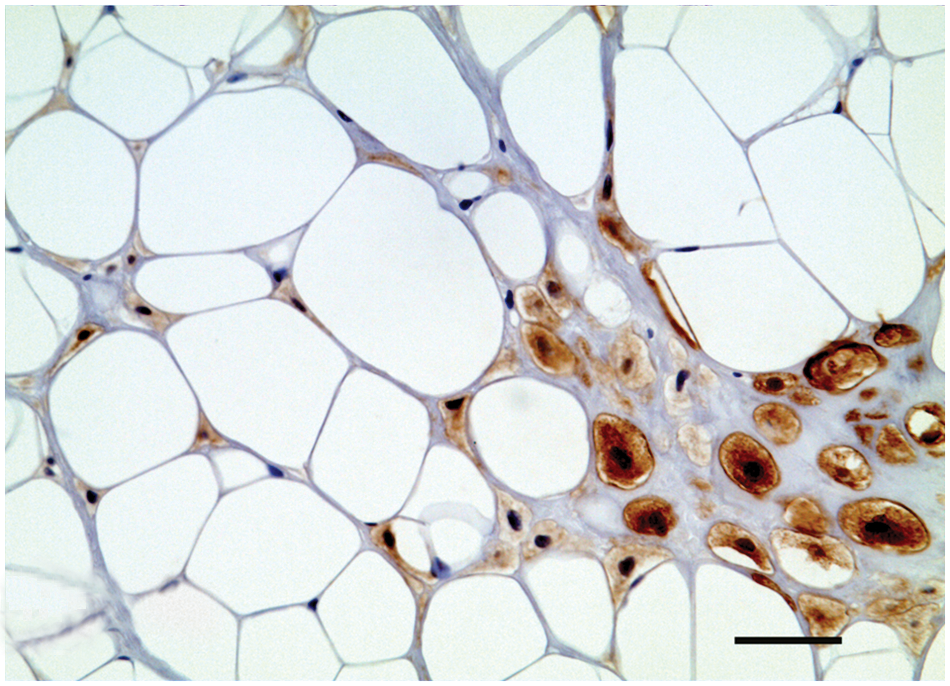
Mammary gland; dog; chondrolipoma. S-100 immunolabeling located in cartilaginous cells. Biotin–streptavidin–peroxidase method, hematoxylin counterstain. Bar = 50 µm.
Human chondrolipoma is a rare benign neoplasm of middle-aged patients that originates in subcutaneous or deeper soft tissues. 13 Despite its low frequency, chondrolipoma is included in the WHO classification of soft tissue tumors. 8 Some cases of chondrolipoma located in mammary gland have been described in human patients.1,2,4,5 Chondrolipoma is not yet included in the WHO histological classification of mesenchymal tumors of domestic animals. 6 Nevertheless, neoplasms with histological features resembling those reported for chondrolipoma in human beings have been published.9,10,12 Differential diagnosis of these tumors includes chondroid lipoma, mesenchymoma, hamartoma, and chondroid metaplasia.
Human chondrolipoma appears as a multilobulated mass that histologically consists of lobules of mature adipose tissue separated by fibrocollagenous septa containing foci of chondroid metaplasia and, occasionally, encapsulated masses of mature hyaline cartilagine. 13 In contrast, chondroid lipoma is characterized by nests and cords of abundant uni- or multivacuolated lipoblasts embedded in prominent myxoid to hyalinized chondroid matrix admixed with a variable amount of mature adipose tissue. 8 Chondroid lipoma is highly vascular and may contain hemorrhage and fibrosis. In the current case, chondroid foci were composed of chondrocytes and chondroblasts with some cellular signs of atypia. Vacuolated cells were widely scattered and only represented a small portion of the cellular population. Furthermore, the tumor had reduced vascularity and lacked foci of hemorrhage.
In 1948, the term
The process that leads to the formation of cartilaginous tissue among adipose lobules is not clearly explained. The cartilage component may originate from chondroid metaplasia of some lipomatous cells 13 or from the presence of pluripotential mesenchymal cells in the neoplasm. Pluripotential stem cells are located in the bone marrow but can move to subcutaneous tissue in which they can differentiate in mature mesenchymal cells. These cells may lose the ability to differentiate in the appropriate tissue and to acquire the specific phenotype during migration via systemic circulation. 10 Chondroid metaplasia often is a sequel of local trauma such as in lipomas located close to bone. 10 In the case described herein, the lesion was distant from bone, so this possibility was rejected.
Cartilaginous cells of the lesion described herein were reactive to S-100 antibody, as previously reported in the literature.9,13 Singe or small groups of chondroblasts located within the connective septa were S-100 immunoreactive as well. The absence of immunoreactivity to myoepithelial markers (P63, CK 5/6, and CK 14) excludes an origin from mammary myoepithelial cells. The lesion also lacked surrounding mammary tissue.
Atypical features of cartilaginous cells, the location of the lesion, and immunohistochemical staining results supported the diagnosis of chondrolipoma. This neoplasm consists of adipose tissue with scattered islands of moderately atypical cartilagenous cells. Due to location and consistency of the mammary chondrolipoma, it may be confused on palpation with a complex or mixed mammary tumor. Despite cellular atypia, chondrolipoma is a benign tumor that does not metastasize or recur after adequate surgical excision. 13
Footnotes
Acknowledgements
The authors thank Dr Paolo Viacava, head of the Department of Pathology at the “Ospedale Versilia” of Camaiore (Lucca), for providing slides and diagnosis revision.
a.
ImmPACT DAB, Vector Laboratories, Burlingame, CA.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
