Abstract
Routinely diagnosed simple solid carcinoma (SSC) of the canine mammary gland comprises a heterogeneous group of tumors. Seventy-two cases that had been diagnosed as SSC based on hematoxylin and eosin–stained tissue sections were reclassified immunohistochemically on the basis of myoepithelial markers p63 and α-smooth muscle actin, as well as a luminal epithelial marker cytokeratin 8. Only 23 cases (32%) were true SSC, composed only of luminal epithelial cells, whereas 11 cases (15%) were malignant myoepithelioma (MM), composed predominantly of myoepithelial cells, and 38 cases (53%) were biphasic carcinoma (BC), characterized by biphasic proliferation of luminal epithelial and basal/myoepithelial components. As the pathological parameters were compared between the reclassified tumor types, infiltrative potential, vascular/lymphatic invasion, lymph node metastasis, and Ki-67 labeling index were higher in true SSC compared with MM and BC, suggesting that the former may exhibit a poorer prognosis compared with the latter two.
Canine mammary simple carcinoma is defined as a carcinoma composed of one type of either luminal epithelial cells or myoepithelial cells and is further subdivided on the basis of growth patterns as tubulopapillary, solid, and anaplastic carcinoma according to the World Health Organization (WHO) classification published in 1999. 6 In the past, most simple solid carcinomas (SSCs) were considered to comprise luminal epithelial cells. However, in 2003 Gama and colleagues 2 examined the expression of a basal/myoepithelial marker, p63, in SSC cases that had been diagnosed based on hematoxylin and eosin (HE)–stained tissue sections, and they found that 7 of 10 cases exhibited p63+ tumor cells. One case presented diffuse p63 expression, and the remaining 6 cases were characterized by a dual immunoreactivity with p63– cell nests bordered by p63+ cells. In 2004, a similar observation was reported by Martín de las Mulas and colleagues 4 using calponin, a marker for cells with myoid differentiation, including myoepithelial cells. Calponin immunoreactivity revealed that myoepithelial cells were a major component in 6 of 8 SSC cases that had been previously diagnosed based on HE-stained tissue sections. These studies demonstrate the presence of basal/myoepithelial tumor cells that are morphologically similar to luminal epithelial tumor cells but differ from the classic myoepithelial morphology of spindle shape, arranged in a stellate, reticulated pattern with intercellular mucinous substance in complex carcinoma and arranged in a sarcoma-like pattern in spindle cell carcinoma. 5 In 2011, Goldschmidt and colleagues 3 proposed a modified WHO classification that included an important addition for diagnosis: immunohistochemical markers that distinguish between luminal epithelial and myoepithelial components. However, the degree of malignancy of these basal/myoepithelial cell marker–positive solid tumors was less well understood. 10
In this study, we analyzed archival specimens from 72 cases that were diagnosed as SSC based on HE-stained tissue sections and divided these cases into 3 tumor types according to immunostaining analysis; we then compared the differences in pathological parameters with regard to malignancy grade among these tumor types.
The evaluation was performed semi-quantitatively using serial sections with HE staining and immunostaining (LSAB method) using mouse monoclonal antibodies against p63 (clone 4A4, IgG2a, 1:150; Neomarkers, Fremont, CA), α-smooth muscle actin (SMA, clone 1A4, IgG2a, 1:400; DAKO, Glostrup, Denmark), and cytokeratin 8 (CK8, clone Ks8.7, IgG1, 1:10; PROGEN, Heidelberg, Germany). In addition, to clearly demonstrate the biphasic proliferation of 2 types of cells, double-enzyme immunohistochemistry for p63/CK8 or SMA/CK8 was performed using Histofine Simple Stain MAX-PO and Histofine Simple Stain AP (NICHIREI, Osaka, Japan). In some cases, p63/SMA double immunostaining was applied to differentiate myoepithelial cells from myofibroblasts. In concordance with previous reports and our own experience, luminal epithelial tumor cells were consistently negative for p63 and SMA and usually positive for CK8. 1,2,9,10,12 On the other hand, myoepithelial tumor cells were consistently positive for p63, frequently positive for SMA, and negative or weakly positive for CK8. 1,2,9,10,12 Based on these immunohistochemical properties, 72 cases that had been diagnosed as SSC were divided into the following 3 tumor types: true SSC of luminal epithelial cell type, malignant myoepithelioma (MM), and biphasic carcinoma (BC). The definitions are as follows: true SSCs do not contain p63/SMA-positive tumor cells, and at least 60% of the tumor cells are positive for CK8; MM is a tumor in which 90% or more of the tumor cells are positive for p63 and/or SMA; and BC is characterized by a dual mixture of p63 and/or SMA-positive cells and both p63 and SMA-negative cells, and at least 60% of the p63/SMA-negative cells express CK8.
The age at the time of surgery, the largest diameter of tumor mass, and the presence or absence of the widespread central necrotic/acellular zones on cut surface of the tumor mass were obtained from review of the records of each case. The HE sections were evaluated for pathological parameters of the tumor growth, such as marginal infiltration, vascular/lymphatic invasion, and metastasis to the neighboring lymph node. The number of mitoses was counted per 10 high-power fields at the tumor periphery. Carcinomas were graded according to Peña’s method, which is a canine adaptation of the human Elston and Ellis grading system. 7 Briefly, each carcinoma was evaluated by tubule formation, mitotic activity, and nuclear pleomorphism of carcinoma cells and was assigned to grade I, II, or III. The stromal proportion was measured within high-power fields in each of the 8 areas selected randomly from the whole tumor area using area-measurement software (Photoshop CS3 Extended; Adobe Systems, San Jose, CA). The Ki-67 labeling index was examined in 8 high-power fields (approximately 1000 cells) in sections immunostained with anti–Ki-67 antibody (clone MIB-1, IgG2a, 1:100; DAKO) and defined as the ratio (%) of Ki-67–positive tumor cells to the total number of tumor cells.
Statistical analyses were conducted between the 3 tumor types for the clinical and pathological parameters. Student’s t-test for continuous variables, Mann-Whitney’s U test for nonparametric variables, or Fisher’s exact probability test for categorical variables were used to examine significant differences. Bonferroni’s procedure was used to adjust for multiple comparisons. Differences were considered significant when P < .05.
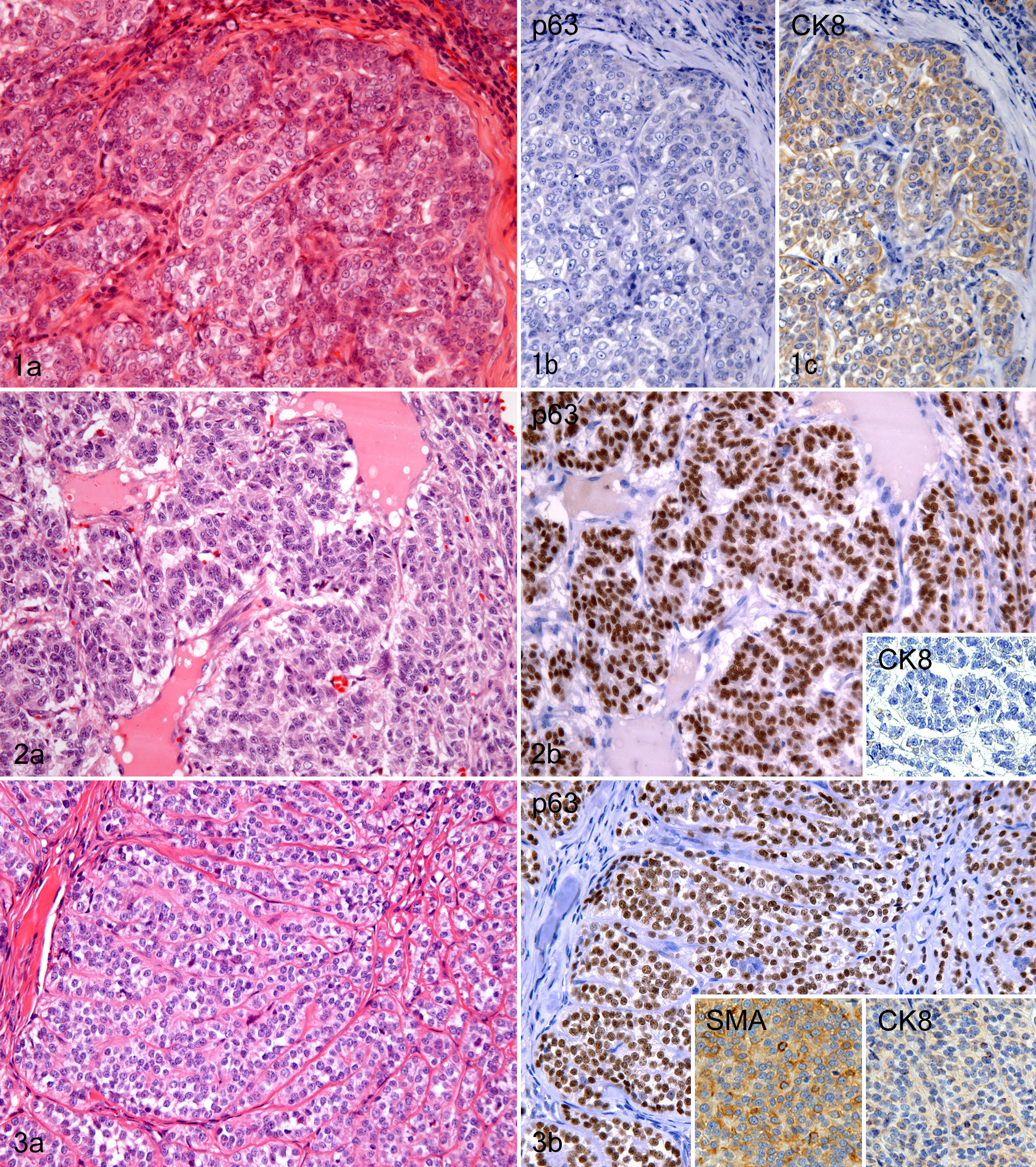
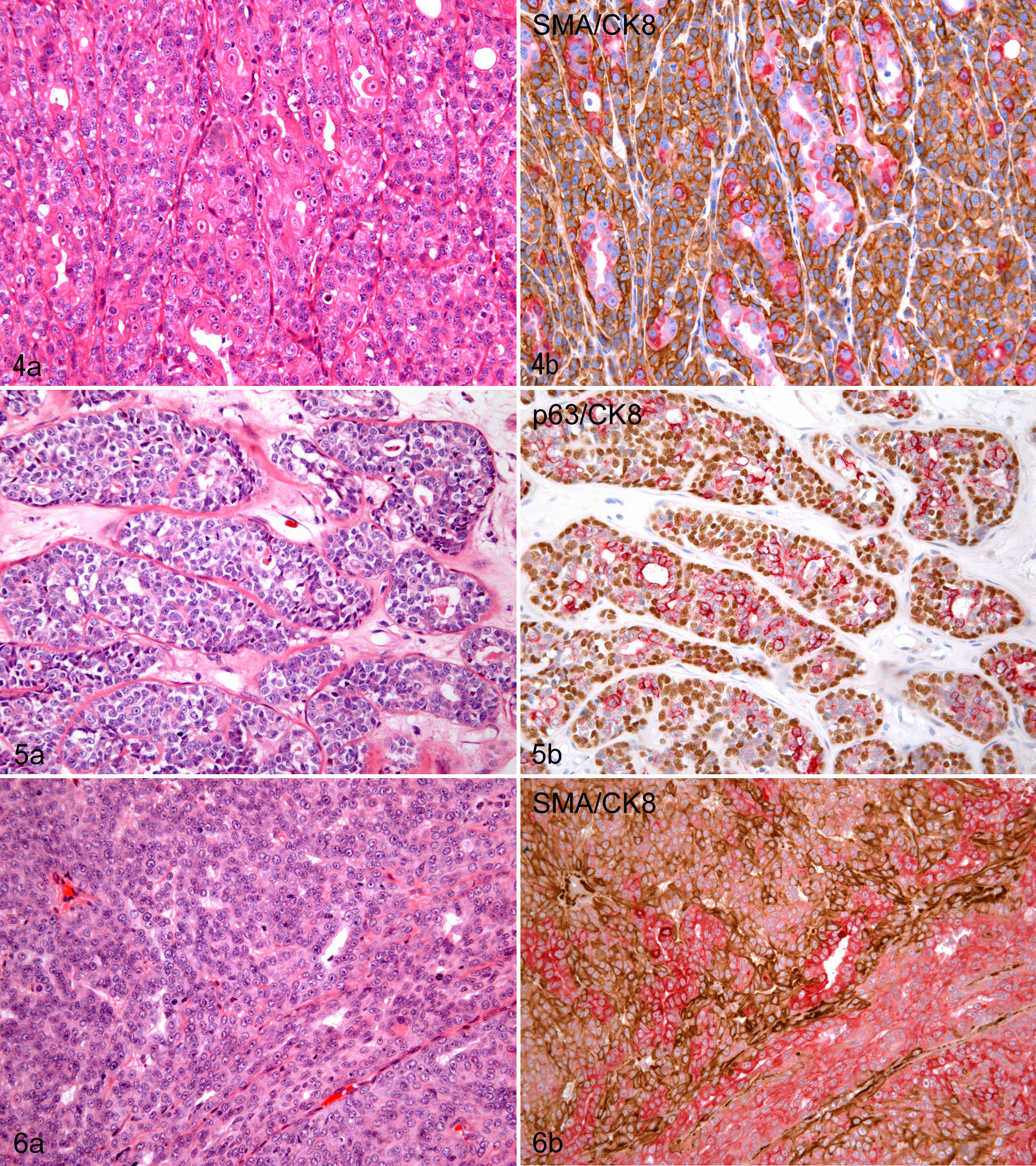
Of the 72 cases of SSC that had been routinely diagnosed by HE, only 23 (32%) immunohistochemically exhibited features of SSC composed only of luminal epithelial tumor cells defined in the WHO classification (Fig. 1a–c). 6 Eleven cases (15%) were rediagnosed as MM. They were morphologically and immunohistochemically similar to epithelioid cell type (Fig. 2a, b) or clear cell type (Fig. 3a, b) of MM (synonym: myoepithelial carcinoma) arising in human breast and salivary gland. 1,12 All 11 cases expressed p63 diffusely. It is known that human MMs also express p63 consistently, but do not always retain SMA expression, and is often weakly positive for luminal cytokeratins. 1,9,12 Six (55%) and 9 (82%) of 11 canine mammary MMs were positive for SMA and CK8, respectively, in variable proportions of tumor cells, although the intensity of CK8 staining was relatively low compared with that in luminal epithelial cells of normal glands. These results are compatible with the proportion of SMA-positive and CAM5.2 (CK7/8)–positive cases (50% and 89% respectively) in a series of MM of the human salivary gland. 9 Thirty-eight cases (53%) were reclassified as BC, meaning a heterogeneous group of tumors composed of both luminal epithelial and basal/myoepithelial tumor cells (Figs. 4a, b and 5a, b). The basal/myoepithelial tumor cells of this tumor were often indistinguishable from luminal epithelial tumor cells when examined only on HE-stained tissue sections (Fig. 6a). However, a double-enzyme immunohistochemical technique with anti-p63 or anti-SMA and anti-CK8 antibodies clearly revealed the admixture of the 2 types of cells (Fig. 6b).
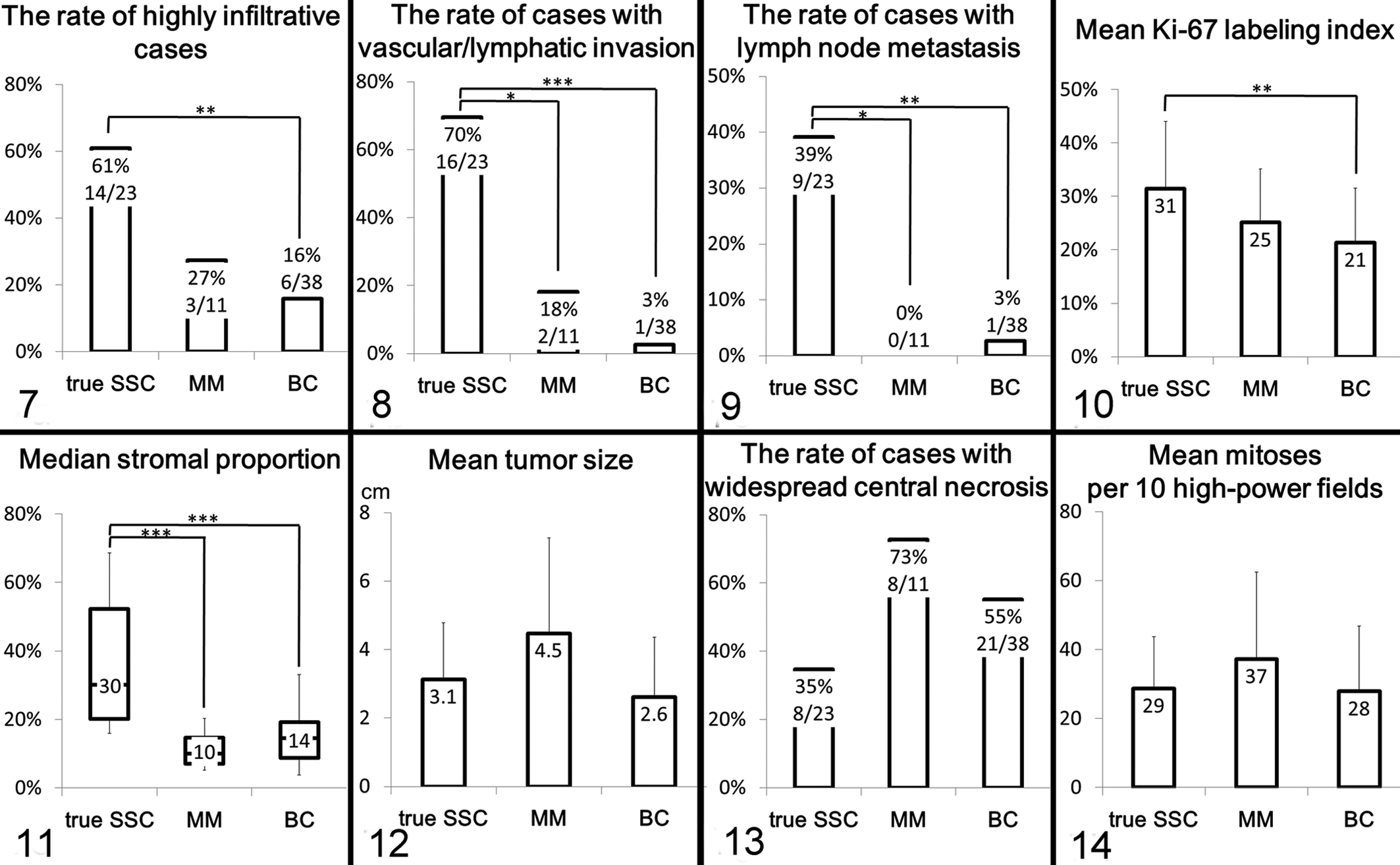
A comparison of pathological parameters between the 3 tumor types is shown in Figs. 7 to 16. True SSC usually exhibited highly infiltrative growth (14 of 23 cases, 61%; Fig. 7), whereas MM and BC exhibited less of such growth (3 of 11 cases, 27%; 6 of 38 cases, 16%, respectively; SSC vs BC, P < .01). The frequency of cases with vascular/lymphatic invasion was higher in true SSC (16 of 23 cases, 70%; Fig. 8) compared with MM and BC (2 of 11 cases, 18%; 1 of 38 cases, 3%, respectively; SSC vs MM, P < .05; SSC vs BC, P < .001). In true SSC, metastasis to the lymph node was noted in 9 of 23 cases (39%; Fig. 9), but this was rare for MM and BC (0 of 11 cases, 0%; 1 of 38 cases, 3%, respectively; SSC vs MM, P < .05; SSC vs BC, P < .01). It has been previously demonstrated that infiltrative growth, vascular/lymphatic invasion, and lymph node metastasis are good criteria for indicating malignant behavior in canine mammary carcinomas. 6,8 The Ki-67 labeling index, known as an independent and objective prognostic factor in disease-free and overall survival, 8 was also higher in true SSC (mean rate: 31%; Fig. 10) compared with MM and BC (25% and 21%, respectively; SSC vs BC, P < .01). True SSC was accompanied by a prominently desmoplastic stromal reaction, whereas most cases of MM and BC exhibited a high tumor cellularity and scarce reactive stroma. There was a larger stromal proportion in the tumor in the true SSC (median rate: 30%; Fig. 11) compared with the MM and BC (10% and 14%, respectively; SSC vs MM, BC, P < .001). The higher proportion of stroma in the true SSC could possibly reflect the potential for greater infiltration, because tumor stroma is most likely related to tumor progression. 11 Our previous study indicated that the appearance of stromal myofibroblasts is associated with poor prognosis for canine mammary simple carcinoma. 11
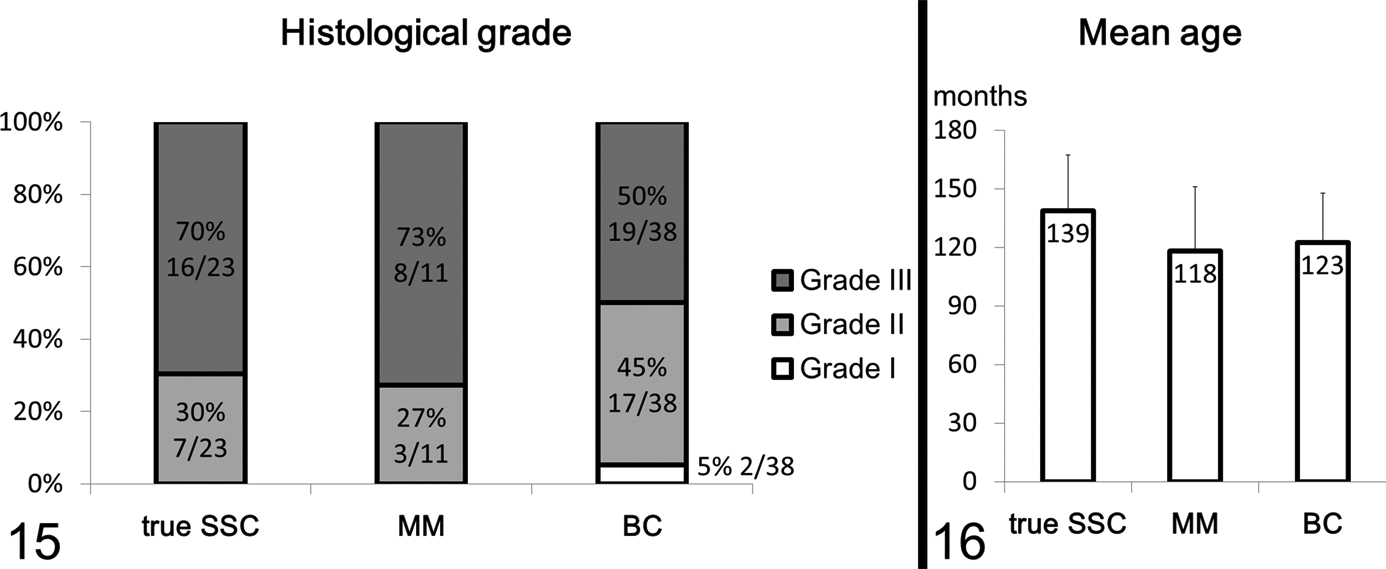
The tumor masses of MM were characterized by large size, well-circumscribed spherical shape, and widespread central necrotic/acellular zones on their cut surface. The mean tumor size (4.5 cm; Fig. 12) of MM was larger compared with true SSC and BC (3.1 cm and 2.6 cm, respectively), but the differences were not statistically significant. MM and BC often exhibited widespread central necrosis (8 of 11 cases, 73%; 21 of 38 cases, 55%; Fig. 13). In true SSC, this type of necrosis was relatively uncommon (8 of 23 cases, 35%).
The number of mitoses and histologic grade were generally high in all 3 tumor types, and there were no marked differences among them (Figs. 14, 15). The mean age was the highest in dogs with true SSC, but the difference was not statistically significant (Fig. 16).
As stated above, MM and BC exhibited rare vascular/lymphatic invasion and lymph node metastasis, regardless of large tumor size, widespread necrosis, and high histologic grade. This finding indicates that these tumors possessed a lower grade of malignancy compared with true SSC, in which invasive and metastatic features were frequently observed. In humans, most carcinomas with basal/myoepithelial differentiation in the salivary gland have been known to exhibit a relatively low grade of malignancy and prolonged survival. 1,12 Similar observations have long been noted in complex and mixed mammary tumors of dogs. 6 Some data have indicated that myoepithelial cells contribute to local growth and inhibition of metastasis via secretions, such as anti-invasive proteinase inhibitors. 12
In conclusion, we demonstrated that there are significant differences in certain indicators of malignancy between true SSC of luminal epithelial cell type, MM, and BC, which were divided according to immunohistochemical evaluation. The findings that we present in this study support the application of the modified WHO classification of canine mammary tumors 3 and suggest that the use of immunohistochemistry in the diagnosis of canine mammary tumors is warranted to obtain a more accurate prognosis.
Footnotes
Acknowledgments
We thank Dr Yoko Matsuda, Dr Toshiyuki Ishiwata, and Dr Zenya Naito from Departments of Pathology and Integrative Oncological Pathology, Nippon Medical School for helpful discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
