Abstract
Spontaneous basaloid adenomas occurred in four out of 354 dogs with mammary tumors. Affected dogs were pure-bred, intact females between 6 and 8 years of age. Three dogs were nuliparous, two had pseudopregnancies, and none had received contraceptive steroids. The tumors were multiple (three cases) or unique, less than 1 cm in diameter, well delineated, and composed of uniform cords and clusters of monomorphic epithelial cells with focal signs of squamous or glandular differentiation. A basal cell immunophenotype (cytokeratins 5 and 14 positive) without either glandular epithelial (cytokeratins 8, 18, and 19 negative) or myoepithelial (calponin negative) differentiation was observed in the majority of tumor cells. No recurrence or metastasis was recorded after follow-up periods between 3 and 24 months. In spite of the hormone-dependent nature of this tumor in female Beagles given experimental contraceptive steroids, spontaneous basaloid adenomas lacked estrogen receptor alpha and progesterone receptors.
Basaloid adenoma of the mammary gland of the dog is a benign tumor composed of uniform cords and clusters of monomorphic basaloid epithelial cells.8 This type of tumor was first reported in female Beagles receiving experimental oral steroids for 5–7 years.4,5 Spontaneous basaloid adenomas of the mammary gland have been described in two dogs exclusively, and categorization of clinical and morphologic features is lacking.3,8 Here we describe the epidemiologic, clinical, and morphologic features of four cases of spontaneous basaloid adenomas of the mammary gland of the dog. In addition, we have analyzed the immunophenotype and the steroid hormone receptors content of the tumor cells using tumor markers and immunohistochemical techniques.
Clinical and epidemiologic data of the four dogs are shown in Table 1. Biopsy specimens were fixed in formalin and processed routinely according to the standard histologic procedures. Hematoxylin and eosin, periodic acid–Schiff, and Alcian blue histochemical techniques, as well as the avidin–biotin–peroxidase complex (ABC) immunohistochemical technique were performed. Technical data about specific antibodies, as well as their immunoreactivity pattern in the normal mammary gland (internal positive control), are shown in Table 2. Normal canine tissues known to contain the antigens sought were used as external positive controls as follows: canine skin for S100 protein and glial fibrillary acidic protein antibodies (peripheral nerves) as well as for RCK-102 (squamous epithelium), NCL-5D3 (apocrine gland epithelium), cytokeratin 14 (basal cells of squamous epithelium), and vimentin (dermal fibroblasts) antibodies; and canine uterus for estrogen and progesterone receptors (myometrial and endometrial—stromal and epithelial—cells) antibodies. Tissue sections under study treated with either normal rabbit serum (for polyclonal antibodies) or mouse ascites fluid (for monoclonal mouse antibodies) instead of corresponding specific antibodies were run as negative controls. The immunohistochemical techniques of detection of estrogen and progesterone receptors have been detailed elsewhere.7
Epidemiologic and clinical data of the four dogs with basaloid adenomas.

∗ Mammary, glands numbered 1 (pectoral) to 5 (inguinal).
† L: left; R: right.
The antibodies employed to analyze the immunophenotype and the hormone dependence of spontaneous canine basaloid adenomas, their technical features, and their immunoreactivity with epithelial and myoepithelial cells of the normal mammary gland using the avidin–biotin–peroxidase complex technique are shown.
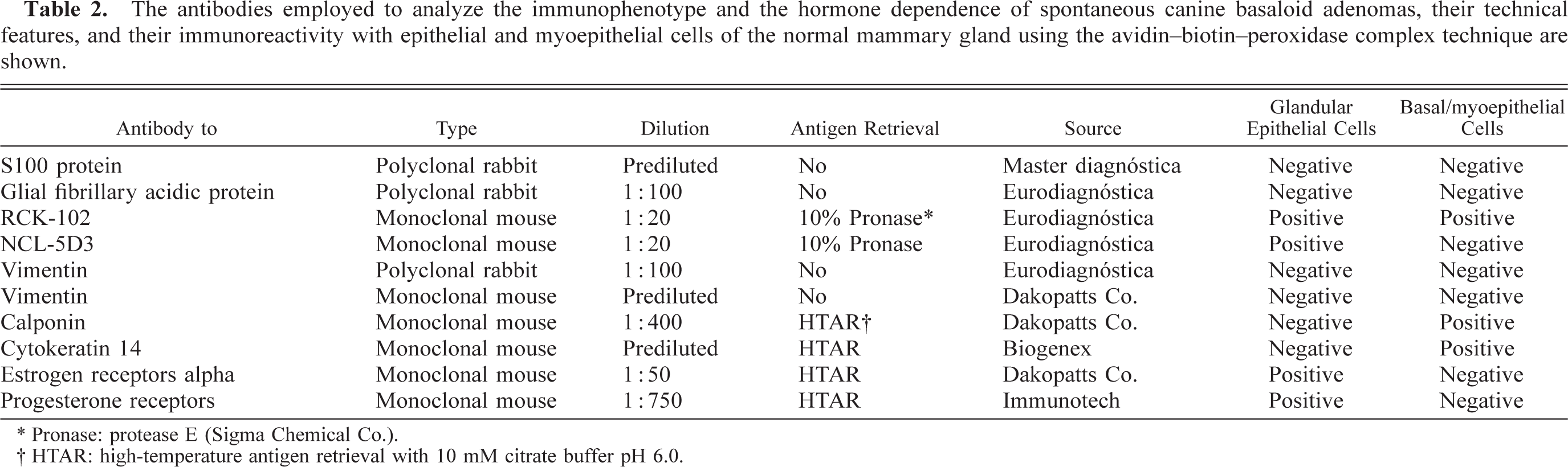
∗ Pronase: protease E (Sigma Chemical Co.).
† HTAR: high-temperature antigen retrieval with 10 mM citrate buffer pH 6.0.
Grossly, multiple (case Nos. 1, 3, and 4) or single (case No. 2) small, discrete nodules, usually less than 1 cm in diameter, were observed. They affected one (case Nos. 2–4) or more glands (left 2nd, 3rd, and 4th in case No. 1) and were well circumscribed, firm, and grayish or yellowish on the cut surface. The nodules formed two large conglomerates in case No. 1, one 8 cm in diameter affecting the left 2nd and 3rd mammary glands and the other 5.5 cm in diameter affecting the 4th mammary gland and ulcerating the overlying skin.
Histologic examination of the tissue specimens revealed four (case No. 1), one (case No. 2), seven (case No. 3), and two (case No. 4) well-circumscribed nodules composed of round to oblong epithelial cells surrounded by a prominent fibrous stroma (Fig. 1). The cells were arranged into small nests and cords and had uniform, round to ovoid euchromatic nuclei without mitoses, and scant cytoplasm. Some cells were aligned perpendicular to the rest of the cells, and periodic acid–Schiff staining demonstrated that they were palisaded against a thin basal lamina. In some nodules, split spaces without content were observed. In some other nodules, foci of laminated keratin were present, and the surrounding cells had rounded nuclei and abundant pink cytoplasms with occasional keratohyaline granules (Fig. 2). The stroma was minimal but was sometimes edematous creating cystic spaces and giving the impression of false lumens (Fig. 2). At these locations, Alcian blue staining revealed a large amount of mucopolysaccharide. Each group of the round to oblong epithelial cells surrounded by a prominent fibrous stroma was defined as a nodule, and each nodule was identified as a basaloid adenoma. Solid proliferations of epithelial cells with or without atypia and with occasional foci of laminated keratin were observed within ducts and acini in the vicinity of basaloid adenomas in case Nos. 1 and 3 (Fig. 3).
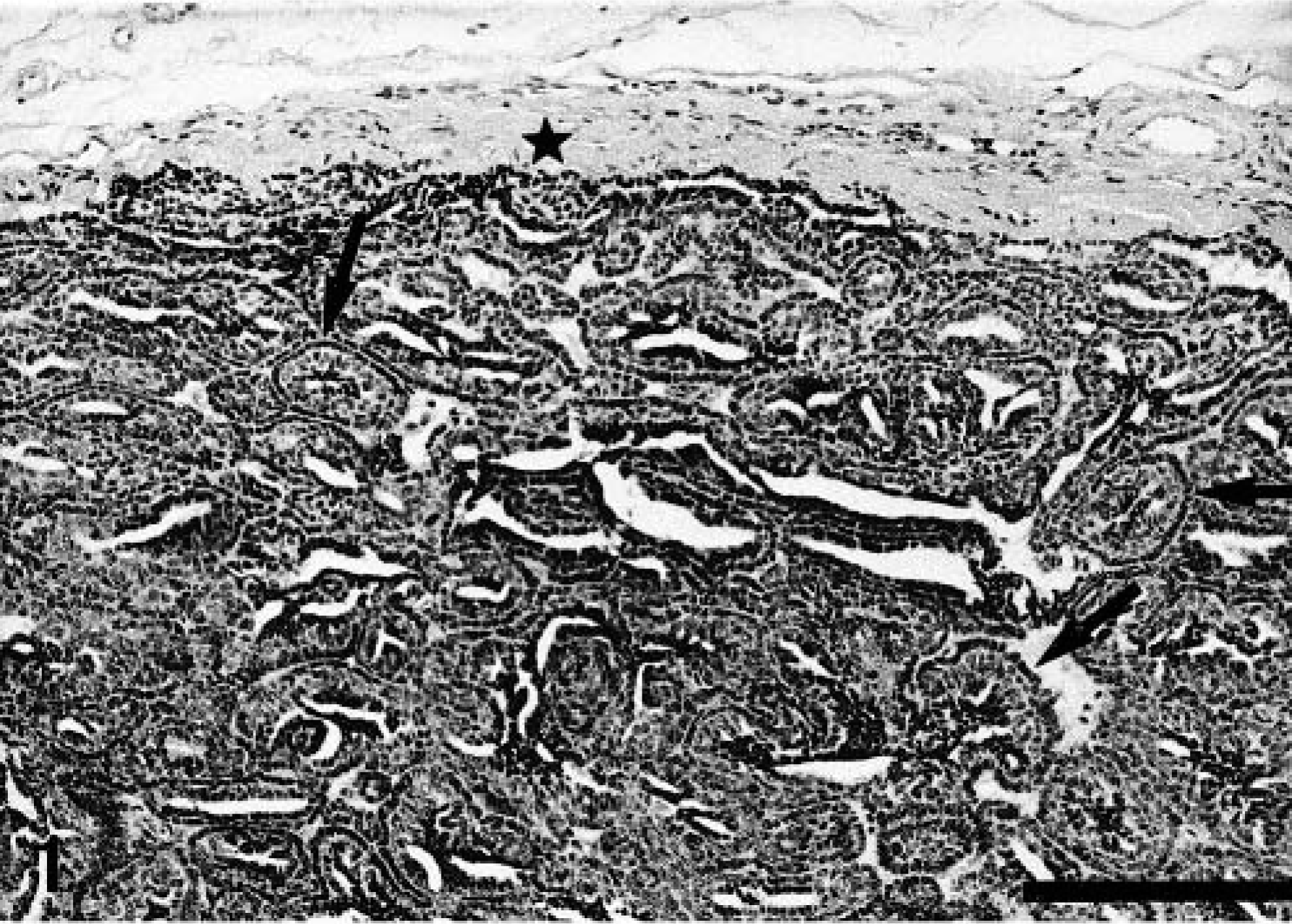
Mammary gland; dog No. 2. Basaloid adenoma. Well-circumscribed nodule surrounded by fibrous stroma (star) and composed of round to oblong epithelial cells arranged into small nests and cords with split spaces without content between them. Some cells are aligned perpendicular to the rest of the cells (arrows). HE. Bar = 100 μm.
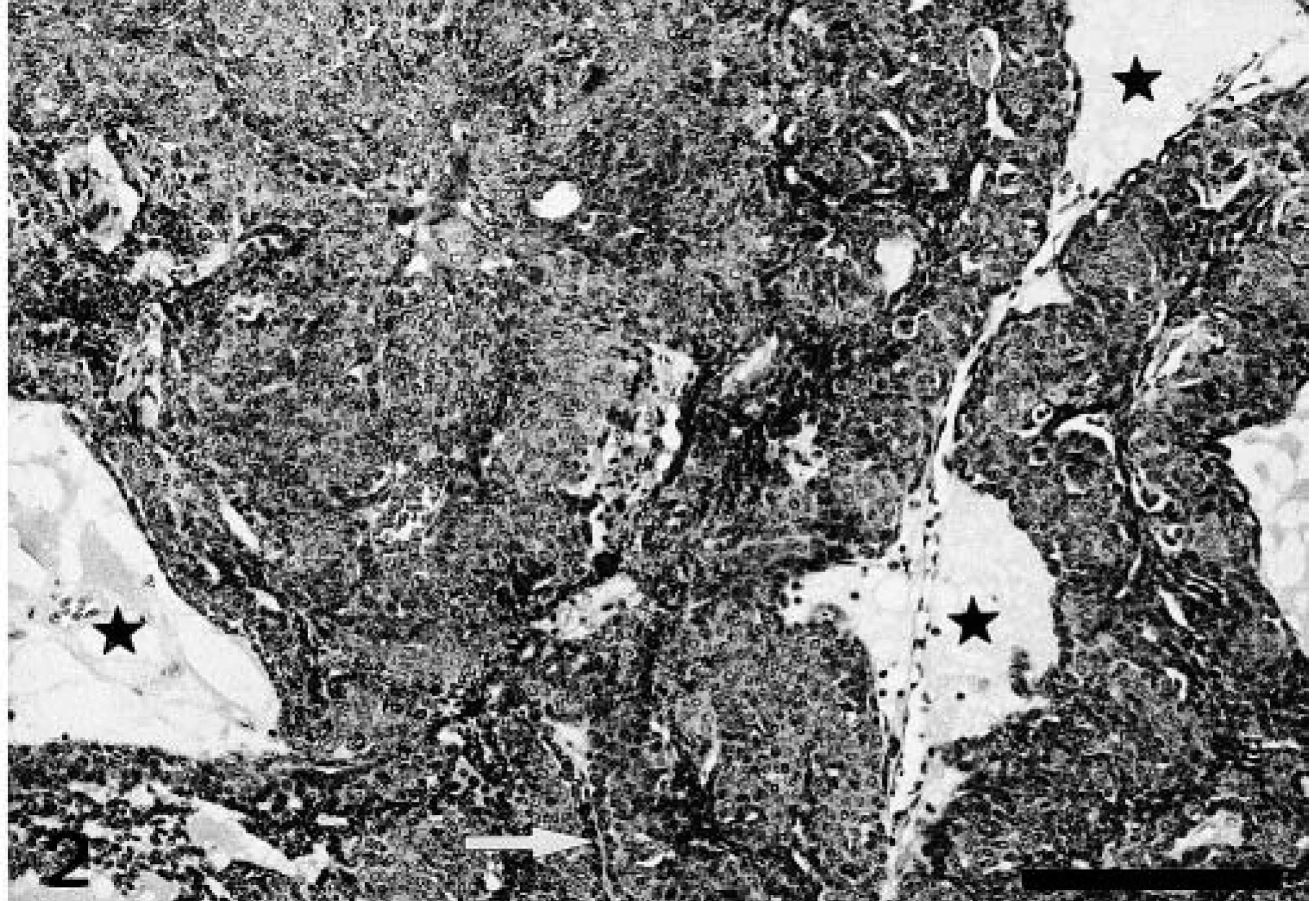
Mammary gland; dog No. 3. Basaloid adenoma. Nests of tumor cells with rounded nuclei and abundant cytoplasms indicative of squamous differentiation. Some basaloid epithelial cells are aligned perpendicular to the squamous cells (arrow). The stroma is edematous, creating cystic spaces and giving the impression of false lumina (stars). HE. Bar = 100 μm.
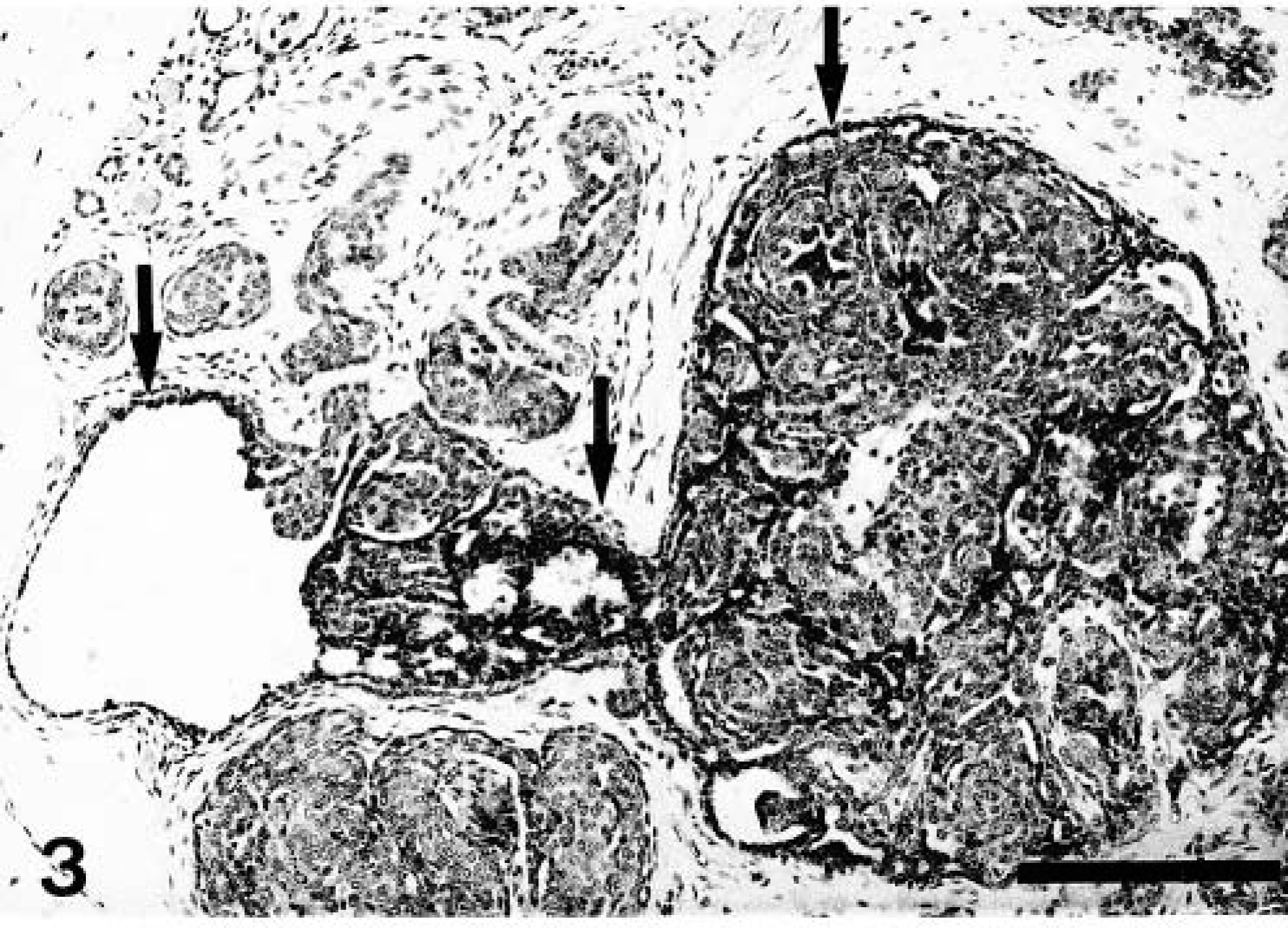
Mammary gland; dog No. 3. Mammary gland tissue in the vicinity of basaloid adenomas. A dilated duct (arrows) shows an intraductal proliferation of basaloid epithelial cells and cells with squamous differentiation. The edge of another basaloid growth is observed below. HE. Bar = 100 μm.
Basaloid adenoma was the only type of tumor observed in case Nos. 2 and 3. In case No. 1, tumors were observed in the left 2nd and 3rd mammary glands as well as around a simple solid carcinoma observed in the left 4th mammary gland and composed of solid masses of cells with severe atypia and numerous mitoses that infiltrated the skin (Fig. 4). The regional lymph node of case No. 1 had no metastases. In case No. 4, a complex carcinoma and a complex adenoma were additionally found.
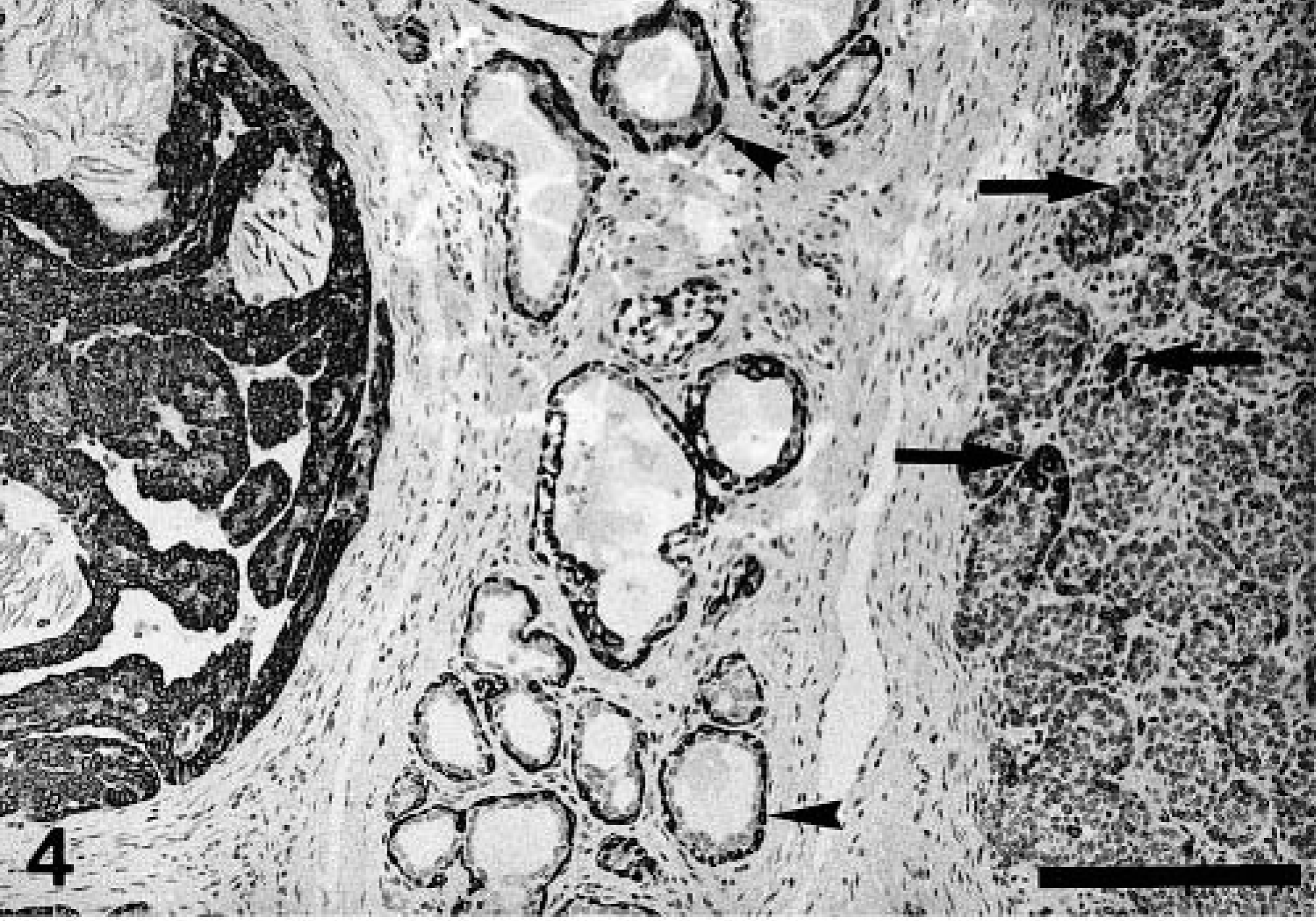
Mammary gland; dog No. 1. Simple solid carcinoma (right-hand side) in the vicinity of a basaloid adenoma (left-hand side). The malignant tumor has an expansive, noncapsulated type of growth and is composed of solid masses of atypical epithelial cells. The majority of cells of the basaloid adenoma are stained with cytokeratin 14 antibody, whereas only isolated cells are stained in the solid carcinoma (arrows). These positive cells are located at the periphery of the solid growth and disappear progressively toward its center. The basal/myoepithelial cell layer of dilated acini located between the two lesions is uniformly stained with cytokeratin 14 antibody (arrowheads). ABC immunohistochemical method for cytokeratin 14 antibody, counterstaining with Mayer's hematoxylin. Bar = 100 μm.
Immunohistochemically, all cells of basaloid adenomas reacted with the antibody that recognizes cytokeratins 5 and 8 (RCK-102 antibody) and the majority of cells with the cytokeratin 14 antibody (Figs. 4, 5a). Cells negative for the latter lined either collapsed lumens (single rows facing each other) or open empty lumens (Fig. 5a). Some cytokeratins 5 and 8–positive cells and cytokeratin 14–negative cells reacted with the antibody that recognizes cytokeratins 8, 18, and 19 (NCL-5D3 antibody), which also haphazardly stained isolated cells. Calponin immunoreactivity was heterogeneous among different nodules. Thus, a single incomplete row of positive cells next to the edematous stroma was seen in some of them, whereas in some others either isolated positive cells or negative cells were found (Fig. 5b). In addition, some spindle cells of the tumor's capsule were identified as myofibroblasts because they expressed calponin but not cytokeratin 14 (Fig. 5a, b). Estrogen receptors alpha and progesterone receptors were not detected in basaloid adenomas but adjacent ductal and acinar proliferative lesions showed heterogeneous nuclear staining. The rest of the antibodies employed did not react with either the normal mammary gland duct and acini or the basaliod adenomas. The immunohistochemical staining pattern of the solid carcinoma found in case No. 1 differed from that of the basaloid adenomas in that cytokeratin 14–positive cells were less and located at the periphery of the solid masses exclusively, with a progressive disappearance toward the center of the tumor (Fig. 4).
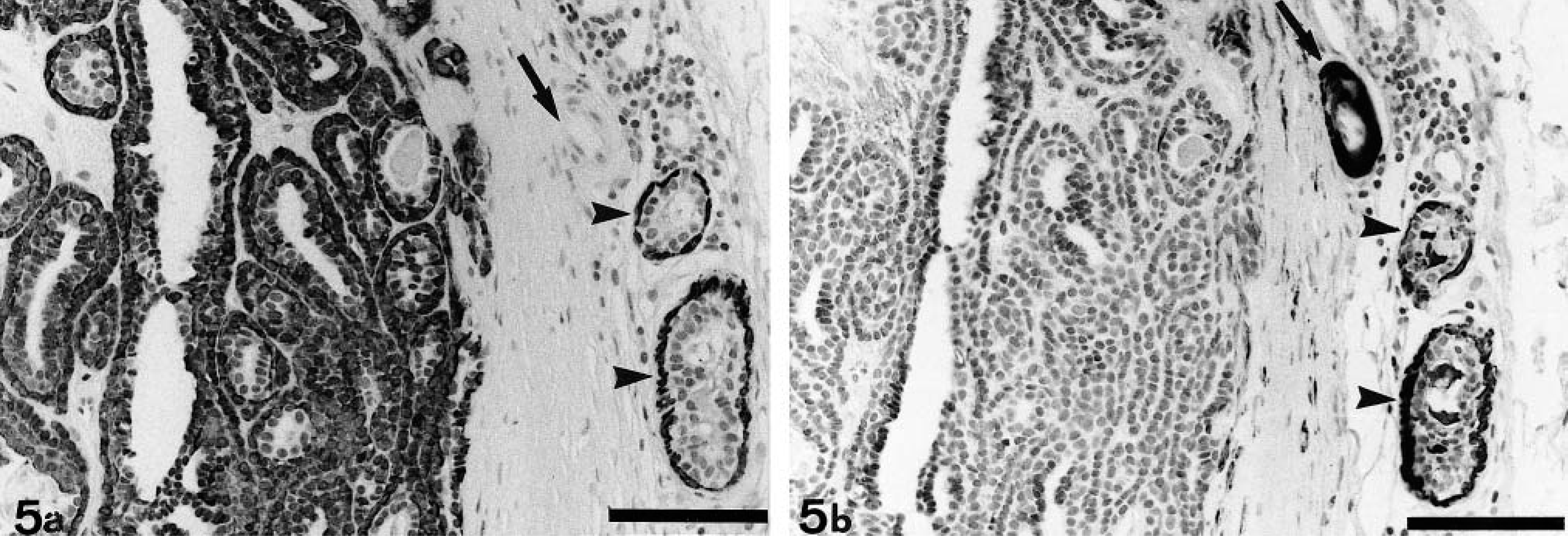
Mammary gland; dog No. 2. Basaloid adenoma.
The histologic appearance of the tumors was consistent with the mammary basaloid adenomas.5,8 The tumors clearly originated in the mammary gland, and the presence of ductular and acinar proliferative lesions of similar characteristics in the vicinity of the tumors in two cases suggested a multicentric and simultaneous origin in the lobule and the terminal duct that enters the lobule (Fig. 3) as the origin of the tumors.5 Histologically, the tumor cells had a basaloid appearance with occasional signs of glandular or squamous differentiation.3,5,8 This appearance was confirmed by our immunohistochemical findings: the majority of tumor cells had a basal cell immunophenotype (cytokeratins 5 and 14 positive). The immunoreactivity observed with RCK-102 antibody in the predominant population of tumor cells can be attributed to the expression of cytokeratin 5 exclusively. The isolated cells expressing markers of glandular (cytokeratins 8, 18, and 19) or myoepithelial (calponin) differentiation could be remnants of preexisting structures or differentiated tumor cells.1,10 The presence and role of undifferentiated cells in the mammary gland and their relationship to the luminal epithelium and the myoepithelium is not entirely clear, although a stem cell origin has been suggested for many dysplastic and tumorous lesions.8 In our study, the predominant cells of basaloid adenomas had a basal cell immunophenotype identical to that of basal cells of squamous epithelia, which is in accordance with the finding of squamous differentiation in the great majority of basaloid adenomas.1 Basaloid adenomas have not been described in the human breast, but basaloid adenoma of the canine mammary gland resembles human salivary gland basaloid adenoma, and the immunophenotype of this tumor mimics the embryonic development of the tissues of the salivary glands.2,6
The usual form of presentation of spontaneous basaloid adenomas was that of multiple small nodules affecting one or more mammary glands in pure-bred intact females between 6 and 8 years of age.3,5 These multiple mammary nodules may form a single palpable mass on clinical examination, as in case No. 1.3 Reproductive data indicate that three out of four patients were nuliparous, two had pseudopregnancies, and none of the dogs had been given oral contraceptives. Our cases represented less than 1% of the 354 mammary tumors recorded since 1998, whereas basaloid adenomas in toxicologic studies comprised between 5% and 10% of the mammary nodules that developed during the studies.4,5 Comparison of clinical and epidemiologic data of our group of animals with mammary tumors indicates that dogs with basaloid adenomas (Table 1) are younger than dogs with other types of mammary neoplasms (medium age is 6.75 versus 9.45 years), but other data retrieved from the group of animals with mammary tumors indicate that differences with respect to ovariectomy status (21%), parity (77% nuliparous), pseudopregnancy (54%), and the administration of steroid contraceptives (18%) are small. Prevalence of spontaneous basaloid adenomas has not been published, and available clinical history includes that one of the two cases described was a 6-year-old spayed female Husky that had received estradiol on two occasions for mismating, whereas the other was a 6-year-old intact female Poodle.3 Basaloid adenomas are benign tumors of the mammary gland, although a rare malignant counterpart has been recognized.8 In our study, basaloid adenomas were found in the vicinity of a solid carcinoma (case No. 1). The histologic appearance suggested a malignant transformation from the former, although the relationship between the two tumors could not be ascertained.
Basaloid adenomas are described in Beagle dogs administered investigational progesterone and synthetic progestin compounds for long periods of time.4,5 The four dogs bearing spontaneous basaloid adenomas were intact females and had immunoreactive products to estrogen receptors alpha and progesterone receptors antibodies in the nuclei of acinar and ductal epithelial cells of the surrounding mammary gland. Some nuclei in the intraductal and intracinar proliferative lesions found in the surroundings of basaloid adenomas (case Nos. 1 and 3) also had immunoreactive products to both antibodies but the tumor cells of basaloid adenomas did not. The steroid hormone receptor content of either experimental or spontaneous basaloid adenomas has not been previously studied. The progestin-induced increase in incidence of mammary tumor formation in the dog has been attributed to the ectopic production of growth hormone in the mammary gland.9 An explanation based on our findings would be that progestins acting upon differentiated, steroid hormone–responsive epithelial cells would induce growth hormone production by these cells, which in turn would act upon undifferentiated stem cells giving rise to different proliferative lesions. The findings of Kwapien et al. (1977), which reported that the tumors usually remained static after an initial short period of time, support this explanation.5
Footnotes
Acknowledgements
This work has been supported by grant number PM98-0164 from the Dirección General de Enseñanza Superior e Investigación Científica, Ministerio de Educación, Cultura y Deportes, Spain.
