Abstract
Infection with influenza A (H5N1) virus, which has not been associated with respiratory disease in humans previously, caused clinical signs of acute respiratory distress syndrome and multiple-organ dysfunction syndrome with high mortality in humans in Hong Kong in 1997. To study the pathogenesis of this disease, we infected four cynomolgus monkeys (Macaca fascicularis) with 2.5 × 104 median tissue culture infectious dose (TCID50) of influenza virus A/Hong Kong/156/97 (H5N1) and euthanatized them 4 or 7 days after infection. The main lesion was a necrotizing broncho-interstitial pneumonia (4/4) similar to those found in primary influenza virus pneumonia in humans, with desquamation of respiratory epithelium (4/4), intra-alveolar hemorrhage (4/4), hyaline membrane formation (2/4), and infiltration with neutrophils and macrophages (4/4). Lesions in other organs consisted of a suppurative tonsillitis (2/4) and necrosis in lymphoid organs (1/4), kidney (1/4), and liver (1/4). By immunohistochemistry, influenza virus antigen was limited to pulmonary tissue (4/4) and tonsils (2/4). Based on these results, we suggest that the cynomolgus monkey is a suitable animal model for studying the pathogenesis of human H5N1 virus infection and that multiple-organ dysfunction syndrome in this disease may be caused by diffuse alveolar damage from virus replication in the lungs alone.
Keywords
Influenza A (H5N1) virus infection in Hong Kong in 1997 caused respiratory disease in 18 humans, of whom six died. 3 , 5 , 19 This was unusual because this subtype had not been associated previously with respiratory disease in humans, 5 and infection appeared to have been transmitted directly from chickens to humans in all the cases without reassortment of the virus genome. 3 , 19 The main clinical findings were those of acute respiratory distress syndrome (ARDS), caused by a severe primary virus pneumonia, and multiple-organ dysfunction syndrome (MODS). 23 Pulmonary lesions in two patients who died 1 month after onset of illness consisted of organizing diffuse alveolar damage, interstitial fibrosis, and cystically dilated air spaces. Changes in the bone marrow and lymphoid organs were considered to be consistent with reactive hemophagocytic syndrome. 20 Mice inoculated with human H5N1 virus isolates developed not only pneumonia but also myocardial necrosis, encephalitis, 9 , 10 , 12 and fat necrosis. 12 These lesions were associated with the presence of influenza virus antigen as determined by immunohistochemical analysis.
Influenza A virus infection of the subtypes H1N1, H2N2, and H3N2 caused pandemic disease outbreaks in humans in 1918, 1957, and 1968, respectively. 11 Influenza A virus in humans infects the respiratory tract, causing epithelial necrosis with associated inflammation. Infection usually is limited to trachea and bronchi but may extend to bronchioles and alveoli, resulting in an interstitial pneumonia. Bacterial superinfection is common. 21 Extrarespiratory diseases associated with influenza include myocarditis, myositis, parotitis, encephalopathy, and Reye-Johnson syndrome. However, the role of influenza virus in the pathogenesis of these lesions is poorly substantiated. 4
Although the mouse model of influenza virus infection is used widely because of its predictability and convenience of use, the nature of the disease in mice is somewhat different from that of influenza in humans. 18 To examine the pathogenesis of H5N1 virus infection in an animal species more closely related to humans, we infected cynomolgus macaques (Macaca fascicularis) with influenza virus A/Hong Kong/156/97 (H5N1), the isolate obtained from the index case in Hong Kong in 1997. 3 , 5 The clinical and virologic results of this experimental infection have been published previously. 17 Here, we report the detailed pathologic and immunohistochemical findings.
Materials and Methods
Experimental protocol
The experimental protocol was performed as described previously. 17 Briefly, four cynomolgus macaques were placed in a negatively pressurized glove box in pairs of one male and one female and were infected with 2.5 × 104 median tissue culture infectious dose (TCID50) of influenza virus A/Hong Kong/156/97 (H5N1), the isolate obtained from the index case in Hong Kong in 1997. Virus suspension was applied intratracheally, on the tonsils and on each of the conjunctivae. Before infection, these monkeys were seronegative for influenza A and B viruses, as demonstrated by hemagglutination inhibition assay, 13 using all the human influenza A and B virus vaccine strains from the past two decades, as well as the reference H5 strains including influenza virus A/Hong Kong/156/97. The absence of virus-specific antibodies before infection was further confirmed by immunofluorescence assay using Madin Darby Canine Kidney cells infected with influenza A and B viruses. 16 Macaque Nos. 1 and 2 were euthanatized at 4 days postinfection (dpi) and macaque Nos. 3 and 4 at 7 dpi by exsanguination under ketamine anesthesia.
Necropsy, histology, and immunohistochemistry
Necropsies were carried out according to a standard protocol. Histologic and immunohistochemical examinations were performed as described previously. 17 Briefly, samples for histologic examination were stained with hematoxylin and eosin (HE) or with an avidin–biotin complex immunoperoxidase method, using a monoclonal antibody to the nucleoprotein of influenza virus A as the primary antibody. Lung samples also were stained with Gram's and periodic acid–Schiff stains. The following tissues were examined by light microscopy for the presence of lesions (HE) and for the detection of influenza viral antigen (immunoperoxidase method): conjunctiva, nasal concha, larynx, oropharynx, trachea, primary and secondary bronchus, lung, tonsil, tracheo-bronchial lymph node, inguinal lymph node, mesenteric lymph node, spleen, heart, liver, kidney, cerebrum, cerebellum, brain stem, soleus, and gastrocnemius muscle. The adipose tissue attached to trachea, larynx, kidney, spleen, tracheo-bronchial lymph node, inguinal lymph node, and mesenteric lymph node was examined specifically for evidence of fat necrosis. 12 Lung, tonsil, tracheo-bronchial lymph node, inguinal lymph node, mesenteric lymph node, spleen, heart, liver, and meninges of cerebrum, cerebellum, and brain stem were examined specifically for proliferation of benign hemophagocytic histiocytes, indicative of reactive hemophagocytic syndrome. 20
Results
Macroscopic findings
Gross lesions in all four macaques were restricted to the respiratory tract and associated lymph nodes. The macaques euthanatized at 4 dpi had focal pulmonary consolidation, involving the caudal part of the left caudal lobe in macaque No. 1 and the ventral part of the right medial lobe in macaque No. 2. The consolidated tissue was red-purple, firm, slightly depressed, and less buoyant than normal, although it did not sink in water. The tracheo-bronchial lymph nodes of macaque No. 2 were slightly enlarged (up to 5 mm in diameter), whereas those of macaque No. 1 appeared to be normal (up to 2 mm in diameter).
The macaques euthanatized at 7 dpi had extensive pulmonary consolidation with a cranio-ventral distribution, involving about 20% of the right lung and 5% of the left lung in macaque No. 3 and 10% of the right lung and 25% of the left lung in macaque No. 4. In addition, macaque No. 3 had disseminated pulmonary hemorrhages (1–5 mm in diameter) and mild subpleural emphysema caudo-dorsally, and macaque No. 4 had a large area of necrosis, characterized by brown-red, dry tissue and a rough pleura, in the central part of the right lung. Trachea and bronchi contained foamy fluid, which was scant and clear in macaque No. 3 and abundant and red in macaque No. 4. There was moderate enlargement (up to 10 mm in diameter) of the tracheo-bronchial lymph nodes in macaque No. 3 and of the tracheo-bronchial lymph nodes and tonsils in macaque No. 4.
Histopathologic findings
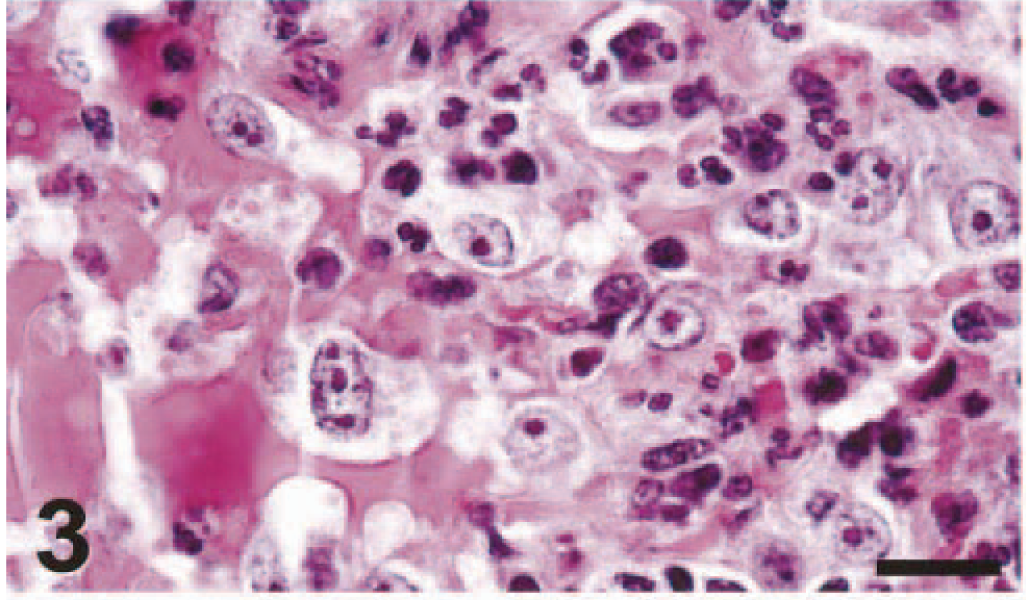
The main lesion in the consolidated pulmonary tissue of macaque Nos. 1 and 2, euthanatized at 4 dpi, involved the alveoli and bronchioles (Figs. 1–4), whereas the trachea and bronchi were less severely affected (Figs. 5, 6). The lumina of alveoli and bronchioles were variably filled with protein-rich edema fluid, fibrin, erythrocytes, and cell debris, admixed with many neutrophils and macrophages, and rare eosinophils and lymphocytes (Fig. 3). Many macrophages had a large nucleus with a prominent nucleolus, and some were binucleate. There was extensive loss of epithelium from alveolar and bronchiolar walls, which were thickened by the presence of edema fluid, fibrin, neutrophils, and mononuclear cells. Multifocally in nonconsolidated pulmonary tissue, there were a moderate number of macrophages and fewer neutrophils in the lumina of alveoli and bronchioles. The changes in bronchi, trachea, and larynx were relatively mild (Fig. 5). In multiple small areas the palisading arrangement of the epithelium was disrupted, there were vacuoles in and between epithelial cells, and a few neutrophils were present in the epithelium and the subjacent connective tissue. In other areas there was loss of epithelium to the basal cell layer or to the basement membrane, with infiltration of neutrophils in the underlying connective tissue. In the lumen there were a few neutrophils, macrophages, and sloughed epithelial cells.
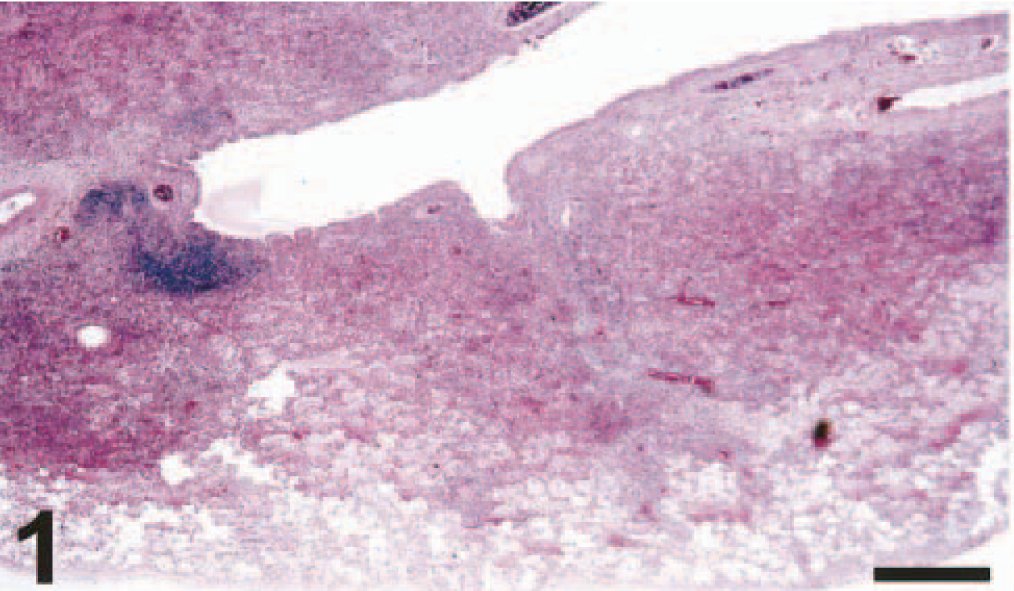
Lung; cynomolgus macaque No. 2. The bronchioles and alveoli are flooded with inflammatory exudate, with a peribronchial distribution (broncho-interstitial pneumonia). HE. Bar = 1,500 μm.
Lung; cynomolgus macaque No. 2. The alveolar architecture is obliterated by the presence of macrophages, neutrophils, erythrocytes, and protein-rich edema fluid in the alveolar lumina. HE. Bar = 25 μm.
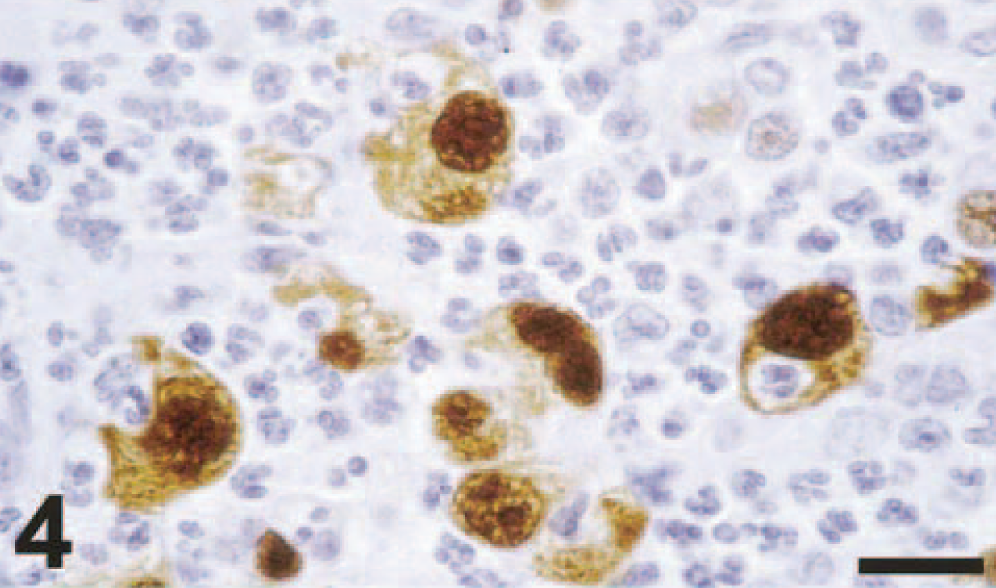
Lung (duplicate section of Fig. 3); cynomolgus macaque No. 2. There is positive nuclear and cytoplasmic staining of alveolar macrophages. Avidin–biotin–peroxidase complex method for nucleoprotein of influenza A virus counterstained with hematoxylin. Bar = 25 μm.
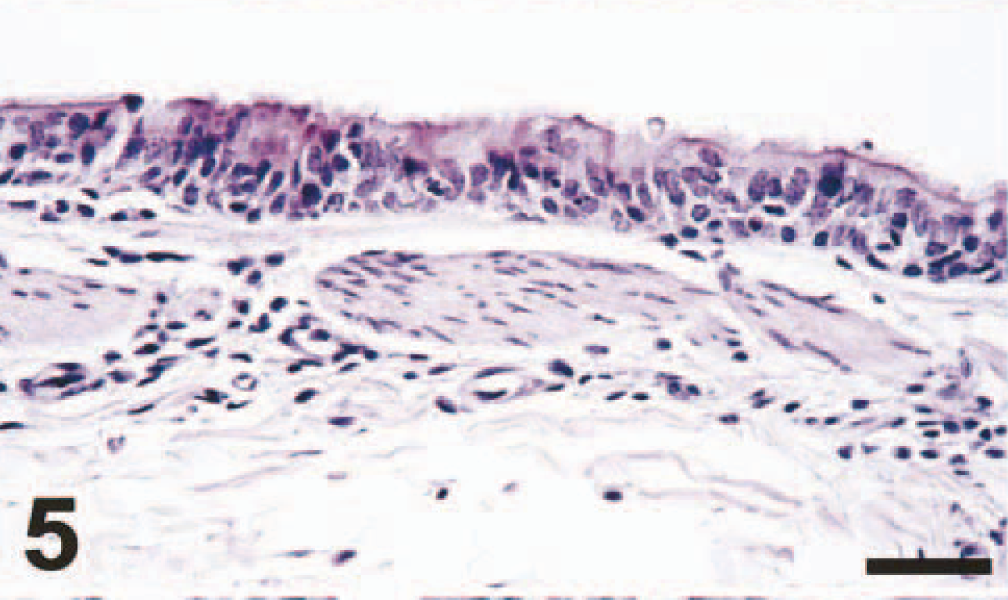
Bronchus; cynomolgus macaque No. 2. There is epithelial disorganization, loss of cilia, intra- and intercellular vacuolization, and infiltration of a few neutrophils in the epithelium and the subjacent submucosa (bronchitis). HE. Bar = 100 μm.
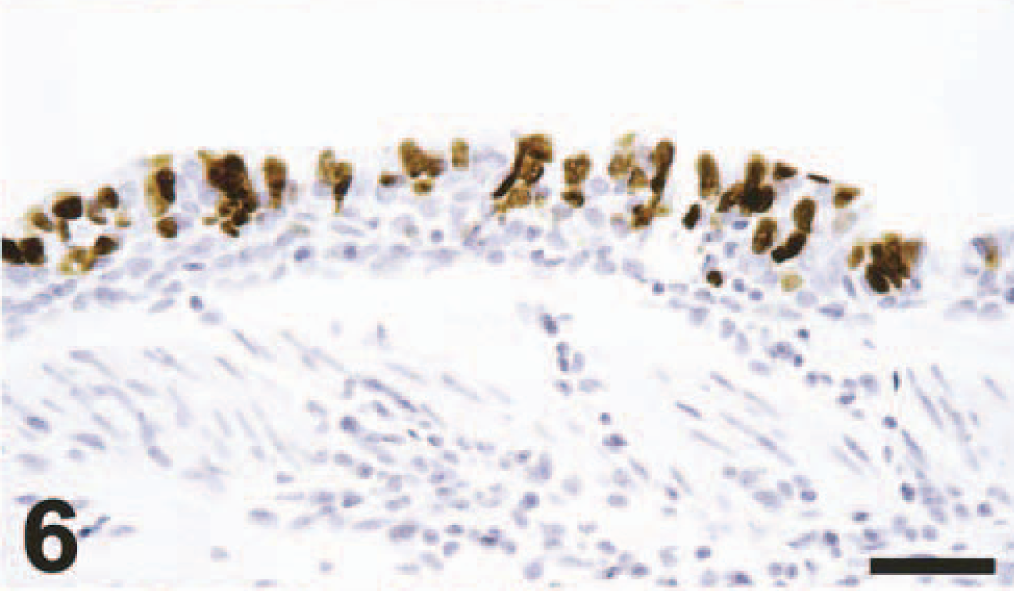
Bronchus (duplicate section of Fig. 5); cynomolgus macaque No. 2. There is positive nuclear and cytoplasmic staining of ciliated epithelial cells. Avidin–biotin–peroxidase complex method for nucleoprotein of influenza A virus counterstained with hematoxylin. Bar = 100 μm.
The main lesion in the consolidated pulmonary tissue of macaque Nos. 3 and 4, euthanatized at 7 dpi, again involved alveoli and bronchioles. The changes were similar to those found at 4 dpi, except that there were relatively more macrophages and lymphocytes and fewer neutrophils in the lumina, and there was a distinct eosinophilic, periodic acid–Schiff-positive layer (hyaline membrane), characteristic of diffuse alveolar damage, 4 apposed to the wall of some alveoli (Fig. 7). Also, alveolar walls in several areas were lined by small cuboidal cells with hyperchromatic nuclei (type 2 pneumocyte hyperplasia) (Fig. 8). There was regeneration of epithelium in some bronchioles, visible as a single layer of squamous to cuboidal cells with hyperchromatic nuclei. There were similar, but more extensive, changes in bronchi, trachea, and larynx compared with those found at 4 dpi. In addition to the areas of epithelial necrosis and infiltration with neutrophils, there were areas of epithelial regeneration, visible as multiple layers of basal cells under a single layer of columnar epithelial cells. There was marked edema and infiltration with moderate numbers of lymphocytes and plasma cells in the connective tissue around pulmonary blood vessels. The necrotic pulmonary tissue in macaque No. 4 consisted of a well-demarcated area of coagulation necrosis, involving bronchial, bronchiolar, and alveolar tissue, suggestive of a pulmonary infarct, although an intra-arterial thrombus was not found. The associated pulmonary blood vessels had a marked necrotizing vasculitis, characterized by intramural and perivascular infiltration by neutrophils, endothelial hypertrophy and necrosis, and perivascular edema.
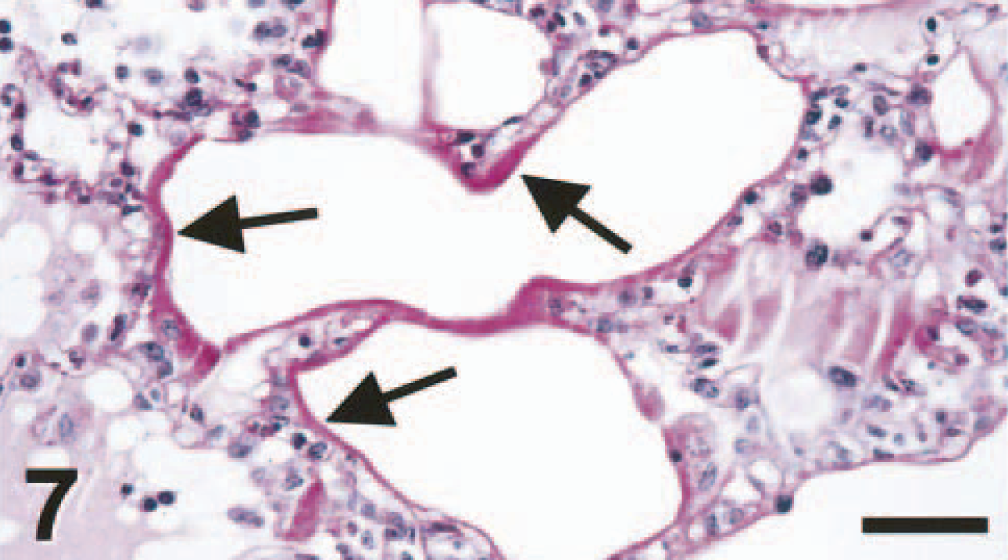
Lung; cynomolgus macaque No. 4. The alveolar walls are lined by a periodic acid–Schiff (PAS)-stained hyaline membrane (arrows). PAS. Bar = 100 μm.
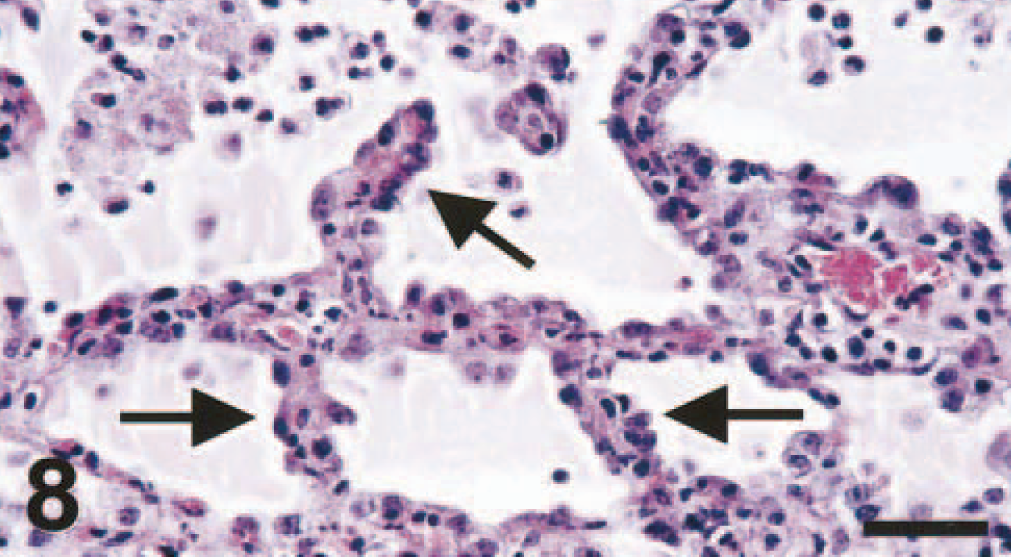
Lung; cynomolgus macaque No. 4. The alveolar walls are thickened and lined by cuboidal epithelial cells (arrows), indicative of type 2 pneumocyte hyperplasia. HE. Bar = 100 μm.
In macaque Nos. 3 and 4, lesions also were found in nonpulmonary tissues. Both macaques had a mild suppurative tonsillitis, characterized by the presence of sloughed epithelial cells, cell debris, fibrin, and a moderate number of neutrophils in the tonsillar crypts (Figs. 9–12). Macaque No. 4 had extensive lymphocytic necrosis in spleen, tonsil, and lymph nodes, visible as follicular depletion, karyorrhectic debris, and occasional pools of protein-rich edema fluid in germinal centers. In addition, macaque No. 4 had focal hepatic necrosis, consisting of an aggregate of hypereosinophilic pyknotic hepatocytes admixed with a few neutrophils, and multifocal renal tubular necrosis, with hypereosinophilic karyorrhectic tubular epithelial cells and intraluminal protein casts. No bacteria were seen in the HE- or Gram-stained sections of pulmonary tissue in any of the macaques. Evidence for neither hemophagocytic syndrome nor fat necrosis was observed in any of the tissues examined.
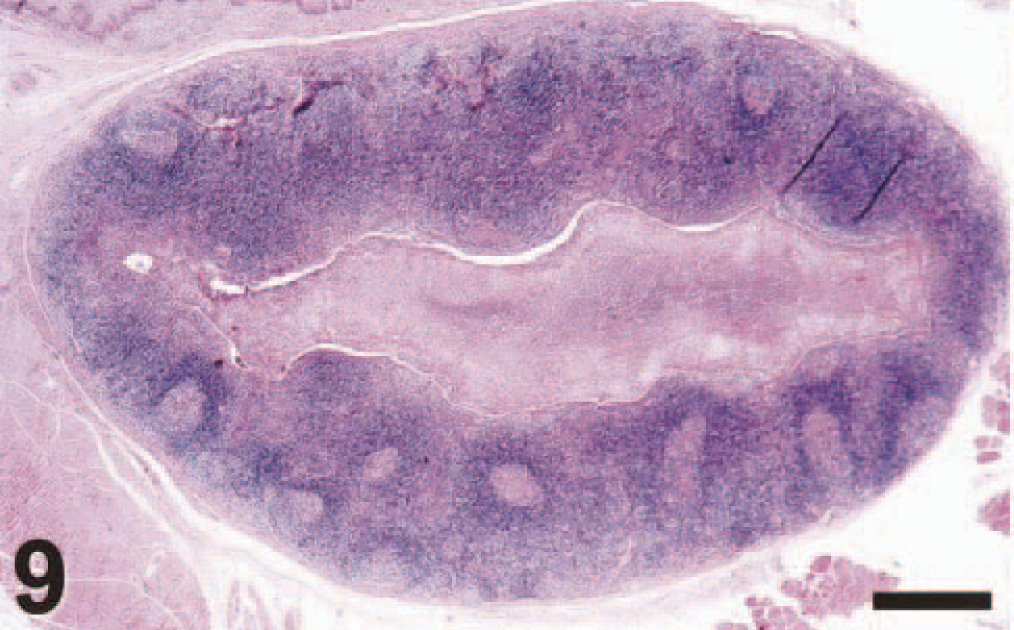
Tonsil; cynomolgus macaque No. 3. The tonsillar crypt is filled with inflammatory exudate, and the germinal centers are prominent (suppurative tonsillitis). HE. Bar = 100 μm.
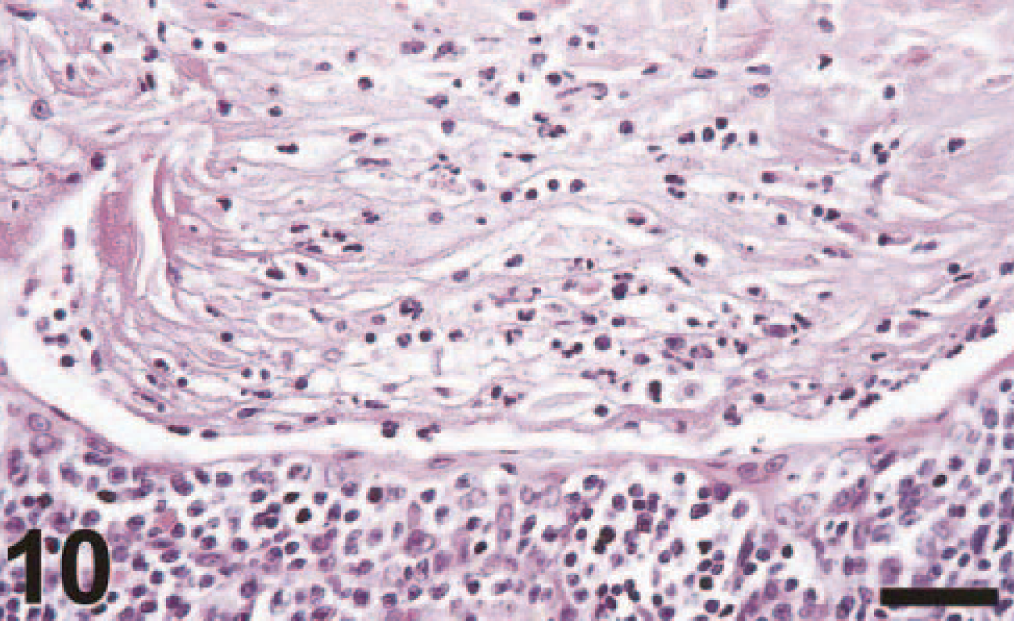
Tonsil (higher magnification of Fig. 9); cynomolgus macaque No. 3. The tonsillar crypt is filled with neutrophils, sloughed epithelial cells, and cellular debris (suppurative tonsillitis). HE. Bar = 100 μm.
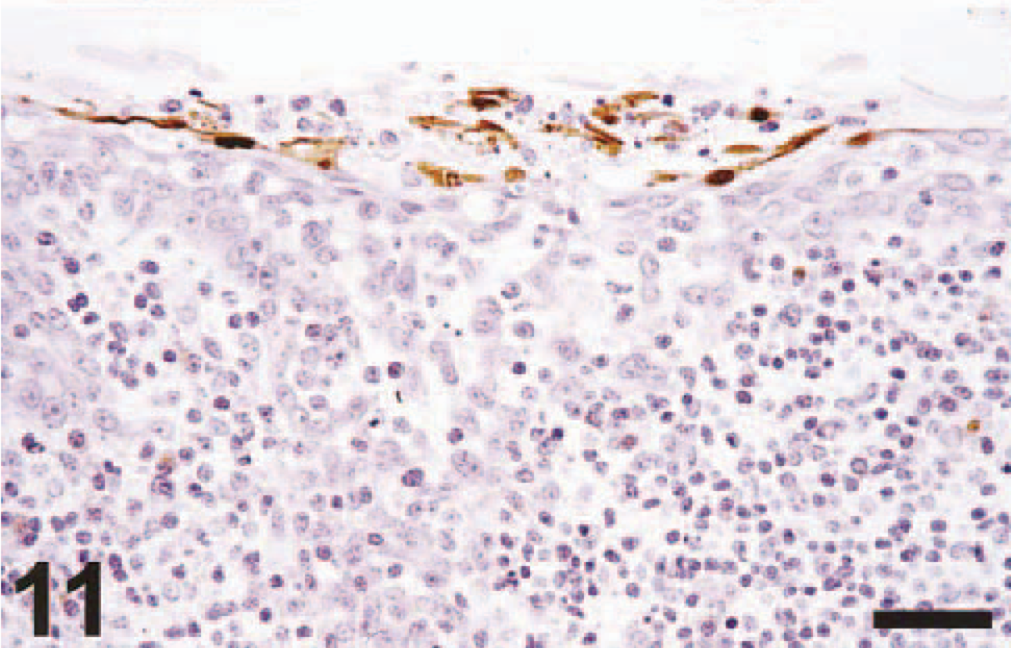
Tonsil; cynomolgus macaque No. 4. There is positive nuclear and cytoplasmic staining of cryptal epithelial cells, some of which are sloughed. Avidin–biotin–peroxidase complex method for nucleoprotein of influenza A virus counterstained with hematoxylin. Bar = 100 μm.
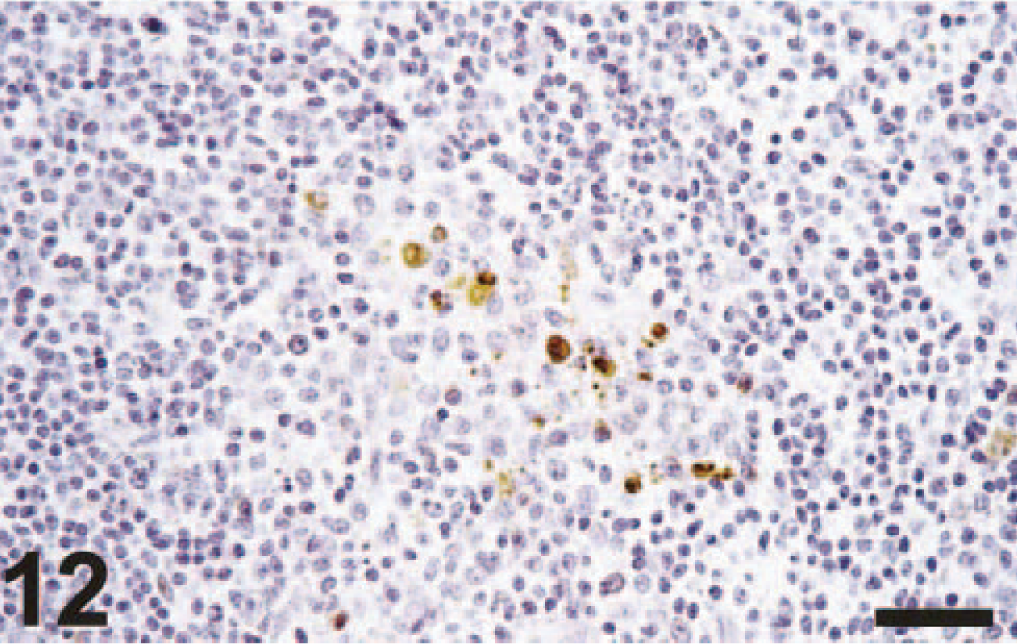
Tonsil; cynomolgus macaque No. 3. There is positive nuclear and cytoplasmic staining of dendritic cells in the germinal center. Avidin–biotin–peroxidase complex method for nucleoprotein of influenza A virus counterstained with hematoxylin. Bar = 100 μm.
Immunohistochemical findings
Positive staining in tissues of macaque Nos. 1 and 2, euthanatized at 4 dpi, was limited to the respiratory tract. A moderate number of alveolar macrophages, a few type 1 and type 2 pneumocytes, neutrophils, and unidentified mononuclear cells, and occasional cellular debris in the pneumonic areas stained strongly positive (Figs. 2, 4). Positive staining was visible as diffuse to granular brown staining, which was usually darker in the nucleus than in the cytoplasm. No positive staining was visible in normal lung tissue. Multifocally in bronchioles and in small and large bronchi, there was strong positive staining of occasional individual epithelial cells or groups of epithelial cells and rare intra-epithelial neutrophils (Fig. 6). The nucleus of epithelial cells stained darker than the cytoplasm. In some columnar epithelial cells, there was a band of dark brown staining subjacent to the cilia. The mucoid and basal cells of the bronchial epithelium did not stain positive. Rare sloughed epithelial cells, macrophages, and neutrophils in the bronchial lumen also stained positive.
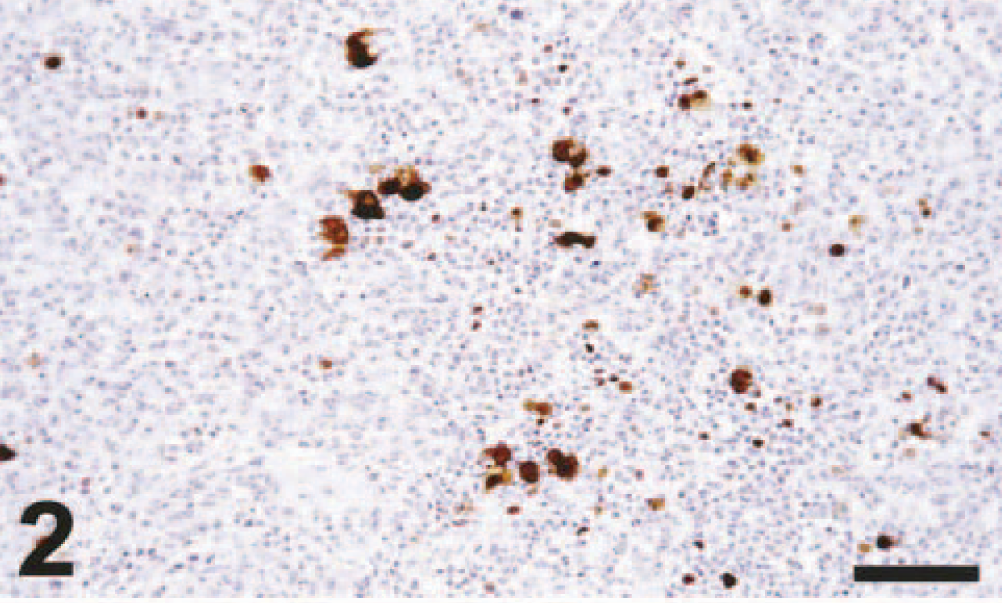
Lung (duplicate section of Fig. 1); cynomolgus macaque No. 2. There is abundant positive nuclear and cytoplasmic staining of cells in the pulmonary tissue. Avidin–biotin–peroxidase complex method for nucleoprotein of influenza A virus counterstained with hematoxylin. Bar = 200 μm.
Positive staining in tissues of macaque Nos. 3 and 4, euthanatized at 7 dpi, was limited to the respiratory tract and tonsils. Positive staining in the alveoli, bronchioles, and bronchi was similar to that seen at 4 dpi, except that fewer cells stained positive and staining was weaker. In the tonsils of both macaques, the nucleus and cytoplasm of a moderate number of cryptal epithelial cells, sloughed epithelial cells, and cell debris in the tonsillar crypt stained strongly positive (Fig. 11). The nucleus or cytoplasm, or both, of a few large dendritic cells in germinal centers of the tonsillar lymphoid tissue stained weakly to moderately positive (Fig. 12). No positive staining was observed in any of the other tissues, including the attached adipose tissue, of the macaques euthanatized at 4 or 7 dpi.
Discussion
The primary lesion of experimental H5N1 virus infection in macaques was a severe necrotizing broncho-interstitial pneumonia similar in character and severity to those found in primary influenza virus pneumonia in humans. 21 The virus etiology of this lesion was confirmed by positive staining for influenza virus in lesional tissues by immunohistochemistry (this study) and by virus isolation and reverse transcription–polymerase chain reaction procedures. 17 Bacterial superinfection was ruled out by the absence of visible bacteria in Gram-stained sections of pulmonary tissue and the histologic character of the pulmonary lesions. Had the cynomolgus macaques survived, the regenerative changes in their lungs observed at 7 dpi (Fig. 8) would likely have developed into the organizing diffuse alveolar damage with interstitial fibrosis seen in two humans who died 1 month after onset of H5N1 virus infection. 20 The failure to detect influenza virus in postmortem lung samples of those patients was likely due to the chronicity of the disease in combination with antiviral treatment.
Lesions in the extrarespiratory organs of experimentally infected macaques corresponded in part with those of humans with fatal H5N1 virus infection. Necrotic lesions in kidney, liver, spleen, and lymph nodes, without evidence of viral infection, were found both in macaque No. 4 and in two humans with fatal H5N1 virus infection. 20 These necrotic lesions are the pathologic correlates of MODS, which was a common clinical finding in humans with H5N1 virus infection. 23
However, none of the experimentally infected macaques had lesions suggestive of reactive hemophagocytic syndrome, which was the most prominent pathologic feature in the two autopsies of humans. 20 Virus-associated hemophagocytic syndrome is a benign disorder characterized by proliferation of cytologically benign hemophagocytic histiocytes in bone marrow, lymph nodes, spleen, liver, and leptomeninges in association with an acute systemic viral infection, 15 including influenza virus infection. 14 The failure to detect this syndrome in the macaques may be related to the shorter duration of their illness–a few days compared with 1 month in the humans with this syndrome.
The suppurative tonsillitis in macaque Nos. 3 and 4 was associated with the presence of influenza virus antigen in epithelial cells in the crypts and in dendritic cells in the germinal centers (Figs. 9–12). Infection of the tonsils with influenza virus may either have been primary or have spread from the respiratory tract through expulsion of infected sloughed epithelial cells. Influenza virus infection is known to cause tonsillitis in humans, 22 but to our knowledge, this lesion has not been described pathologically. To et al. 20 did not mention tonsillar lesions in their autopsy results of two humans with fatal H5N1 virus infection.
The pulmonary lesions in these cynomolgus macaques resembled those found in Burnet's report, 2 which is, to our knowledge, the only previous pathologic study of influenza in cynomolgus macaques. Burnet 2 infected 10 cynomolgus macaques by the intratracheal route with “W.S. egg (monkey passage),” an egg-adapted strain of human influenza virus A (H1N1), passaged through cynomolgus macaques. Five of these animals, killed at 2–3 dpi, had multifocal to extensive pulmonary consolidation. Two of the remaining five animals, in which the natural progression of the disease was followed, died at 3 and 6 dpi, with pulmonary consolidation involving almost the whole of the lungs, whereas the other three survived. Histologic findings consisted of a broncho-interstitial pneumonia, with necrosis of the bronchiolar epithelium and flooding of the alveoli with fibrinous exudate containing polymorphs and mononuclear cells.
Mice have been used extensively as a model of human infection with H5N1 virus. The same isolate, influenza virus A/Hong Kong/156/97 (H5N1), that we used in this macaque model was inoculated into BALB/c 8 and ddY mice 12 for histologic and immunohistochemical studies. BALB/c mice had a necrotizing broncho-interstitial pneumonia, with positive immunohistochemical staining for influenza A nucleoprotein predominantly in respiratory epithelial cells and no evidence of influenza virus antigen in visceral organs or brain. These results are similar to those in the macaques. It is interesting to note that in BALB/c mice at 4 dpi, influenza virus antigen was more prevalent in respiratory epithelium than in alveolar macrophages, whereas it was the reverse in the macaques. In contrast to the macaques, these mice had no lesions in extrarespiratory organs. In addition to respiratory tract lesions, ddY mice had fat necrosis in perivisceral and spinal fat, with positive staining of adipocytes, and mild encephalitis in brain stem and cerebrum, with positive staining of neurons, glial cells, and ependymal cells. Furthermore, positive staining without associated lesions occurred in cardiac myofibers and hepatocytes. These results differ from those in the macaques, in which we found neither lesions in brain or perivisceral fat nor positive staining in any extrarespiratory tissue except tonsil.
Why H5N1 virus infection caused such a severe disease in humans is not clear. Soon after the human cases had occurred in Hong Kong in 1997, Yuen et al. 23 speculated that H5N1 virus in humans could replicate in organs outside the respiratory tract. However, support for this hypothesis was provided neither by subsequent pathologic studies of humans by the same research group 20 nor by our experimental studies in macaques.
Alternatively, we suggest that virus replication in the respiratory tract and the resulting diffuse alveolar damage were the primary insult from H5N1 virus infection in humans and macaques and could explain the clinical signs and lesions in other organs in the absence of virus replication in extrarespiratory tissues. ARDS, which is the clinical correlate of diffuse alveolar damage, commonly progresses to MODS. 1 The hepatic, renal, central nervous, gastrointestinal, hematologic, and cardiac systems are most commonly affected. 7 MODS is a consequence of medical progress and has become unmasked because of the advances in critical care medicine. 6 Initial reports of MODS date from the 1970s, 6 which may explain why this syndrome is not mentioned in the reports of previous human influenza pandemics, the last of which occurred in 1968. 11
The pathogenesis of MODS has not yet been elucidated. The primary defects are thought to involve the microcirculation and mitochondrial metabolism, and mechanisms may include the release of cytokines into the circulation. 6 This is supported by the data of To et al., 20 who found elevated levels of interferon-γ, soluble interleukin-2 receptor, and interleukin-6 in sera of two humans with fatal H5N1 virus infection. We are currently investigating the role of different cytokines in the pathogenesis of H5N1 virus infection in our macaque model.
In conclusion, H5N1 virus infection in cynomolgus macaques caused a primary viral pneumonia and lesions in extrarespiratory organs similar to those in humans in Hong Kong in 1997. This resemblance suggests that the cynomolgus macaque is a suitable animal model for studying the pathogenesis of H5N1 virus infection in humans. Based on the lack of extrarespiratory virus replication in these macaques and in humans, we suggest that MODS in human H5N1 virus infection is caused by diffuse alveolar damage from virus replication alone. Further studies on the pathogenesis of ARDS and MODS resulting from H5N1 virus infection may help to prevent or treat these serious complications in future influenza outbreaks.
Footnotes
Acknowledgements
This study was partly funded by the Foundation for Respiratory Virus Infections, notably Influenza (SRVI). We thank Frank van der Panne and Frida van der Ham for excellent technical assistance.
