Abstract
The purpose of this study was to evaluate the effects of estrogen and progesterone on the vaginal mucosa and their role in vaginal transmission of simian immunodeficiency virus. Incidentally, endometrial hyperplasia was observed in estrogen-treated monkeys at necropsy. Six adult female rhesus monkeys (Macaca mulatta) were ovariectomized and 120 days later received two subcutaneous implants, each containing 200 mg estradiol. The animals were sacrificed 17–27 months later and the uterus examined at necropsy. All the monkeys had simple endometrial hyperplasia, some with polyps or adenomyosis, at the time of necropsy. The severity of the changes correlated with the time between implantation and necropsy. The lesions were similar to endometrial hyperplasia caused by unopposed estrogen in women, but occurred over a time period that is suitable for experimental manipulation. Rhesus monkeys could be used as a model to test the safety of various combinations of sex steroids for the prevention of postmenopausal symptoms in women.
The endometrium of both human and nonhuman primates responds to changes in levels of steroid sex hormones by modifying its structure. Although the normal anatomic changes that occur during reproductive life are well known in women14 and macaque monkeys,1,5,13 the effects of various combinations of exogenous hormones can be unpredictable. Unusual combinations of proliferative, secretory, and atrophic changes involving the epithelium, stroma, and vessels of the endometrium can occur, depending on the particular hormones administered, as well as the dosage and duration of therapy.7
In normally cycling women, shedding of the endometrial lining during menstruation is followed by estrogen-induced endometrial proliferation. Glands, stroma, and blood vessels actively grow for about 2 weeks, until ovulation occurs. After ovulation, progesterone inhibits endometrial proliferation and induces secretory activity. After menopause, the endometrium usually becomes inactive and eventually atrophic. Without ovulation, no progesterone is produced. Estrogen, however, may continue to be produced after menopause because of the conversion of androgens secreted by the ovaries and adrenal cortices into estrogens. Metabolic disorders such as obesity and diabetes may enhance extragonadal estrogen production by aromatization of androgens. The endometrium may respond to prolonged estrogen stimulation unopposed by progesterone with hyperplasia and possibly neoplasia. Anovulatory cycles in younger women may produce a similar result.7
Increasingly, postmenopausal women are being treated with hormone-replacement therapy to relieve postmenopausal vasomotor symptoms and to prevent urogenital atrophy, osteoporosis, and possibly cardiovascular disease. Initially, estrogens were used for this purpose. It eventually became clear, however, that prolonged administration of unopposed estrogen was associated with endometrial hyperplasia, endometrial polyps, and Type I (endometrioid) endometrial carcinoma.2,7,11,17 A relationship between estrogen, endometrial hyperplasia, and endometrial cancer has been suspected for over 50 years,11 and numerous studies have documented an increased risk of well-differentiated adenocarcinoma of the endometrium with unopposed estrogen.4 Because of these undesirable effects, lower dosages of different estrogens combined with continuous or intermittent progesterone are now recommended for this purpose. Surprisingly, unopposed estrogen is still used by some physicians today.19
Although reducing the dose of estrogen will lower the risk of hyperplasia and carcinoma, reducing the dosage may also reduce the beneficial effects on the physical and psychological symptoms of menopause and on the conservation of bone mass. However, recent evidence suggests that long-term use of lower dosages of estrogen may be a beneficial alternative to standard doses.10
The reproductive tracts of female macaque monkeys are anatomically and physiologically similar to those of women, although there are some differences. Female macaques typically undergo menarche at approximately 2.5–3 years of age, although the initial menstrual cycles are irregular and often anovulatory.15 Macaques reach menopause sometime in their mid to late 20s.20,21 In addition, rhesus monkeys (Macaca mulatta) typically display seasonal variation in their reproductive cycles, showing less reproductive activity during the summer months.6
Surveys of the spontaneous pathology of the macaque female reproductive tract have not described endometrial hyperplasia.3,8,9 We have not observed spontaneous endometrial hyperplasia in several thousand necropsies of adult female rhesus monkeys from the breeding colonies at the Tulane Regional Primate Research Center. Endometrial changes have been induced in macaques by experimental exposure to a variety of hormonally active agents.3 We report here simple endometrial hyperplasia, adenomyosis, and endometrial polyp formation in ovariectomized rhesus monkeys exposed to exogenously administered estrogen.
Materials and Methods
Six adult female rhesus monkeys (Macaca mulatta) between 7 and 20 years old and weighing 5–9 kg underwent bilateral ovariectomy as part of a simian immunodeficiency virus (SIV) transmission study.18 Two months later two 30-mm silastic implants, each of which contained 200 mg of estradiol-17β, were implanted subcutaneously in each monkey. Estradiol levels were measured 2 and 9 months after implantation, as described previously.18 In brief, serum was separated from clotted blood and was stored at −70 C until assayed. Hormone assays were performed by Dr. David Hess at the Oregon Regional Primate Research Center using previously described methodology.12 The limits of detection for the progesterone and estrogen assays were 0.10 ng/ml and 5 pg/ml, respectively. The monkeys were sacrificed between 16 and 27 months after implantation with estrogen, and complete necropsy examinations were done. All studies received prior approval from the Institutional Animal Care and Use Committee of the Tulane Regional Primate Research Center and were conducted in compliance with the Guide for the Care and Use of Laboratory Animals and with Public Health Service Policy.
Results
Progesterone was not detected in the serum of any monkey. Estradiol levels are given in Table 1.
Effects of unopposed estrogen on the endometrium of rhesus monkeys.

At necropsy, the uteri of all six monkeys were enlarged two to three times normal, measuring between 2.5 and 7.0 cm in length (measured from the anterior tip of the uterus posterior to the beginning of the cervix) and 2.0–5.0 cm in diameter. The endometrium was between 1 and 17 mm thick (Table 1), whereas the endometrium in untreated ovariectomized monkeys was about 1 mm thick. In all cases, the epithelial surface was smooth and glistening. The endometrium was flat in monkey Nos. 1 and 2, but was thrown into convoluted folds and polypoid projections in the other monkeys. A polyp with a fibrous core was present in monkey No. 4. The thickened endometrium contained a few to numerous round cysts that were up to about 1 mm in diameter in all monkeys. The cysts contained clear fluid (Fig. 1). Dark-red blood filled the lumen of the uterus of monkey No. 1. The endometrial epithelium was focally denuded in this animal.
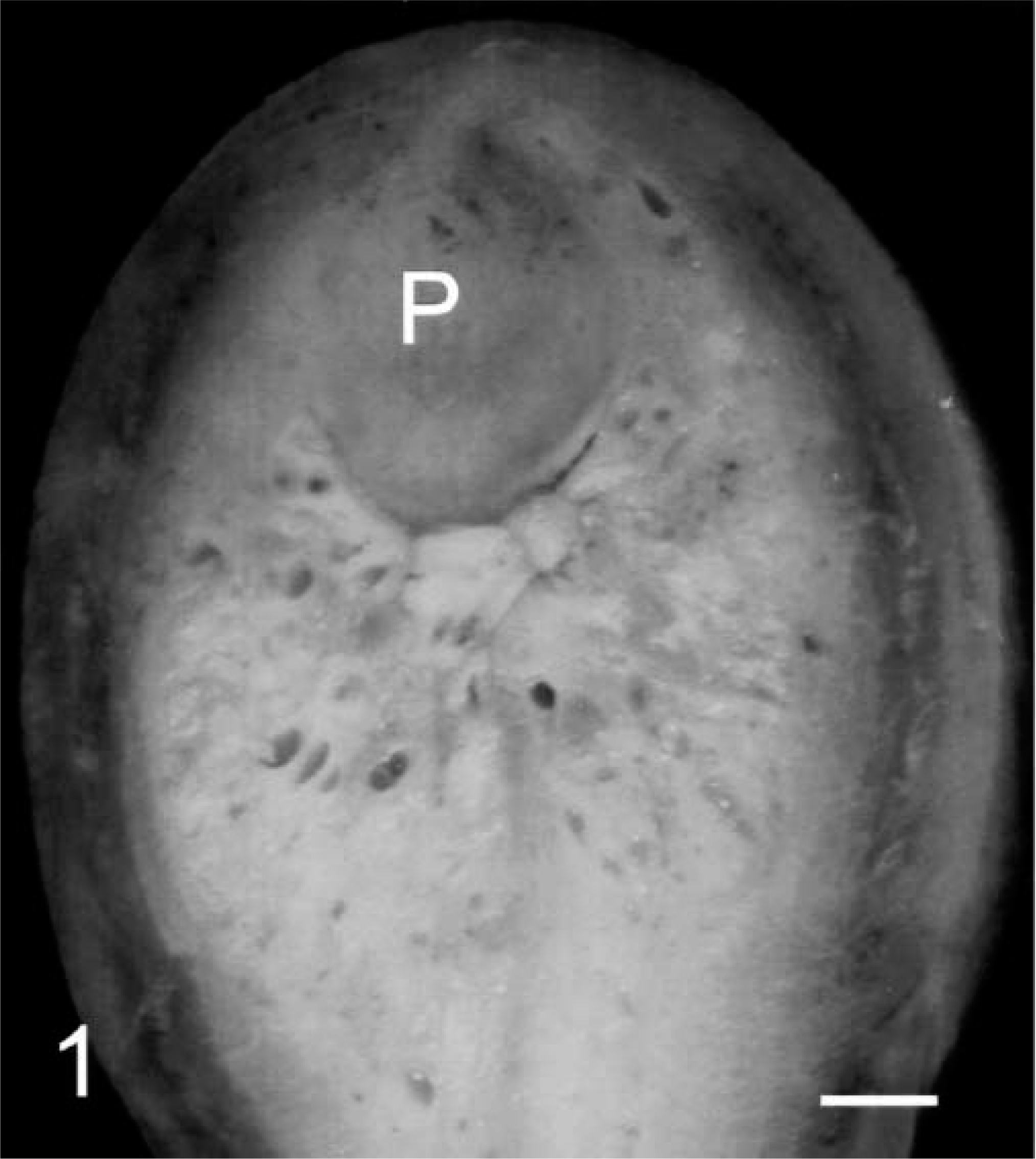
Endometrial hyperplasia. Sagittal section of the uterus of monkey No. 4. Note the thick endometrium, endometrial cysts, and endometrial polyp (P). Bar = 0.33 cm.
Microscopically, there was proliferation of glands and stroma (Fig. 2). The glands varied in density but were never crowded. Most were round and were often cystically dilated. A few appeared to have rudimentary outpouchings, but these could have been the result of collapse. The glandular epithelium was simple columnar to pseudostratified columnar without overt vacuolation. The lumens of the glands were often empty, but some contained amorphous secretion.
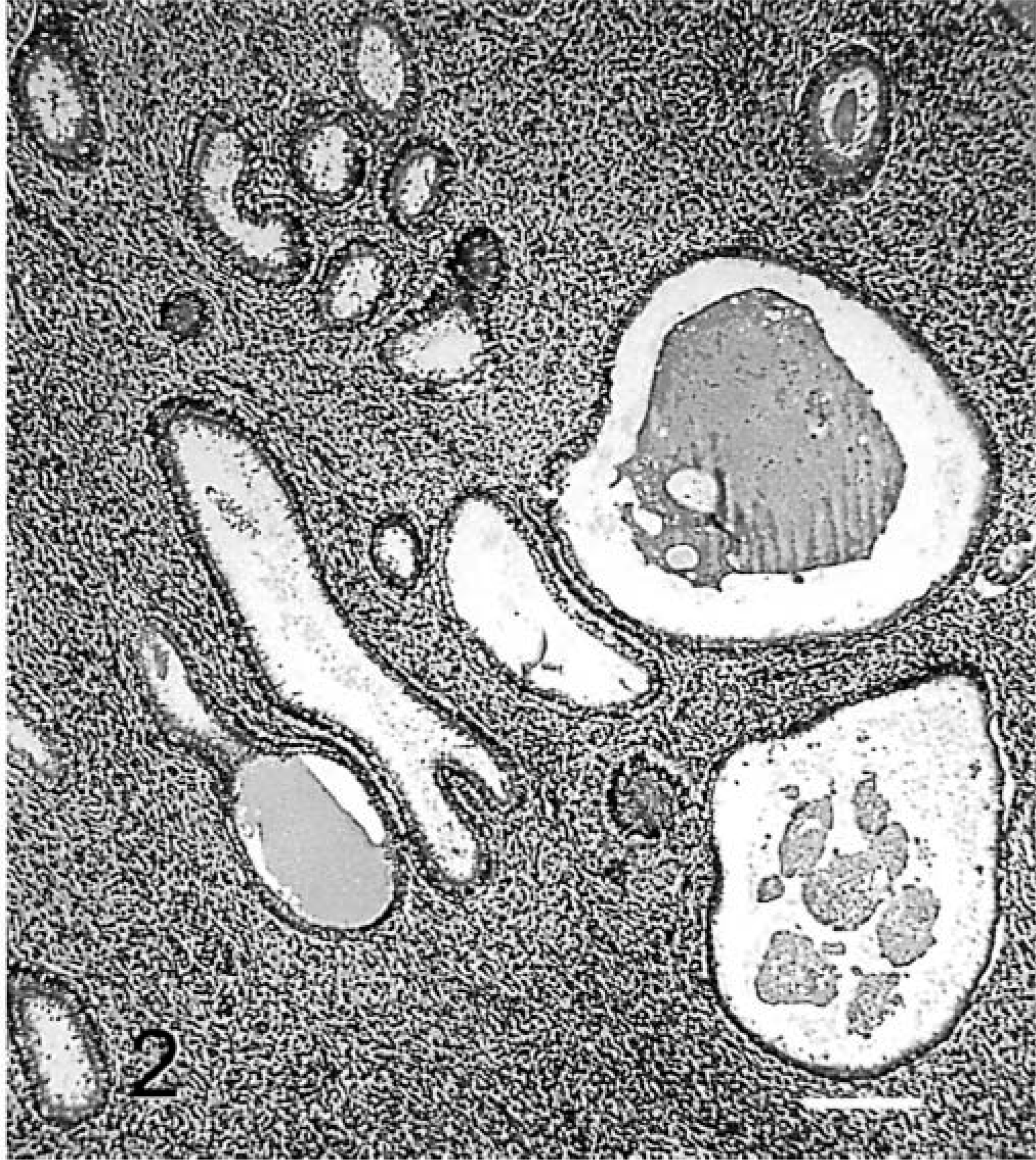
Endometrial hyperplasia. Histologic section of endometrium of monkey No. 4. Note cystically dilated round endometrial glands and stromal proliferation. Bar = 200 μm. (HE, 100×).
The endometrial stroma was diffusely cellular and generally had evenly distributed small vessels. The density of stromal nuclei varied because some areas were quite edematous, whereas other areas were not. There were a few mitotic figures in the stroma. Several small foci of adenomyosis, characterized by glands deep within the myometrium, were observed in monkey No. 2. There was no evidence of epithelial atypia or of stromal invasion. In some areas there were proliferated, thick-walled, spiral arteries.
Discussion
The study was conducted to evaluate the effects of estrogen and progesterone on the vaginal epithelium and on the rate of transmission and viral replication after intravaginal inoculation of SIV.18 Rhesus monkeys develop a markedly thickened vaginal epithelium in response to estrogen, whereas the epithelium becomes very thin in the absence of estrogen stimulation. The thickened epithelium effectively blocks vaginal transmission of SIV.18
The endometrial changes associated with unopposed estrogen appear to be quite similar in monkeys and humans, except that the monkey endometrium may develop hyperplasia within a shorter time period. The endometrial changes associated with unopposed estrogen in humans are partially dependent on the length of exposure.7 In our monkeys, endometrial hyperplasia was present 16 months after estradiol implants were inserted, when the first monkey was examined at necropsy. The monkeys we studied had estradiol levels from 89 to 897 pg/ml, whereas progesterone was not detectable. These estrogen levels are within the physiologic range, although the exposure was continuous and unopposed by progesterone. We know neither the minimum level of estradiol nor the minimum time period required to induce endometrial hyperplasia in rhesus monkeys. It is apparent from Table 1 that the severity of endometrial hyperplasia in these monkeys correlated with the length of exposure, but not with the level of serum estradiol. The rhesus macaque is a useful model for studying these factors. Data from humans suggest that most hyperplasias without atypia probably represent early, reversible lesions.
We did not observe any nuclear atypia, complex hyperplasia, or stromal invasion in the monkeys we studied. We do not know whether these changes or overt endometrial carcinomas can be induced in rhesus monkeys with unopposed estrogen over a longer time period. The risk of developing endometrial carcinoma associated with exogenous estrogen use in postmenopausal women is related to duration of exposure, with approximately 10-fold increases associated with each decade of use.16 Additional longer-term studies in rhesus monkeys might result in tumor formation. If so, this would be a relevant model in which to study the pathobiology of some types of endometrial carcinoma.
In summary, we have described the induction of simple endometrial hyperplasia, adenomyosis, and endometrial polyp formation in ovariectomized rhesus monkeys with exogenous estrogen unopposed by progesterone. Investigators using female macaques as a model for mucosal transmission studies of viruses should be aware of this long-term complication. In addition, the rhesus monkey should be a useful model for the study of various dosages and combinations of hormonally active drugs and other substances being developed for use in women and could possibly be developed as a model for the pathogenesis of endometrial carcinomas associated with unopposed estrogen.
Footnotes
Acknowledgements
This work was supported by Grants AI-41952 and RR-00164 from the National Institutes of Health and the Peggy Levitz Fund of St. Michael's Medical Center.
