Abstract
Neurocutaneous melanosis (NCM) is a rare phakomatosis characterized by proliferation of melanin-producing cells in both the skin and the brain. In this study, we describe the clinical and pathologic features of NCM in a 4.5-year-old female cynomolgus macaque. Histopathologically, skin lesions showed foci of nests and cords of pigmented cells in the dermis similar to blue nevi in humans. In the brain, focal pigmented cell infiltration was observed in the connective tissue under the leptomeninges and in the brain parenchyma. The pigmented cell was moderately reactive with a pan-melanoma antibody (melanoma(pan)) in the skin. In the brain, the pigmented cell was moderately to strongly positive for melanoma(pan) in subleptomeningeal areas and in the cerebral cortex. Melanosomes were observed in pigmented cells in the brain by electron microscopic examination. Based on the histologic, immunohistochemical, and electron microscopic results, the diagnosis of NCM was made. This case is possibly the first report of the condition in animals.
Neurocutaneous melanosis (NCM) is a rare phakomatosis characterized by proliferation of melanin-producing cells in both the skin and the brain. 4–7 At present about 100 human cases have been reported in the literature. Most cases are sporadic and no gender or racial predilection is evident. Kadonaga and Frieden 5 in 1991 proposed clinical criteria for the diagnosis of NCM. 1) The presence of large (one of which is at least 20 cm in diameter) or multiple melanocytic nevi with benign (melanosis) or malignant (melanoma) central nervous system (CNS) tumors. 2) The absence of malignant melanoma in any organ (including skin) other than in the CNS. 5 Two-thirds of patients with NCM have giant congenital melanocytic nevi, and the remaining one third have numerous lesions but no giant lesions. A survey of the literature, to the best of our knowledge, does not document evidence of NCM in any species of animal. We have necropsied nearly 2,000 cynomolgus monkeys at our facility and this was the first time we have seen these histopathologic lesions. In this article, we describe the histologic, immunohistochemical, and ultrastructural characteristics of the first case of spontaneous NCM in a cynomolgus macaque.
Clinically and a thorough gross examination of the skin showed multiple black–blue pigmented areas in a generalized distribution over the body of a 4.5-year-old female cynomolgus macaque, with a diameter from approximately 0.5–3.0 cm with areas of normal skin interspersed. The monkey was provided by the Academy of Military Medical Sciences affiliated with the Chinese People's Liberation Army. The major organs, including liver, spleen, lung, kidney, adrenal gland, urinary bladder, and skin, were obtained for histopathologic examination by Motac Collaborative Laboratory, Institute of Laboratory Animal Science, Chinese Academy of Medical Science. Neuropathologic gross examination showed pigmentation coating the convexities, base of the brain, the interpeduncular fossa, the ventral surface of the pons, medulla, and cerebellum without any mass. The entire brain was almost black. 1,5,9 This finding was similar to those in human patients. Liver, spleen, lung, kidney, adrenal gland, and urinary bladder had a grossly normal appearance.
Histopathologic findings in the skin lesions showed nests and cords of pigmented cells in the dermis (Fig. 1), which is identical to that seen with blue nevi. These cells were heavily pigmented, spindle or polygonal (Fig. 2). Nuclei were oval and uniform, with fine chromatin. In addition, elongated pigmented cells were also observed within the deep dermis. No other skin abnormality was noted. Although the proliferation of pigmented cells could be observed in the skin of normal monkeys, these lesions do not involve the entire body as it did in our macaque. The histologic appearance of the brain lesion was focal pigmented cell infiltration in the connective tissue under the leptomeninges and in the brain parenchyma (Fig. 3). Pigmented cells were arranged either in small clusters or occurred individually and, in some places, were parallel to the surface (Fig. 3). Within the brain parenchyma, Virchow-Robin spaces were infiltrated by pigmented cells of spindle (Fig. 4) or polygonal pattern, which would be classified as S-type or spindle lesions according to the typical histopathologic features of patients with NCM, as proposed by Kadonaga and Frieden. 5 Proliferation of pigmented cells was diffuse under leptomeninges and in multiple sites in the brain parenchyma, which differed from small clusters of melanin-containing cells in meninges and Virchow-Robin spaces in the normal cynomolgus monkey. No other histologic features of malignancy, including cellular pleomorphism, nuclear atypia, hemorrhage, or necrosis, were noted. All of these findings are consistent with those in human patients with NCM.
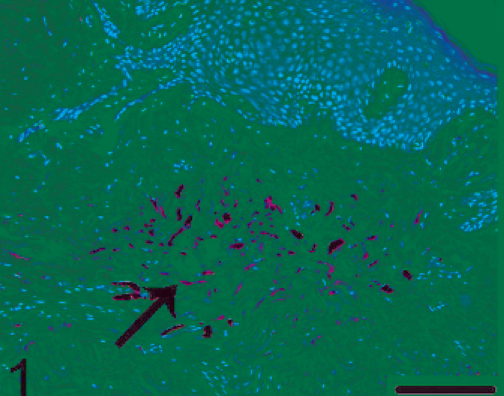
The blue nevus in the skin. Focal pigmented cell proliferation was observed in the dermis (arrow). HE. Bar = 120 µm.

The blue nevus in the skin. The blue nevus consisted of nests of well-differentiated spindle and polygonal pigmented cells (arrow), most of which contained variable amounts of granular, brown to black intracytoplasmic pigment. HE. Bar = 60 µm.
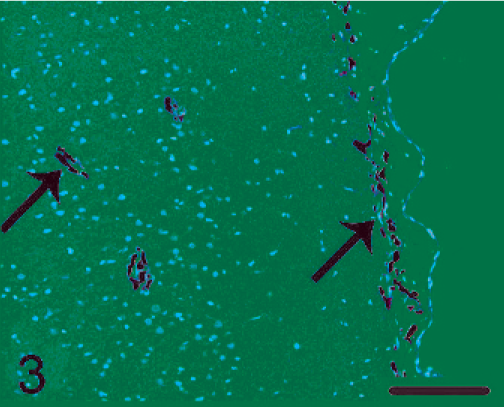
Brain. Pigmented cell proliferation was observed in the brain parachyma (left arrow) and under the leptomeninges (right arrow). HE. Bar = 120 µm.
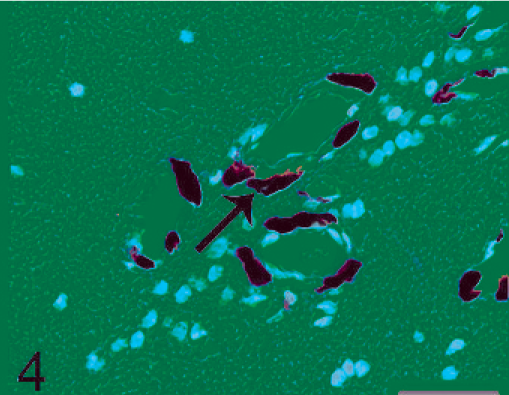
Brain. Pigmented cell proliferation in the brain parachyma. The microscopic appearance of the parenchyma was that of pigmented cell proliferation of spindle pattern within the Virchow-Robin spaces. Pigment could be easily observed (arrow). HE. Bar = 30 µm.
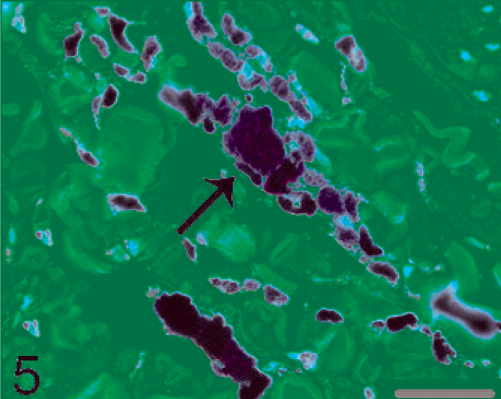
Skin. Pigmented cells indicated by the arrow reacted moderately with melanoma(pan) antibody in the dermis. Immunohistochemistry, melanoma(pan), hematoxylin counterstain. Bar = 30 µm.
To increase the specificity of the histopathologic diagnosis, tissue samples were also studied with immunohistochemical staining. Immunohistochemistry was performed according to the following method. Deparaffinized sections were incubated with 3% hydrogen peroxide for 20 minutes to block endogenous peroxidase activity. After microwave retrieval for 10 minutes, slides were allowed to cool at room temperature, rinsed in deionized water and washed in phosphate-buffered saline. The tissues were blocked with normal goat serum for 20 minutes, followed by incubation with the primary antibody overnight at 4°C. Then the slides were incubated with the anti-mouse second antibody (Ready-to-use, catalog Number: PV-6002; Zhongshan Biotechnology Co., Beijing, China) for 30 minutes at room temperature. To avoid confusion between the melanin pigment and the brown staining of the substrate, a red chromogen, 3-amino-9-ethylcarbazole (AEC, catalog number: ZLI-9036, Zhongshan Biotechnology Co.), acted as substrate for visualization. Sections were counterstained with Harris's hematoxylin.
The pigmented cells were moderately immunopositive for a pan-melanoma antibody (melanoma(pan)) (Ready-to-use, clone number: M2-7C10+M2-9E3+MAQ02; Maixin-Bio Co., Fuzhou, China) in skin (Fig. 5). It is an anti-human melanoma(pan) antibody but can cross-react with monkeys. The melanoma(pan) is combination of HMB45, MART-1, and tyrosinase, which is a useful marker for labeling melanin. 10 In the brain, pigmented cells were moderately to strongly positive for melanoma(pan) under the leptomeninges (Fig. 6) and in the cortex (Fig. 7). The electron microscopic examination revealed electron-dense ellipsoid and spheroid melanosomes that measured 0.1–0.5 µm in the pigmented cells in the frontal lobe of the brain (Fig. 8). In addition, the red immunoreaction was observed in the neurons of the normal brain tissue, but not in the cell with heavily pigment when the neurofilament antibody acted as primary antibody (data not shown), which further confirmed our result.
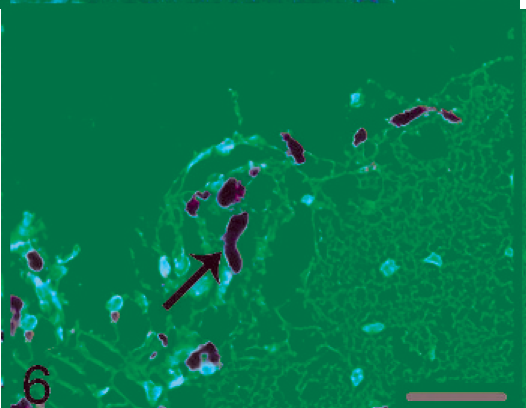
Brain. Melanoma(pan) staining was strongly positive in pigmented cells indicated by the arrow under the leptomeninges Immunohistochemistry, melanoma(pan), hematoxylin counterstain. Bar = 30 µm.

Brain. Melanoma(pan) staining was strongly positive in the pigmented cells (arrow) in the Virchow-Robin spaces. Immunohistochemistry, melanoma(pan), hematoxylin counterstain. Bar = 30 µm.
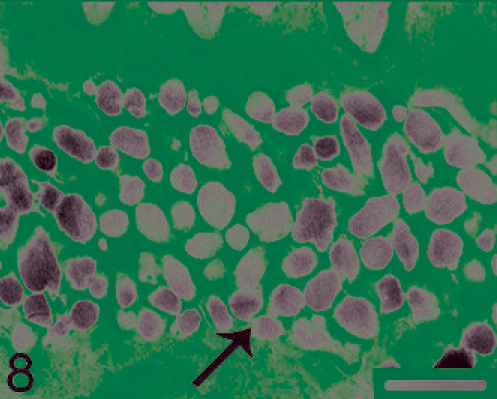
Frontal lobe of the brain. Ellipsoid and spheroid melanosomes (arrow) of varying size. Electron microscopy, uranyl acetate and lead citrate. Bar = 1 µm.
Most previous research has focused on clinical manifestations and magnetic resonance imaging of human NCM, with the histopathology rarely described. 2,3,8,9,11 However, confirmation of the diagnosis of NCM is still based on the histologic findings. Here, we describe the pathologic, immunohistochemical, and ultrastructural characteristics of spontaneous NCM in a cynomolgus macaque, which has not previously been described. The details in this report will be helpful in making a histopathologic diagnosis of NCM in monkeys, and also provides some important clues for the diagnosis of NCM in other species.
Footnotes
Acknowledgements
We thank Dr. Huan Zhao for technical assistance. We also thank The Chinese Public Fund Project of Institute of Laboratory Animal Science (DWS 200701), The Chinese Academy of Medical Sciences, and The Peking Union Medical College for financial support.
