Abstract
The almost simultaneous initial detections of avian influenza A H5N1 viruses in central Europe in February 2006, at a time devoid of migratory bird activity, raised the question of the origin of these viruses. This report presents molecular data from Europe providing evidence for multiple and spatially overlapping H5N1 introductions into Bavaria, Germany.
Since 2003, highly pathogenic avian influenza A (HPAI) subtype H5N1 viruses have spread and become endemic in several Southeast Asian countries, 3 , 5 , 7 , 8 , 10 causing more than 200 human infections with fatal outcomes in more than half of them. In 2005, a novel epidemiologic trend was observed for the first time when thousands of wild birds died at Qinghai Lake in central China, a congregational place for hundreds of thousands of migratory birds. By August 2005, HPAI outbreaks in poultry were reported along migratory bird flight routes in northwest China, Russia, and Kazakhstan. It was feared that the virus could spread further west toward Europe via migratory birds. This became evident when HPAI subtype H5N1 was detected in poultry and wild birds in Turkey, Romania, and Croatia during October 2005. Thereafter, coinciding with the end of the migratory bird season, there were no further reports from countries further west, and it was widely believed that central Europe would not be in danger until the following spring, when migratory birds were expected to return from their winter quarters in Africa.
It was therefore quite unexpected when, in the absence of migratory bird activity, the first HPAI detections were almost simultaneously reported during 1 week of mid-February 2006 in Germany, Austria, and France. Hence, the role of migratory birds was questioned, as well as that of possible illegal imports of bird products from other affected countries as an explanation for these almost simultaneous reports. In an effort to elucidate the origin and the relatedness of HPAI viruses, the molecular phylogeny of the hemagglutinin and neuraminidase genes of 21 H5N1 HPAI samples from 10 different species of dead wild birds from throughout Bavaria were analyzed. Bavaria represents the largest state of Germany, covering 70,547 km2 in central Europe. The data were compared with those of HPAI viruses from neighboring and other countries involved in the current avian flu pandemic.
For this study, the Bavarian State Ministry of the Environment, Public Health and Consumer Protection coordinated the collection of dead birds, which were screened at the Bavarian Health and Food Safety Authority in Erlangen and Oberschleissheim for the presence of influenza A in tracheal swabs and internal organs. RNA extractions using Trizol reagent a were done according to the instructions of the manufacturer and were followed by a real-time screening PCR for the matrix protein gene using the OneStep RT-PCR kit b with primers, probes, and cycling conditions according to Spackman et al. 9 Positive cases were further examined with H5 and N1 subtype-specific real-time PCR for which primer and probe sequences as well as the cycling conditions were provided by the German Reference Center for Avian Influenza (Friedrich Loeffler Institute, Boddenblick 5a, 17493 Greifswald-Insel Riems, Germany, unpublished data, available from the Reference Center upon request). Between the first case in February 2006 and the last H5N1 detection in May 2006, 7,825 birds were screened. Out of 93 (1.2%) influenza A virus positive birds, 74 (0.9%) tested positive for the H5N1 subtype (Table 1). For 21 of these birds for which additional material was available and which were chosen to best possibly represent the affected host species and their geographic distribution, the complete coding sequences of their hemagglutinin and neuraminidase genes were determined by 4 and 3 PCR amplifications, respectively, targeted to overlapping gene sections. These newly developed primers (Table 2) could also be used for directly sequencing the PCR products, thus facilitating the simultaneous determination of the entire coding regions without subcloning. In detail, after reverse transcription with random primers, a PCR reactions were completed in a total volume of 50 μl containing 2.5 units of HotStar Taq DNA polymerase (Qiagen), 250 μM of each dNTP and 1 μM of each primer. Amplifications were performed with an initial 14.5 minute Taq polymerase activation step at 95°C followed by 50 cycles of denaturation at 95°C for 1 minute, annealing at 45°C for 1 minute, and elongation at 72°C for 1 minute. PCR products were purified from agarose gels using the QIAquick Gel Extraction kit b and sequenced using the same primers. All polymorphic sites were confirmed by at least 1 additional DNA sequence originating from an independent PCR amplification to rule out PCR polymerase artifacts. Sequences were aligned using the CLUSTAL W algorithm (http://www.ebi.ac.uk/clustalw), and phylogenetic analysis was done using the PHYLIP phylogeny package (version 3.5c) 2 employing the maximum parsimony method using the DNAPARS algorithm, with gaps counted as 1 event each. Support for phylogenies was measured by bootstrapping 1,000 replicates with the programs SEQBOOT and CONSENSE. Trees were drawn with the programs RETREE and DRAWGRAM from the same PHYLIP package.
Summary of influenza A/H5N1 screening results.
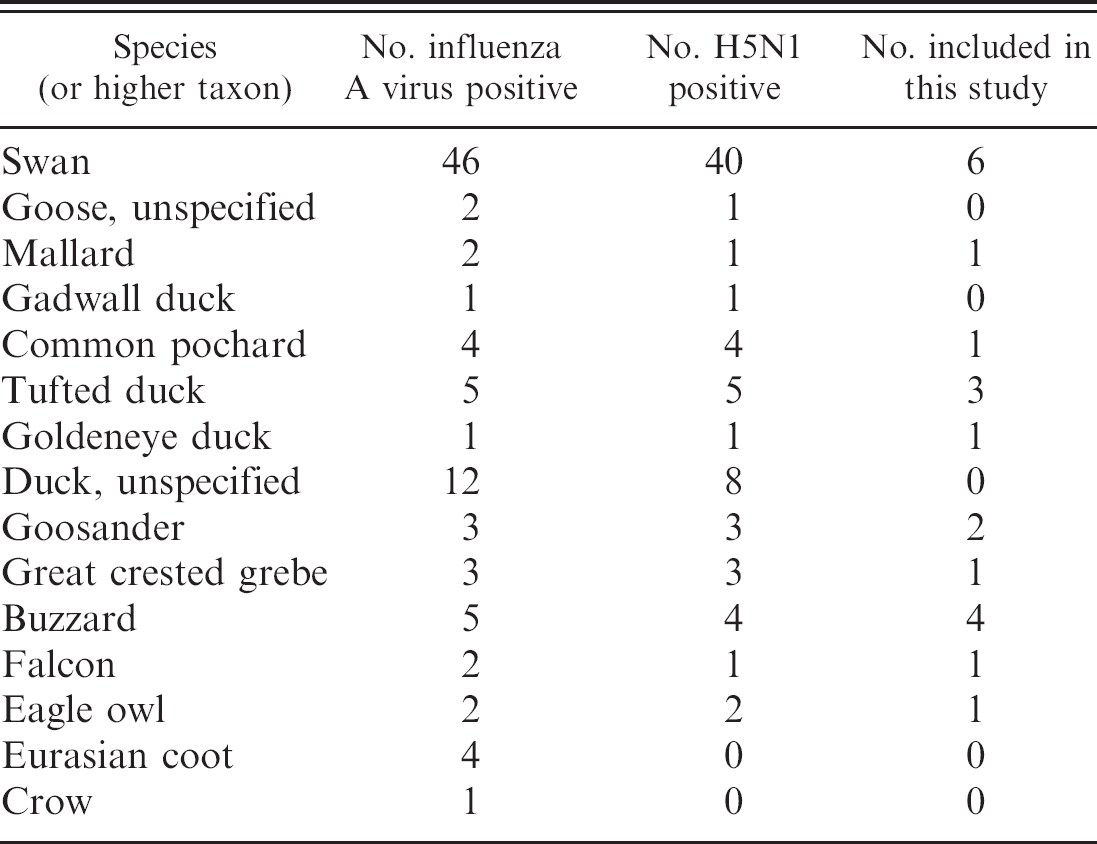
Sequence analysis demonstrated the HPAI cleavage site PQGERRRKKRGLF (single-letter amino-acid notation) in all 21 specimens. Alignment of the complete coding regions showed 2 separated genetic groups with 18 and 3 specimens for both the hemagglutinin genes (GenBank accession numbers DQ458992, DQ659679, EF165048-EF165066) and the neuraminidase genes (DQ458993, DQ659680, EF165571-EF165588). The first genotype (Bavaria 1) was found throughout Bavaria and in all host species (Table 1). The second genotype (Bavaria 2) was found in 1 swan, 1 great crested grebe, and 1 buzzard from eastern Bavaria. For the hemagglutinin genes, the identities within the 2 groups were >99.65% and >99.77%, identities between these groups were 98.65% to 98.89%. For neuraminidase, the identities within the 2 groups were >99.70% and >99.93%, identities between these groups were 98.74% to 98.96%.
The demonstration of 2 genotypically distinct, nonoverlapping groups of isolates is indicative of separate introductions of HPAI H5N1 viruses into Bavaria. Phylogenetic analysis of the hemagglutinin genes (Fig. 1) resulted in a clustering of these isolates within the previously described genotype V 6 and showed close relationships of the Bavaria 1 genotype (type isolate A/mallard/Bavaria/1/2006, GenBank accession number DQ458992) with previously reported sequences from Russia (A/chicken/Volvograd/236/06) and Mongolia (A/whooper swan/Mongolia/3/05 and A/whooper swan/Mongolia/6/05). Genotype Bavaria 2 (type isolate A/common buzzard/Bavaria/2/2006, GenBank accession number DQ659679) was closely related to isolates from the Czech Republic (A/Cygnus olor/Czech Republic/5170/2006), Turkey (A/turkey/Turkey/1/2005), Egypt (A/chicken/Egypt/5612NAMRU3-S/2006), and Nigeria (A/chicken/Lagos. NIE/8.06/SO300). For the above mentioned isolates, neuraminidase gene sequences have been reported only for the 2 Mongolian isolates A/whooper swan/Mongolia/3/05 and A/whooper swan/Mongolia/6/05 (GenBank accession number AB239311 and AB239325, respectively), which also clustered with genotype Bavaria 1.
Forward (for) and reverse (rev) PCR primers used for amplification and sequencing of the hemagglutinin gene (HA, primers INF-1 to INF-8) and the neuraminidase gene (NA, primers INF-23 to INF-28).

Locations of primer positions are relative to the start of the coding regions of the hemagglutinin gene (reference sequence: GenBank accession number DQ458992) and the neuraminidase gene (reference sequence: DQ458993).

Phylogenetic analysis of the H5 hemagglutinin genes, based on 1,559 base pairs of commonly reported coding region (position 64 to 1,652), for 21 Bavarian isolates from this study (written in bold) in relation to closely related and representative strains: Cygnus olor/Czech Republic/5170/2006 (Genbank accession number DQ515984), chicken/Lagos.NIE/8.06/SO300 (AM262546), chicken/Egypt/5612NAMRU3-S/2006 (DQ837589), turkey/Turkey/1/2005 (DQ407519), chicken/Volvograd/236/06 (DQ864719), whooper swan/Mongolia/3/05 (AB233322), whooper swan/Mongolia/6/05 (AB233320), cat/Germany/606/2006 (DQ643982), swan/Germany/R65/2006 (DQ464354), duck/Tuva/01/2006 (DQ861291), Cygnus olor/Italy/742/2006 (DQ412997), chicken/Crimea/08/2005 (DQ650663), migratory duck/Jiangxi/2295/2005 (DQ320920), Hong Kong/213/03 (AB212054), crested eagle/Belgium/01/2004 (DQ182483), duck/Hong Kong/821/02 (AY676033), and goose/Guangdong/1/96 (AF144305).
The distribution of the above mentioned countries (Russia and Mongolia vs Czech Republic, Turkey, Egypt, Nigeria) is suggestive of the 2 main bird migratory routes to central Europe, 1 being East-West from Asia, the other being South-North from Africa (via Spain, Italy, or Turkey). This finding supports the role of migratory birds as the main vector for the H5N1 HPAI virus import to central Europe and suggests that the current outbreak results from separate introductions. Multiple imports of HPAI H5N1 virus by different migratory bird routes have previously been reported from Nigeria 1 but not from Europe, where thus far only an outbreak with a single genotype was reported for the Czech Republic. 4
The observed time lag between the main migratory season in this study and the time of detections supports the view that these introductions can go unnoticed for periods of several months until, possibly after accumulation of large numbers of birds at still unfrozen watering places, virus transmission increases. It can further be speculated that the extraordinarily hard winter season of 2005/2006 increased fatalities that might have been unnoticed in previous years when monitoring activities were less intense.
Acknowledgements. This work was supported by a grant from the Bavarian State Ministry of the Environment, Public Health and Consumer Protection (project 05/19). The authors thank Heike Lang, Oberschleissheim, for excellent technical assistance and Dr. Peter Kreuzer, Erlangen, for coordinating laboratory examinations.
Footnotes
a.
Invitrogen Corp., Carlsbad, CA.
b.
Qiagen, Hilden, Germany.
