Abstract
A total of 15 adrenocortical carcinomas with myxoid differentiation from 15 ferrets were evaluated in this retrospective study. Six of these ferrets (40%) either were euthanatized or died due to invasive and/or metastatic disease. The myxoid component was a variable part (between 5% and 95%) of the adrenal cortical neoplasm and consisted of sheets and cords of small, polygonal neoplastic cells that formed lumenlike spaces. Such spaces contained a variable amount of alcian blue (pH 2.5)-positive mucinous product (i.e., acidic mucopolysaccharides). Neoplastic cells were negative for the argentaffin reaction, but immunohistochemically they were strongly positive for vimentin and α-inhibin and lightly positive for synaptophysin. Proliferating cell nuclear antigen (PCNA)-labeling indices (LI) of adrenal cortical neoplastic cells within the myxoid component of the neoplasm were significantly elevated (P < 0.05) compared with those of typical neoplastic adrenal cortical cells or the adjacent nonneoplastic zona reticularis. Ultrastructurally, cells in the myxoid component exhibited a typical adrenocortical phenotype characterized by cytoplasmic lipid vacuoles, prominent rough and smooth endoplasmic reticulum, and zonula adherens. This lesion was interpreted as an adrenal cortical carcinoma with myxoid differentiation and appeared to be highly malignant based on PCNA LI, rate of invasion into adjacent tissue, and metastasis (6/15). This report is the first description of this histologic variant in the ferret, which morphologically resembled the rare myxoid variant of adrenocortical carcinoma described in humans.
Keywords
Introduction
Adrenal cortical neoplasms occur frequently and have been reported as the second most common neoplasm of the domestic ferret, following only pancreatic islet tumors in prevalence. 6 , 8 , 9 , 12–14 , 16–18 , 20 , 22–25 A significant increase in the incidence of adrenocortical neoplasia has been reported in ferrets neutered at an early age (i.e., 2–4 months) when compared with ferrets neutered later in life. 20 This finding has been attributed to chronic trophic stimulation of zona reticularis cells by luteinizing hormone.
Adrenocortical adenomas and carcinomas in ferrets are usually functional, with clinical signs of hyperestrogenism and feminization in male ferrets. 6 , 8 , 9 , 12–14 , 16–18 , 20 , 22–25 Lesions reported in association with the estrogen excess include bilateral to diffuse alopecia, bone marrow toxicity, 13 squamous metaplasia of prostatic ductular epithelium, and cystic prostatic disease in male ferrets 6 and mammary gland hyperplasia/development in castrated male ferrets. 14 In addition to elevated levels of 17-β-estradiol, other steroid hormones, such as androstenedione, dehydroepiandrosterone sulfate, and 17-hydroxyprogesterone, may be secreted by the zona reticularis. 17 These hormones also can be elevated in ferrets with functional adrenocortical neoplasms. 17
A common feature of adrenocortical carcinomas in ferrets is the presence of a separate spindle cell component variably associated with neoplastic adrenal cortical cells. These cells have been shown by immunohistochemical staining 10 to express smooth muscle actin (SMA) and either arise from smooth muscle cells within or beneath the capsule or are a morphologically distinct type of adrenocortical cell. It has been debated whether this cell population is neoplastic, metaplastic, or a reactive hyperplasia; however, these spindle cells can be invasive into adjacent adrenal tissue and have occasionally been misdiagnosed as leiomyosarcomas. More likely this process is similar to the subcapsular spindle cell hyperplasia as seen in the adrenal glands of mice. 11
This report describes a myxoid variant of adrenocortical carcinoma in ferrets that was not present in cases of adenocortical nodular hyperplasia or adenomas. The histopathologic, histochemical, and immunohistochemical features of the adrenocortical carcinomas described in this report are similar to those of the myxoid adrenocortical carcinoma in human beings. There have been two case reports and a case series of adrenocortical carcinomas with a myxoid component in human patients. 2 , 7 , 21
Materials and Methods
The neoplasms in this report were from surgical biopsy and necropsy submissions obtained from the archives of The Ohio State University Department of Veterinary Biosciences for the years 1992–2002 (13 cases) and from the archives of the Michigan State University Animal Health Diagnostic Laboratory, Lansing, Michigan (two cases). These 15 neoplasms with myxoid differentiation were selected after a review of ferret adrenal lesions from the respective department archives, which included a total of 33 adrenal cortical neoplasms.
Adrenal gland lesions were evaluated microscopically. Adrenocortical carcinoma was diagnosed in cases where there was a proliferative lesion of cortical cells with variable cellular atypia that invaded beyond the adrenal capsule and/or had evidence of distant metastasis. 3 , 4 , 19 Well-demarcated, variably encapsulated, proliferative lesions that did not extend beyond the adrenal capsule were diagnosed as cortical adenomas. 3 , 4 , 19 Multiple areas of unencapsulated nodular proliferation confined to the adrenal cortex were diagnosed as nodular hyperplasia. 3 , 4 , 19 Out of a total of 33 adrenal gland masses in 33 ferrets that were examined, 19 (58%) were adrenocortical carcinomas (of which 15 had significant myxoid differentiation), 10 (30%) were adrenocortical adenomas, three (9%) were adrenocortical nodular hyperplasia, and one (3%) was a malignant pheochromocytoma.
Eight of the adrenal tumors were submitted as surgical biopsies. One tumor was a referral biopsy from a ferret with a limited history and signalment. Four ferrets were electively euthanatized, and two ferrets died naturally. Complete necropsies were performed on these ferrets.
Formalin-fixed, paraffin-embedded archival adrenal gland tissue was evaluated. Sections were cut at 5 μm and stained routinely with hematoxylin and eosin (HE) stain. Sections were stained histochemically by the alcian blue (pH 2.5) and Churukian-Schenck methods for acidic mucopolysaccharides and chromaffin reaction, respectively. Sections were stained immunohistochemically for: α-1-fetoprotein (AFP; polyclonal), chromogranin-A (CGA; clone DAK-A3), cytokeratin cocktail (CKC; i.e., AE1/AE3, MAK-6, and CAM.2), proliferating cell nuclear antigen (PCNA, clone PC10), SMA (clone 1A4), synaptophysin (polyclonal), vimentin (clone V9) (DAKO, Carpinteria, CA), and α-inhibin (clone R1) (Serotec, Inc., Raleigh, NC). The primary monoclonal antibodies were anti-human but were reliably cross-reacted with ferret tissue based on internal controls. Sections were stained using a DAKO Autostainer™. Briefly, sections were deparaffinized and hydrated. Sections were steam-heated in a citrate-based target retrieval solution (DAKO) for 20 minutes at 94 C, allowed to cool, and placed in phosphate-buffered saline (PBS). Endogenous peroxidases were blocked by incubation with 10% H2O2 in PBS for 5 minutes at room temperature (RT), and the sections were blocked with serum-free protein block for 10 minutes at RT. Sections were incubated with primary antibody diluted in antibody diluent for 30 minutes at RT. Primary antibodies were diluted as follows: α-inhibin: 1:40, AFP: 1:3,000, CGA: 1:50, CKC: 1:50, PCNA: 1:30, SMA: 1:75, synaptophysin: 1:50, and vimentin: 1:100. Sections were incubated with 1:200 diluted biotinylated secondary antibody (Vector Laboratories, Burlingame, CA) at RT for 30 minutes followed by incubation with avidin-biotin complex (Vector Laboratories) for 30 minutes at RT. The chromogen was diaminobenzidine.
PCNA staining was performed on representative adrenal masses. Labeling indices (LI; No. positive cells/100 total cells in three random fields per histologic phenotype) were determined for areas of myxoid component, areas of polygonal neoplastic cells, spindle cell component, and normal adrenal cortex (i.e., zona reticularis, the putative site of origin of the adrenocortical carcinomas). The Student's t-test was used to statistically compare PCNA LI between different adrenal cell types. The level of significance was set at P < 0.05. The mean percentage involvement (i.e., total area) of neoplasms by myxoid-differentiated neoplastic cells in representative cross-sectional HE sections at the level of the widest aspect of the mass was determined by histomorphometric techniques using the BioquantNova™ instrument and software system (R&M Biometrics, Inc., Nashville, TN).
In two cases formalin-fixed tissue was postfixed in 3% glutaraldehyde in 0.1 M sodium cacodylate buffer and 1.33% OsO4 in S-collidine buffer. It was then dehydrated, cleared, and embedded in Eponate™ plastic (Electron Microscopic Supplies Inc.; Fort Washington, PA). One-micron sections were cut to identify areas of interest for thin sections. Thin sections were collected on grids and stained with 1.5% uranyl acetate and 1.5% lead citrate. Sections were observed through a Philips 300™ transmission electron microscope.
Results
The mean age of the 15 ferrets evaluated was 5.38 ± 1.50 years (range = 3–9 years). Seven ferrets were spayed females. Six ferrets were castrated males, whereas two ferrets did not have an available history. All ferrets with clinical signs (disease) were neutered early in life (i.e., < 6 months of age).
Presenting clinical signs were variable but were generally associated with hyperestrogenism and/or space-occupying mass effect of the neoplasm. In animals with a history (13/15), clinical signs included polyuria/polydipsia (3/13), symmetric alopecia (13/13), enlarged vulva (3/7), enlarged mammary glands (1/6 males), abdominal distension (4/13), stranguria (3/6 males), and tenesmus/constipation (1/13).
Adrenal gland masses were present in all 15 ferrets and varied from 0.75 to 5.50 cm in diameter, with the larger masses completely effacing the normal architecture of the adrenal gland (Fig. 1). Hepatic metastases were present in three ferrets that underwent postmortem examination. The multilobular, metastatic masses varied from 1 to 5 cm in diameter (Fig. 2). There was no apparent predilection for neoplasm development in either the left or the right adrenal gland in this limited group of ferrets. The site of the adrenal mass in the referral biopsy case was not specified. Adrenocortical carcinomas were bilateral in two ferrets, and one ferret had contralateral adrenocortical nodular hyperplasia of the zona fasciculata.
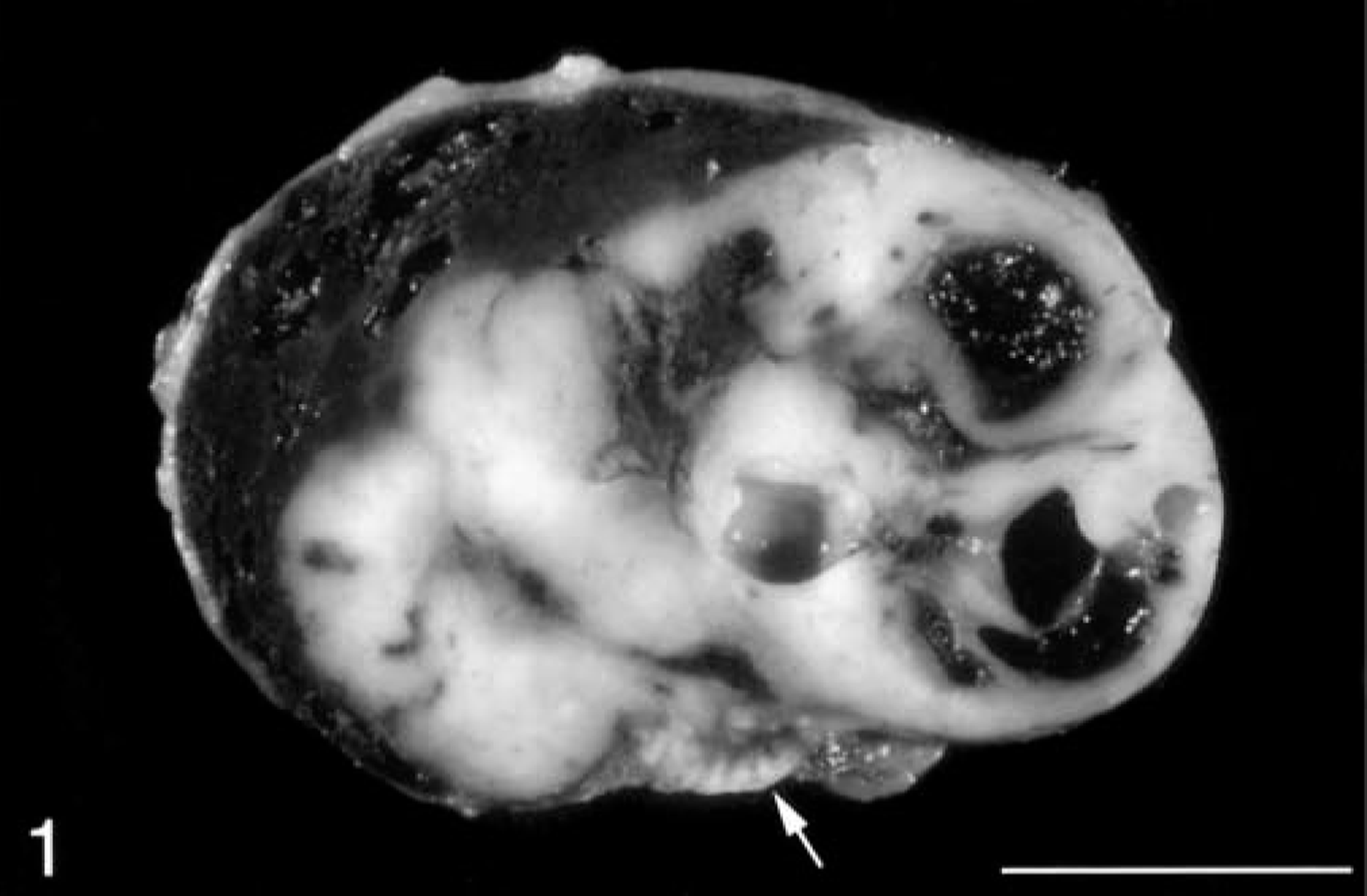
Adrenal gland mass; ferret. A cross section of an adrenocortical carcinoma with myxoid differentiation. Note the residual, normal adrenal cortex (white arrow) and the prominent cystic spaces in the tumor. Bar = 1 cm.
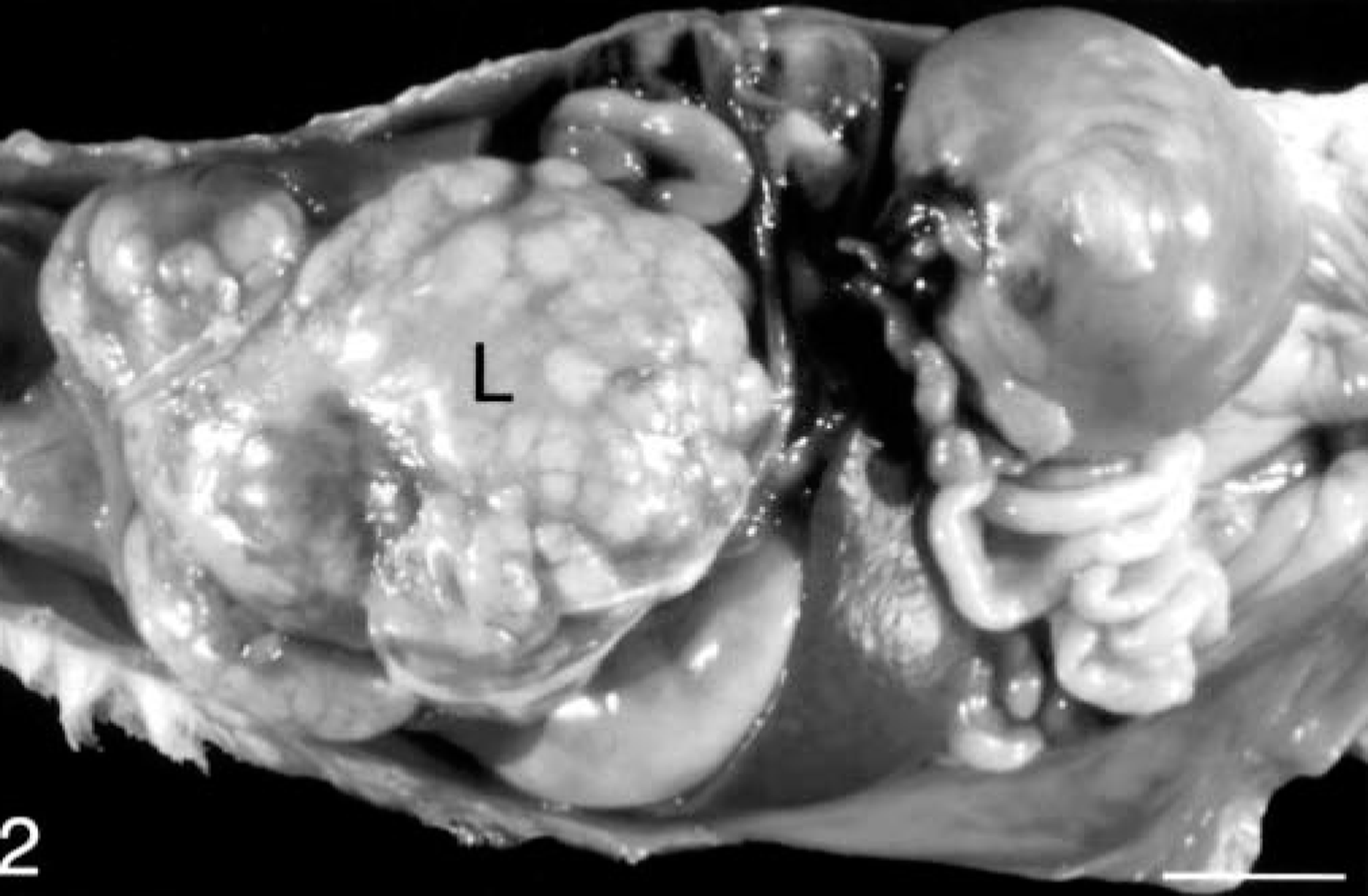
Peritoneal cavity; ferret. In situ photograph of a ferret with a large, multilobular, metastatic adrenocortical carcinoma involving the liver (L). Bar = 1 cm.
Two ferrets had direct visceral invasion, the first into the caudal vena cava and the second into the liver. Evidence of metastasis to the liver was present in four ferrets (Fig. 2), and evidence of metastasis to the lung (Fig. 3) and regional lymph nodes (not shown) was present in one ferret.
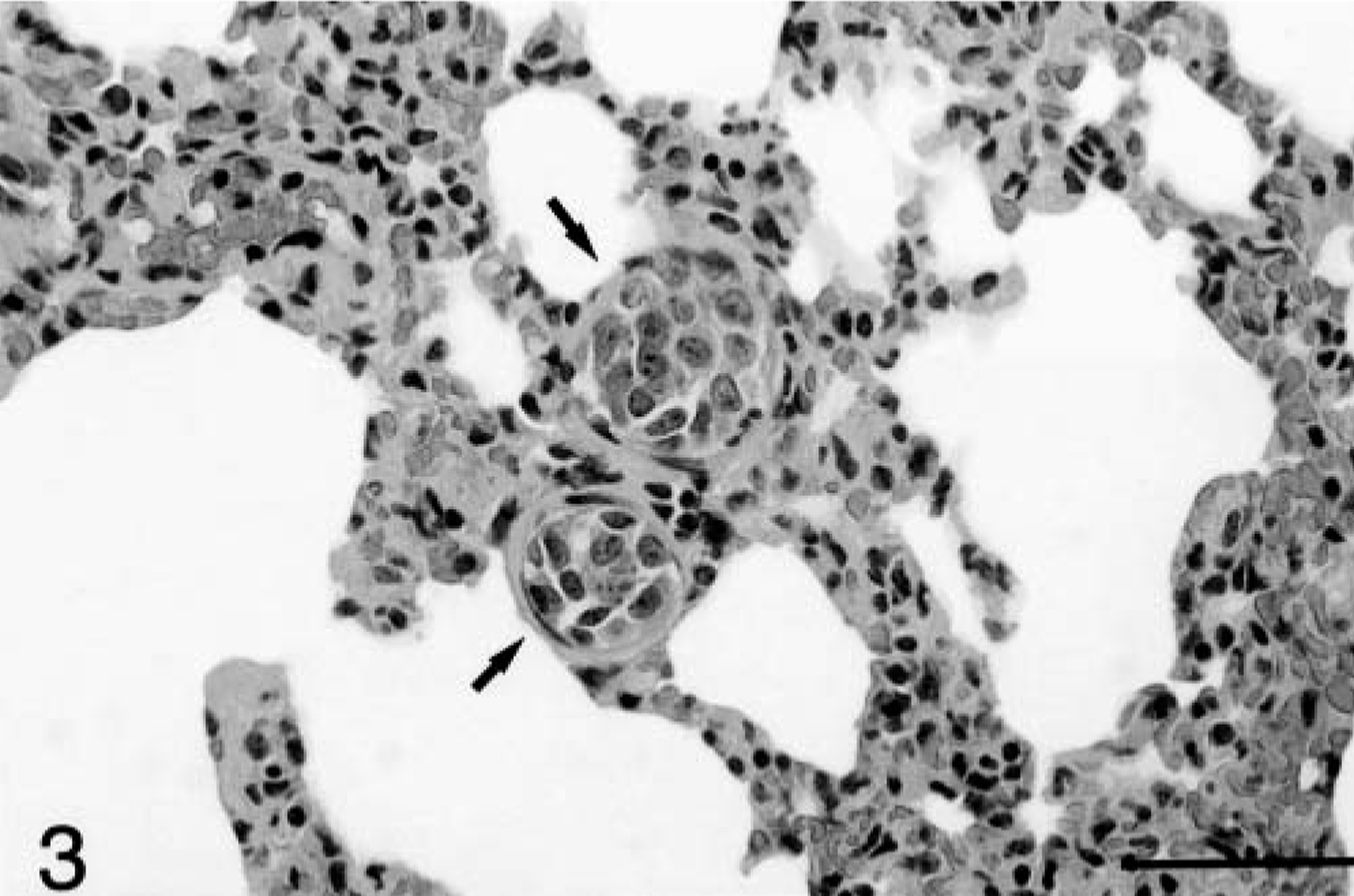
Lung; ferret. Tumor emboli (arrows) within the pulmonary vessels in a ferret with adrenocortical carcinoma. HE. Bar = 50 μm.
In most cases the adrenal gland masses were modest in size (i.e., < 2.5 cm), without visceral invasion or metastasis; however, in the invasive and metastatic masses the myxoid differentiation was more extensive than in the others (50–95% of the tumor). The invasive part of the mass in two ferrets and the metastases in four other ferrets were myxoid, suggesting more malignant behavior of this component of the adrenocortical carcinoma. In general, there was a more prominent myxoid phenotype in larger adrenal masses (i.e., > 2.5 cm), and they were associated with an increased rate of metastasis and death of the animal.
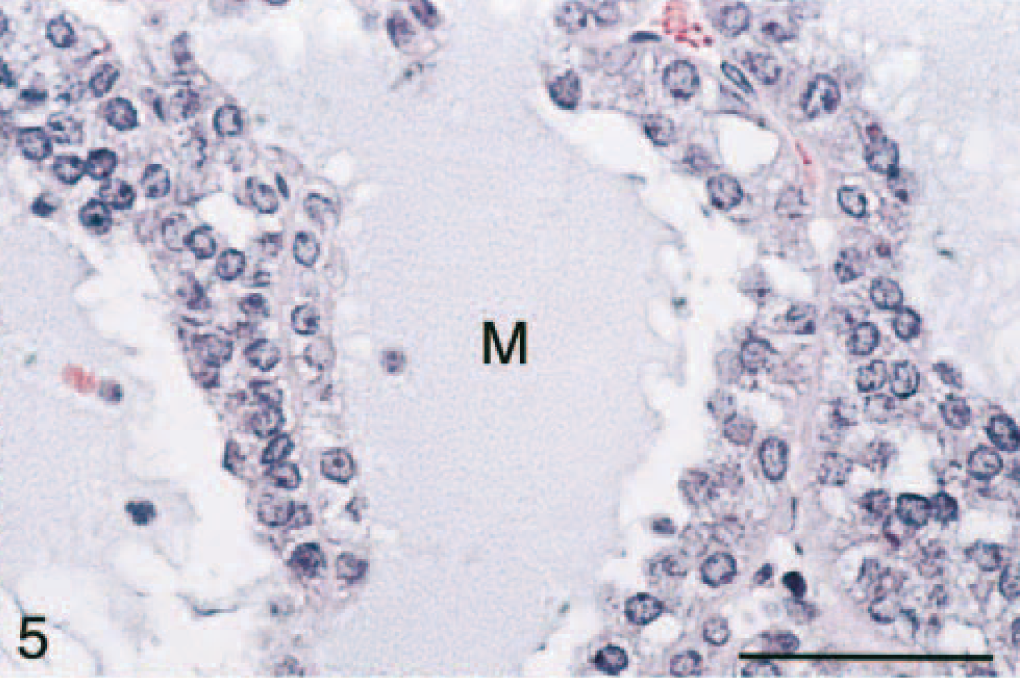
All tumors had areas of myxoid differentiation characterized by sheets, trabeculae, and cords of cuboidal cells with abundant eosinophilic to amphophilic, granular cytoplasm and a central round nucleus with little cellular/nuclear atypia (Figs. 4, 5). Mitotic figures were rare (0–2/20× hpf). These cells frequently formed lumenlike structures or spaces, with minimal fibrovascular stromal tissue. Lumenlike structures contained a mucinous material that also formed lakes between sheets of cells. These cells usually were intimately associated with areas of more typical neoplastic adrenocortical cells (i.e., cuboidal cells with an abundant vacuolated eosinophilic cytoplasm with a central nucleus with little atypia). In very large adrenal masses, there was moderate central necrosis and hemorrhage. The spindle cell component varied from being prominent to nonexistent. Occasionally, large cystic cavities were present at the periphery of the masses. The cysts were lined by an attenuated layer of cells and contained a central eosinophilic material that was variably alcian blue (pH 2.5) positive, and they were interpreted as developmental cysts.
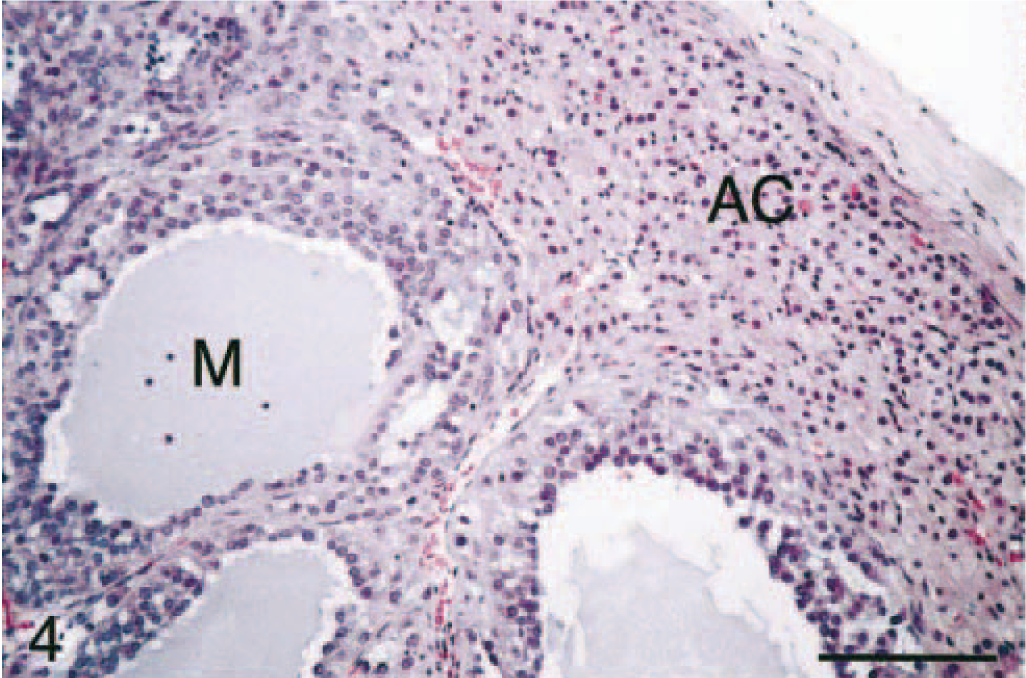
Adrenal gland mass; ferret. Adrenocortical carcinoma with myxoid differentiation. Cystic spaces lined by neoplastic cells contain abundant mucinous product (M). Note the overlying normal adrenal cortex (AC; zonae fasciculata and glomerulosa). HE. Bar = 50 μm.
Adrenal gland mass; ferret. Adrenocortical carcinoma with myxoid differentiation illustrating typical cellular morphology, fibrovascular stroma, and prominent mucinous product (M). HE. Bar = 50 μm.
Histochemically, only the adrenal medullary cells within the sections stained positively with the Churukian-Schenck argentaffin stain (not shown). These cells had multifocal, fine, black granular cytoplasmic staining (i.e., neuroendocrine granules) and were variably present based on the size and orientation of the section of the adrenal mass lesion. The mucinous matrix in the lumenlike structures within areas of myxoid differentiation in all adrenocortical carcinomas stained positively with alcian blue pH 2.5 (Table 1; Fig. 6). Therefore, neoplastic cells within areas of the myxoid component were not of neuroendocrine origin and produced a mucinous product consisting of acidic mucopolysaccharides.
Immunohistochemical and histochemical staining characteristics of neoplastic cells in areas of myxoid differentiation.

∗ 0 = no staining; +/− = variable positive staining; +++ = strong positive staining.
† Diffuse cytoplasmic staining.
‡ Staining of scattered adrenal medullary cells only.
§ Staining of spindle cell component only.
‖ Alcian blue staining of mucinous matrix/product associated with myxoid neoplastic cells.
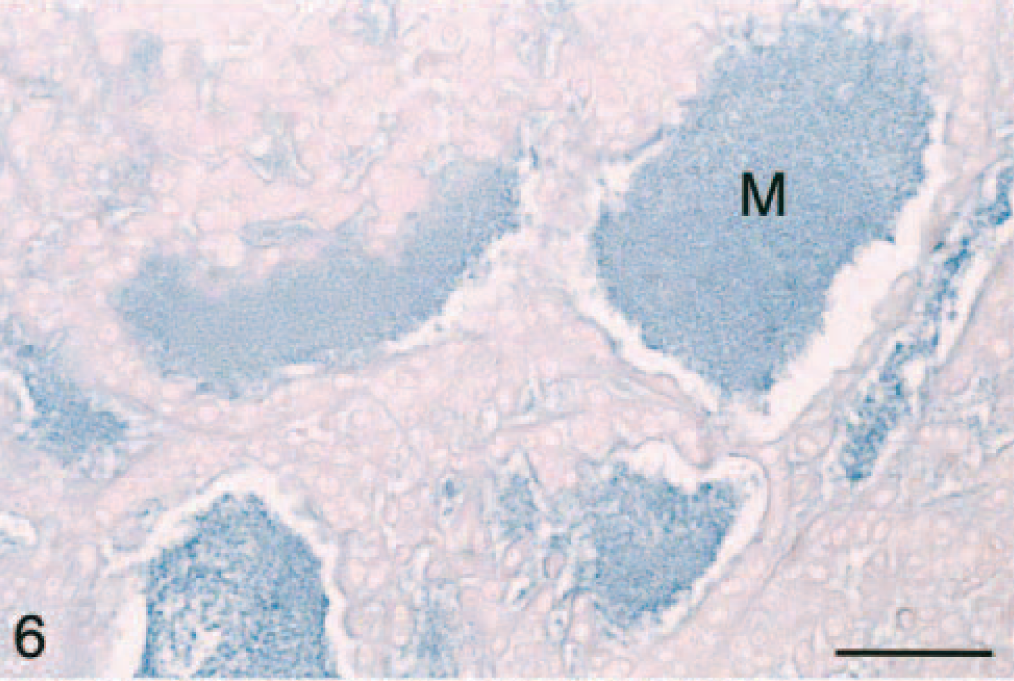
Adrenal gland mass; ferret. Adrenocortical carcinoma with myxoid differentiation. Note the alcian blue staining of the mucinous product (M). Alcian blue (pH 2.5) with nuclear fast red counterstain. Bar = 50 μm.
Immunohistochemical evaluation of the adrenocortical carcinomas showed strong positive cytoplasmic staining of the cell population in the myxoid component with anti-vimentin antibody (Table 1; Fig. 7) and anti–α-inhibin (Table 1; Fig. 8). The cytoplasm of these cells also stained lightly and variably positive with anti-synaptophysin antibody (Table 1), whereas it was negative with anti-CK, anti-CGA, and anti-AFP antibodies (Table 1). Only adrenal medullary cells stained positively for CGA (Table 1). The spindle cell component of these adrenocortial carcinomas stained strongly positive with anti-SMA antibody (Table 1), but there was no staining of the myxoid areas of the masses with this antibody (Table 1). A variable number of nuclei stained strongly positive for PCNA depending on the neoplastic cell phenotype (Fig. 9). PCNA LI in areas of myxoid component were significantly increased (P < 0.05) over those in areas of normal zona reticularis, the spindle cell component, and areas of more typical cuboidal neoplastic cells (Table 2).
Proliferating cell nuclear antigen (PCNA) labeling indices (LI) of neoplastic cells within adrenocortical carcinomas with myxoid differentiation and associated nonneoplastic cell types.
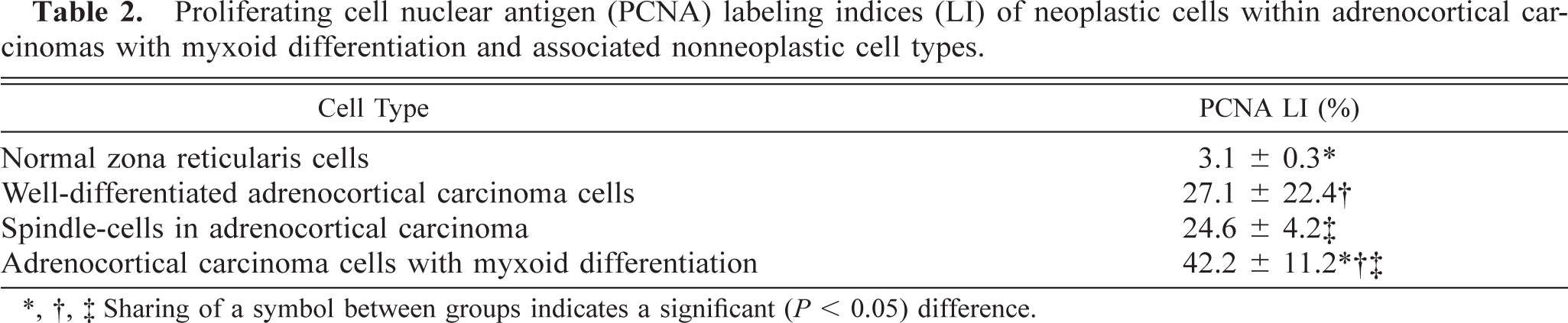
∗, †, ‡ Sharing of a symbol between groups indicates a significant (P < 0.05) difference.
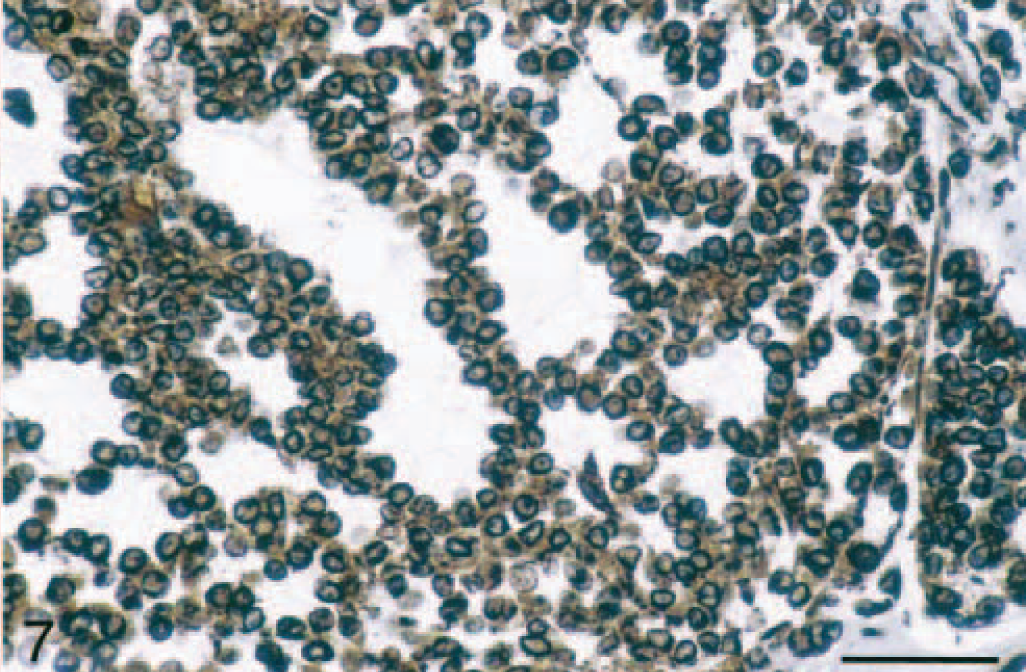
Adrenal gland mass; ferret. Vimentin expression in the myxoid component of adrenocortical carcinoma indicated by diffuse cytoplasmic staining of neoplastic cells. Vimentin, avidin-biotin complex peroxidase method. Bar = 50 μm.
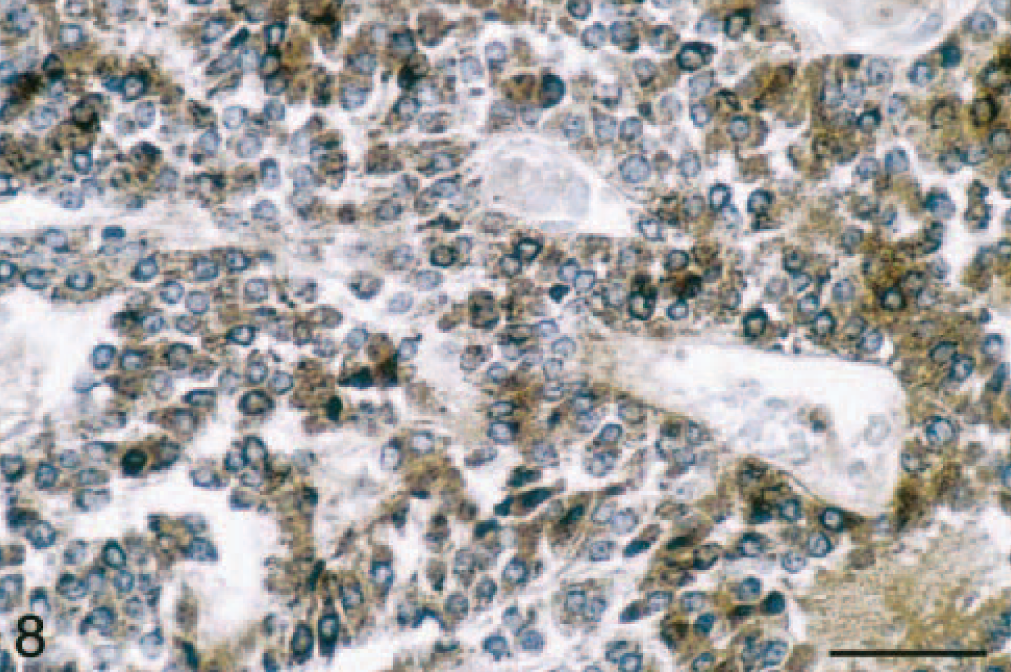
Adrenal gland mass; ferret. Intracytoplasmic α-inhibin expression in adrenocortical carcinoma with myxoid differentiation. α-Inhibin, avidin-biotin complex peroxidase method. Bar = 50 μm.
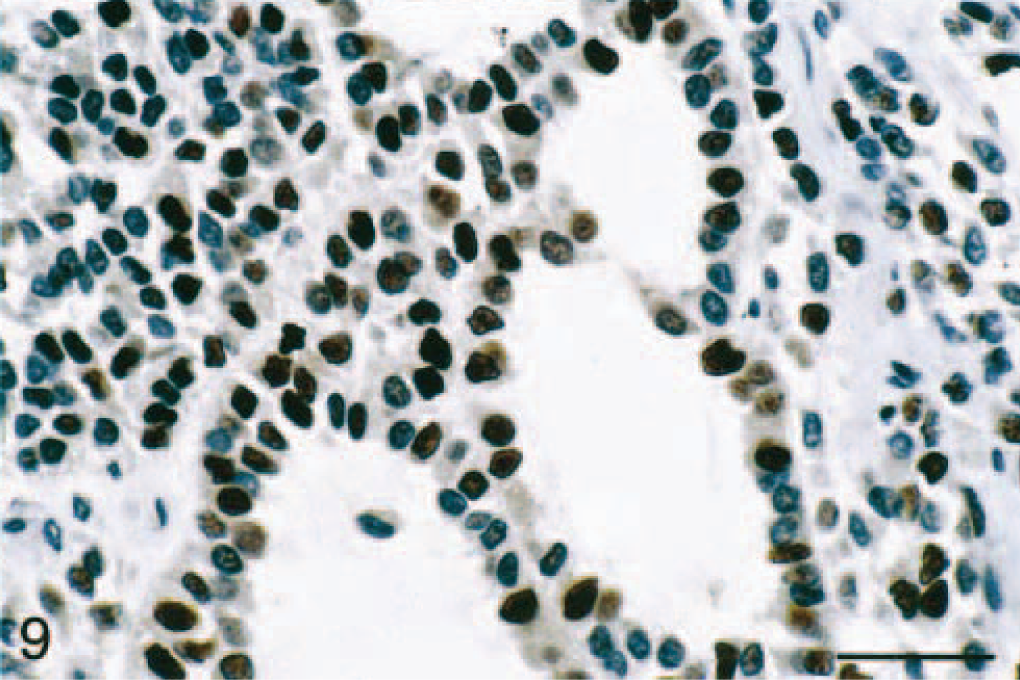
Adrenal gland mass; ferret. Frequent proliferating cell nuclear antigen (PCNA)-stained nuclei in an adrenocortical carcinoma with myxoid differentiation. PCNA, avidin-biotin complex peroxidase method. Bar = 50 μm.
Ultrastructurally, neoplastic cells within areas of the myxoid component were similar to well-differentiated neoplastic adrenocortical cells and had cytoplasmic lipid vacuoles, prominent rough and smooth endoplasmic reticulum, occasional mitochondria, free polyribosomes, and zonulae adherens (Fig. 10).
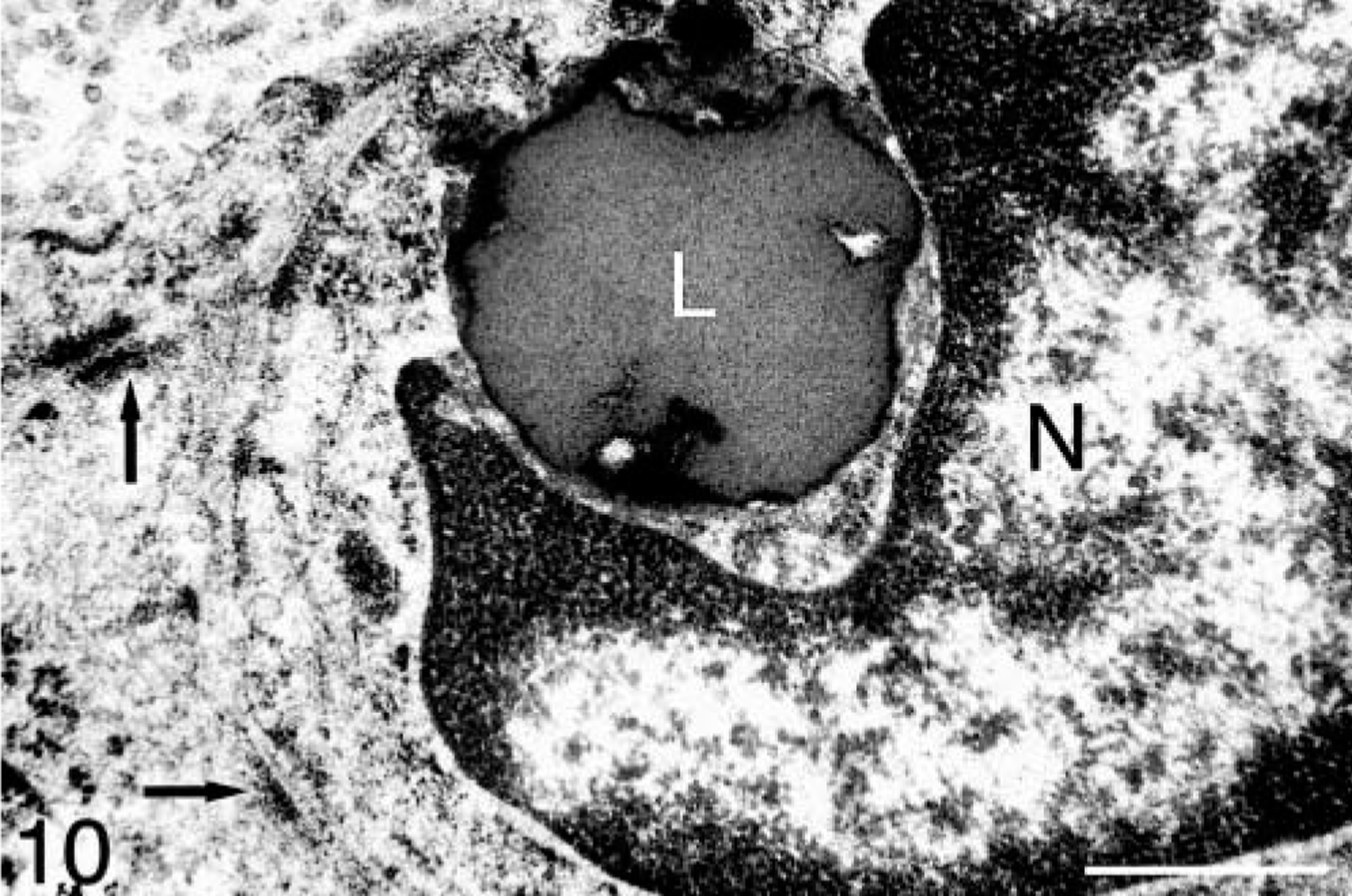
Adrenal gland mass; ferret. Zonula adherens (arrows) between adjacent cells in an adrenocortical carcinoma with myxoid differentiation. A lipid droplet (L) is present adjacent to the nucleus (N). Bar = 900 nm.
Discussion
This article describes a variant of adrenocortical carcinoma in 15 ferrets characterized by myxoid differentiation, which appears to be more malignant than well-differentiated adrenocortical carcinomas. Immunohistochemical and histochemical staining of neoplastic cells within areas of the myxoid component confirmed an adrenal cortical origin of the neoplastic cells. The positive cytoplasmic staining with vimentin, α-inhibin, and synaptophysin supports an adrenocortical and, more specifically, a zona reticularis origin of the neoplasm. Vimentin is expressed primarily by cells of mesenchymal origin, but epithelial cells in adrenocortical carcinomas in humans stain positively with vimentin. 2 Synaptophysin is a marker of neuroendocrine cells and is expressed primarily by adrenal medullary cells, but adrenocortical carcinoma cells also can express this protein. 2 α-Inhibin is the alpha subunit of inhibin and is expressed in numerous cell types, including adrenocortical cells within the zona reticularis and gonadal stromal cells. α-Inhibin has been shown to be more prominent in human adrenocortical carcinomas that secrete sex hormones. 1 , 2 , 15
The origin of neoplastic cells with myxoid differentiation in adrenocortical carcinomas of ferrets is unknown. Differentiation of neoplastic cells from the zona reticularis into cells capable of producing mucin is the most likely explanation. An alternative explanation would be that this phenotype reflects degeneration of neoplastic adrenocortical cells and associated stroma; however, there was no evidence of degenerative neoplastic cells or abnormal fibrovascular stroma in nonmyxoid components of these neoplasms. The least likely origin of these neoplastic cells, as suggested by some investigators of the human neoplasm, 2 , 7 , 21 would be from ectopic rests of gonadal stromal granulosa or sertoli cells or their precursors. Adrenal cortical cells and gonadal stromal (coelomic) epithelial cells are both derived from splanchnic mesoderm. The adrenal cortical cells originate in the medial aspect of the gonadal/urogenital ridge, whereas gonadal stromal cells are from the lateral aspect of the gonadal/urogenital ridge. 5 Negative AFP immunohistochemical staining of the adrenocortical carcinomas with myxoid differentiation in ferrets ruled out this potential origin of the tumor cells.
Neoplastic adrenocortical cells with myxoid differentiation are similar histomorphologically to neoplastic cells comprising myxoid adrenocortical carcinomas in humans beings. Adrenocortical neoplasms in ferrets also share clinical features with those in human patients (sex hormone production) and are thought to arise in the zona reticularis of the adrenal gland. Feminization associated with hyperestrogenism is not always reported in human patients as in male ferrets. A masculinizing effect is seen frequently in women and feminization in men and children. The myxoid component is present in both adrenocortical adenomas and carcinomas in humans; 2 however, we have observed this feature only in carcinomas in ferrets.
In summary, we describe a variant of adrenocortical carcinoma in the domestic ferret with prominent mucin production, interpreted as myxoid differentiation. The neoplasm was shown to be of adrenal cortical origin by histochemical and immunohistochemical methods. This variant of adrenocortical carcinoma in the ferret appeared to be more malignant than its well-differentiated counterpart. This hypothesis was supported by the presence of myxoid differentiation only within carcinomas, a more aggressive biologic behavior with invasion and distant metastasis, a significantly elevated PCNA LI compared with that in normal and other adrenocortical carcinomas, and an increased mortality in ferrets as a result of the tumor. The myxoid variant of adrenocortical carcinoma in ferrets was similar to the rare myxoid adrenocortical carcinoma in human beings. It is uncertain why this phenotype had not been described previously in adrenal neoplasms of ferrets. Possiblities include an interpretation that this change represented either mucinous degeneration in a large adrenal mass or a nonadrenocortical origin of this cell population in the neoplasm. Further studies are needed to determine the origin of mucin-producing cells in adrenocortical carcinomas in ferrets and whether this variant is consistently associated with a more aggressive biologic behavior.
Footnotes
Acknowledgements
Portions of this manuscript were previously presented in abstract form at the 52nd ACVP Meeting in Salt Lake City, UT, on 1–5 December 2001. We thank fellow residents in the Ohio State University Department of Veterinary Biosciences for their submission of case material for this case series and Dr. Barbara Oglesbee for her clinical input. We also thank the following people for their excellent technical assistance: Alan Flechtner, BS, HTL (ASCP), Ann Saulsberry, HT (ASCP), Evelyn Handley (transmission electron microscopy), Kim Partee, HT (ASCP), Liane Peterson, BS, and Marc Hardman (figures).
