Abstract
A 6-year-old male Pointer dog was presented with a 4-week history of progressive hind-limb stiffness. Magnetic resonance imaging demonstrated a focal intramedullary lesion at T1 level with a pattern of ring contrast enhancement. At necropsy, a circumscribed intramedullary reddish-gray tumor was observed. Microscopically, the tumor was composed of thin-walled capillaries lined by endothelial cells and separated by pleomorphic cells (stromal cells) with a moderate degree of anisokaryosis. Immunohistochemically, the endothelial cells were positive for factor VIII–related antigen and the stromal cells were positive for neuron-specific enolase and vimentin. GFAP-positive astrocytes were occasionally observed within the tumor. Both endothelial and stromal cells were negative for synaptophysin, S-100 protein, pankeratin, smooth muscle actin, CD34, CD68, α1-antichymotrypsin, and lysozyme. The tumor showed considerable morphologic and immunohistochemical similarities with human hemangioblastoma, and hence the inclusion of this tumor type within the primary neoplasms of the canine central nervous system is suggested.
Hemangioblastomas are highly vascular neoplasms of uncertain origin. 11 The frequency of human hemangioblastomas varies between 1% and 2% of intracranial tumors with a peak incidence in middle-aged adults and a male predominance of 2:1. Most originate in the cerebellum, rarely are supratentorial tumors, and 3.2% are encountered in the spinal cord. 11 Hemangioblastomas may be single or multiple and they can occur in association with Von Hippel-Lindau syndrome, a familial multisystem disorder, which includes retinal hemangioblastoma and a number of extraneural lesions including renal cell carcinoma, pheochromocytoma, cysts of the kidneys, liver, pancreas, adrenal glands, epididymis, and mesosalpinx. 5 The tumor is composed of two main components: endothelial cells forming a vascular network and larger interstitial cells, termed “stromal cells,” whose histogenesis is obscure.
To our knowledge, only one case of intramedullary neoplasm with features of hemangioblastoma has been diagnosed in the dog. 1 Here, we describe the clinical, neuroradiologic, and pathologic features of a hemangioblastoma of the spinal cord of a dog.
A 6-year-old male Pointer dog was presented with a 4-week history of slowly progressive hind-limb stiffness and exercise intolerance. Neurological evaluation showed a mild hind-limb paresis associated with stiffness of the forelimbs. Proprioception was decreased in all limbs. Cranial nerves were normal and the spinal reflexes were decreased in the forelimbs. The lesion was clinically localized in the cervicothoracic intumescence of the spinal cord (C6-T2). Myelography demonstrated an ill-defined intramedullary lesion at T1 level. Cerebrospinal fluid analysis was normal. A magnetic resonance (MR) imaging of the spinal cord was performed. T1-weighted images were obtained in transverse, sagittal, and axial planes with a spin echo (SE) pulse sequence before and after paramagnetic contrast infusion. T2-weighted sagittal images were achieved with a fast-SE pulse sequence. T2-weighted images showed a round-shaped intramedullary hypointense mass at T1 level. After paramagnetic contrast infusion, the lesion showed a pattern of ring contrast enhancement. At surgery, the presence of a presumptive intramedullary tumor was confirmed and the dog was euthanatized because of poor prognosis.
A complete necropsy was performed and no gross lesions were observed, except for the spinal cord at the level of the first thoracic vertebra. On external examination, there was a red, round, bulging mass of approximately 4 mm in diameter on the left side of the spinal cord, accompanied by prominent congestion of meningeal vessels (Fig. 1). The spinal cord was sampled and fixed in 10% neutral buffered formalin solution. In transverse sections the mass was circumscribed, intramedullary, reddish-gray, and approximately 6 mm in diameter. The mass caused displacement of the midline spinal cord structures. The sampled tissue was routinely processed for histology and sections were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), trichrome, reticulin, and toluidine blue methods. Immunohistochemistry was performed using the avidin–biotin–peroxidase complex (ABC) method (Vector Laboratories, Burlingame, CA, USA). The primary antibodies used were rabbit antibodies against glial fibrillary acidic protein (GFAP, prediluted; Ventana Medical Systems, Inc., Tucson, AZ, USA), S-100 protein (prediluted; Ventana), factor VIII-related antigen (FVIII-rag, prediluted; Ventana), α1-antichymotrypsin (prediluted; Ventana), lysozyme (prediluted; Ventana), synaptophysin (prediluted; Ventana), and mouse monoclonal antibodies against vimentin (prediluted; Ventana), neuron-specific enolase (NSE, prediluted; Ventana), pankeratin (prediluted; Ventana), CD34 (prediluted; Ventana), CD68 (1:100; Dako, Carpinteria, CA, USA), and smooth muscle actin (SMA, 1:50; Dako). Before incubation with the antibodies against S-100, FVIII-rag, synaptophysin, vimentin, and NSE, the sections were submitted to microwave antigen retrieval in citrate buffer at pH 7.2 for 15 min at 500 W. For the other antibodies, no microwave treatment was used.

Spinal cord, hemangioblastoma; dog. There is an intramedullary bulging mass abutting the pia mater. Note marked congestion of meningeal vessels.
Microscopically, the mass was highly cellular, vascularized, and composed of haphazardly oriented small capillaries lined by plump endothelial cells separated by larger pleomorphic stromal cells. This latter cell component had indistinct cytoplasmic borders and occasionally contained cytoplasmic vacuolizations. The nuclei, which were large and vesicular with prominent nucleoli, showed a moderate degree of anisokaryosis (Fig. 2). Mitotic figures were occasionally observed at the periphery of the mass. An abundant network of reticulin fibers surrounded groups of stromal cells and supported the vascular architecture. In the central area of the tumor the vascular structures were less clearly defined or markedly compressed and the tissue was more fibrillar. Plasma cells were scattered throughout the neoplastic tissue and were found, admixed with lymphocytes, in large perivascular aggregates at the edge of the mass. The surrounding neuroparenchyma was not infiltrated by the neoplastic cells, but was compressed, edematous, and gliotic, with myelin vacuolization and occasional axonal swelling. All endothelial cells stained with FVIII-rag, whereas stromal cells were negative (Fig. 3). Stromal cells stained strongly positive with NSE (Fig. 4) and many cells also stained with vimentin. Some cells, interpreted as reactive astrocytes, stained with GFAP. Both endothelial and stromal cells were negative for synaptophysin, S-100 protein, pankeratin, SMA, CD34, CD68, α1-antichymotrypsin, and lysozyme.
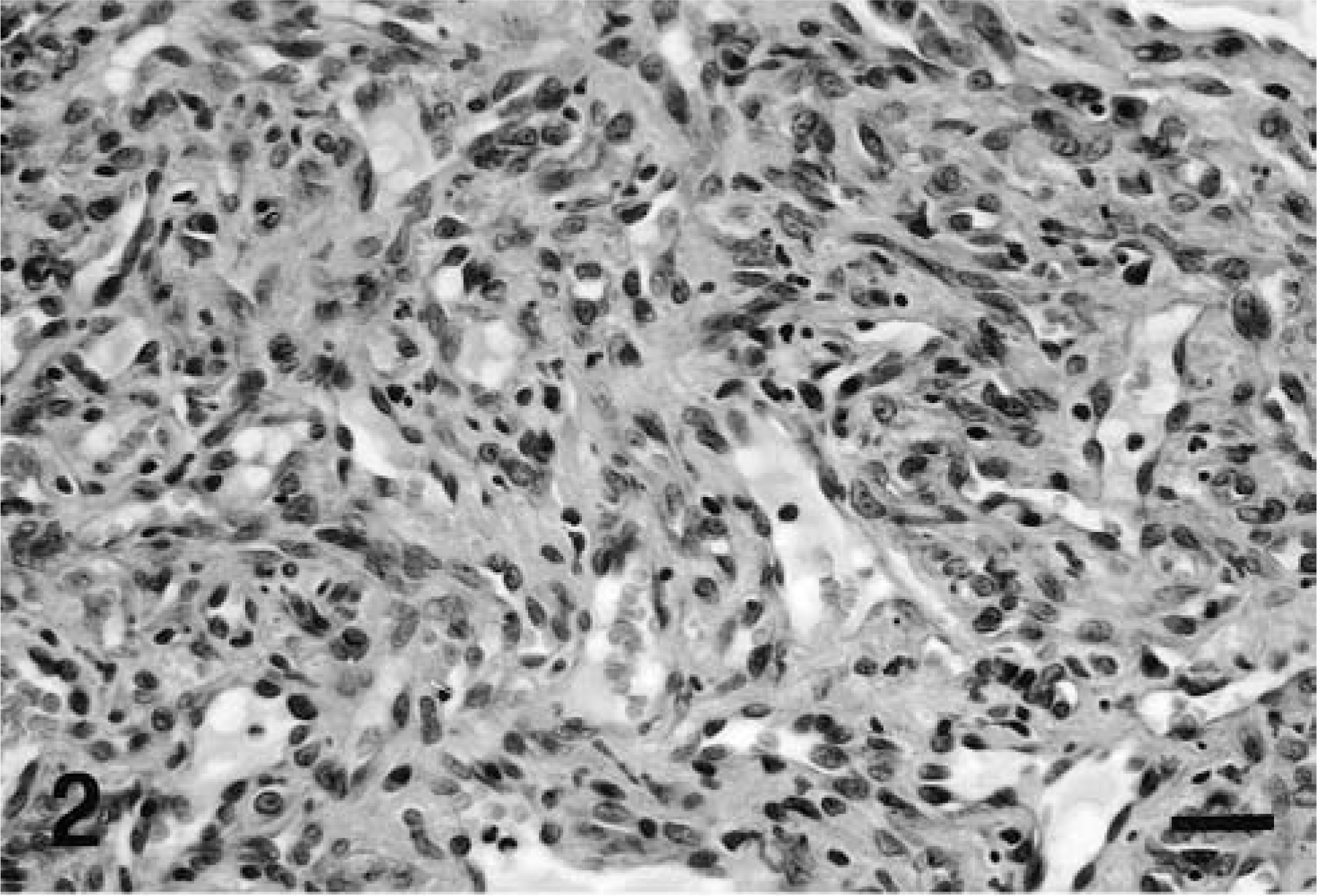
Spinal cord, hemangioblastoma; dog. There are vascular channels lined by endothelial cells and separated by stromal cells with large nuclei. HE. Bar = 50 μm.
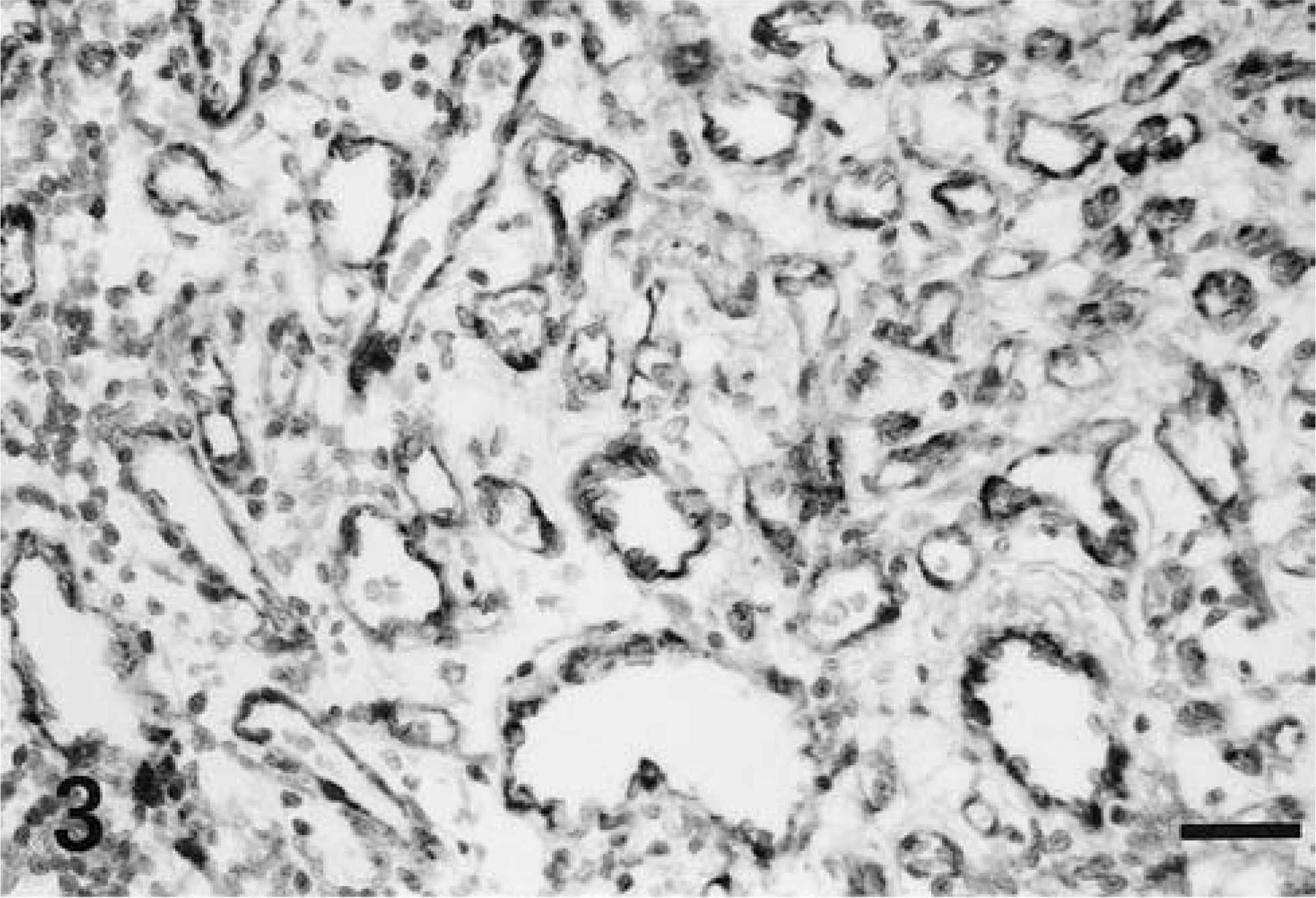
Spinal cord, hemangioblastoma; dog. Immunoreactivity for FVIII-rag is uniformly confined to the endothelial cells. The stromal cells are consistently negative. Avidin–biotin–peroxidase complex method. Bar = 50 μm.
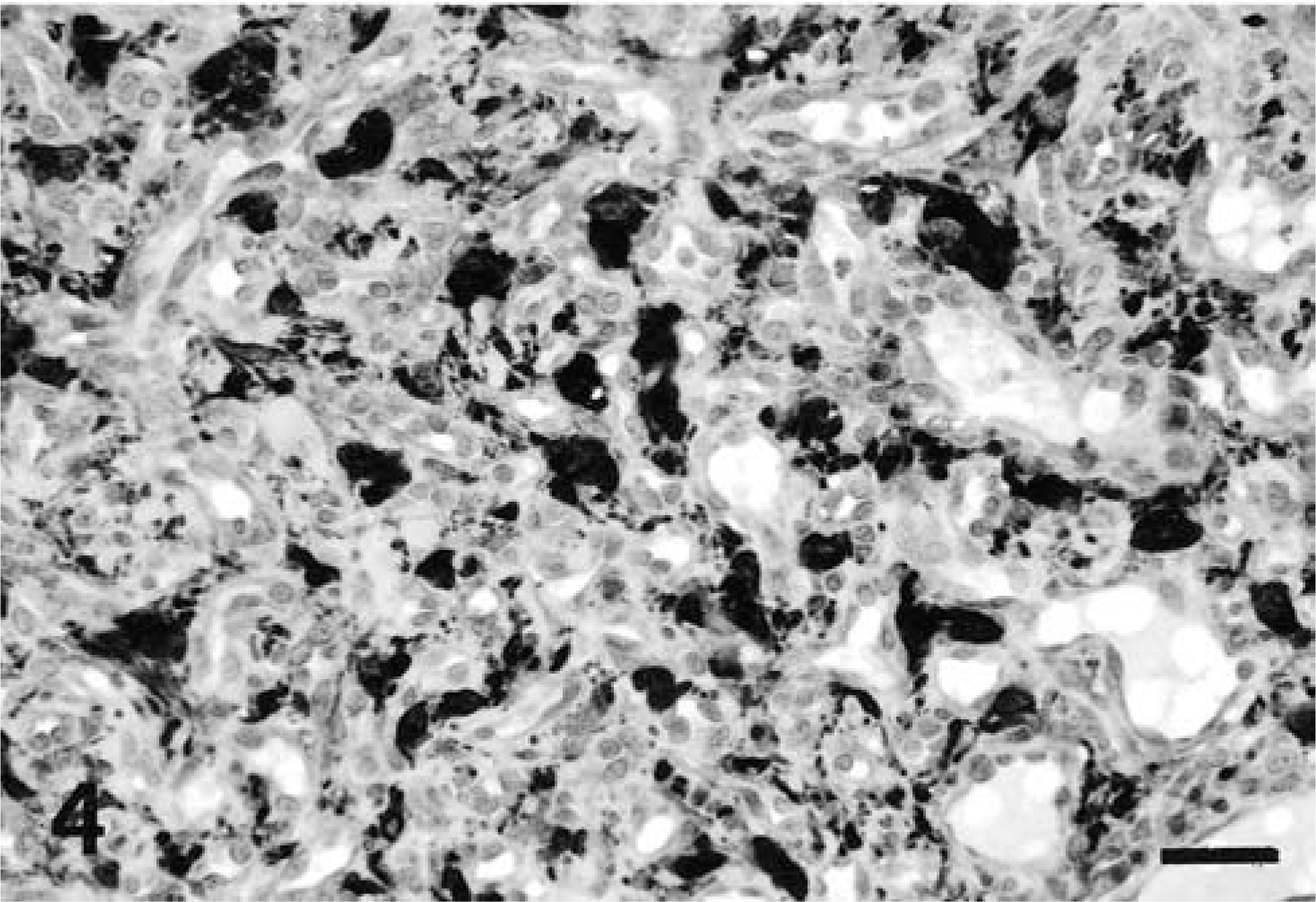
Spinal cord, hemangioblastoma; dog. Many stromal cells are strongly positive with NSE. The endothelial cells stain negative. Avidin–biotin–peroxidase complex method. Bar = 50 μm.
The morphologic features of this tumor closely resemble the description of human hemangioblastoma, 11 as well as that of the canine case diagnosed by the Armed Forces Institute of Pathology (AFIP). 1 The tumor is characterized by the presence of large polygonal interstitial cells packed between abundant and randomly oriented capillary vessels, lined by normal to plump endothelial cells. The tumor is also rich in reticulin that separates the vascular cells from the stromal cells. At least two histologic variants have been recognized in human hemangioblastomas: in the reticular variant the stromal cells are evenly distributed around the vascular network, whereas in the less common cellular variant the stromal cells are arranged in larger sheets or clusters. 5
Two cases of intramedullary vascular tumor of the spinal cord have been reported in dogs. 6 Although those tumors have been diagnosed as hemangiomas, their microscopic description was highly suggestive of hemangioblastoma because at least two cell types have been described: endothelial cells lining numerous capillaries and stromal fusiform to multipolar cells in about equal amounts. Scattered plasma cells and large perivascular cuffs of lymphocytes at the margins of the neoplastic mass have been described both in the case submitted to the AFIP 1 and in the tumors reported by Cordy. 6 The presence of a less vascular central area with abundance of collagen fibers, as well as a reticulin-rich fiber network, have also been described. 1,6 In all cases including this one, the tumors were intramedullary, abutted the pia mater, were centered to one side of the midline, and occurred in the caudal cervical to cranial thoracic regions. This spinal cord region, namely C6-T2, is the most commonly reported site for, at least, glial cell tumors in dogs. 16 The neurologic signs were progressive forelimb paresis, hemiparesis, 1,6 and progressive tetraparesis and neck rigidity in one case. 6 The histologic features of our tumor were also consistent with the MR findings, which showed a pattern of ring contrast enhancement indicative of pronounced vascularization and a less vascularized central zone.
Microscopically, differential diagnosis included other primary tumors with vascular components that have been rarely reported in the central nervous system of animals. A cavernous angioma has been described in the spinal cord of a dog and consisted of thin-walled dilated vessels without intervening tissue. 21 Vascular hamartomas have been described in the canine brain and consisted of excessive proliferation of thin-walled blood vessels. 18 One single report of such malformation has been reported in the spinal cord of a dog. 6 Because human hemangioblastomas present most often as masses attached to the dura, the differential diagnosis with angioblastic (or angiomatous) meningioma should be considered. 5 This variant has been occasionally described in dogs and is characterized by prominent blood vessels of different size surrounded by whorls of neoplastic meningeal cells. 15 Meningioangiomatosis has been rarely reported in the dog brain and is characterized by proliferated blood vessels and spindle-shaped concentrically arranged meningothelial cells. 17,19 Meningeal hemangiopericytoma is probably related to the soft tissue hemangiopericytomas and is composed of branching blood vessels surrounded by lobules or sheets of tightly packed polygonal cells. 4 Hemangioendothelioma of the brain is a multinodular tumor comprised of clusters of epithelioid cells often containing intracellular vacuoles and interstitial fibromyxoid matrix. 13 These last two tumor types have not been recognized in animal central nervous system up to now.
Our immunohistochemical results are widely comparable with those of most human hemangioblastomas
7–11
,
14,20
and with the case diagnosed by the AFIP.
1
Despite many investigations, the histogenesis of the tumor and particularly the origin of the stromal cells is still uncertain. Most immunohistochemical studies on human hemangioblastomas have reported frequent positivity of stromal cells for vimentin, S-100 protein, and NSE.
2,9,10,14,20
Endothelial markers (FVIII-rag and
Although the tumor of our report and human hemangioblastoma share many morphologic and immunohistochemical similarities, a few differences should be noted. Spinal hemangioblastomas in human beings are sometimes accompanied by a syrinx, and cerebellar hemangioblastomas are frequently cystic. 11 Stromal cells are often characterized by vacuolated lipid-rich cytoplasm and moderate amounts of glycogen. 5 An additional feature of human hemangioblastoma is the presence of mast cells containing tryptase and chymase. 12 None of these features were observed in our case, nor have they been described in similar canine tumors. 1,6 Conversely, central fibroplasia of the tumor and extensive peritumoral inflammation have not been described in human hemangioblastomas.
In conclusion, a novel spinal cord tumor in a dog is described on the basis of the histologic and immunohistochemical features. Although a complete overlapping of canine and human hemangioblastoma cannot be delineated, the inclusion of this tumor type within the primary tumors of canine central nervous system is suggested.
Footnotes
Acknowledgements
This study was supported by grants from the Italian Ministry of Education, University and Scientific Research (MIUR) and the University of Pisa.
