Abstract
Spinal cord glioma is uncommonly reported in dogs. We describe the clinicopathologic and diagnostic features of 7 cases of canine spinal cord glioma and briefly review the veterinary literature on this topic. The median age at presentation was 7.2 y. Six females and 1 male were affected and 4 dogs were brachycephalic. The clinical course lasted from 3 d to 12 wk, and clinical signs were progressive and associated with multiple suspected neuroanatomic locations in the spinal cord. Magnetic resonance imaging of 6 cases revealed T2-weighted hyperintense lesions with variable contrast enhancement in the spinal cord. All dogs had a presumptive clinical diagnosis of intraparenchymal neoplasia or myelitis based on history, advanced imaging, and cerebrospinal fluid analysis. Euthanasia was elected in all cases because of poor outcome despite anti-inflammatory or immunosuppressive treatment or because of poor prognosis at the time of diagnosis. Tumor location during autopsy ranged from C1 to L6, with no clear predilection for a specific spinal cord segment. The diagnosis was based on histopathology and the immunohistochemistry expression of glial fibrillary acidic protein, oligodendrocyte lineage transcription factor 2, 2′,3′-cyclic-nucleotide 3′-phosphodiesterase, neuron-specific enolase, synaptophysin, and Ki-67. Diagnoses consisted of 4 cases of oligodendroglioma, 2 cases of gliomatosis cerebri, and 1 astrocytoma. This case series further defines the clinicopathologic features of canine spinal glioma and highlights the need for comprehensive immunohistochemistry in addition to routine histopathology to confirm the diagnosis of these tumors.
Spinal cord tumors are classified as extradural (when arising from outside the dura mater), intradural or extraparenchymal (when located between the dura mater and the spinal cord), and intraparenchymal (when located in the spinal cord parenchyma).2,6 Most canine spinal cord tumors are extradural and include neoplasms of musculoskeletal origin, myeloma, lymphoma, hemangiosarcoma, liposarcoma, and metastatic tumors. Intradural and intraparenchymal tumors are less common and typically include cases of meningioma or nerve sheath tumor and glioma, respectively.2,22,27,31,42 Glioma is one of the most common primary central nervous system (CNS) neoplasms of dogs. 19 Gliomas arise from glial cells in the brain or spinal cord and are grouped into astrocytic, oligodendroglial, and ependymal tumors. Multiple glioma subtypes are also recognized, including gliomatosis cerebri, in which neoplastic glial cells disseminate diffusely through the parenchyma.23,34 Most cases of gliomatosis cerebri in the dog are believed to be astrocytic in origin, but other types of glioma can also be associated with disseminated spread. 34 Canine glioma typically affects older individuals, especially those of brachycephalic breeds, and tumors occur mainly in the frontal, parietal, or temporal telencephalic lobes, and less frequently in the brainstem and spinal cord. 19 Descriptions of canine spinal cord glioma are uncommon and typically consist of single case reports or cases included in large population studies that simultaneously evaluate other primary or secondary spinal cord tumors.22,25,26,28,29,31,33,36,43,47 In addition, most of these studies concentrate on the clinical aspects and clinical diagnosis of spinal cord neoplasms, with little emphasis given to the histopathology, grading, and immunohistochemistry (IHC) of such tumors.2,16,21,22,27,43,44,47
In this study, we describe the clinicopathologic and diagnostic features of 7 cases of canine spinal cord glioma retrospectively identified at the University of Georgia, Athens Veterinary Diagnostic Laboratory (AVDL; Athens, Georgia) and Cornell University, College of Veterinary Medicine, Section of Anatomic Pathology (CU-SAP; Ithaca, New York) between January 2000 and July 2015. Cases were retrieved from the archives, and all relevant information (signalment, history, clinical presentation, diagnostic workup, outcome, and pathology findings) was collected. All cases had a full neurologic examination and were studied at autopsy, which was performed within 24–72 h after death. Tissues were collected, fixed in 10% neutral-buffered formalin, sectioned at 5 μm, and stained using hematoxylin and eosin. IHC for glial fibrillary acidic protein (GFAP), oligodendrocyte lineage transcription factor 2 (Olig2), neuron-specific enolase (NSE), and synaptophysin (SYP) was performed according to AVDL standard procedures 35 (cases 1–4) and to CU-SAP procedures 9 (cases 5–7). In addition, IHC for and (CNPase) 2′,3′-cyclic-nucleotide 3′-phosphodiesterase and Ki-67 was performed according to CU-SAP procedures. 9 Positive controls consisted of normal canine spinal cord tissue. Hematoxylin and eosin–stained slides and IHC sections were reviewed by 2 pathologists (DR Rissi and AD Miller), and cases were graded according to the World Health Organization (WHO).18,23 Given the low frequency of canine spinal cord glioma, a review of the veterinary medical literature was conducted using the PubMed/MEDLINE database with the following key words: canine, dog, spinal cord, glioma, astrocytoma, oligodendroglioma, ependymoma, and gliomatosis cerebri.
The signalment and relevant clinical information were retrieved from selected cases (Table 1). The age of affected dogs varied from 6 to 10 y (median age: 7.2 y). Most dogs were female spayed (6 of 7), and brachycephalic breeds were slightly overrepresented (4 of 7). Clinical signs were progressive over 3 d to 12 wk and consistent with neuroanatomic localization to C6-T2, T3-L3, L6-S3, caudal spinal cord segments, or a combination of these. Magnetic resonance imaging (MRI) a of 6 cases revealed ill-defined multifocal or single lesions that were hyperintense on T2-weighted and fluid-attenuated inversion recovery images and hypointense on T1-weighted images with variable contrast enhancement (Fig. 1). The computed tomography scan of 1 dog (case 5) was normal. A clinical diagnosis of neoplasia or myelitis was made in 4 cases (1, 2, 6, and 7) and of neoplasia in 3 cases (3–5).
Signalment and clinicopathologic features of 7 spinal cord gliomas in dogs.*
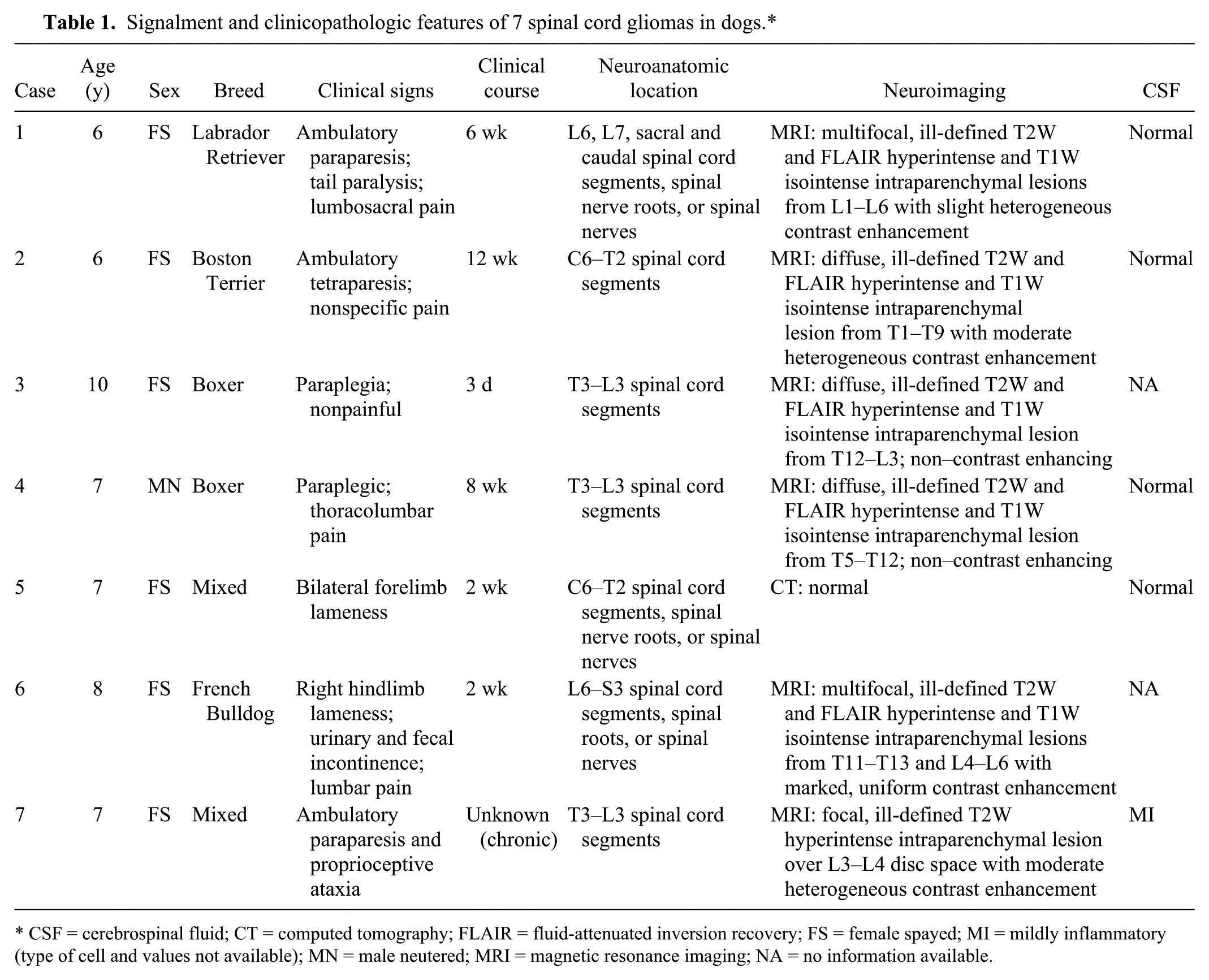
CSF = cerebrospinal fluid; CT = computed tomography; FLAIR = fluid-attenuated inversion recovery; FS = female spayed; MI = mildly inflammatory (type of cell and values not available); MN = male neutered; MRI = magnetic resonance imaging; NA = no information available.
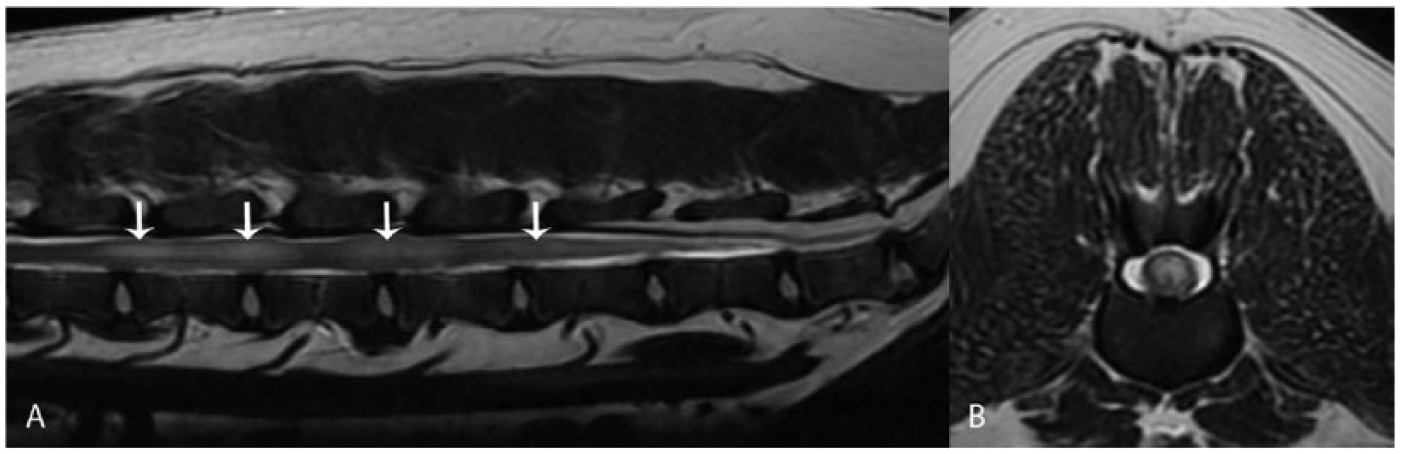
Magnetic resonance imaging of spinal cord oligodendroglioma in dog 1.
All affected dogs were submitted for autopsy after being euthanized because of poor outcome despite anti-inflammatory or immunosuppressive treatment or because of poor prognosis at the time of diagnosis. The main pathologic changes are summarized in Table 2. No apparent predilection for a specific spinal cord segment was evident. Gross changes were observed in 3 cases (3, 5, and 7) and consisted of poorly demarcated, dark red to gray, soft, gelatinous areas that mildly expanded the spinal cord parenchyma.
Pathologic features, immunohistochemistry, and final diagnosis of 7 spinal cord gliomas in dogs.*
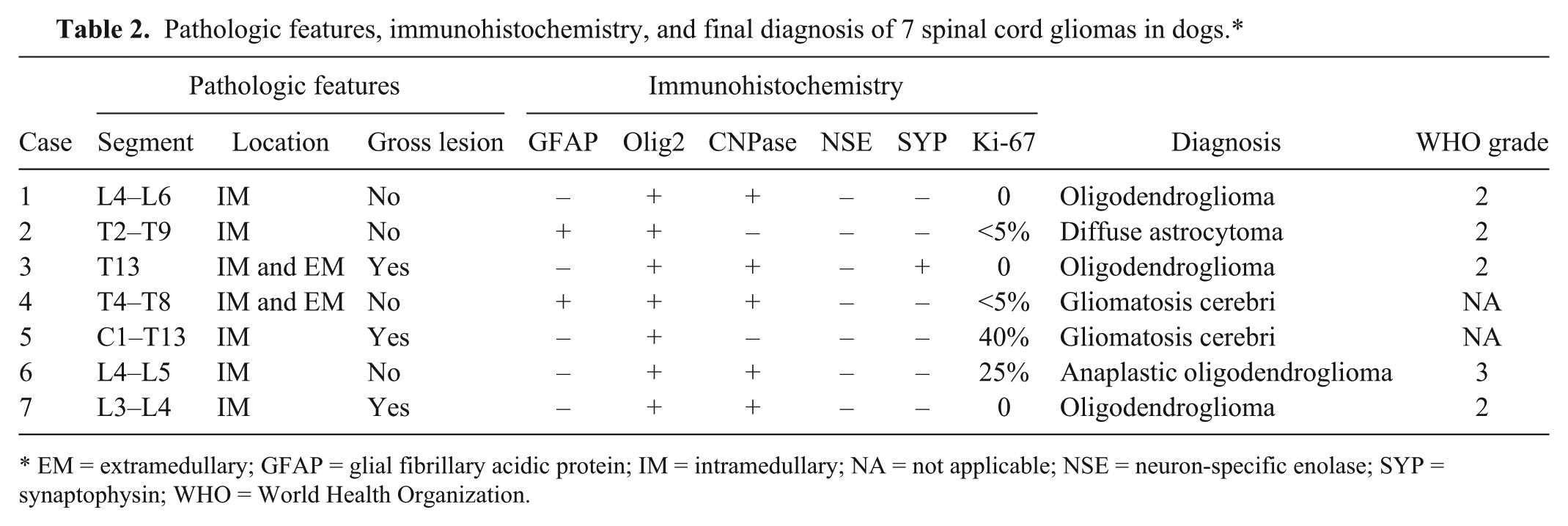
EM = extramedullary; GFAP = glial fibrillary acidic protein; IM = intramedullary; NA = not applicable; NSE = neuron-specific enolase; SYP = synaptophysin; WHO = World Health Organization.
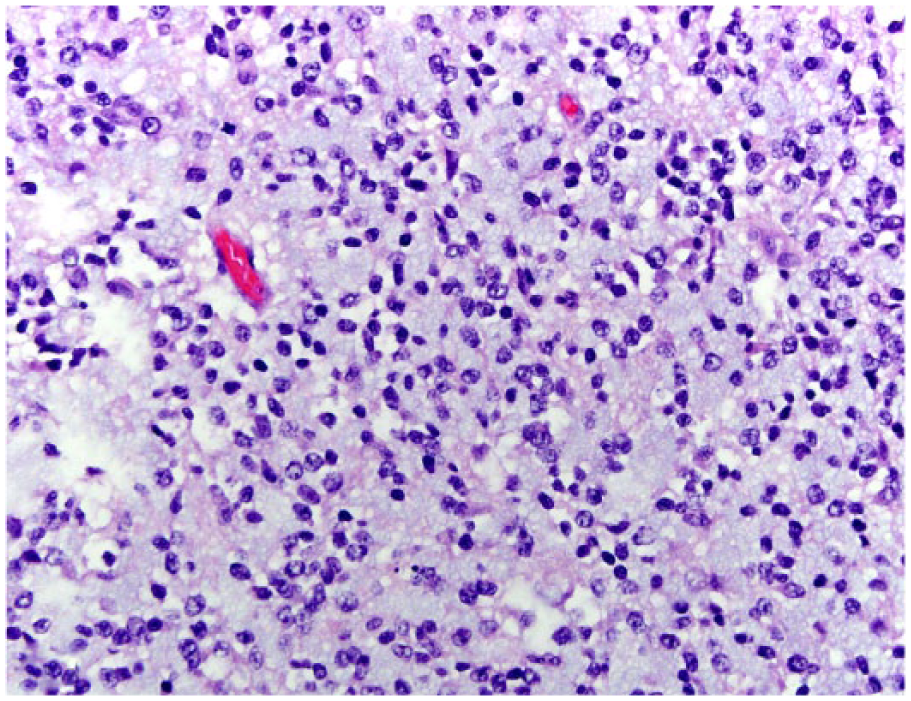
Histologically, all tumors were poorly demarcated, often infiltrative, and affected mainly the spinal gray matter. Oligodendrogliomas (cases 1, 3, 6, and 7) were poorly demarcated and affected primarily the gray matter, but also extended into the adjacent white matter (Fig. 2). Neoplastic cells were arranged in sheets or cords supported by preexisting parenchyma or a fine fibrovascular stroma with occasional microvascular clusters (glomeruloid vessels; cases 3 and 6). Neoplastic cells were mildly to moderately pleomorphic and had a moderate amount of cytoplasm, with distinct borders, that was either clear (forming a perinuclear halo) or contained basophilic, fibrillar, mucinous material. Nuclei were round-to-oval and had finely stippled to dense chromatin and 1–4 nucleoli. Overall mitotic activity was absent or low. Small areas of necrosis and hemorrhage were present in all 4 tumors. Case 6 was highly cellular and had increased cellular pleomorphism, with extensive areas of necrosis (Fig. 3) and 19 mitoses in ten 400× fields. Cases of gliomatosis cerebri (cases 4 and 5) were characterized by extensive, segmental, unilateral infiltration of neoplastic glial cells throughout the dorsal, lateral, and ventral funiculi and ventral horn of the spinal cord with no discrete mass formation (Fig. 4). There was a lack of distinction between the white and gray matter in the affected areas. Neoplastic cells were supported by preexisting neural tissue or small numbers of capillaries and had a moderate amount of round-to-polygonal, eosinophilic, vacuolated cytoplasm with indistinct borders. Nuclei were round-to-oval and had finely stippled chromatin with indistinct nucleoli. Mitotic index was 7 (case 4) and 11 (case 5) in ten 400× fields. Areas of hemorrhage or small perivascular accumulations of lymphocytes and plasma cells were observed occasionally. The astrocytoma (case 2) was poorly demarcated and affected both gray and white matter, although it appeared to arise primarily from the gray matter (Fig. 5). Neoplastic cells were widespread throughout the affected areas and were supported by preexisting neural tissue. These cells had moderate pleomorphism and scant, round-to-polygonal, eosinophilic cytoplasm with distinct borders. Nuclei were round to slightly elongate and had densely coarse chromatin with indistinct nucleoli. Mitoses were absent. IHC findings are summarized in Table 2 and shown in Figures 6–9. The main features of canine glial tumors of the spinal cord reported in the veterinary literature are summarized in Supplemental Table 1 (available at http://vdi.sagepub.com/content/by/supplemental-data).
Spinal oligodendroglioma in dog 3. Sheets of neoplastic cells admixed with mucinous material or a perinuclear clear halo efface the gray matter. H&E.
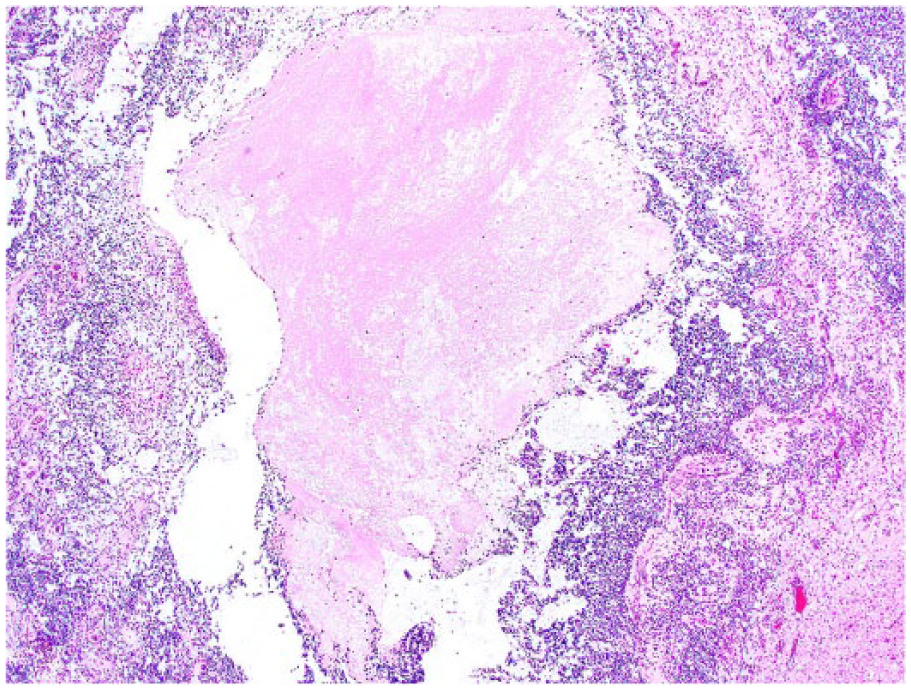
Spinal anaplastic oligodendroglioma in dog 6. The tumor is highly cellular with extensive areas of necrosis (center). H&E.
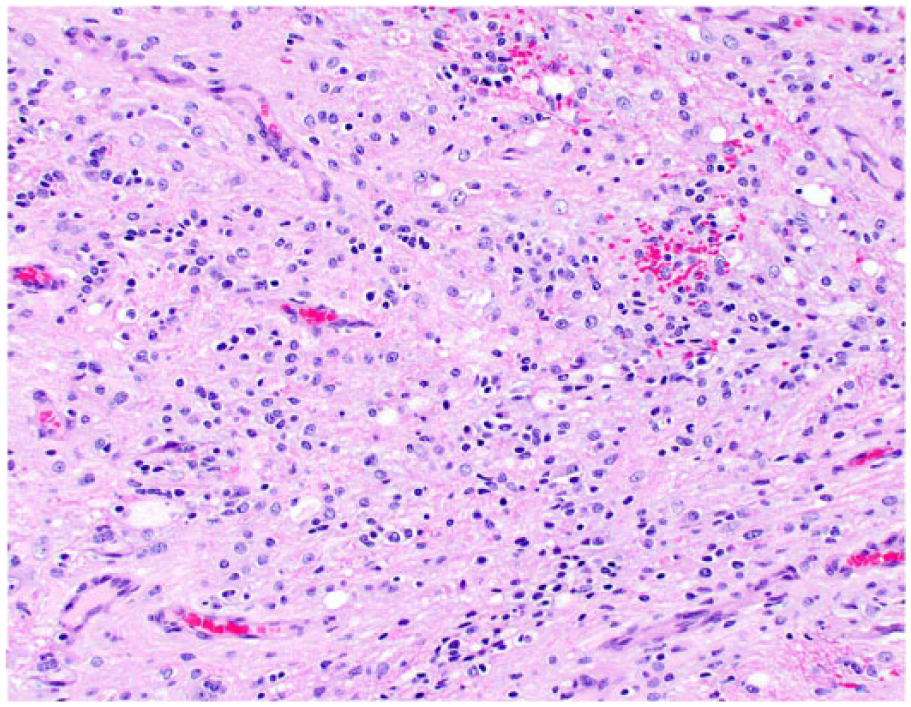
Spinal gliomatosis cerebri in dog 4. Neoplastic cells with indistinct cytoplasm and round nuclei are distributed throughout the gray matter and are admixed with small areas of hemorrhage. H&E.
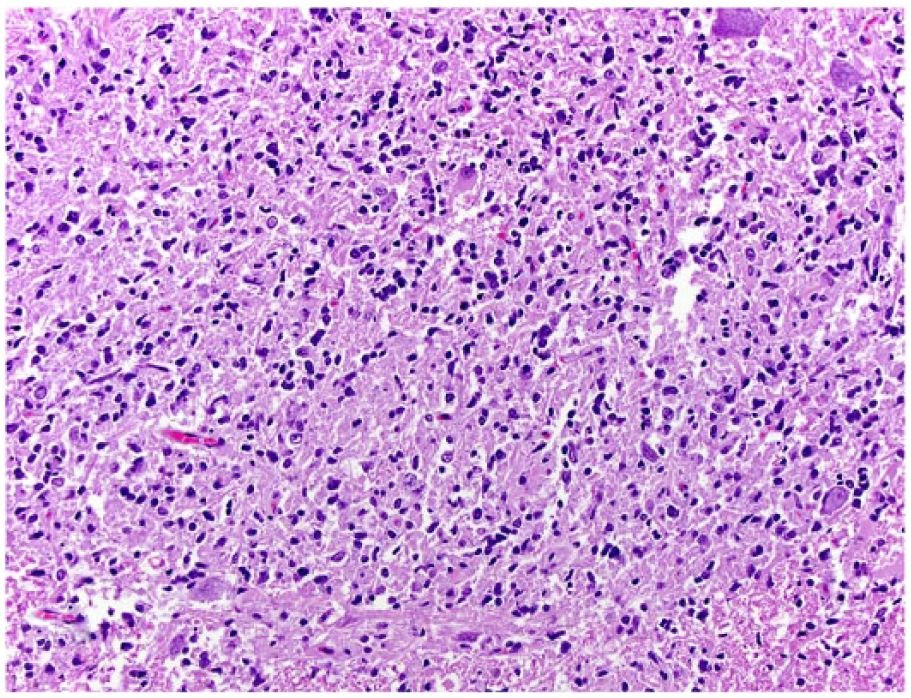
Spinal astrocytoma in dog 2. Neoplastic cells with indistinct cytoplasm and round-to-elongated nuclei efface the gray matter. H&E.
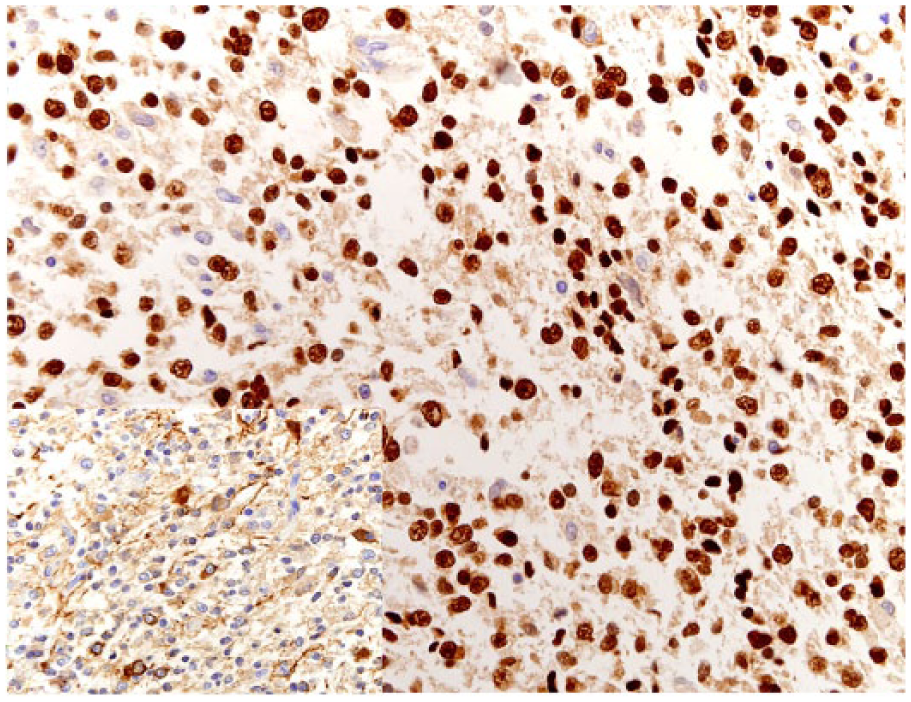
Spinal oligodendroglioma in dog 1. Neoplastic oligodendrocytes have strong nuclear immunostaining for Olig2. Bar = 100 µm. Inset: Spinal oligodendroglioma in dog 3. Neoplastic oligodendrocytes also exhibit membrane immunostaining for synaptophysin.
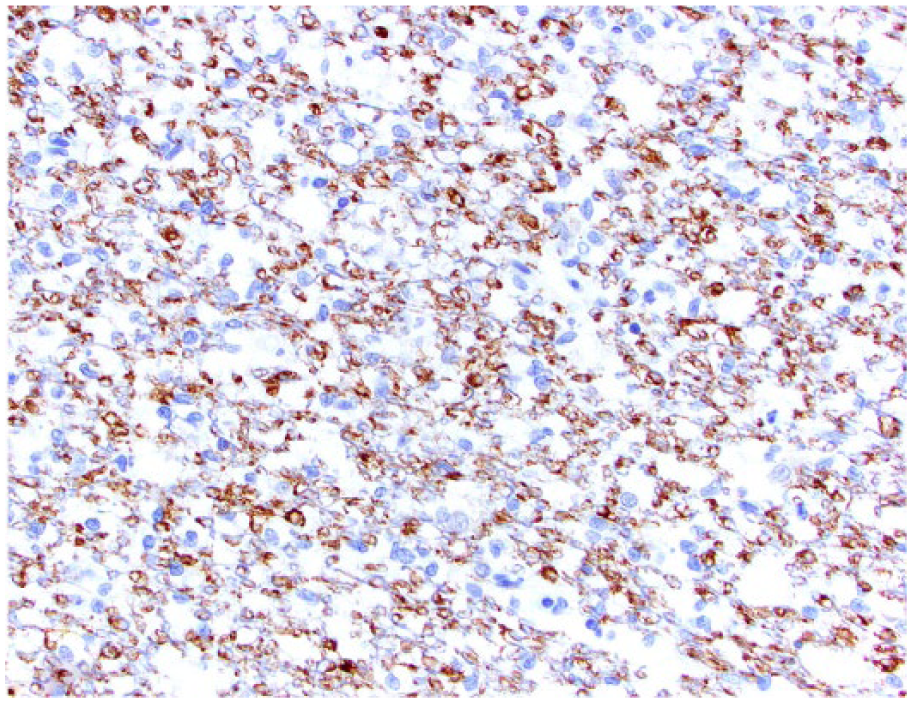
Spinal oligodendroglioma in dog 1. Neoplastic oligodendrocytes have cytoplasmic immunostaining for CNPase.
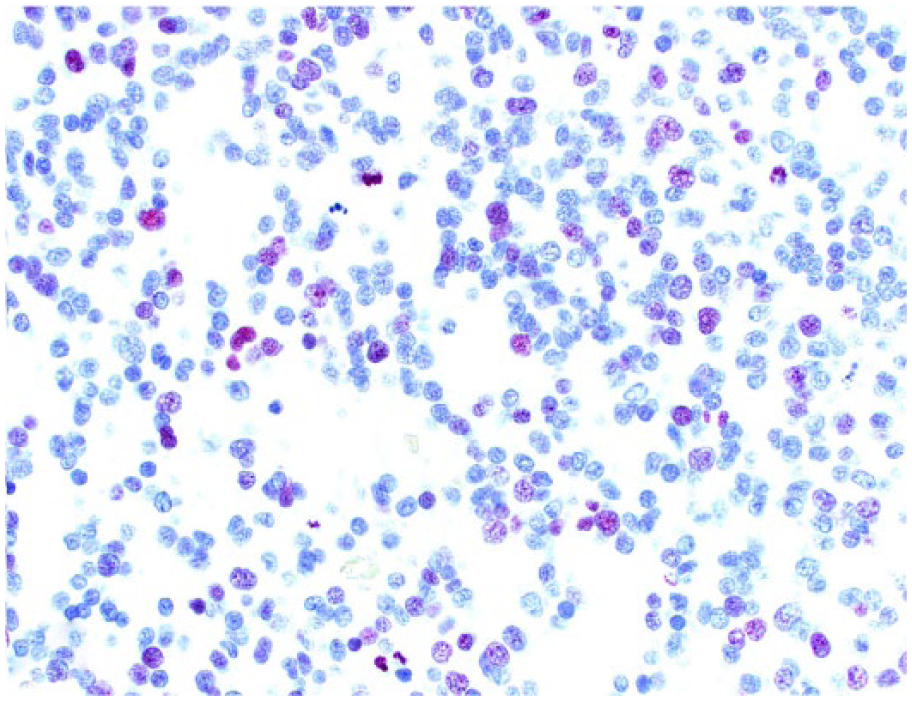
Spinal anaplastic oligodendroglioma in dog 6. Scattered neoplastic oligodendrocytes exhibit nuclear immunostaining for Ki-67.
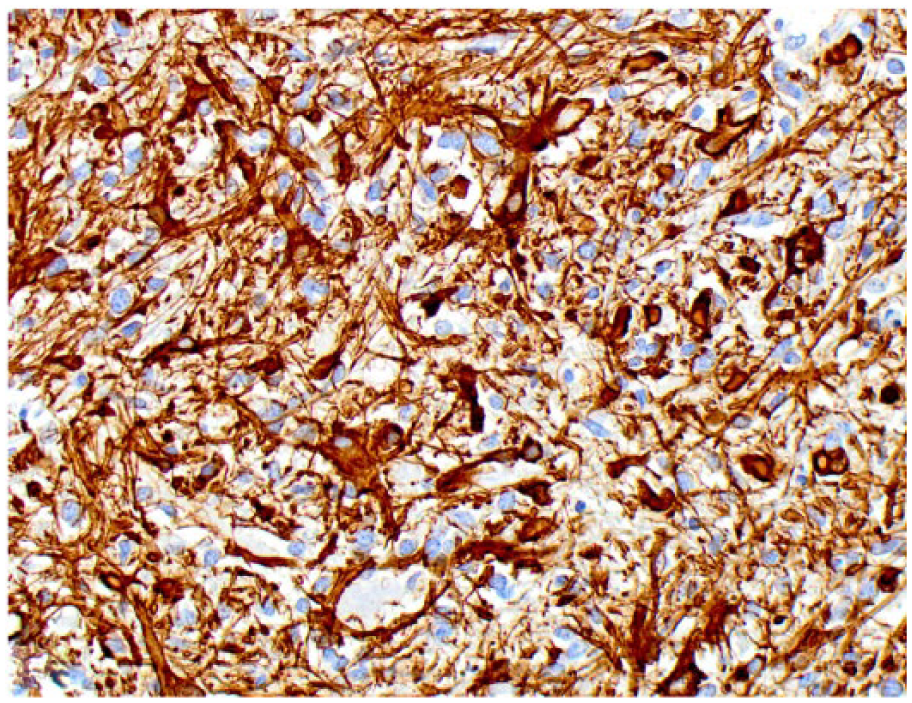
Spinal astrocytoma in dog 2. Neoplastic astrocytes have cytoplasmic immunostaining for glial fibrillary acidic protein.
Spinal cord glioma is an uncommon neoplasm of dogs, and its low prevalence is consistent with the AVDL diagnostic service routine, in which ~40 diagnoses of cerebral glioma and only 4 of spinal cord glioma (cases 1–4) have been identified over the past 15 y. Based on the reviewed veterinary medical literature, the most common type of canine spinal cord glioma is the ependymoma, with at least 24 reported cases.4,7,11,12,28,31,38,40,42,45 Ependymal tumors are followed by astrocytoma (14 cases),5,25,29,31,36,47 oligodendroglial tumors (10 cases),17,20,26,31,41 gliomatosis cerebri (4 cases),3,33,34 and unclassified glioma (3 cases).10,14,30 Although IHC staining can aid immensely in refining the diagnosis of brain and spinal cord tumors, such tests were not done in many of the reported cases, which could alter the actual frequency of these glioma types in the canine population. Our report describes 7 additional cases of canine intramedullary spinal cord glioma diagnosed by histopathology and IHC. We systematically classified and graded these tumors according to the WHO 23 and demonstrated the usefulness of an IHC panel in further defining the cell lineage in these cases. In addition, we confirmed CNPase IHC as a useful immunomarker in the diagnosis of oligodendroglioma in dogs.
Similar to our observations, spinal cord glioma tends to affect adult to older dogs, with no sex predisposition. Interestingly, ependymoma appears to affect younger dogs more commonly when compared to other gliomas.7,28,38,40,46 Although cerebral glioma typically occurs in brachycephalic breeds, 19 no such predisposition has been identified in cases of spinal cord glioma reported elsewhere. 31 Although 4 of our dogs were brachycephalic, this result should be interpreted with caution given the low number of cases in our study.
The clinical diagnosis and treatment of intramedullary spinal cord neoplasia may be challenging because of the rapidly progressive clinical course and neuroanatomic location of these tumors. 31 Although lesions may be initially intraparenchymal, thus making surgical resection difficult, tumors can expand and infiltrate other sites.16,31 This feature was observed in cases 3 and 4 of this report, in which the primary intraparenchymal tumors extended into and expanded the intradural spaces. The clinical diagnosis of glioma in humans relies heavily on neuroimaging, which is a noninvasive and accurate method that can be used not only to determine neuroanatomical location of the tumor, but also tumor type and grade. 15 As observed in the current study, diagnostic imaging is useful in detecting canine and feline cerebral and spinal cord tumors (including gliomas),9,15,39,44 but no distinct features have been identified in these cases that could reliably distinguish between tumor type or grade. 44
Canine glioma occurs more frequently as a solitary lesion in the brain and less frequently in the spinal cord.18,19 Affected spinal cord segments in our report were, in decreasing order of frequency, thoracic (3 cases), lumbar (3 cases), and cervical (1 case). Primary canine spinal cord tumors have been described more frequently affecting the cervical spinal cord. Spinal cord glioma tends to occur more often at the cervical and thoracic segments, followed by the lumbar and sacral areas.22,31 The available veterinary medical literature confirms a predisposition for these areas, with multiple cases of glioma reportedly occurring mainly in the cervical and thoracolumbar segments.3,5,7,10–12,14,17,20,25,26,28–31,33,34,36,38,40–42,46,47
Only 3 cases in our study had grossly visible spinal cord lesions and, therefore, diagnostic confirmation was achieved based on histopathology and IHC in all cases. The absence of gross changes in 4 cases may be related to the fact that tumors were poorly demarcated and not clearly distinct from the adjacent spinal cord parenchyma. The most common spinal cord glioma in our study was the oligodendroglioma. Although case 3 was histologically consistent with an oligodendroglial tumor, the remaining oligodendroglioma cases (1, 6, and 7) had morphologic features that could be common to both oligodendroglial and astrocytic neoplasms, indicating the need for IHC for diagnostic confirmation. Oligodendrogliomas were uniformly immunoreactive for Olig2 and immunonegative for GFAP and NSE. In addition, neoplastic cells had variable immunoreactivity for CNPase; case 3 was also immunoreactive for SYP. The astrocytoma was immunoreactive for GFAP and Olig2 and negative for CNPase, NSE, and SYP. Olig2 and GFAP are not exclusive markers for oligodendrocytes and astrocytes, respectively, and thus both oligodendrogliomas and astrocytomas can concurrently express Olig2 and GFAP immunopositivity, making the final diagnosis difficult in a subset of cases. However, expression of Olig2 and CNPase, as observed in our cases, is a solid indicator of the diagnosis of oligodendroglioma. 13 Similarly, positive GFAP immunostaining does not unequivocally confirm the diagnosis of astrocytoma, but it supports it when in combination with a negative IHC for Olig2 and CNPase.13,35 The occasional expression of neuronal markers (such as SYP and NSE) by cerebral or spinal cord gliomas indicates that, in addition to a glial cell origin, these tumors may share a neuronal–oligodendroglial progenitor cell of origin.32,35 Neoplastic cells in case 4 (gliomatosis cerebri) diffusely infiltrated the gray and white matter, but maintained the spinal cord architecture and had no discrete mass formation. These are regarded as the defining features of disseminated gliomatosis cerebri in human and veterinary medicine and can be used to distinguish gliomatosis cerebri from other gliomas, such as astrocytoma and oligodendroglioma, where an individual mass can be identified grossly or histologically, without evidence of parenchymal dissemination.34,37 The histogenesis of human gliomatosis cerebri remains controversial and, although cases with oligodendroglial differentiation have been reported occasionally, an astrocytic origin is supported in most cases based on the constant immunoreactivity of neoplastic cells to GFAP.1,24,37 The origin of these neoplasms is even more debatable in companion animals, with cases being reported to arise from astrocytes, oligodendrocytes, and microglia.8,9,34 The IHC profile in cases 4 and 5 in our study supports an oligodendroglial origin for the tumors. Ki-67 IHC staining was variable and more pronounced in cases 5 (gliomatosis cerebri) and 6 (anaplastic oligodendroglioma). Similarly, although the presence of microvascular proliferation and increased cellularity and pleomorphism are suggestive of malignancy in cases of glioma, evaluation of proliferative cell markers together with routine histopathology has been shown to be helpful when grading feline spinal cord gliomas. 9
The cases presented in our study provide additional data regarding the clinicopathologic and diagnostic features of canine spinal cord gliomas. An IHC panel can be useful in further defining the cell lineage in cases of glioma, and the evaluation of proliferative cell markers may be useful for the final diagnosis and grading of these tumors.
Footnotes
Acknowledgements
We thank Patricia Rowe from the Histology Laboratory at the Athens Veterinary Diagnostic Laboratory (UGA) for her technical assistance with immunohistochemistry.
Authors’ contributions
DR Rissi contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. R Barber contributed to design of the study; contributed to analysis and interpretation of data; and drafted the manuscript. A Burnum contributed to acquisition and interpretation of data. AD Miller contributed to design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
GE 3.0 Tesla Signa HDx, GE Healthcare, Milwaukee, WI.
Declaration of conflicting interests
The author (s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author (s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
