Abstract
In routine diagnostic activity, pathologists may be confronted with nervous system tumors. The lack of clinical information, economic restrictions for additional testing, and the lack of expertise in neuropathology may render the diagnosis challenging. The goals of this study were to assess the agreement in diagnosing nervous system tumors in domestic carnivores among 4 board-certified surgical pathologists without particular expertise in neuropathology and a neuropathologist expert, and to investigate the utility of special stains frequently used in routine diagnostic laboratories. Forty-six tumors (7 cats, 38 dogs, and 1 unknown carnivore) were retrieved and 1 hematoxylin and eosin–stained slide per tumor was selected. Diagnoses (tumor type and subtype) were formulated based on histological features and available clinical information. Confidence in the diagnosis was also scored. Subsequently, a panel of histochemical and immunohistochemical stains (Gordon Sweet silver stain and immunohistochemistry for AE1/AE3, vimentin, glial fibrillary acid protein, S100, neuron-specific enolase and neurofilament) was evaluated by the pathologists, who either confirmed or changed their original diagnoses. Intraobserver and interobserver agreement and confidence in relation to diagnosis before and after analysis of special stains were assessed. The use of special stains increased the complete agreement among surgical pathologists, with regard to tumor type, from 63% to 74%. Cases with a high confidence score had a higher interobserver agreement than cases with a low confidence score. These results suggest that pathologists without expertise in neuropathology agree in the diagnosis of most nervous system tumors, and special stains available in most laboratories only slightly increase this agreement.
Primary tumors of the nervous system are histopathologically and ontogenically heterogeneous. Among domestic animal species, they occur most frequently in dogs and cats, while other species are only sporadically affected. 5 Data on prevalence of nervous system tumors in dogs and cats are sparse. The prevalence of canine nervous system tumors, based on necropsy data, is 2% to 4.5%. 6,17 In dogs, over 50% of all gliomas occurs in brachycephalic breeds (eg, Boxer, Boston Terrier, English and French Bulldog, English Toy Spaniel), while dolichocephalic breeds are more commonly affected by meningiomas. In cats, the frequency of intracranial neoplasia is 0.01% and meningiomas are more frequently reported. 14,19
In human medicine, the latest edition of the World Health Organization classification 10 of central nervous system tumors is largely based on morphological, immunohistochemical, and genetic analyses. In veterinary medicine, since data on molecular and genetic markers are still limited, histopathology remains the gold standard for tumor diagnosis. Diagnostic veterinary neuropathology, being considered a highly specialized branch of veterinary pathology, has been performed on a regular/routine basis in few specialized centers worldwide and by few pathologists with a personal interest in this subject. The increasing level of specialization of veterinary neurologists and neurosurgeons and the availability of advanced surgical and imaging procedures are enabling a rising number of nervous system specimens to be analyzed by pathologists working in diagnostic veterinary laboratories. Unfortunately, pathologists working in these institutions, even when board certified and with expertise in surgical pathology, may struggle to diagnose nervous system tumors correctly, due to the lack of familiarity with the pathological diagnosis of nervous system diseases, insufficient clinical information, and, at times, limited availability of (immuno)histochemical stains.
Pathologist interobserver agreement is a main concern in human neuropathology. Several studies have been performed, demonstrating variable diagnostic discrepancy, depending on tumor type and grade. 13,15,20 To our knowledge, in veterinary medicine, no such studies have been performed for nervous system tumors. The goal of the present study was to assess the agreement among 4 board-certified surgical pathologists without thorough expertise in neuropathology and a neuropathology expert in diagnosing nervous system tumors in domestic carnivores.
Materials and Methods
Study Design
This study was set up to investigate the ability of surgical pathologists without specific expertise in neuropathology to diagnose nervous system tumors, without use of neuropathologically relevant immunohistochemical stains. To this end, 46 tumors, previously diagnosed as nervous system tumors, were retrieved from the histopathology archive of VetAgro Sup, Lyon (France). Inclusion criteria were nervous system tumor (based on the diagnosis present on the original histopathology report), domestic carnivore, and any location except for skin. For each tumor, sections were stained with hematoxylin and eosin (HE) and a panel of histochemical and immunohistochemical stains that was predefined based on the general availability of stains in routine veterinary diagnostic laboratories. Tumor sections were microscopically evaluated by 4 surgical pathologists without expertise in neuropathology, defined as receiving fewer than 15 central nervous system cases per year, and 1 board-certified pathologist with longstanding expertise in neuropathology. Each pathologist formulated a tumor-type and tumor-subtype diagnosis based on HE stains only (“first diagnosis”). Clinical information (species, breed, age, sex, and location of the tumor) was provided when available. Each pathologist rated her or his confidence in the provided diagnosis as either low or high. Subsequently, each pathologist reevaluated the HE-stained slides together with the provided histochemical and immunohistochemical stains (and with knowledge of his or her first diagnosis) and confirmed or changed the diagnosis accordingly (“second diagnosis”). All participants (surgical pathologists and the neuropathologist) were blinded to the other pathologists’ diagnoses during the entire evaluation of tumor slides. The inter- and intraobserver agreement was analyzed at the end of the study.
Tumor Tissues and Stains
For each tumor, eight 4-µm-thick sections were obtained. One was stained with HE. In another section, reticulin fibers were stained with the Gordon Sweet technique (Kit Gordon Sweet; Bio-optica, Milano, Italy). This technique aids in differentiating peripheral nerve sheath tumors (PNSTs), meningiomas, and gliomas based on the presence of basement membranes around single tumoral cells in PNSTs, reticular fibers around groups of cells in meningiomas, and the lack of a stroma and a basement membrane in gliomas. 2 The remaining 6 sections were immunolabeled for vimentin, wide-spectrum cytokeratin, S100, glial fibrillary acid protein (GFAP), neurofilament, and neuron-specific enolase (NSE). Vimentin positivity combined with variable positivity for cytokeratin may support the diagnosis of meningioma. Combined positivity to GFAP, S100, and vimentin may indicate the diagnosis of astrocytoma, while oligodendrogliomas may exhibit S100 positivity accompanied by lack of vimentin and GFAP expression. PNSTs may be variably positive to S100, GFAP, and NSE. 3,4,7,8 Neurofilament and NSE may support neuronal origin of tumor cells. 6
Immunohistochemistry was performed with the avidin-biotin-peroxidase complex method, using antibodies against cytokeratin AE1/AE3 (clone AE1/AE3, diluted 1:50; Dako, Carpinteria, CA), GFAP (rabbit polyclonal antibody, diluted 1:200; Dako), neurofilament (clone 2F11, diluted 1:100; Cell Marque, Sigma Aldrich, Rocklin, CA), NSE (clone BBS/NC/VI-H14, diluted 1:100; Dako), S100 (S100B rabbit polyclonal antibody, diluted 1:400; Dako), and vimentin (clone V9, 1:50; Dako). For anti-GFAP and antineurofilament, no antigen retrieval was applied. For the remaining antibodies, antigen retrieval was performed by heating tissue sections at 90°C for 40 minutes in citrate buffer at pH 6, followed by a 20-minute cooling. Labeling was amplified by the last-step product of the ultraTek HRP (antipolyvalent) ready to use kit (ScyTek Laboratories, Logan, UT) (30 minutes at 20°C) and revealed with Vector NovaRED Peroxidase (HRP) Substrate kit (Vector, Burlingame, CA) (5 minutes). Hematoxylin counterstain (5 minutes) was applied, and then sections were dehydrated and mounted. The negative control consisted of the replacement of the primary antibody with the antibody diluent solution (Emerald diluent antibody; Cell Marque, Sigma Aldrich).
Tumor Diagnosis and Interpretation of Stains
Diagnoses (tumor type and subtype) were formulated independently by 4 board-certified surgical pathologists and the neuropathologist before (“first diagnosis”) and after (“second diagnosis”) evaluation of special stains. Tumor types and subtypes were defined on the basis of the classification in Tumors in Domestic Animals 6 with 4 adaptations: tumors of neuroepithelial origin were called gliomas, pituitary tumors were included in the tumor type called tumors of the sellar region, glioblastoma was considered a tumor subtype, and orbital meningioma was included in the meningioma tumor type category (Suppl. Table S1). Confidence in the first diagnosis was also scored (low or high) by each pathologist. To avoid bias of the pathologists in the tumor diagnosis, there was no decisional algorithm for interpretation of (immuno)histochemical stains. Each pathologist interpreted the stained slides on her or his own, using Tumors in Domestic Animals 6 as the main reference and peer-reviewed articles if needed.
Analysis of Results
The diagnoses obtained from the 4 surgical pathologists and the neuropathologist were analyzed considering the following parameters: (1) interobserver agreement on tumor type diagnosis, among surgical pathologists, and between surgical pathologists and the neuropathologist, at first and second diagnosis; (2) interobserver agreement among surgical pathologists and between surgical pathologists and the neuropathologist on tumor subtype at the first and second diagnosis; and (3) interobserver and intraobserver agreement in relation to the confidence score given by each pathologist. The agreement among surgical pathologists was classified as complete (agreement among all 4 surgical pathologists), major (accordance between 3/4 surgical pathologists), partial (accordance between 2/4 surgical pathologists), and absent (4 different diagnoses).
Interobserver agreement on tumor type at first and second diagnosis, among surgical pathologists and among the whole panel of pathologists (surgical pathologists and the neuropathologist), was evaluated by statistical analysis calculating κ for categorical nominal using a web-based program (www.statstodo.com). A κ of 1 indicates a perfect agreement, whereas a κ of 0 indicates an agreement equivalent to chance. 22 The κ cutoff values for different levels of agreement are reported in Supplemental Table S2.
The raw data analyzed in this study can be requested from the author.
Results
Of the 46 examined tumors selected for the study, 7 were from cats, 38 were from dogs, and in 1 case the carnivore species was unknown. Twenty-two animals were females, 21 were males, and in 3 cases the sex was unknown. Tumor location, as defined by the clinician, was intracranial in 24 cases, intraspinal in 16 cases, extracranial and extraspinal in 3 cases, intra- and extracranial in 1 case, and unknown in 2 cases (Suppl. Table S3).
Interobserver Agreement in Tumor Type Diagnosis
At first diagnosis, agreement between surgical pathologists was complete in 29 cases out of 46 (63%), major in 9 cases (20%), partial in 7 cases (15%), and absent in only 1 case (2%). At second diagnosis, the complete agreement increased to 74% (Table 1). Hence, the use of (immuno)histochemical stains increased the complete agreement between surgical pathologists by 11%. Statistical analysis identified a strong agreement both at first (κ = 0.66, P < .0001) and second diagnosis (κ = 0.76, P < .0001) with the latter being slightly higher.
Interobserver Agreement Among Surgical Pathologists at First and Second Diagnosis, in 46 Nervous System Tumors.
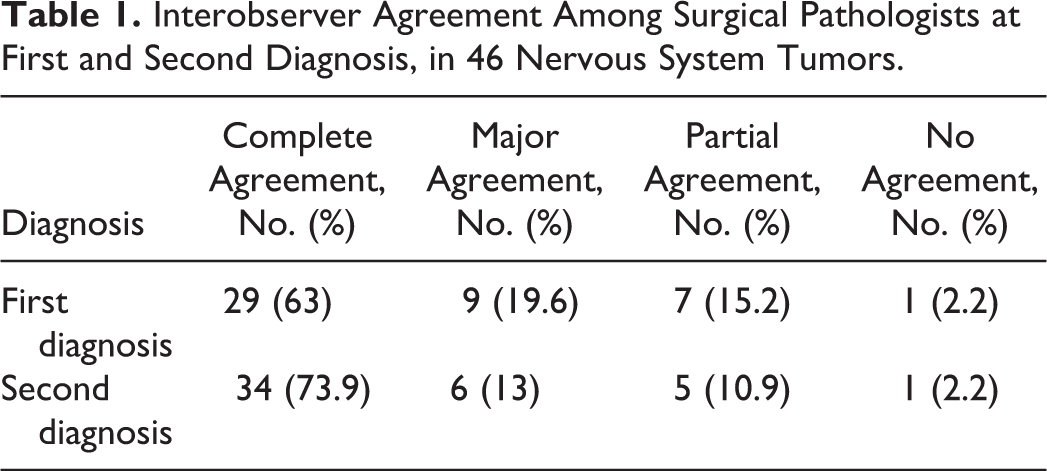
Thus, major to complete agreement was obtained in 38 cases (83%) at first diagnosis and in 40 cases (87%) at second diagnosis. The 38 cases with major to complete agreement at first diagnosis included 16 meningiomas (42%), 15 gliomas (40%), 3 PNSTs (8%), 2 ectopic nephroblastomas of the canine thoracolumbar spinal cord (5%), and 1 sarcoma (3%). Among the 40 cases with major to complete agreement at second diagnosis, there were 15 meningiomas (38%), 14 gliomas (35%), 5 PNSTs (13%), 2 ectopic nephroblastomas of the canine thoracolumbar spinal cord (5%), 2 sarcomas (5%), and 1 pituitary tumor (3%). Considering the neuropathologist diagnosis as reference, the tumor type for which surgical pathologists mostly agreed with the neuropathologist on the first diagnosis were gliomas (100% and 93% of agreement, respectively, at first and second diagnosis), meningiomas (94% and 88% of agreement, respectively, at first and second diagnosis), and ectopic nephroblastomas of the canine thoracolumbar spinal cord (100% of agreement at first and second diagnosis). Special stains increased the agreement significantly for PNSTs, which passed from 43% of agreement at first diagnosis to 71% at second diagnosis (Suppl. Table S4).
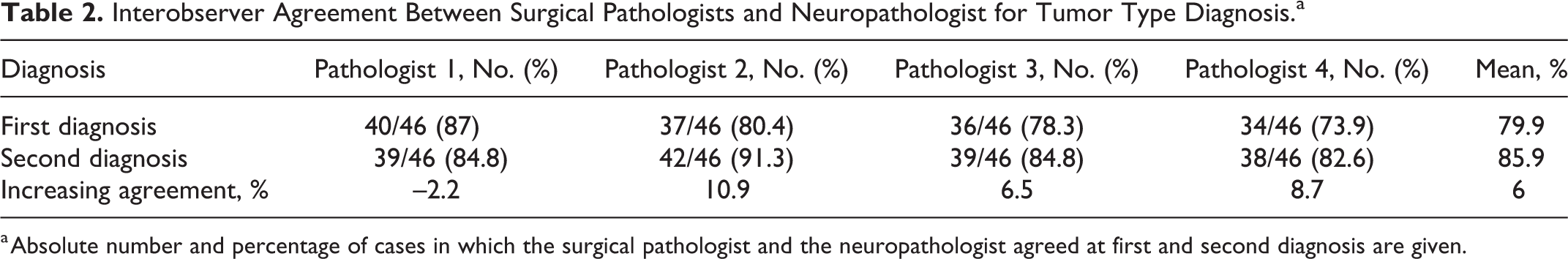
Each surgical pathologist agreed with the neuropathologist in more than 73% of the cases at first diagnosis, while at second diagnosis, interobserver agreement slightly increased for 3 pathologists and decreased for the fourth (Table 2). Statistical analysis identified a strong agreement between the neuropathologist and the surgical pathologists both at first (κ = 0.6804, P < .0001) and second diagnosis (κ = 0.7763, P < .0001), with a slight increase at second diagnosis.
Interobserver Agreement Between Surgical Pathologists and Neuropathologist for Tumor Type Diagnosis.a
a Absolute number and percentage of cases in which the surgical pathologist and the neuropathologist agreed at first and second diagnosis are given.
Interobserver Agreement in Tumor Subtype Diagnosis
Considering only cases with major to complete agreement in tumor type, surgical pathologists agreed on tumor subtype in 28 out of 38 cases (74%) at first diagnosis and in 26 out of 40 cases at second diagnosis (65%). The neuropathologist agreed with the majority of the surgical pathologists in 18 out of 38 (47%) cases at first diagnosis and in 20 out of 40 cases at second diagnosis (50%).
In summary, special stains did not increase the interobserver agreement for tumor subtype.
Confidence in Tumor Diagnosis
Confidence in tumor type diagnosis was rated by all pathologists (surgical pathologists and the neuropathologist) at first diagnosis. Two surgical pathologists (1 and 2) were confident in a high number of cases (29 and 32, respectively), but after evaluation of the special stains, they did not confirm the original diagnosis in 3 and 4 cases, respectively (Table 3). One pathologist (pathologist 2) changed only the subtype for all 3 cases. The neuropathologist agreed with the first subtype diagnosis in 2 cases and with the second subtype diagnosis in the third one. The other pathologist (pathologist 1) changed the tumor type in 2 cases and the subtype for the other 2 cases. The neuropathologist agreed with the first diagnosis in 2 cases and proposed a different diagnosis in the other 2.
Diagnostic Changes Before and After Special Stain Analysis for the Cases With a High Confidence Rate.
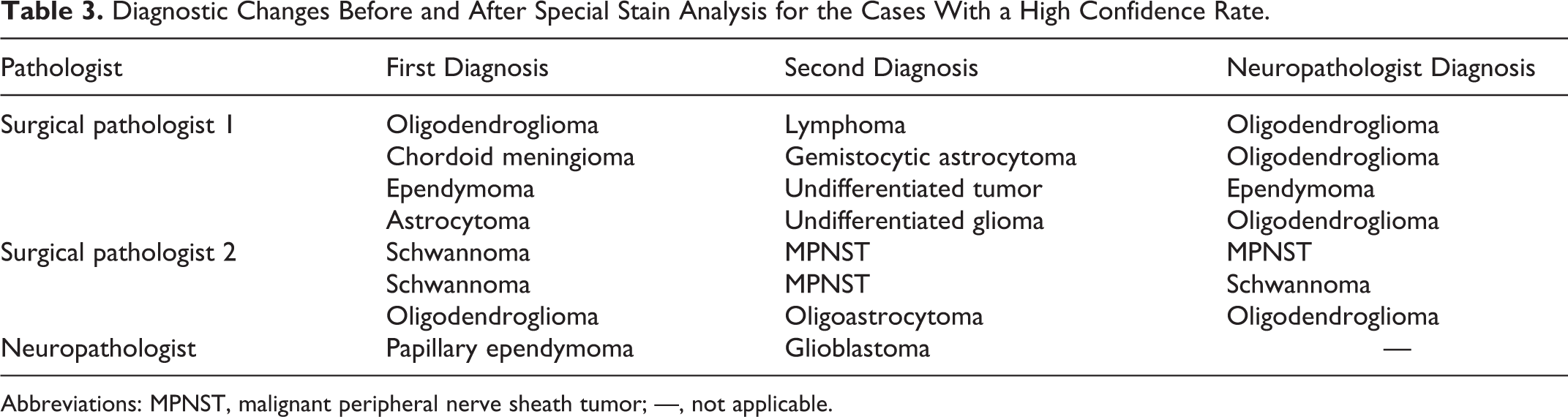
Abbreviations: MPNST, malignant peripheral nerve sheath tumor; —, not applicable.
Two other surgical pathologists (3 and 4) had a lower number of highly confident diagnoses (23 and 24 cases, respectively), but these were always confirmed after examination of the special stains. The neuropathologist had the greatest number of highly confident diagnoses (39), which were all confirmed for tumor type at second diagnosis (Suppl. Table S5).
When comparing the confidence score to the agreement between surgical pathologists at first diagnosis, most of the cases with a high confidence score were the cases with a complete agreement. Most of the diagnoses with a major agreement were considered of low confidence.
Discussion
The aim of the study was to investigate the ability for board-certified pathologists without longstanding expertise in neuropathology to diagnose nervous system tumors in dogs and cats and to evaluate how special stains commonly available in routine diagnostic laboratories could improve nervous system tumors diagnosis.
With regard to the tumor type, surgical pathologists obtained a complete or major interobserver agreement in 83% of the cases at first diagnosis. Each pathologist agreed with the neuropathologist at first diagnosis in 74% to 87% of cases, which statistically resulted in a strong agreement. These results suggest that surgical pathologists, despite their subjective difficulty to diagnose nervous system tumors, are able to type the tumor in most of cases.
According to the neuropathologist and in agreement with the current literature, 6,16,17,19 the most common tumor types in the present study were meningiomas and gliomas (oligodendroglioma and glioblastoma), and their high prevalence could explain the major or complete interobserver agreement in the majority of the cases.
There might be various reasons for the interobserver disagreement in the remaining 17% of tumor cases. Lack of clinical information, particularly precise localization of the tumor and unavailability of magnetic resonance imaging (MRI) and computed tomography (CT) scan results, might have affected the pathological diagnosis. Availability of anamnestic data depends on the compliance of veterinarians to submit detailed clinical information, on their access to advanced imaging techniques, and on economical restrictions set by the owners. In this context, this study supports the notion that in neuropathology, close collaboration between clinicians and pathologists is essential for correct tumor diagnosis. Imaging techniques (MRI, CT scan) serve as a surrogate for macroscopic pathology, allowing the precise localization and providing biochemical information on the mass, hence significantly narrowing the possible differential diagnoses. 6,21 Histopathologically speaking, certain nervous system tumors might be more difficult to diagnose than others and might need more expertise. Interobserver agreement was high in tumors such as meningioma, glioblastoma, choroid plexus tumors, and ectopic nephroblastoma of the lumbar spinal cord, potentially because of characteristic morphologic features (Figs. 1–3). In contrast, agreement was particularly low in PNSTs. From our data, PNSTs are apparently at risk of being misdiagnosed by surgical pathologists without expertise in neuropathology as transitional or fibrous meningioma (Figs. 4, 5) when data on exact location are not available because of some overlap in cellular morphology. Interobserver agreement on PNSTs was significantly increased at second diagnosis following assessment of special stains. Features that were used for confirmation of PNST included GFAP and NSE expression and absence of AE1/AE3 expression. 8,9 Subjectively, the staining of reticulin fibers with the Gordon Sweet technique appeared to be very useful for the discrimination of PNSTs and meningiomas, given that in PNSTs, neoplastic cells are individually surrounded by basement membranes, while in meningiomas, reticulin fibers surround groups of cells.
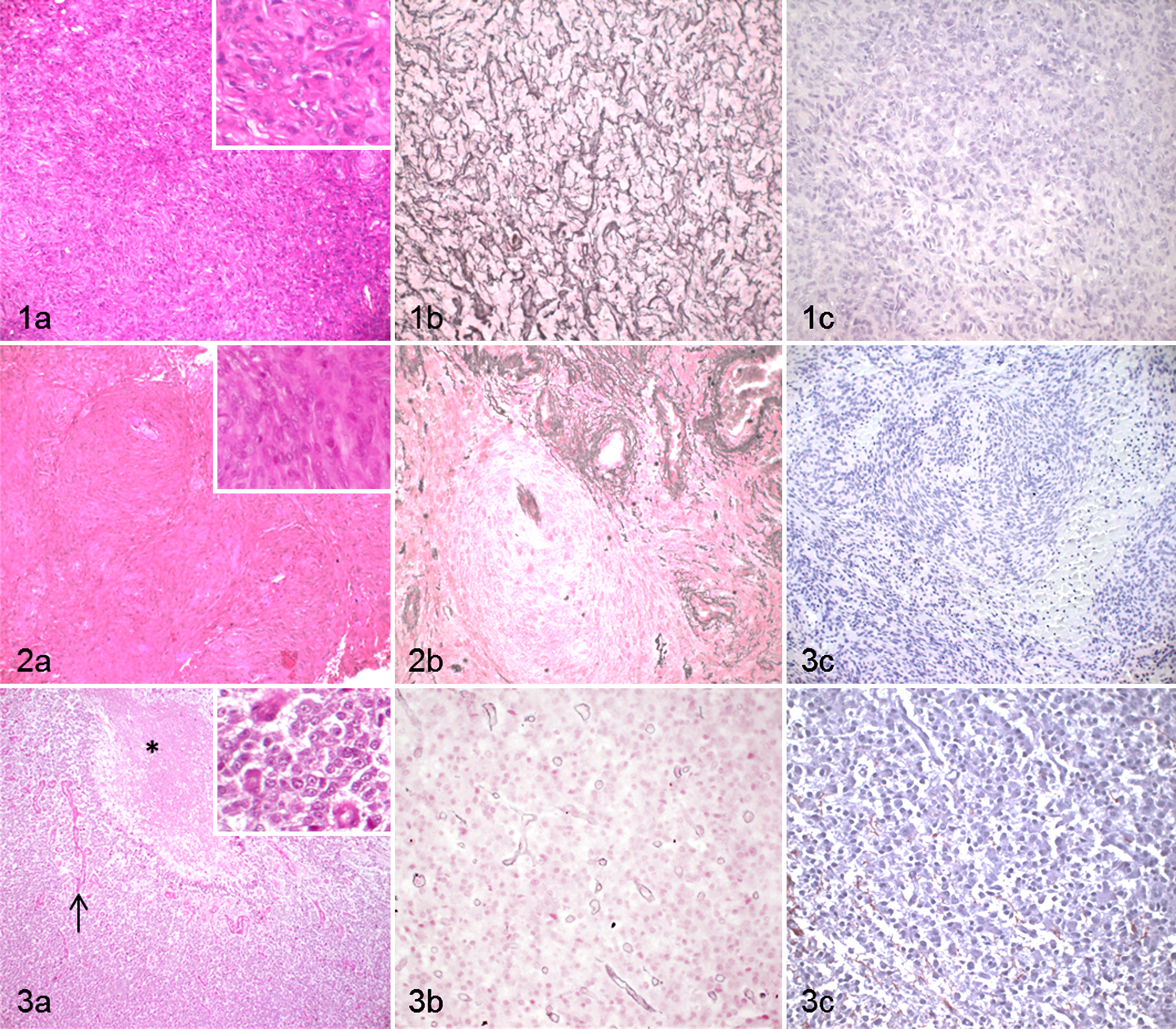
Examples of cases with a high agreement for tumor type among surgical pathologists and the neuropathologist.
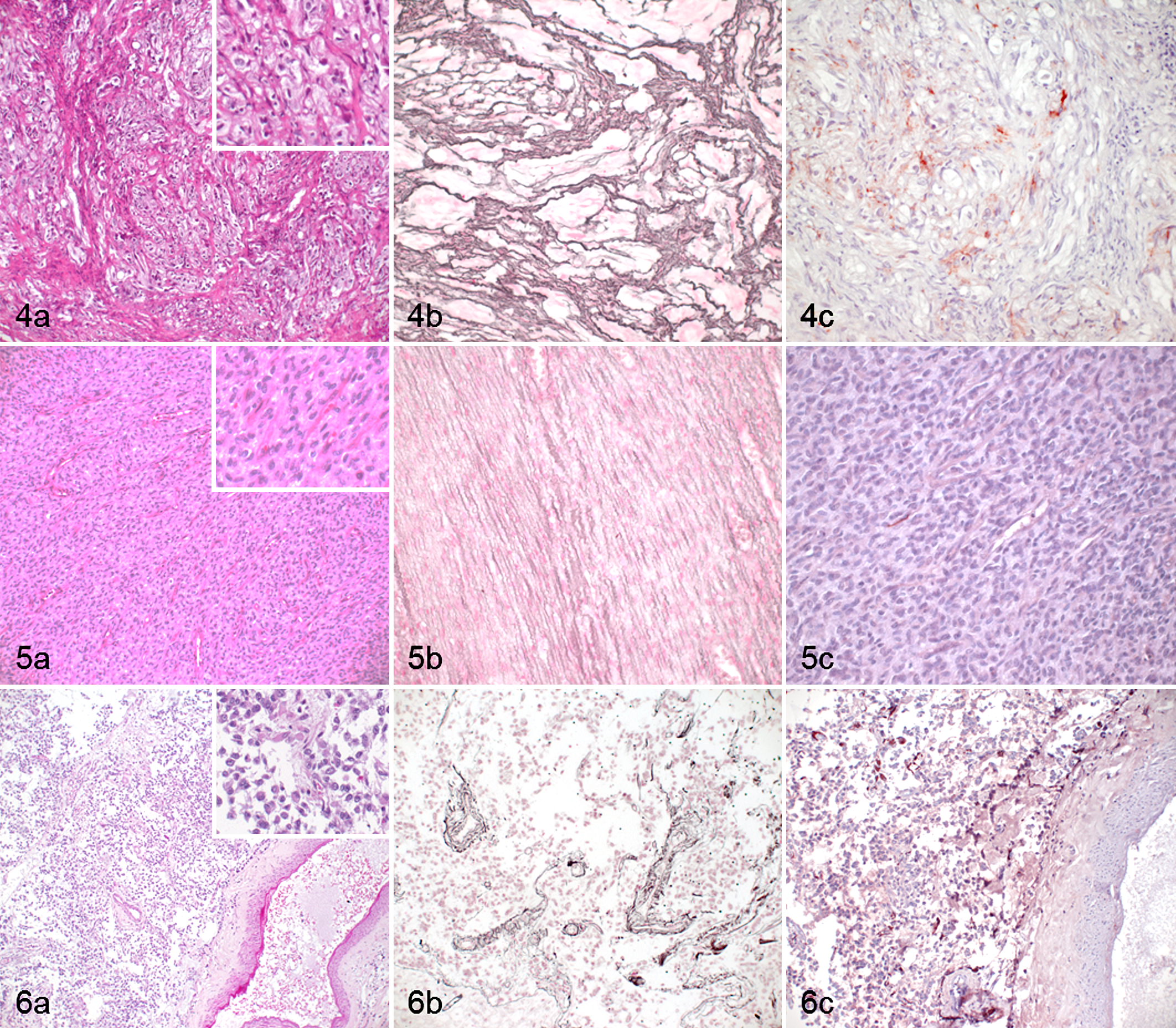
Examples of cases with a low agreement among surgical pathologists and the neuropathologist.
Overall, the panel of special stains increased interobserver agreement among surgical pathologists only from 63% to 74%. This low increase might be due to the lack of specificity of the antibody panel applied, as antibodies were selected based on their general availability in routine diagnostic laboratories and not based on their specificity for nervous system tumors. Thus, the interobserver agreement could be improved when applying more specific antibodies (eg, Olig-2, Kir7.1) (Fig. 6). In summary, we recognize the value of routinely available special stains for the diagnosis of nervous system tumors, such as PNSTs. In most tumors, however, morphological features in HE sections appear to be sufficient for correct diagnosis.
Tumor subtype agreement was lower than the tumor type agreement, and the neuropathologist agreed with the surgical pathologists on tumor subtype only in 50% of the cases. Disagreement was common with regard to meningioma subtypes, as also reported in human pathology studies on meningiomas, indicating that diagnosis of the tumor subtype is more challenging. 15 Grade I meningiomas are subdivided into different histopathological subtypes depending on the predominant pattern, and different histological patterns may be present in the same tumor. 6 Disagreement is therefore likely due to the subjective identification of the predominant histological pattern. Likewise, criteria for grade II tumors may be subjective, including nuclear atypia, macronucleoli, hypercellularity, and small cells. Due to lack of knowledge of the biological and prognostic relevance of grading in nervous system tumors and unavailability of tumor-specific therapies, the accuracy of tumor subtype/grade diagnosis currently has a low impact in veterinary medicine. However, because of the significant developments in nervous system tumor diagnosis and treatment in dogs and cats, it is expected that accurate nervous system tumor classification and grading will rapidly gain similar importance as in human medicine. In humans, tumor classification and grading are relevant and affect therapy, survival, and enrollment in clinical trials. 15,20 Therefore, larger studies investigating interobserver agreement on tumor grades that are based on prognostically relevant characteristics are highly desirable in veterinary medicine.
Another aspect investigated in the present study was the subjective confidence of pathologists in the accuracy of their first diagnosis. As expected, the neuropathologist was highly confident in a much higher proportion of cases compared to surgical pathologists (85% vs 50%–70%). However, it is interesting to note that most of the low-confidence diagnoses of surgical pathologists were then confirmed by special stains, suggesting that sometimes the frustration of surgical pathologists could simply reflect their lack of confidence rather than an actual inability to reach a correct diagnosis. Confidence is obviously subjective and is influenced by the background, personality, and experience of the pathologist. In few cases, despite a high level of confidence, a surgical pathologist changed the tumor type diagnosis after examining the special stains; in half of these cases, the neuropathologist agreed with the first diagnosis. The lack of expertise may render surgical pathologists more susceptible to rely on special stains than on tumor morphology in HE-stained sections. However, immunohistochemistry might be misleading. For instance, the antigen-expressing cells may not be homogeneously distributed in the tumor and therefore may be absent in a biopsy. In addition, labeling intensity may decrease with increasing malignancy of the tumor, 1,18 and even well-differentiated tumors may not express a marker, as it is reported for AE1/AE3 in meningiomas 1,8,11 and for GFAP in choroid plexus tumors. 12 Thus, despite the potential advantage of special stains, morphology remains fundamental for nervous system tumor diagnosis.
In conclusion, despite a variable level of confidence, surgical pathologists are able to achieve an accurate diagnosis in a large number of cases. Therefore, neuropathology should not be seen as a mysterious discipline as many pathologists think when dealing with neoplastic diseases. For the few complicated cases, a second opinion could be easily asked. Special stains available in most veterinary laboratories only slightly increase the diagnostic agreement, but they represent a good tool to support the diagnosis of malignant PNSTs and increase the level of confidence. Interobserver agreement may be further increased by the use of more specific immunohistochemical stains such as Olig-2 or Kir7.1. To our knowledge, this is the first study on interobserver agreement on nervous system tumor diagnosis in dogs and cats. More studies involving a larger number of tumors and enrolling a larger number of surgical pathologists and neuropathologists are needed to confirm our results and to investigate interobserver agreement on tumor subtypes/grades.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818824952 - Inter- and Intraobserver Agreement of Canine and Feline Nervous System Tumors
Supplemental Material, DS1_VET_10.1177_0300985818824952 for Inter- and Intraobserver Agreement of Canine and Feline Nervous System Tumors by Sara Belluco, Giancarlo Avallone, Stefano Di Palma, Roberta Rasotto and Anna Oevermann in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
