Abstract
A 7-year-old male castrated Yorkshire Terrier dog developed slowly progressive neurologic disturbances consisting of difficulties in moving the neck, lack of proprioception, and tetraparesis 4 months prior its death. Neurologic examination, computer tomography, and myelography resulted in the tentative diagnosis of intramedullary cervicothoracic spinal cord lesion. At necropsy, an intramedullary cervical spinal cord mass between C5 and C6 was noticed. Histologically, cells of this well-demarcated, nonencapsulated neoplasm were arranged in sheaths or cords separated by a fine fibrovascular stroma. The polygonal to round tumor cells were characterized by moderate pale, basophilic, and vacuolar cytoplasm and round to slightly oval, centrally located nuclei with fine-stippled heterochromatin, a single nucleolus, and a very low mitotic activity. Tumor cells lacked glial fibrillary acidic protein, vimentin, factor VIII-related, and cytokeratin antigen expression. Histologic and immunohistochemical findings led to the diagnosis of a cervical spinal cord oligodendroglioma.
Intramedullary spinal cord tumors originating from neuro-ectodermal cells have been described only rarely. 1,2,4,5,9 Spinal cord tumors in dogs have been classified according to their location into three categories: extradural, intradural-extramedullary, and intramedullary tumors. 1,3,6 Most canine spinal cord tumors are found extradurally, whereas primary intradural tumors are less common. 1,3,9 In a recent study of 33 spinal cord tumors in dogs it was reported that five cases (15%) were classified as intramedullary tumors, such as two nephroblastomas, a glioma, an astrocytoma, and a metastasis of a carcinoma. 1,2 Intramedullary neuroectodermal tumors include astrocytomas, ependymomas, and oligodendroglioma. 4,5 Clinical signs are mostly due to the space-occupying effect of the tumor. 4
This report describes the histologic, immunohistochemical, and clinical findings of a spinal cord mass of a 7-year-old male castrated Yorkshire Terrier. The animal had a 4-month history of increasing inability to move the neck. The dog's condition deteriorated despite supportive treatment, and the animal showed circling and paresis of the forelimbs. The dog was submitted for a diagnostic work-up to the Clinic for Small Animals, School of Veterinary Medicine in Hanover, Germany. Neurologically, the animal displayed mildly to moderately reduced posture reactions, neurologic deficits of both forelimbs and hindlimbs, and hyperesthesia between C1 and C7. Computer tomography revealed dilatation of the lateral ventricles. The disks and bones of the spinal column were without significant changes. The cerebrospinal fluid was clear, and the total leukocyte and red blood cell numbers were without significant changes. Myelography showed a blockage before C5. Because of rapid progression of the clinical signs and the poor prognosis, the animal was euthanatized and submitted for necropsy.
Gross pathology revealed mild dilatation of the lateral ventricles and a mild swelling of the cervical spinal cord between C3 and C6. No significant macroscopic findings were observed in other organs. Tissue samples were fixed in 10% buffered formalin, routinely processed, and embedded in paraffin. Four-micron-thick paraffin sections of various organs including the spinal cord were cut and stained with hematoxylin and eosin and selected slides for luxol fast blue to detect myelin sheaths. For immunohistochemistry antibodies specific for glial fibrillary acidic protein (GFAP: 1 : 800), vimentin (1 : 15), cytokeratin (1 : 100), factor VIII–related antigen (1 : 200), and the avidin–biotin–peroxidase complex method with 3,3-diaminobenzidine-tetra-hydrochloride-dihydrate were used. 1
After formalin fixation a round to oval, 0.3- to 0.8-cm-diameter, intramedullary mass, which compressed the central canal was noticed on cross sections of the cervical spinal cord between C5 and C6 (Fig. 1). This well-circumscribed mass was surrounded by compressed spinal cord tissue and exhibited focal areas of hemorrhages.
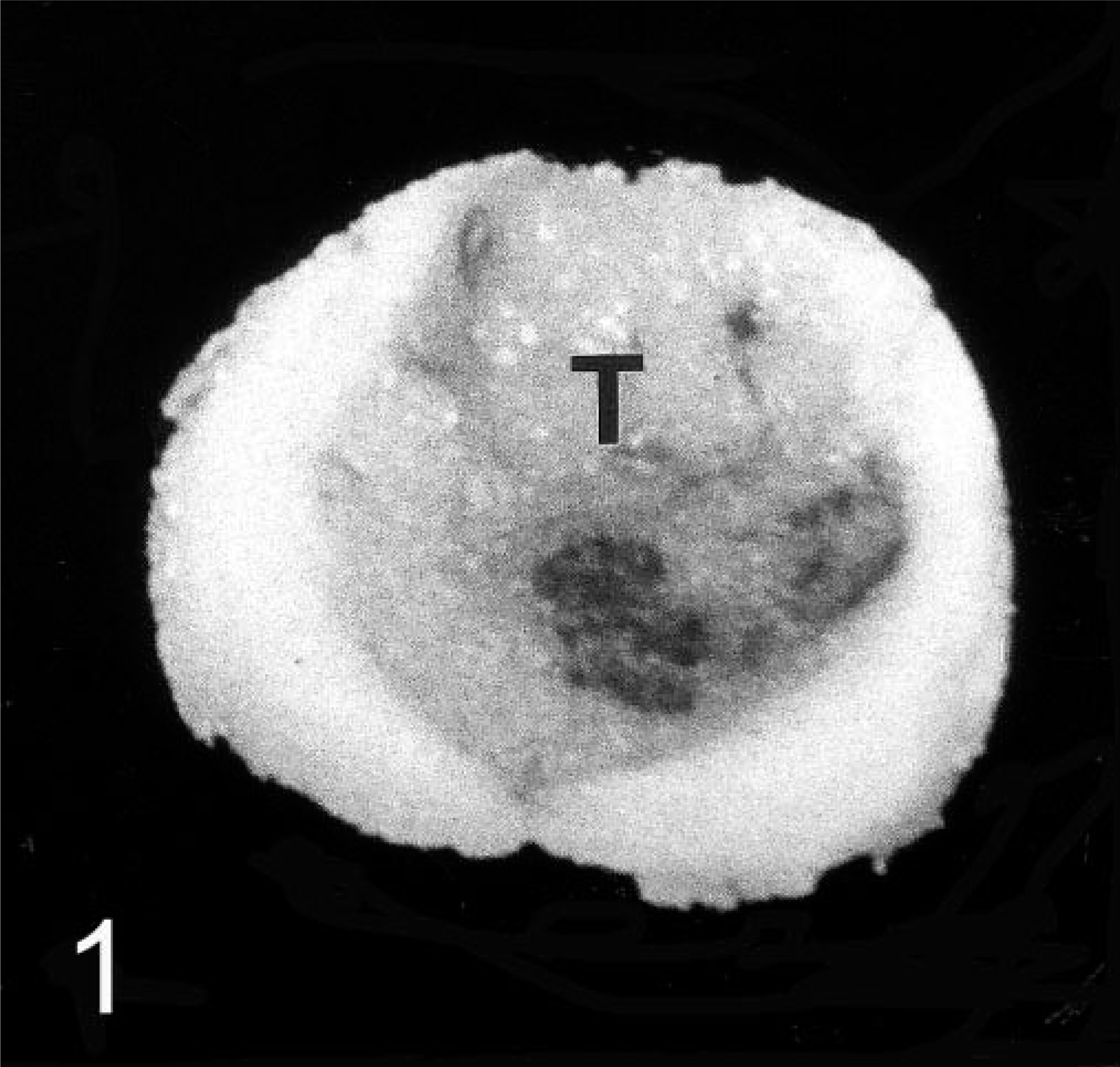
Cervical spinal cord, canine oligodendroglioma. Well-demarcated intramedullary tumor (T) and compression atrophy of the adjacent cervical spinal cord tissue between C5 and C6.
Histologically, this cell-rich, well-demarcated, nonencapsulated tumor occupied about two thirds of the spinal cord and compressed the central canal. The tumor consisted of a homogeneous cell population arranged in sheaths or cords with a fine vascular stroma, areas of multifocal necrosis, hemorrhages, and glomerular-like tufts (Fig. 2). The round tumor cells were characterized by a moderate amount of a pale basophilic cytoplasm, a round to slightly oval nucleus with fine-stippled heterochromatin, and an inconspicuous single nucleolus (Fig. 3). No mitotic figures were observed. Gitter cells and multifocal dystrophic calcifications were frequently observed within areas of necrosis and hemorrhages. Randomly distributed gemistocytes within the tumor and at the periphery of the areas of necrosis were found frequently (Fig. 4). Microcystic areas with accumulation of a pale basophilic mucinous-like material were seen within the neoplasm. At the periphery of the tumor, narrow bands of compressed gray and white matter were observed. Spinal cord segments cranial from the neoplasm displayed mild periventricular malacia, congestion, multifocal hemorrhages, and distortion of the ventral median fissure. No significant histologic findings were detected in the remaining spinal cord, brain parenchyma, and other organs.
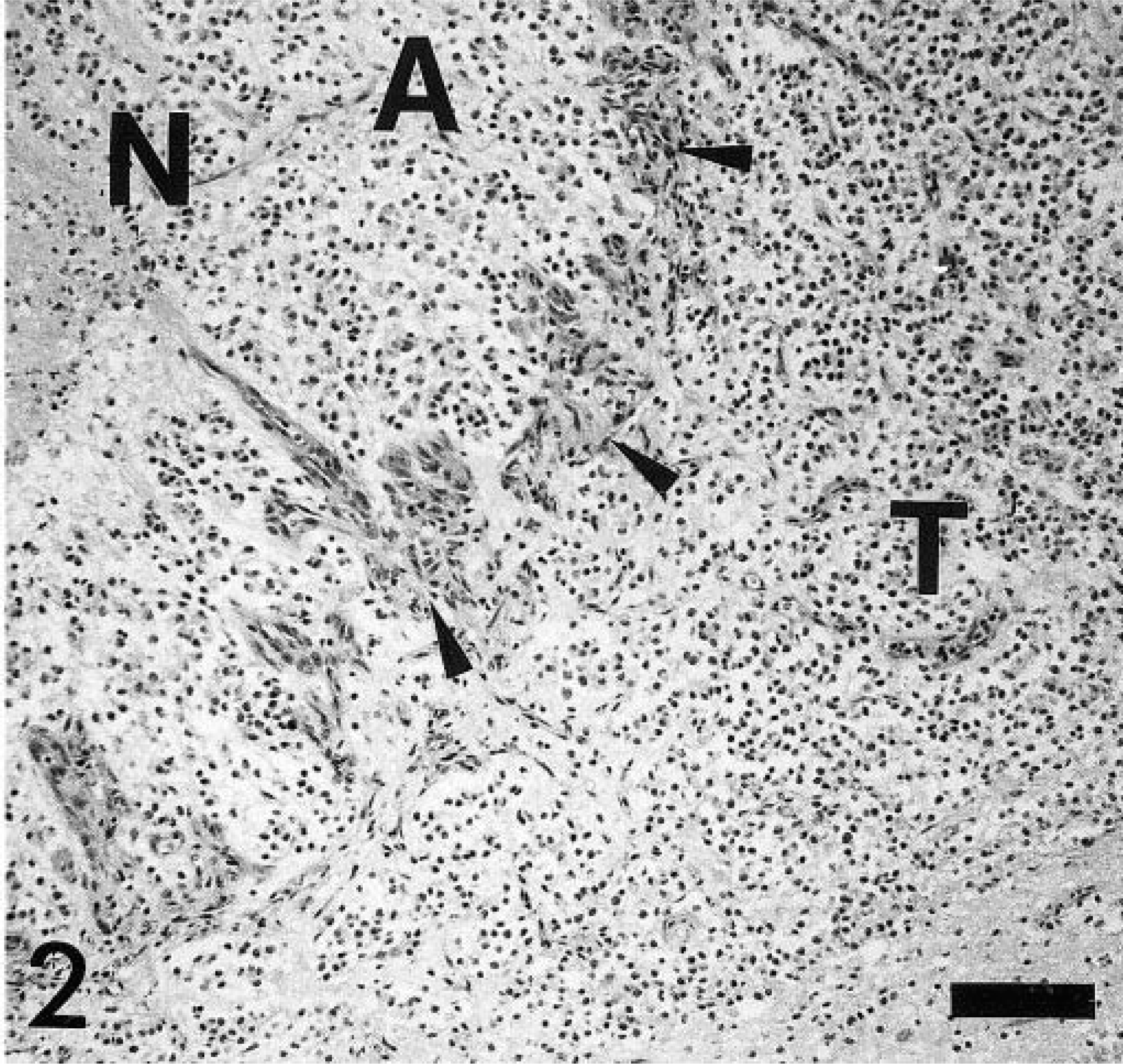
Spinal cord, canine oligodendroglioma. Tumor cell proliferation and glomerular-like vascular proliferation at the periphery and reactive astrocytic response and necrosis. N = necrosis, A = reactive astrocytic response (gemistocytes), T = tumor cells, arrow heads = characteristic glomerular-like tufts. HE staining. Bar = 110 µm.
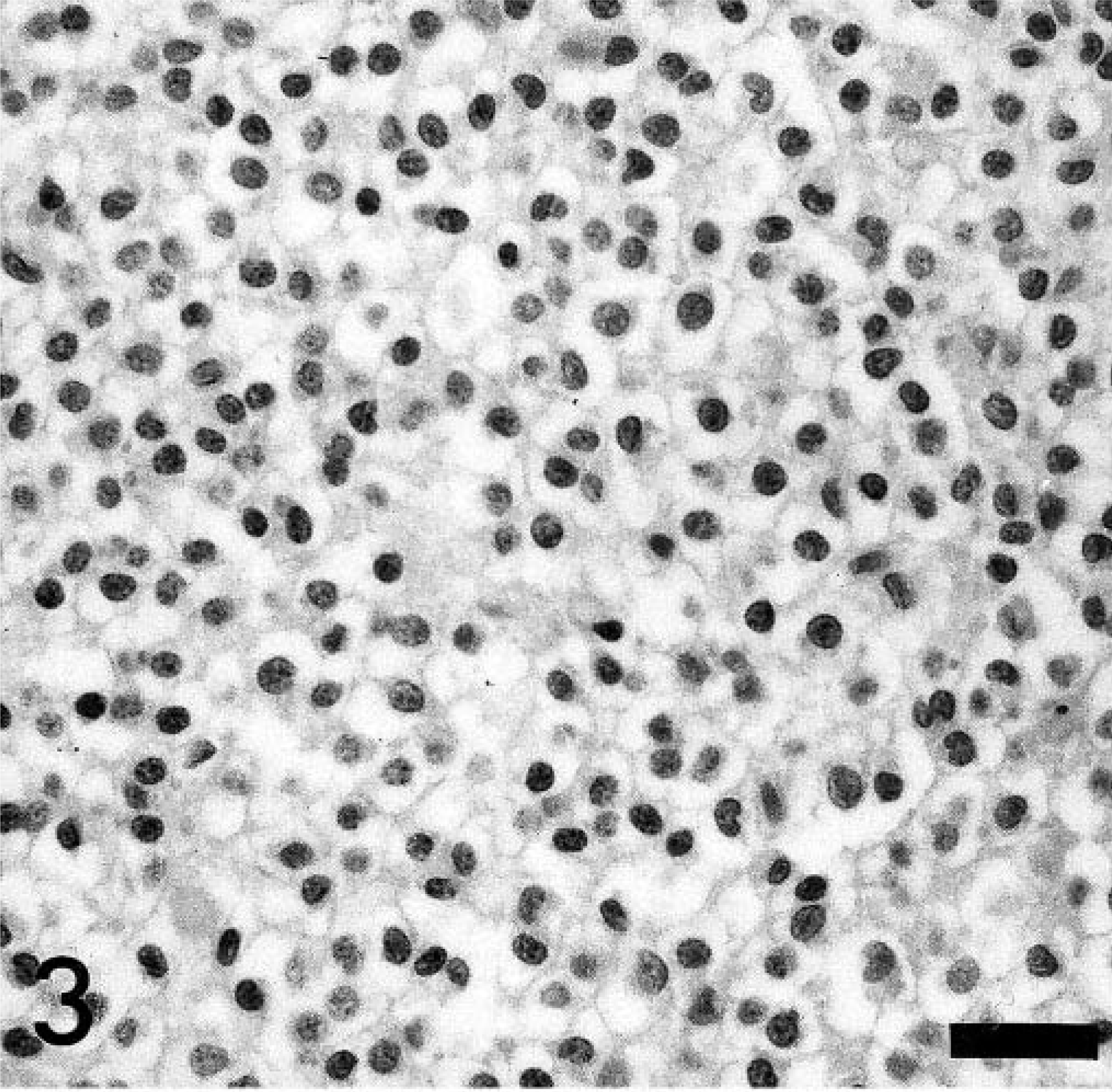
Spinal cord, canine oligodendroglioma. Characteristic “honeycomb” cell pattern of an oligodendroglioma. HE stain, Bar = 30 µm.
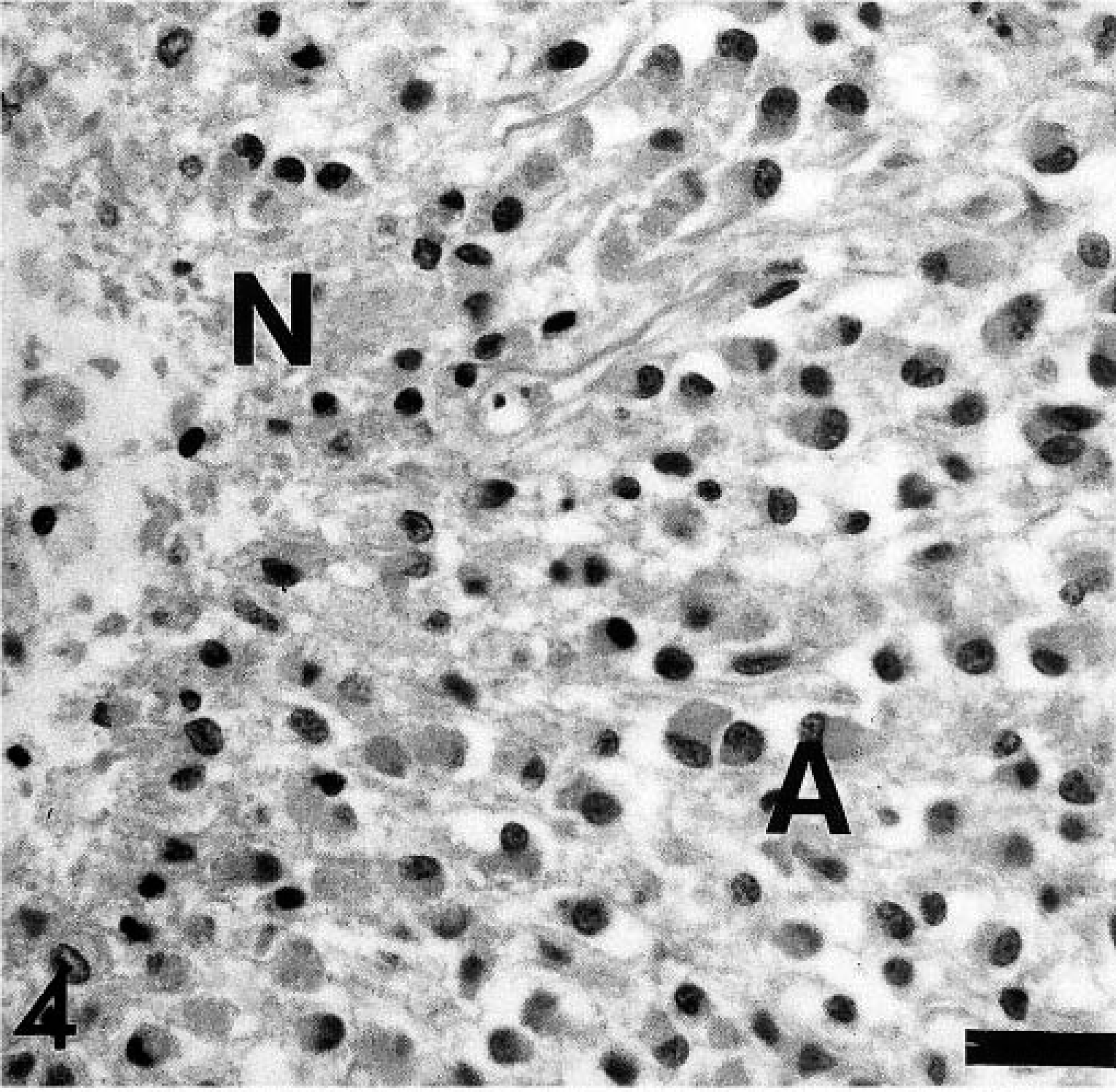
Spinal cord, canine oligodendroglioma. Area of gemistocytic astroglial differentiation and necrosis. A = reactive astrocytic response (gemistocytes), N = necrotic area. HE stain. Bar = 30 µm.
Tumor cells were negative for GFAP, vimentin, cytokeratin, and factor VIII–related antigen. GFAP- and factor VIII–related antigen immunoreactions were found in reactive astrocytes, including gemistocytes and endothelial cells of the glomerular-like tufts, respectively (Figs. 5, 6). The few GFAP-positive cells within the tumor mass with the morphology of gemistocytes were interpreted as secondary astrocytic responses due to tumor-induced tissue damage. Based on the histologic and immunohistochemical findings the neoplasm was diagnosed as an oligodendroglioma.
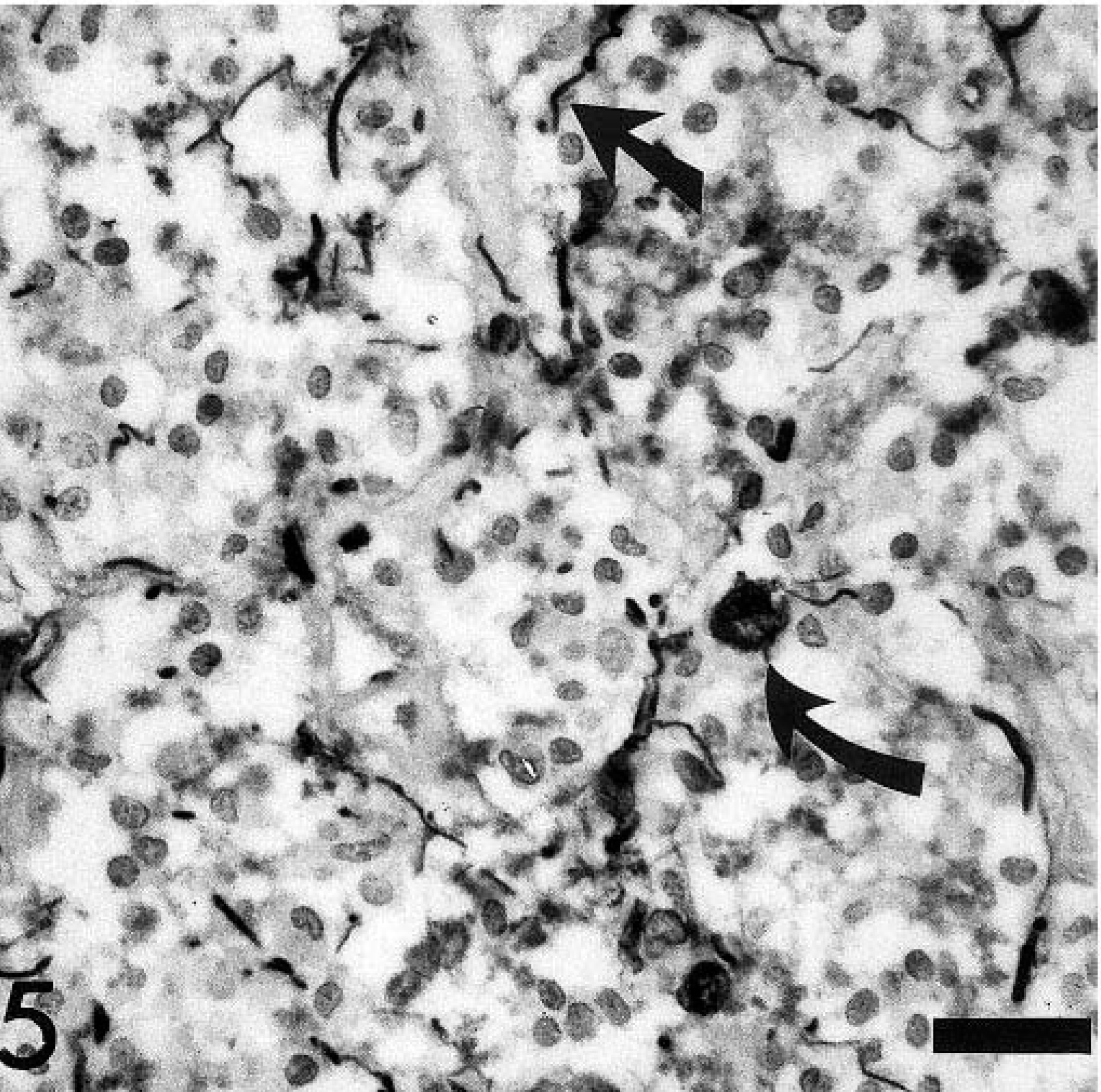
Spinal cord, canine oligodendroglioma. Lack of GFAP immunoreactivity of tumor cells. Note few GFAP-positive cells within the tumor interpreted as reactive astrocytes (arrows). GFAP-specific polyclonal antibody, ABC method, slightly counterstained with hematoxylin. Bar = 30 µm.
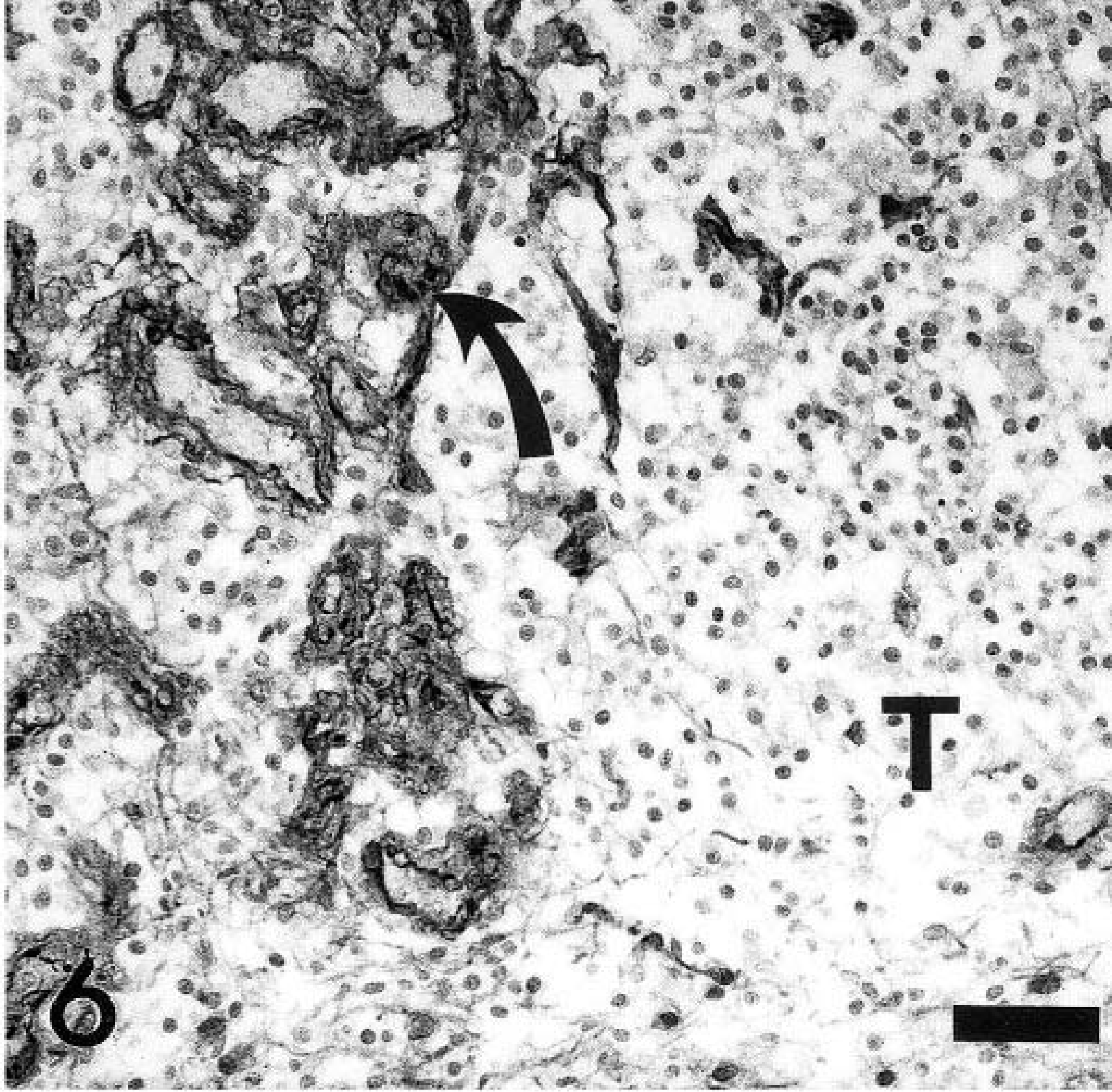
Spinal cord, canine oligodendroglioma. Factor VIII–related antigen expression of glomerular-like tufts (arrows). Note lack of immunoreactivity of tumor cells. T = tumor cells. Factor VII–related antigen specific antibody, ABC method, slightly counterstained with hematoxylin. Bar = 30 µm.
Common sites of canine oligodendrogliomas in dogs are the white and gray matter of the cerebral hemispheres and rarely brain stem and spinal cord. 4 This neuroectodermal tumor has been frequently reported in brachycephalic breeds such as Boxers, Bulldogs, and Boston Terriers. 4,7 According to our literature search, a spinal cord oligodendroglioma has been reported so far only in a 5-year-old Dachshund dog. In the latter, histology revealed a mucinous variant of this type of tumor in the spinal cord. 8 Oligodendrogliomas, usually well demarcated, commonly extended into ventricular spaces and occur in dogs older than 5 years and are more often found in males than in females. 4,7 Similarly, in this case a subependymal origin of the tumor can be assumed, and the tumor also extended into or obliterated the spinal canal.
A benign and malignant (anaplastic) oligodendroglioma can be distinguished in domestic animals according to the new World Health Organization classification of tumors of the nervous system. 5 In this case, tumor cells were well differentiated, and the tumor was classified as a benign oligodendroglioma.
Dystrophic calcifications, hemorrhages, and necrosis have been reported for most canine oligodendrogliomas. 4 Micro-vascular proliferation is an other characteristic feature of this tumor. Similarly, prominent neovascularization has been described for high-grade astrocytomas. 5,7 Renin and platelet-derived growth factor produced by neoplastic astrocytes and vascular endothelium have been implicated as mediators that might trigger vasculogenesis. 7 Multifocal microcystic areas and accumulation of mucinous-like material as observed in this case are also frequent findings in oligodendrogliomas. 4,7,8
To summarize, neurologic disturbances and severe ataxia correlated well with the anatomic location of this rare spinal cord oligodendroglioma.
Footnotes
Acknowledgements
We would like to thank Mrs. B. Behrens, P. Grünig, and K. Rohn for excellent technical and photographic support.
