Abstract
Central neurocytoma is a rare, prognostically favorable neuronal tumor of the human central nervous system, typically located intraventricularly near the foramen of Monro. Extraventricular cerebral neurocytomas and spinal tumors have also been reported. To date, however, neurocytomas have not been documented in domestic animal species. In this report, we describe a spinal cord tumor in a dog. The microscopic examination revealed tumor cells forming loosely packed clusters in some areas and palisades in other areas. In addition, they showed fine fibrillary neuropil-like areas of different sizes, sometimes resembling the “rosettes” of pineocytomas, as well as ependymoma-like perivascular pseudorosettes. The tumor cells had scant eosinophilic cytoplasm, with perinuclear halos, closely resembling the appearance of oligodendroglioma. Immunohistochemical staining showed expression of synaptophysin and neuron-specific enolase by tumor cells and pronounced in fibrillary areas. On the basis of histomorphology and immunohistochemical reactivity, the present tumor was diagnosed as extraventricular neurocytoma.
Central neurocytoma is an uncommon tumor of the central nervous system in humans, first reported by Hassoun et al. 6 Central neurocytoma was included in the World Health Organization (WHO) tumor classification in 1993. 7 Corresponding to the actual WHO tumor classification, the central neurocytoma is a benign neoplasm that affects mostly young adults, and it is assigned to tumors with neuronal differentiation. It is typically located in the lateral ventricles near the foramen of Monro. Many cases of the “ependymomas of the foramen of Monro” in older literature 16 actually represent central neurocytoma. 14 Because of their histopathologic similarity to oligodendrogliomas, historically, these tumors have also been classified as intraventricular oligodendrogliomas. 10 Neurocytomas are well-differentiated tumors, with a benign histologic appearance that displays various architectural patterns, even in the same specimen. The patterns most commonly seen are the honeycomb arrangement of oligodendroglioma and large fibrillary areas that resemble the irregular rosettes of pineocytoma. 2 In other segments, cells may be arranged in palisades or as perivascular rosettes that mimic an ependymoma. Small foci of calcifications are described in half of the cases, usually distributed throughout the tumor. 4,10 On immunohistochemical examination, strong synaptophysin expression, especially in fibrillary zones and perivascular cell-free cuffs, was recognized as the most reliable marker. 3 Chromogranin A and neurofilament (NF) are usually absent. The proliferative index of neurocytomas is usually low. 4,10 The electron microscopic appearance 4,6 and the expression of neuronal markers by tumor cells favor the neuronal origin of central neurocytoma. In contrast, some investigators demonstrated co-expression of glial fibrillary acidic protein (GFAP) and synaptophysin, especially in extraventricular neurocytoma. 10 In addition, it has been demonstrated that cultured human neurocytoma cells show characteristics of astroglial differentiation. 15 Hence, it was hypothesized that neurocytoma originate from bipotential precursor cells, which can exhibit both glial and neuronal differentiation. 13,14
Besides the neurocytoma in its classical location, a series of extraventricular neoplasms with the histopathologic and immunohistochemical properties of the central neurocytoma have been reported. 1,5,12,13 So far, however, neurocytomas have not been documented in domestic animal species. In this report, we present a spinal-cord tumor in a dog with the histopathologic and immunohistochemical characteristics of a central neurocytoma.
A 7-year-old female spayed mixed-breed dog was introduced to the Small Animal Clinic of the Justus-Liebig-University, Giessen, Germany, with a 3-month history of ataxia in the hind limbs. Upon presentation, the ataxia progressed to moderate paresis. Based on the neurologic examination, a right-sided lesion in the lumbosacral intumescence (L4-S2) was suspected. Magnetic resonance imaging was performed with the dog under general anesthesia. T1-weighted images were obtained in transverse, sagittal, and axial planes with a spin echo pulse sequence before and after contrast administration. Sagittal and transverse T2-weighted images showed an intramedullary hyperintense signal that extended from the third to the fourth lumbar segment. Normal gray and white matter structure could not be discerned. The spinal cord was swollen, and the central canal was obliterated. Cranial to this area, a moderate dilation of the central canal up to TH5 was found. After paramagnetic contrast infusion, the lumbar lesion showed irregular contrast enhancement, and a presumptive diagnosis of an intramedullary tumor was made. The dog was euthanized because of poor prognosis, and a complete postmortem was performed.
Postmortem examination revealed an intramedullary neoplastic mass between the last lumbar segment and the sacral bone. The mass was 2 × 1 × 1 cm in size, grayish pink, moderately demarcated, and adhered to the meninges (Figs. 1, 2). Except for an acute alveolar edema of the lung and a moderate pulmonary congestion, no other gross lesions were observed, specifically no other extraspinal tumors were found. The tumor was fixed in 10% formalin and embedded in paraffin for histopathologic processing and immunohistochemical study. Histopathologic sections were prepared and stained with hematoxylin and eosin and Gomori's silver stain. Immunohistochemical staining was performed by using primary antisera against synaptophysin (clone SY 38, DakoCytomation, Hamburg, Germany), chromogranin A (polyclonal, DakoCytomation), neuron-specific enolase (NSE) (clone NSE-1G4, Zymed, Wien, Austria), NF (clone 2F11, DakoCytomation), GFAP (polyclonal, DakoCytomation), S-100 protein (polyclonal, DakoCytomation), and vimentin (clone V9, DakoCytomation). For proliferative index analysis of tumor-cell nuclei, sections were stained for Ki-67 antigen (clone MIB-1, DakoCytomation). Primary antibodies were visualized with the peroxidase anti-peroxidase method (chromogranin A, GFAP, S100) or with the avidin-biotin complex method (synaptophysin, NSE, NF, vimentin, Ki-67 antigen). The positive immunohistochemical controls consisted of tissues that originally expressed these markers. Consecutive sections were used as negative controls where a nonreacting mouse monoclonal antibody against chicken lymphocytes (monoclonal antibodies) and normal rabbit serum (polyclonal antibodies) replaced primary antibodies.
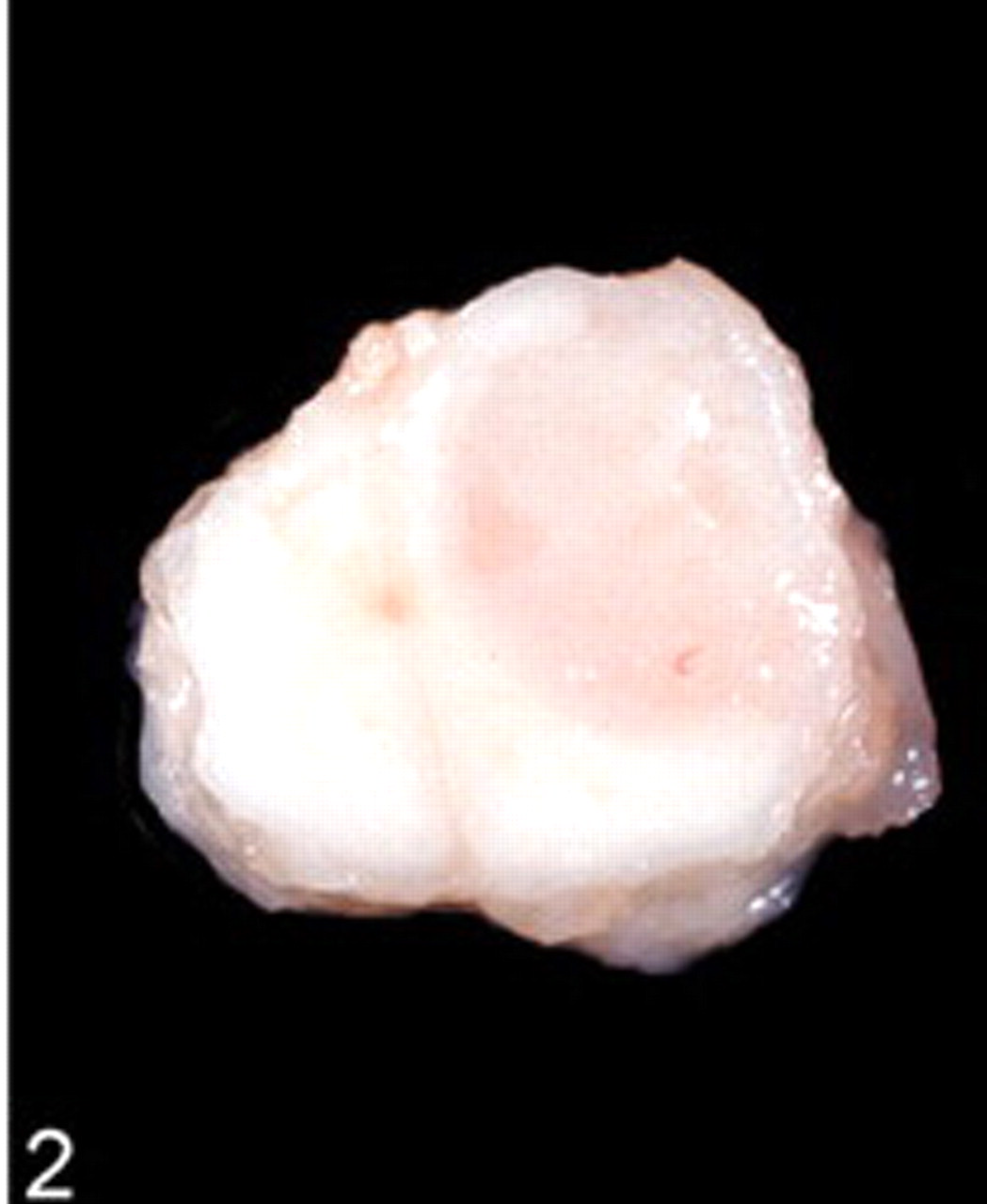
Spinal cord; dog. Transverse sections show the predominant intramedullary location of the tumor.
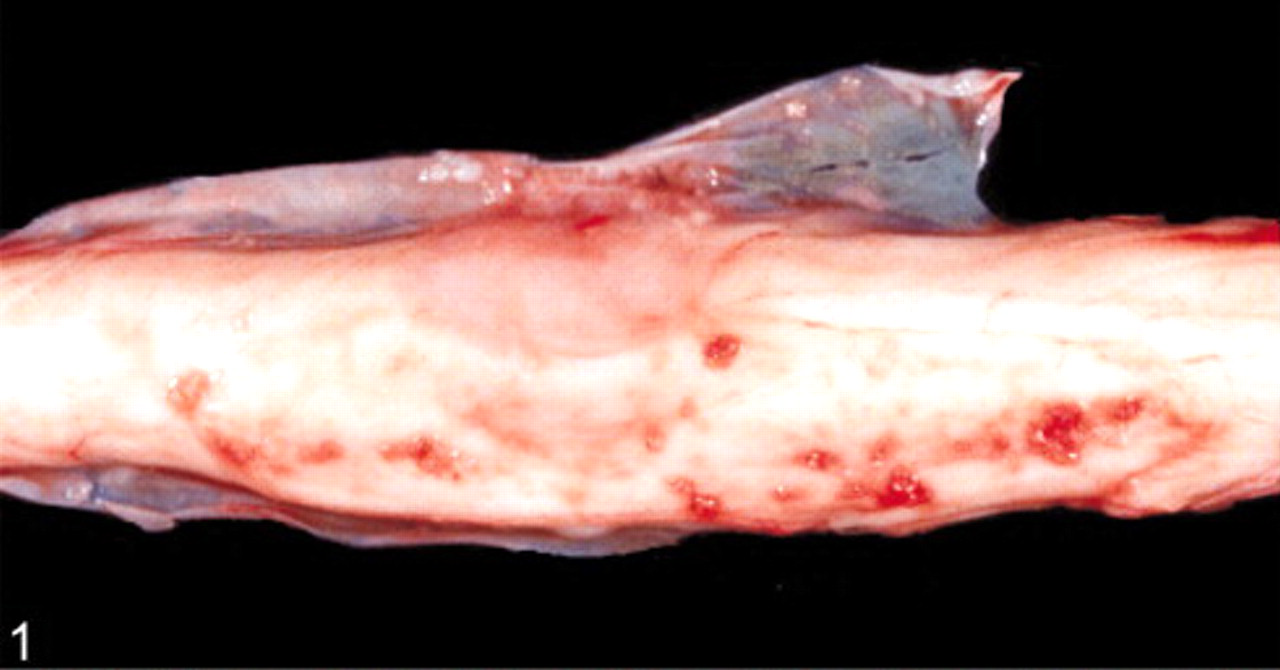
Spinal cord; dog. Gross morphology. Note the grayish pink nodular mass measuring 2 × 1 × 1 cm and adhering to the meninges.
Microscopic examination revealed a highly cellular nodular and expansive mass within the white matter that occupied nearly the whole right side of the spinal cord and displaced the right ventral gray horn. The mass was unencapsulated, well demarcated, and showed mild infiltration of the adjacent tissue at the ventral margin. The tumor cells formed loosely packed clusters in some areas and palisades in others. They lay in a fine, fibrillary neuropil-like matrix with focal acellular areas of different size (Fig. 3). Vascularity was represented by long, thin-walled, capillary-sized vessels surrounded by a cell-free fibrillary network (Fig. 4). Scattered throughout the entire tumor, we found small foci of calcification. The tumor cells were small-to-medium sized and uniform in size and shape, had indistinct cell borders, and scant eosinophilic cytoplasm with perinuclear halos (Fig. 3). The nuclei were round to oval, centrally located, and showed a finely stippled chromatin and inconspicuous nucleoli. The mitotic activity was lower than one mitosis per high power field.
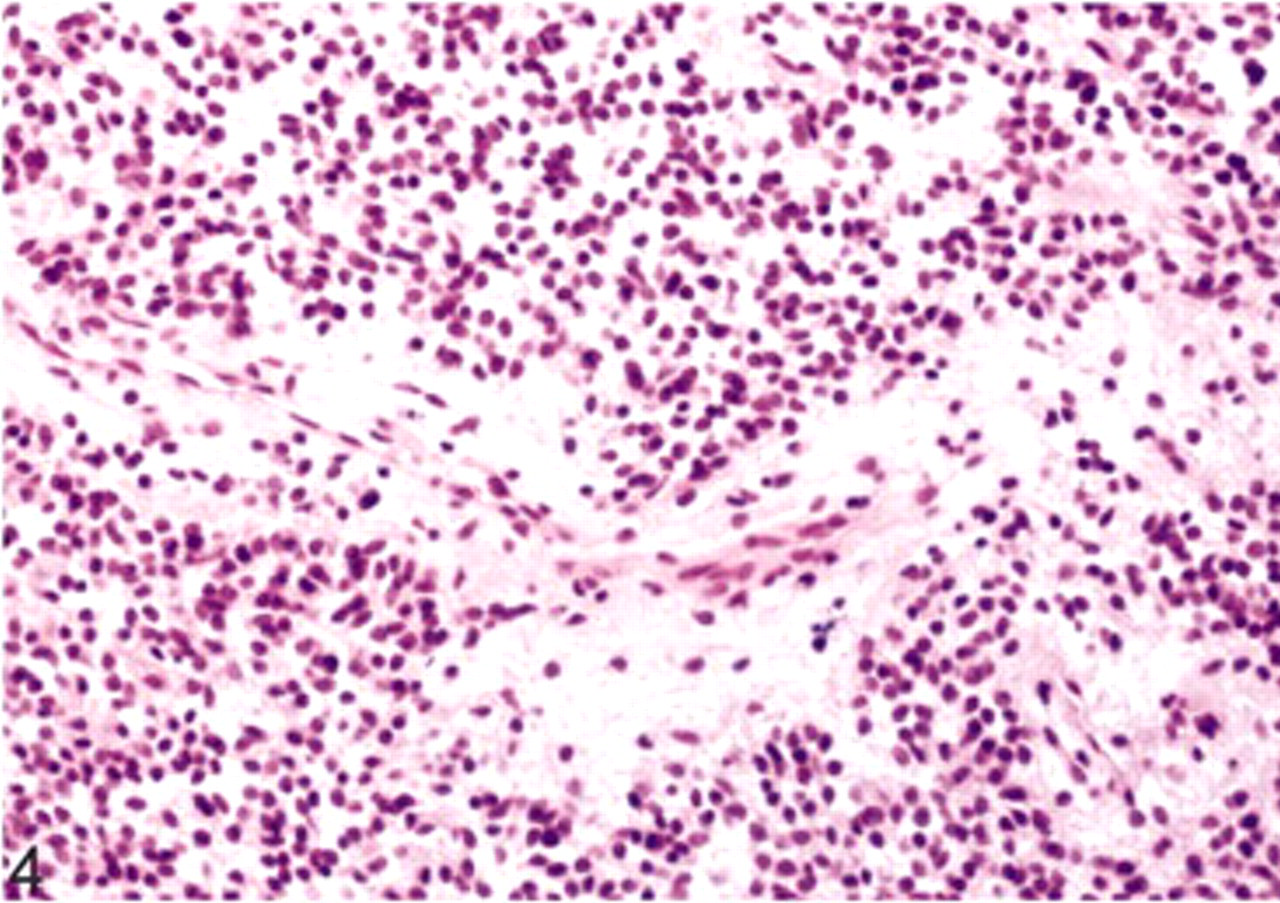
Extraventricular neurocytoma; dog. Perivascular cell-free cuffs and neuropil islands. HE.
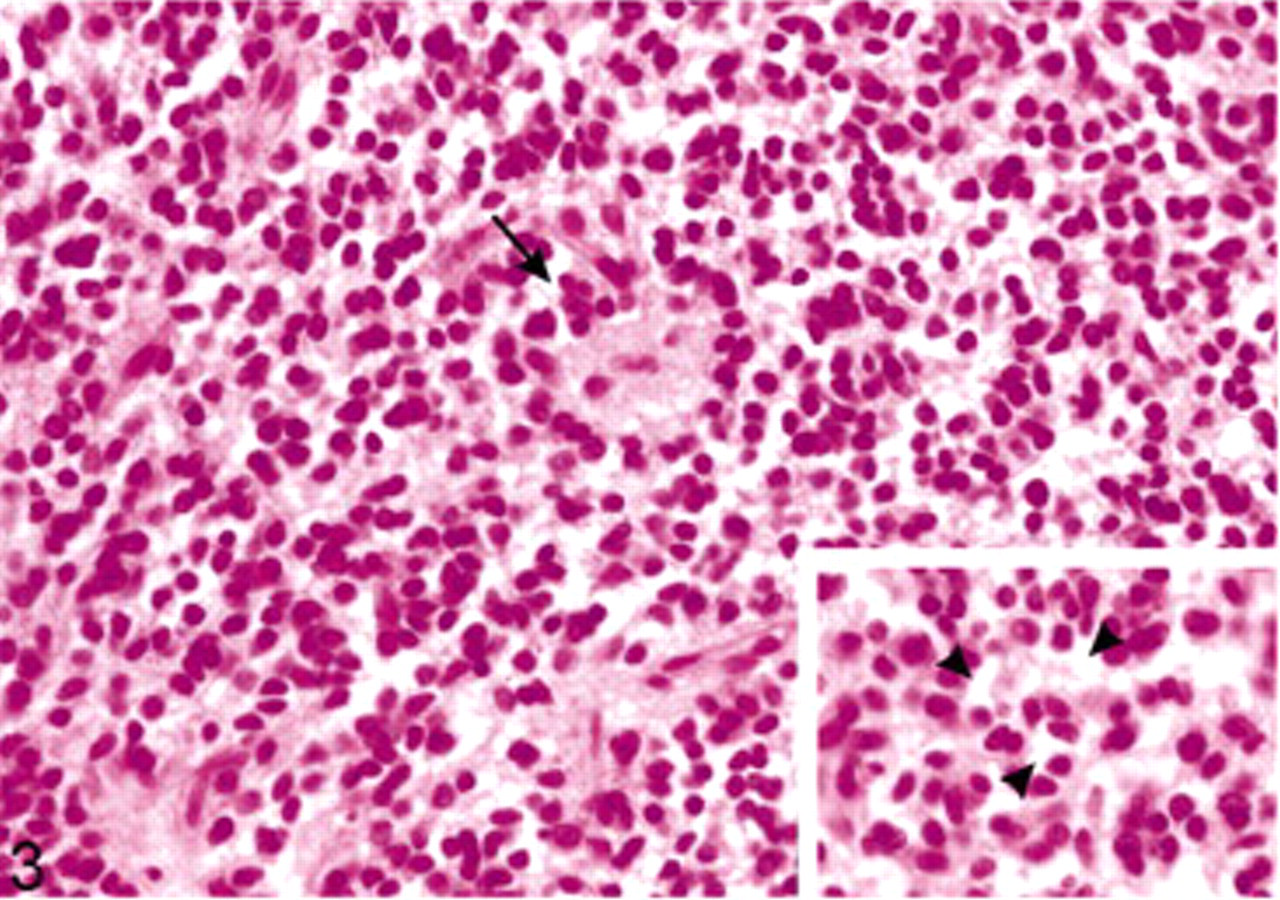
Extraventricular neurocytoma; dog. Tumor cells arranged around large fibrillary areas, resembling irregular rosettes of pineocytoma (arrow), and perinuclear cytoplasmic vacuolations (
On immunohistochemical examination, many tumor cells showed diffuse cytoplasmic positivity for synaptophysin. Especially, the fibrillary zones and perivascular cell-free cuffs showed strong expression (Fig. 5). Staining with antibodies against NSE likewise revealed a positive result in most of the tumor cells (Fig. 6), whereas GFAP could only be observed in cells with long cytoplasmic processes. Because of their morphology, the GFAP-positive cells were rather interpreted as reactive astrocytes than as neoplastic cells (Fig. 7). Detection of chromogranin A, NF, and S-100 protein revealed no positive signal. Vimentin expression could only be seen on capillaries. The Ki-67 labeling index was about 5%.
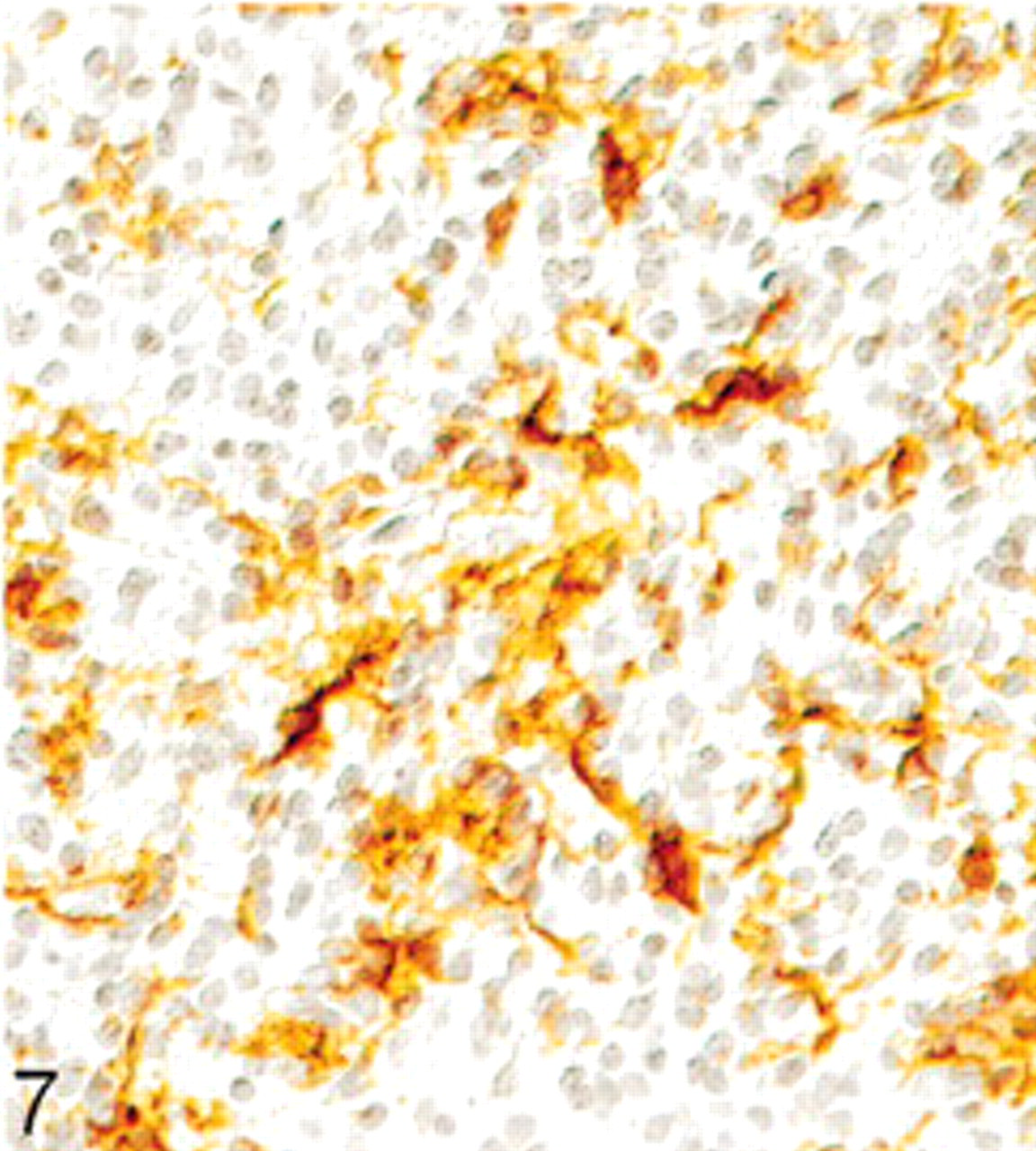
Extraventricular neurocytoma; dog. Positive immunohistochemical labeling for glial fibrillary acidic protein in reactive astrocytes. Peroxidase anti-peroxidase method. Papanicolaou's hematoxylin counterstain.
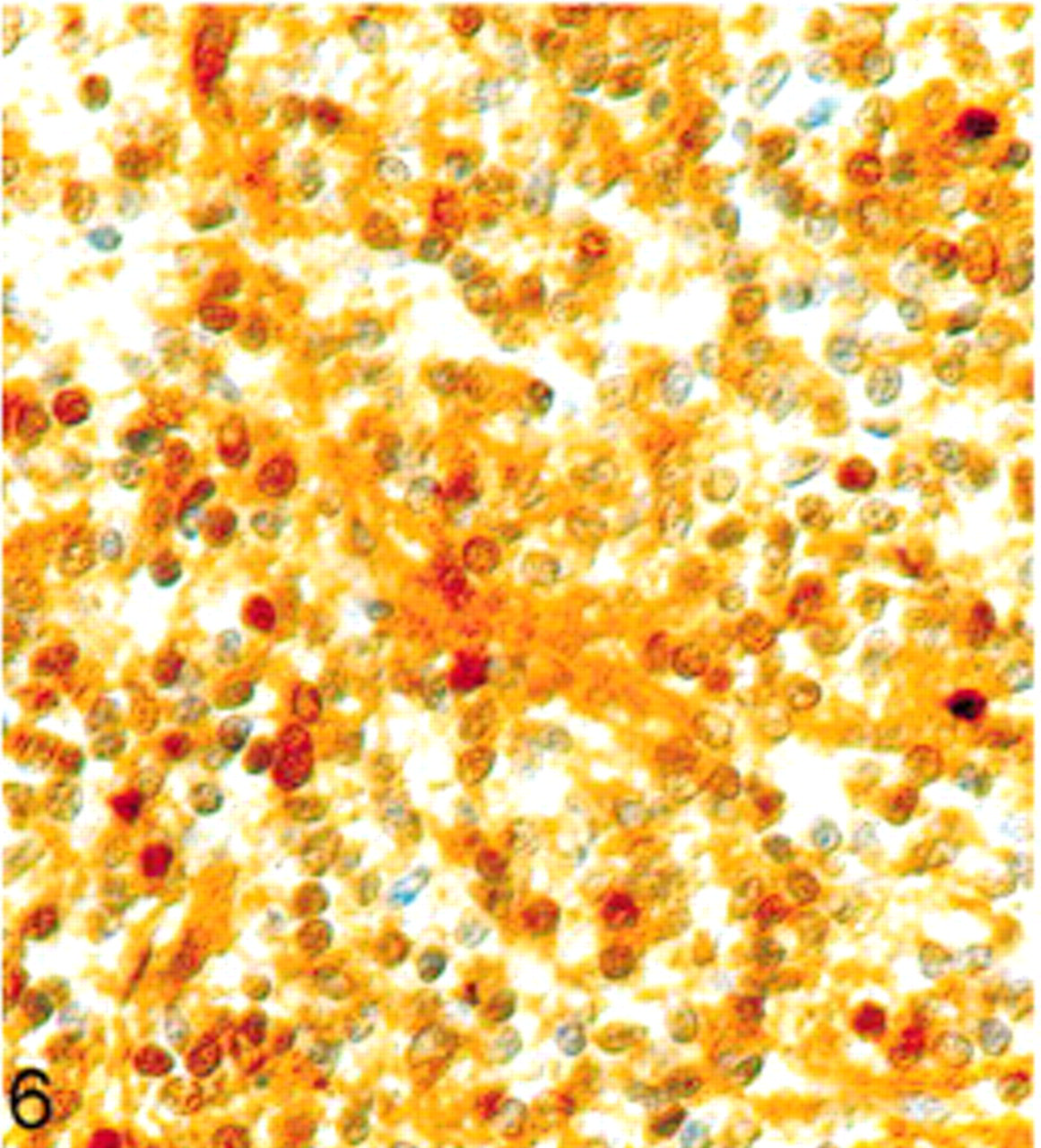
Extraventricular neurocytoma; dog. Positive immunohistochemical labeling for NSE. Avidin-biotin complex method. Papanicolaou's hematoxylin counterstain.
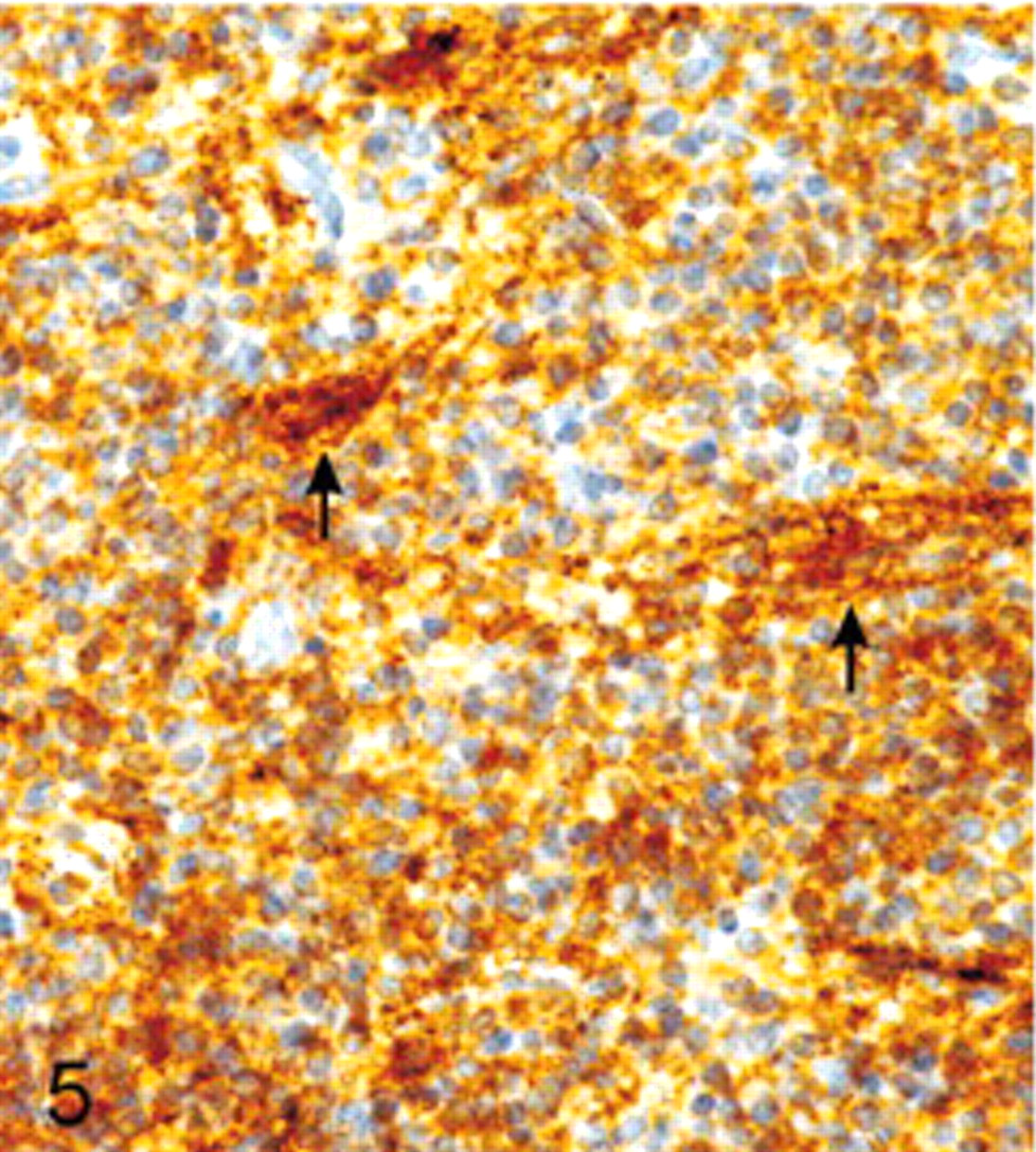
Extraventricular neurocytoma; dog. Positive immunohistochemical labeling for synaptophysin, especially the fibrillary zones and the perivascular cell-free cuffs show strong reactivity (arrow). Avidin-biotin complex method. Papanicolaou's hematoxylin counterstain.
The main differential diagnoses included oligodendroglioma, ependymoma, and pineocytoma. 4 Because of the expression of synaptophysin, with the strongest signal in neuropil islands and in perivascular cell-free fibrillary cuffs (together with similar findings in NSE immunohistochemistry), this neoplasm could clearly be differentiated from oligodendroglioma and ependymoma. The absence of GFAP and vimentin expression on tumor cells substantiates the diagnosis. A pineocytoma was excluded because of the tumor location and the lack of chromogranin expression. A further differential diagnosis is the neuroblastoma, which could be excluded due to the low mitotic activity and Ki-67 labeling indices, the more mature neurocytic tumor cells, the lack of typical Homer-Wright rosettes, 8 the age of the dog, as well as the tumor location. Also, neuroblastomas are described to have positive staining for NF and chromogranin. 9 The immunostaining procedure for chromogranin A was entirely negative in this tumor, which strongly indicated that the lesion was not a paraganglioma. In addition, the tumor formed no lobules (“Zellballen”) 11 surrounded by a well-vascularized fibrovascular stroma. 8 The histopathologic appearance of the tumor, the lack of reticulin fibers, and the lack of vimentin, as well as of S-100 protein-expression excluded a peripheral-nerve-sheath tumor.
The histopathologic and immunohistochemical characteristics of the tumor presented here were all consistent with central neurocytoma. The macroscopic and histopathologic examination of the brain and upper spinal cord, as well as the other organs revealed no evidence of a neoplasm that could have spread to the lumbosacral part of the spinal cord, and the eligible differential diagnoses could be excluded because of the morphologic and immunohistochemical features. In conclusion, we consider that this case represents an extraventricular neurocytoma of the spinal cord in a dog.
