Abstract
Equine pancreatic disease is considered rare, and successful treatment is limited. Additionally, antemortem diagnosis of equine pancreatitis is difficult because of the lack of definitive diagnostic tests. Although a paucity of information exists on this entity in the horse, pancreatic disease has typically been shown to be secondary to other gastrointestinal, hepatic, and endocrine conditions. No predisposition based on age, sex, or breed appears to exist, but several conditions predispose a horse to pancreatitis, including grain overload, endocrine disease, and parasite migration. A retrospective search of cases within the archive of the University of Tennessee necropsy database revealed only 4 cases during a 9-year period (2005–2014). In only 1 case was pancreatic disease considered primary, and in the other 3, pathologies often seen concurrently with pancreatic lesions were identified. These included cecal rupture, colonic displacement, neoplasia (lymphocytic leukemia), and pituitary adenoma. A review of available historical data and associated laboratory results are included with the gross and histologic characteristics of pancreatic lesions, in these 4 newly reported cases.
Pancreatic disease in horses is described as pancreatitis or inflammation of the pancreas but primary necrosis due to vascular compromise is also reported in the horse. Typically, it is a secondary condition that has been associated with other gastrointestinal, hepatic, and endocrine conditions. Its diagnosis is considered rare, partly because of the greater likelihood of primary intestinal or liver disease in this species, as well as because of the difficulty in diagnosing pancreatic disease antemortem.13,14,22 The largest retrospective study characterizing pancreatitis comprised 43 horses over a 25-year period at the University of California, Davis. 22 Additionally, only 13 peer-reviewed case reports on this topic were found, further attesting to its relatively rare occurrence. 13
Unlike in dogs, where old age, reproductive status (spay or neuter), and breed are predispositions for pancreatic disease, 6 in a large retrospective study, there was no predisposition based on breed, sex, or age in affected horses. 22 Overall, pancreatic disease is more commonly reported in adult horses but has been reported less commonly in foals.5,17,19,22
As in other species, several forms of pancreatitis exist in the horse, and these are based on duration: acute, chronic, and chronic active. 22 Acute pancreatitis can be associated with acute colic in the horse and sometimes produces or is the result of duodenal reflux.5,8 Chronic pancreatitis is associated with weight loss, anorexia, lethargy, and mild recurrent signs of colic. Chronic active pancreatitis is a low-grade, “smoldering” condition. 22 Mild manifestations of any of the above forms may be clinically nonspecific and, hence, underdiagnosed.13,22
Although considerable advancement has occurred with development of accurate and sensitive diagnostic tests for pancreatic conditions in dogs and cats, similar advancement has not been seen in farm animal or equine medicine. Antemortem diagnosis of equine pancreatitis is difficult because of the lack of definitive diagnostic tests. Although the accuracy of serum and abdominal fluid amylase and lipase has not been heavily evaluated, in some instances these enzymes are used as preliminary screening tools.17,22 Additionally, trypsin is produced in low amounts in the equine pancreas and other tissues, and hence evaluation of elevated trypsin levels has gained some support in the diagnosis of pancreatitis. 22 Specifically, splanchnic hypoperfusion during shock is proposed to lead to pancreatic damage, release of trypsin into the peritoneal space, and subsequent elevated concentrations in plasma. Trypsin can activate the inflammatory cascade and can play a role in multiple organ failure. 11 In 2 Appaloosa foals, hyperlipemic serum and abdominal fluid, along with elevated serum and peritoneal fluid amylase and lipase activities, aided in the diagnosis of pancreatitis. 17 Elevated gamma-glutamyl transferase 20 can also be used, but none of these values are routinely tested in chemistry profiles.
Additionally, accuracy of these tests for the diagnosis of pancreatitis has not been established in horses, 22 and amylase and lipase levels are often determined from peritoneal fluid3,7,12 as it is routinely collected in colic cases, but for which less-defined reference ranges exist. Several other factors can also affect the value of these tests and make equine pancreatitis difficult to diagnose. For example, damage to intestinal mucosa or renal tubules can contribute to mild elevations of serum amylase and lipase. 7 Additionally, reabsorption of trypsin from the intestine can contribute to serum elevations. 11 Larger increases in trypsin in equine pancreatitis cases were found to be associated with poorer survival. 11 However, rarely, animals with pancreatitis have low amylase levels, especially if they are concurrently hyperlipemic, as hyperlipemia can interfere with the ability of light to scatter in the sample as part of the diagnostic test. 19 Cases of pancreatic disease may also rarely present as hepatobiliary disease due to the close proximity of pancreas tissue to the liver; liberation of pancreatic enzymes that damage liver can complicate antemortem diagnosis. 18
Ideally, measurement of pancreatic enzymes in serum and/or abdominal fluid, ultrasound of the pancreas, and exploratory laparotomy with biopsy would be recommended for pancreatic disease diagnosis confirmation, but these procedures are rarely feasible. 22 Although the pancreas can be difficult to evaluate via laparotomy, except in foals, where the small size of organs allows for easy manipulation, thorough examination of the pancreas is recommended. 5 Masses in the fat at the root of the mesentery or saponified or fibrous plaques within the mesenteric fat identified at laparotomy may indicate concurrent pancreatic disease. In a large retrospective study of pancreatitis in horses, clinicians determined that presenting clinical signs or gross lesions that supported a suspicion of pancreatitis included unexplained abdominal pain, gastric dilation or rupture, peritonitis, and/or the presence of peripancreatic fat necrosis. 22 In that study, moderate to severe abdominal pain was the most common clinical sign in primary or secondary pancreatitis cases. 22 Abdominal pain as a presenting complaint overlaps with presentation for many gastrointestinal and hepatobiliary diseases. Within that retrospective study, primary pancreatitis was diagnosed in 6 out of 43 cases. 22 The majority of the cases were considered secondary to concurrent large colon abnormalities (14/37 cases), with small intestinal abnormalities seen in 10 out of 37 cases, gastric rupture in 4 out of 37 cases, liver abnormalities in 3 out of 37 cases, and other disorders in 6 out of 37 cases. This final category included 1 case of multiple endocrine neoplasia-like syndrome, 1 strychnine poisoning case, 2 immune system dysfunction cases, and 2 cases of pituitary pars intermedia dysfunction (PPID). 22
Although the pancreas is likely routinely assessed at postmortem examination, it is not always routinely assessed by histology, especially if concurrent gastrointestinal or hepatobiliary conditions are identified. It is increasingly important to examine sections of pancreas, especially in cases of equine colic or gastric rupture, as both obvious conditions can also have concurrent pancreatic disease.14,15 Gastric rupture often negates assessment of the pancreas at necropsy, as it is easily deemed the significant lesion that caused death.14,15 Examinations of stomach, bowel, and pancreas should be conducted routinely in horses with supportive historical or laboratory workups for abdominal pain. Additionally, necrotizing panniculitis can be a consequence of pancreatitis. 20
In a previous study in dogs, pancreatitis was associated with a number of concurrent diseases, including diabetes mellitus (14/63 dogs), hyperadrenocorticism (12/63), chronic renal failure (8/63), neoplasia (17/63), congestive heart failure (6/63), and autoimmune disorders (5/63). 7 While few specific pathogenic mechanisms have been studied in and published for the horse, these same disease processes may be relevant in the comparative development of equine pancreatitis. For example, PPID is thought to predispose the horse to pancreatitis by allowing for recurrent bacterial infections owing to compromised immune system function. 22
Other documented predisposing conditions for pancreatitis in horses include grain overload, hyperlipemia, hyperadrenocorticism, and parasite migration.4,7,12,14,22 The mechanism for development of pancreatitis in grain overload situations is complex and might include gastric distension and subsequent endotoxemia, which may induce pancreatic ischemia and release of mediators such as platelet-activating factor, tumor necrosis factor, and bradykinin. 14 A case of carbohydrate overload, reported in a donkey, resulted from ingestion of poultry feed in excess. 14 It is difficult to prove cause and effect, but the donkey had dorsal colonic impaction and gastric dilation. It is likely that either the large bowel lesion or ischemia secondary to gastric dilation led to the pancreatic lesion. The pancreatic changes in the reported case were acute and consisted of edema, necrotic foci, hemorrhage, and mineralization. 14 Antemortem serum lipase and amylase were elevated, supportive of a primary pancreatic disease.
In cases with hyperlipemia, lipase within pancreatic capillaries may cause extensive hydrolysis of triglycerides and release large amounts of unbound free fatty acids, which cause local inflammation and ischemia.7,9 This perpetuates the cycle of pancreatic tissue damage and release of additional enzymes.7,9
Nematodes are most commonly associated in cases of eosinophilic (Sanford SE: 1987, Cross-Canada disease report: Ontario. Chronic eosinophilic pancreatitis and gastroenteritis in a horse. Can Vet J 28:748) and fibrosing pancreatitis, and include intraluminal ascarids (Parascaris equorum), 12 Strongylus equinus, 4 and aberrant Strongylus edentatus migration.3,22 This results in an eosinophilic inflammatory cell response and subsequent fibrosis. Rarely, similar changes have been noted with aberrant Parascaris species migration. 12
Other theories on development of equine pancreatitis include pancreatic hypoperfusion,10,11 bacterial and viral infections, immune dysregulation, 22 gastroduodenal ulceration,5,16 hyperlipemia secondary to PPID,7,14,22 hypovitaminosis A and E, dietary insufficiency of selenium and methionine, hypervitaminosis D, 4 and ascending biliary and/or pancreatic duct reflux or infection.5,22 In a large equine study of bile duct obstruction, the most common cause for the obstruction was cholangiohepatitis with cholelithiasis. This subsequently resulted in reflux of bile into the pancreatic duct and activation of pancreatic enzymes, resulting in autodigestion of the pancreas. 22 Pancreatic necrosis in foals has been associated with gastrointestinal reflux and shock,17,22 vasculitis,19,22 duodenitis that includes gastroduodenal ulceration, 5 duodenal stenosis, 5 chronic cholangiohepatitis, 5 and/or hyperlipemia. 17 Unlike in other species, hypertriglyceridemia has not been associated with pancreatitis in horses.9,21
Successful treatment of equine pancreatitis is limited. 3 Following appropriate medical therapy, continual decreases in serum amylase and lipase were associated with clinical improvement, but many animals are euthanized due to the primary condition or concurrent disease, and refractory abdominal pain. 22 It is important to necropsy all cases of unresolved acute or chronic colic, looking especially to rule out pancreatitis, as the suspicion is that it is often overlooked as a substantial cause of colic.
The following case series, assembled from a retrospective search of the University of Tennessee Veterinary Medical Center (UTVMC; Knoxville, Tennessee) Anatomic Pathology archival database from 2005 to 2014, includes 4 cases of pancreatic disease in horses. The incidence of pancreatic disease compared to the total number of equine necropsies in the same 9-year period was 4 out of 834 (0.4%). Because the necropsy service is used for both in-house and referral cases, some historical and clinical workup information is more extensive (cases 1, 2, and 4) than others (case 3). Although all 4 cases reported herein had concurrent disease, pancreatitis was considered the cause of death in only 1 (case 4).
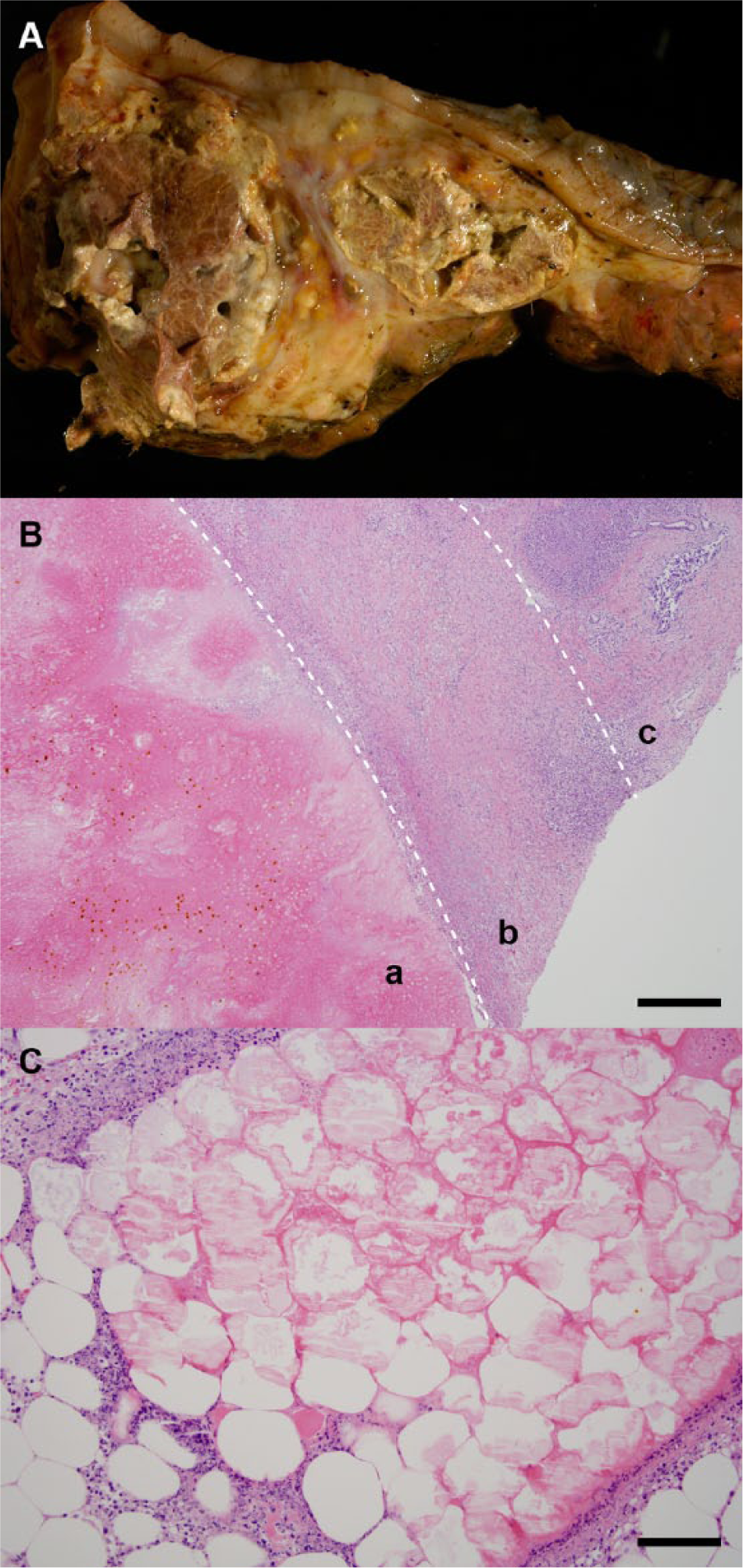
Case 1 was a 10-year-old, female American Quarter Horse that presented to the UTVMC for colic. The horse had an elevated heart rate and a distended abdomen, and on passage of a nasogastric tube, significant reflux was noted. Rectal palpation determined distended small intestinal loops. During exploratory laparotomy, large colon torsion was identified and corrected. Three days following surgery, the horse re-presented for colic, secondary to a colonic displacement, and the decision was made to euthanize. On necropsy examination, in addition to colonic ischemia, multiple white, smooth plaques were present in the fat surrounding the pancreas. Histologic assessment revealed eosinophilic outlines of adipocytes filled with moderate amounts of homogeneous eosinophilic material with a peripheral rim of karyorrhectic debris and rare necrotic neutrophils (Fig. 1C). This was interpreted as acute, marked, multifocal peripancreatic fat necrosis and saponification. The cause of death in this case was colonic torsion.
Case 2 was a 14-year-old, female Tennessee Walking Horse that was referred for colic. On passage of a nasogastric tube, there was significant reflux. Rectal palpation revealed empty bowel, and preliminary blood work values from the referring veterinarian’s workup showed leukopenia, azotemia, elevated fibrinogen, and high packed cell volume and total solids (absolute values not available). Microscopic assessment of free abdominal fluid showed bacteria, food material, and an elevated white blood cell count. Based on a poor prognosis, the horse was euthanized, and a necropsy examination requested. Cecal rupture with acute fibrinous peritonitis was confirmed. A gross description of the pancreas at necropsy was not included, suggesting that it was likely interpreted as normal. However, histological examination of the pancreas revealed cellular dissociation, lack of cytoplasmic zymogen granules, nuclear pyknosis, and karyolysis, consistent with diffuse acute coagulation necrosis. The cause of death was cecal rupture and peritonitis.
Case 3 was a 17-year-old, mixed-breed mare that had been surrendered from a local equine rescue group and euthanized. At necropsy, the animal was in poor body condition (1/9). Grossly, the duodenum was adhered to the small colon via an 8 cm × 6 cm × 4 cm region of edema and fibrin surrounding the pancreas. Histologically, the peripancreatic adipose tissue was characterized by abundant, finely fibrillar eosinophilic material (fibrin) admixed with neutrophils and cellular and karyorrhectic debris, which replaced normal architecture. Multifocally, there was loosely arranged fibrous tissue containing a few perpendicularly oriented small caliber blood vessels lined by plump endothelial cells (early maturing granulation tissue). Moderate numbers of macrophages, lymphocytes, and plasma cells formed multifocal aggregates throughout, and there was scattered mineralization, indicating subacute fibrinonecrotic pancreatitis with early fibroplasia. These lesions were in addition to the finding of neoplastic myeloid cell series (myelocytic leukemia), affecting the kidney, spleen, lymph node, and bone marrow in this patient. The cause of death was attributed to leukemia.
Case 4 was a 21-year-old, Morgan gelding. Presentation was for colic, which had been intermittent over a 2-month period and was characterized by diarrhea and intermittent signs of pain. Previous blood work revealed lipemia, and the animal was a known type I diabetic being treated with insulin daily. Subsequent blood work showed leukopenia (2.75, reference [ref.] interval: 4.6–12), thrombocytosis (299, ref. interval: 107–205), neutropenia (1.26, ref. interval: 2.6–5.5), lymphopenia (1.4, ref. interval: 1.5–4.3), hyponatremia (127), hypochloremia (89); decreased aspartate aminotransferase, creatine kinase blood urea nitrogen, calcium, and albumin; and increased gamma-glutamyl transferase, glucose, and globin. Rectal palpation determined a mass at the dorsal body wall. Ultrasound revealed abdominal fluid, thickened small intestinal walls, and a mass at the level of the right kidney. Cytologic assessment of the abdominal fluid revealed septic suppurative inflammation. Because of a poor prognosis, the animal was euthanized, and a necropsy examination was requested. The pancreas and right kidney were surrounded by soft, amorphous, white material measuring several centimeters thick. The pancreas had a brown, lobular appearance with abundant saponification of the peripancreatic and interlobular adipose tissue (Fig. 1A). Aerobic and anaerobic culture of the pancreas failed to grow bacteria. The pancreatic lesion was characterized histologically by coagulative necrosis adjacent to some regions of viable pancreas. Interlobular fibrosis and marked atrophy were seen throughout the viable regions. Peripancreatic fat was hypereosinophilic and contained foci of hemorrhage, foamy macrophages, neutrophils, cellular debris, and multifocal extensive areas of fibrosis and hemosiderin-laden macrophages (Fig. 1B). Marked chronic active diffuse necrotizing pancreatitis with marked exocrine pancreatic atrophy and interstitial fibrosis was diagnosed. Concurrently, a pituitary microadenoma was identified. In this instance, the cause of death was attributed to the necrotizing pancreatitis.
The concurrent conditions in the cases presented herein are similar to those reported in the larger equine study, in that 2 cases had large bowel conditions: case 1 had a colonic torsion, and case 2 had a cecal rupture. The most commonly associated large bowel lesions in the large study were colonic volvulus in 9 out of 14 horses and right dorsal displacement in 1 out of 14 horses, both of which would be associated with typical intestinal colic signs. 22 The specific association with myelocytic leukemia in case 3 may be considered unusual because bowel, liver, and/or pancreas were not primarily involved, but in other species, particularly dogs, neoplasia is a general risk factor for the development of pancreatitis. 6 Chronic fibrosing pancreatitis has been associated with concurrent hepatic disease in horses, and in the large equine study, was apparent with 3 cases, 2 with choleliths and 1 with suspected, but unproven, biliary obstruction. 22 Concurrent hepatic and pancreatic disease in some reported cases may be secondary to ischemia, following large bowel accidents. 22 Additionally, pancreatitis may result from obstruction of the bile duct from primary biliary disease or, less likely, subsequent to gastrointestinal accidents, both of which could predispose the horse to secondary reflux of bile into the pancreatic duct and subsequent activation of pancreatic enzymes. 22 In cases 1 and 2, the peripancreatic fat showed evidence of acute necrosis, potentially consistent with a recent ischemic event, and possibly associated with a large bowel disorder. 2 The underlying cause of the more subacute lesion in the pancreas of case 3 is unknown but may be related to the long-standing systemic neoplastic process that was confirmed. In case 4, chronic primary pancreatitis was considered the cause of death. While the cause of the pancreatitis was not specifically known, it was considered to be a result of the concurrent diabetes mellitus, a condition that, in dogs, is associated with the development of pancretitis.1,6
All 4 cases in the current study presented with colic, and while 2 had other reasons for gastrointestinal colic, acute pancreatitis often presents with marked abdominal pain, gastrointestinal reflux, and hypovolemic shock. 22 Chronic cases are more likely to show weight loss, inappetence, lethargy, and mild to recurrent signs of colic. 22 Although rare, equine pancreatic disease does occur as an underlying cause of colic. 2 It can be primary, but is more often associated with other conditions, such as those involving the gastrointestinal tract, liver, endocrine system, and/or secondary to neoplasia. Hence, examination, both at postmortem examination and with histology, is recommended, especially in cases without classic equine pathologies associated with colic, to gain more information about the role of pancreatitis in equine death and association with concurrent diseases. Additional development of diagnostic tests may help with differentiation of pancreatic disease in the colic workup.
Footnotes
Acknowledgements
Misty Bailey is acknowledged for technical editing and Ms. Anik Vasington for graphic assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
