Abstract
We herein report a case of phlegmonous gastritis secondary to superior mesenteric artery syndrome. An 80-year-old woman visited the hospital emergency department with the chief complaints of epigastric pain and vomiting. She was hospitalized urgently following the diagnosis of superior mesenteric artery syndrome based on abdominal computed tomography findings. Conservative therapy was not effective, and phlegmonous gastritis was diagnosed based on the findings of upper gastrointestinal endoscopy and biopsy performed on the 12th day of the disease. Undernutrition and reduced physical activity were observed on hospital admission, and proactive nutritional therapy with enteral nutrition was started. An upper gastrointestinal series, performed approximately 1 month later, confirmed the persistence of strictures and impaired gastric emptying. Because conservative therapy was unlikely to improve oral food intake, open total gastrectomy was performed on the 94th day of the disease. Examination of surgically resected specimens revealed marked inflammation and fibrosis, especially in the body of the stomach. Following a good postoperative recovery, the patient was able to commence oral intake and left our hospital on foot approximately 1 month after surgery.
Introduction
Phlegmonous gastritis is an uncommon disease characterized by nonspecific suppurative inflammation of all layers of the stomach wall, which is most profound in the submucosa. The number of cases of phlegmonous gastritis secondary to superior mesenteric artery (SMA) syndrome is extremely low. We recently encountered such a case, wherein conservative therapy was effective in reducing inflammation and improving the patient’s general condition but surgery could not be avoided to treat strictures. Herein, we report this rare case and review the literature on this condition.
Case report
An 80-year-old woman visited the emergency outpatient department of a hospital with the chief complaints of epigastric pain and vomiting. She had no history of smoking and alcohol intake, and hyperlipidemia was the only underlying disease in her medical history. Abdominal computed tomography (CT) showed marked enlargement of the area from the stomach to the descending part of the duodenum, and a change in the caliber of the horizontal part of the duodenum behind the aorta (Figure 1), leading to the diagnosis of SMA syndrome. The patient was urgently hospitalized for conservative therapy that consisted of no oral intake, fluid replacement, administration of an antibiotic (cefmetazole), and nasogastric feeding, but the epigastric pain and severe inflammation persisted. Upper gastrointestinal endoscopy performed on the 12th day of the disease confirmed the presence of ulceration across a wide area of the stomach body (Figure 2(a)). Tissue culture specimens were positive for Enterobacter cloacae and Enterococcus faecium, leading to the diagnosis of phlegmonous gastritis. Replacement of cefmetazole with meropenem (MEPM) normalized the white blood count (WBC) and reduced inflammation. However, endoscopic examination of the extensive ulceration on the 23rd day of the disease did not detect improvement, and the patient was subsequently transferred to our hospital on the 34th day of the disease. The initial abdominal examination detected tenderness but not rebound tenderness, guarding, or a palpable mass. Peripheral blood tests showed normal WBC (6600/µL) and anemia (Hb level: 9.8 g/dL). Blood biochemistry tests indicated undernutrition (total protein level: 6.3 g/dL; albumin level: 2.9 g/dL) and mild inflammation (C-reactive protein (CRP) level: 1.1 mg/dL). No organism was detected in the blood culture, but Enterobacter cloacae and Enterococcus faecium were present in gastric juice culture. Upper gastrointestinal endoscopy, performed on the 38th day of the disease, confirmed the presence of bile-trapping ulceration with a cloudy coating across the entire stomach body, except for the upper part of the greater curvature. It was impossible to insert an endoscope into the duodenum due to stricture in the mid-part of the stomach (Figure 2(b)). Examination of biopsy specimens from the upper part of the greater curvature confirmed severe erosion and granulomatous change (Figure 3). Enterobacter cloacae was detected in the culture of gastric mucosa. Although the infection appeared to be controlled, the patient continued to have undernutrition and reduced physical activity, suggesting that improvement in nutrition status was the priority. No oral intake and antibiotic treatment (MEPM: 4 g/day) were continued while rehabilitation and enteral nutrition were started. MEPM was later replaced by ciprofloxacin based on the results of drug sensitivity tests using the gastric mucosa culture. Repeat endoscopy approximately 1 month later showed that extensive ulceration still affected the entire stomach, although a slight size reduction and partial epithelialization were observed. Furthermore, the stricture in the mid-part of the stomach had worsened (Figure 4). An upper gastrointestinal series confirmed the presence of strictures from the mid-part to the antrum of the stomach, and passage of the contrast agent was problematic. Gastric peristalsis was absent: a small amount of contrast agent came out of the stomach cavity upon changes in position, and significant esophageal reflux was observed (Figure 5). Although an improvement in nutritional status was observed, the strictures and dysfunction (loss of peristalsis resulting in impaired clearance of stomach contents) persisted. Because of the possible long-term problem in oral food intake, a surgical approach (total gastrectomy and Roux-en-Y reconstruction) was employed on the 94th day of the disease. Severe inflammation-associated adhesions affected the center of the upper abdominal cavity. Macroscopically, the greater and lesser curvatures were shortened by 9 and 7 cm, respectively. Increased wall thickness and fibrosis were observed from the upper to the lower part of the stomach body, while antral distension was relatively preserved. The greater omentum was only mildly cloudy (Figure 6). Microscopically, erosion and scarring due to ulceration were observed, particularly in the body of the stomach. Luminal obstruction was observed in a middle-sized artery, while an organized thrombus and obstruction were found in a middle-sized vein in the submucosal and subserosal layers. Active and strong inflammation was found almost exclusively in the mucosa, while fibrosis and xanthogranuloma-like histopathologic features involving multinucleated giant cells were prominent in and below the submucosa. The border of the ulcer in the antrum showed fibrotic foci that were clearly distinguishable from the normal mucosa, suggesting blood supply–dependent ischemia (Figure 7(a)–(d)).

CT examination performed at another hospital before admission into our hospital. Marked enlargement of the area from the stomach to the descending part of the duodenum was observed.
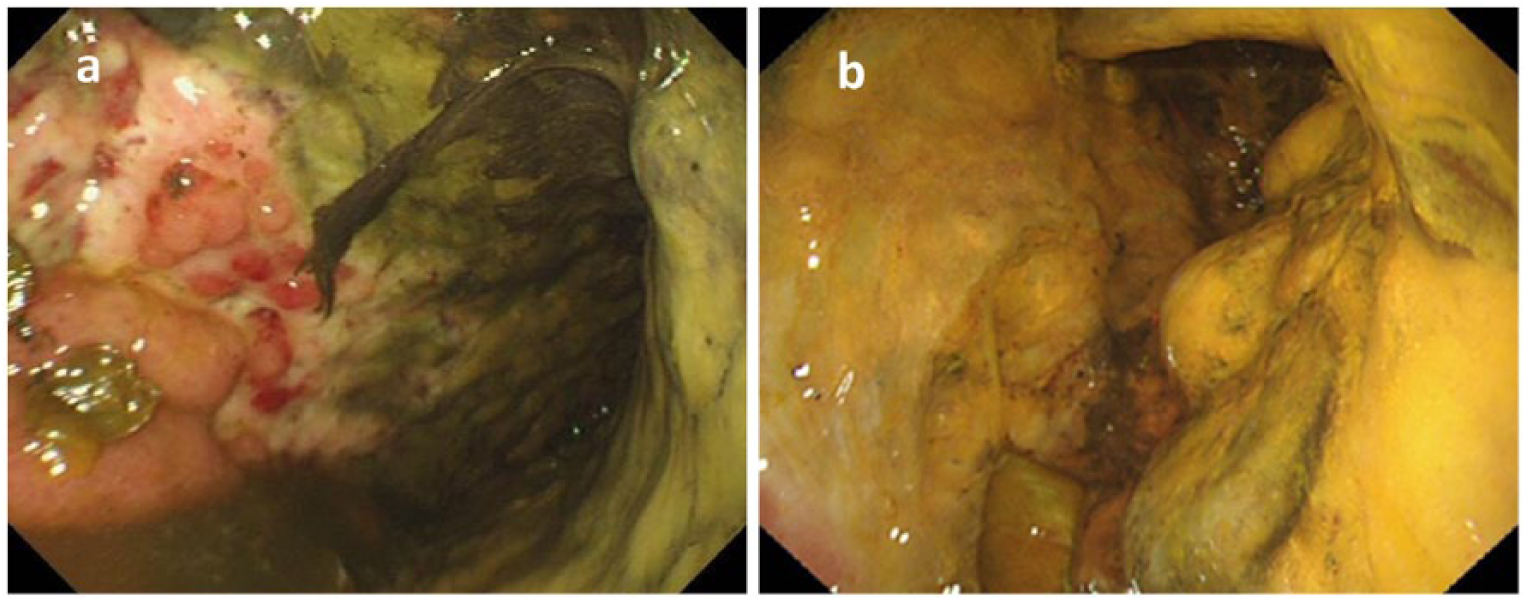
Upper gastrointestinal endoscopy: (a) the 12th day of the disease: ulceration was found across a wide area of the stomach body. A filthy cloudy coating was observed across the entire stomach. Enterobacter cloacae and Enterococcus faecium were detected in culture; and (b) the 38th day of the disease: the ulceration across the entire stomach body had not improved, and bile-trapping and an exudate were observed. A stricture was present in the mid-part of the stomach.

Biopsy: 38th day of the disease (hematoxylin and eosin (HE) staining). Severe erosion was observed, the majority of the epithelium was peeled off, and granulation tissue was present.

Upper gastrointestinal endoscopy findings (90th day of the disease). Gradual epithelialization was observed. An endoscope could not be passed through the stomach cavity because of progressive stricture in the body of the stomach.
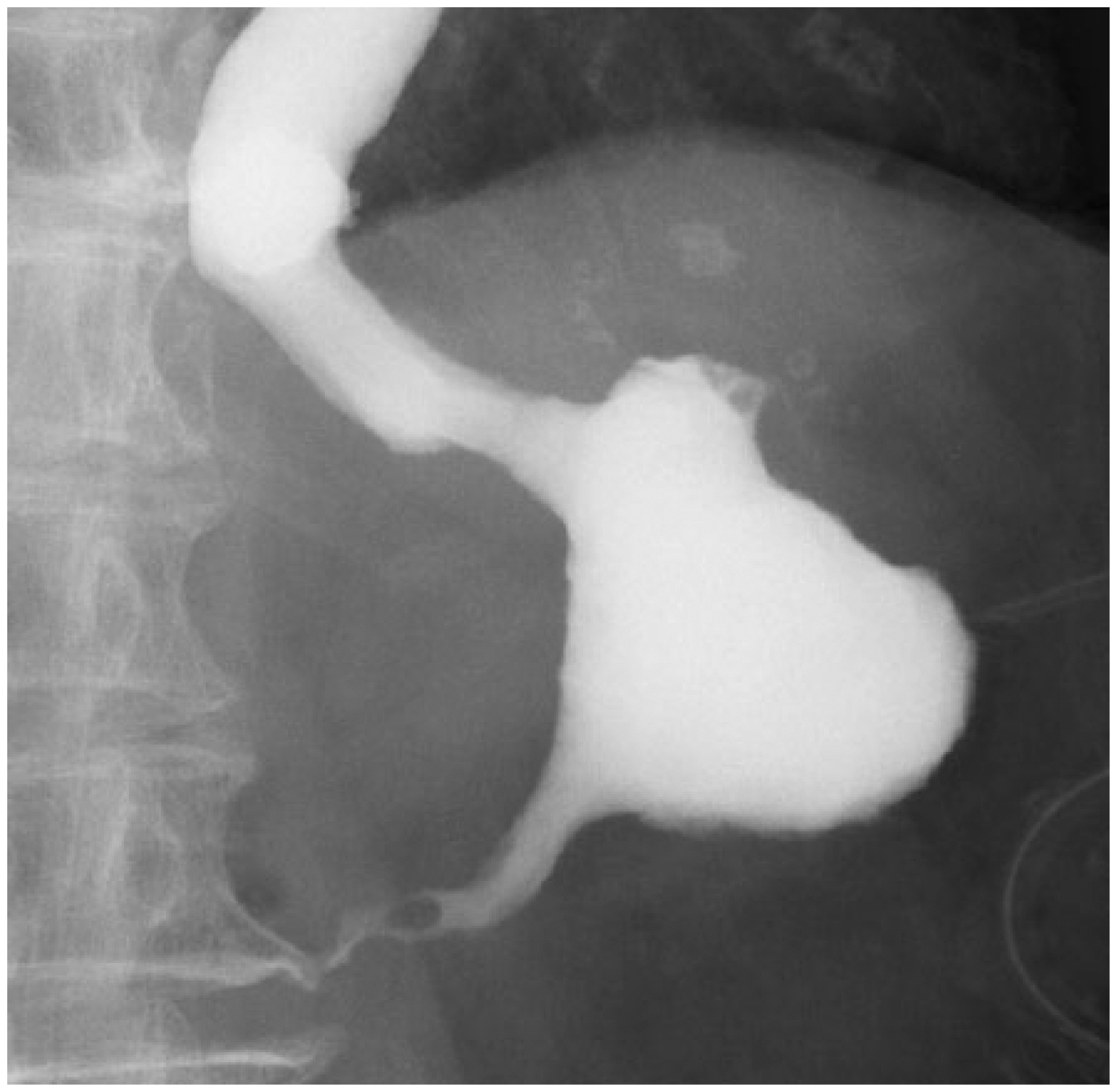
Upper gastrointestinal series findings. Strictures of the stomach body and difficulties in passage of contrast agent were observed. Gastric peristalsis was absent, and a small amount of the contrast agent came out upon changes in position. Significant reflux toward the esophagus was observed.

Macroscopic findings of the surgically resected specimen. Thickening of the entire wall was observed, and the lengths of the greater curvature and the lesser curvature were apparently shortened by 9 and 7 cm, respectively. Increased wall thickness and fibrosis were observed from the upper to the lower part of the stomach body, while antral distension was relatively preserved. The greater omentum was only mildly cloudy.

Pathological findings: (a) a microscopic image of the lesser curvature of the stomach. In the mucosal layer, epithelial shedding and granulation were observed. Fibrosis as well as arterial obstruction (arrow) and organized thrombus (arrow head) were prominent in and below the submucosa; (b) a high-power view of the mucosa. Granulation was observed in the superficial layer; (c) a high-power view of the deeper layer. Foamy cells and multinucleated giant cells, indicative of chronic inflammation and fibrosis, were present; and (d) a microscopic image of the ulcer margin. Active inflammation and erosion were observed in the left part of the section, while preserved epithelium and mild inflammation were seen in the right part of the section. The border of the ulceration was relatively clear.
Following a good postoperative recovery, the patient was able to ingest food orally and left our hospital on foot approximately 1 month after surgery. Her condition has been good during the 2 years of follow-up, allowing her to have a healthy social life.
The patient has consented to the publication of this article. Our institution does not require ethics approval for reporting individual cases.
Discussion
Phlegmonous gastritis is a nonspecific suppurative inflammatory disease characterized by exudative changes and infiltration of inflammatory cells in all layers of the stomach wall, which is most prominent in the submucosa. 1 In 1820, Cruveilheier et al. reported the first case in an autopsy study, 2 and 500 cases have been published. We reviewed 77 cases of phlegmonous gastritis (including those reported in proceedings) registered with the datebase PubMed and the Igaku Chuo Zasshi (database provided by the Japan Medical Abstracts Society) between 2000 and 2014. From the literature review, we found that the disease affects all age groups and is most common in adults in their 50s to 70s, and the male to female ratio is 2:1.
Konjetzny 3 reported three types of phlegmonous gastritis (primary, secondary, and idiopathic) depending on the cause of the disease. Primary phlegmonous gastritis, the most common type, is caused by direct microbial invasion through the mucosa damaged by gastric ulcers, cancer, foreign objects (e.g. nasogastric tubes), biopsy, and increasingly popular endoscopic interventions (e.g. endoscopic mucosal resection, endoscopic submucosal dissection). Secondary phlegmonous gastritis is caused by hematogenous spread from infective foci in other organs (e.g. septicemia, endocarditis), or by direct influence of inflammation in neighboring organs (e.g. pancreatitis, cholecystitis). Idiopathic phlegmonous gastritis has an unknown cause with absence of a primary lesion.
Marked gastric enlargement due to SMA syndrome was observed in this patient, suggesting that ischemia affected a wide area of the gastric wall, which might have led to ulcer formation and subsequent bacterial infection and eventually to phlegmonous gastritis. Thus, the present case was considered to be primary phlegmonous gastritis.
Contributing factors to phlegmonous gastritis include regular alcohol consumption, aging, excessive food intake, extreme exhaustion,3,4 elevated pH of gastric juice, 2 and the use of steroids and other immunosuppressive agents. 5 In particular, Sundberg 6 reported that excessive alcohol consumption and decreased gastric acid levels caused by antacids reduce the sterilizing capacity of gastric juice, thereby contributing to development of the disease. Streptococcus is the most common cause of phlegmonous gastritis, found in approximately 70% of cases, followed by Enterobacter, Escherichia coli, Proteus, and Enterococcus.2,7 In the present case, although blood culture was negative, Enterobacter cloacae was detected in gastric juice and mucosal cultures while Enterococcus faecium was detected in gastric juice culture, suggesting these bacteria could potentially cause phlegmonous gastritis.
Symptoms of phlegmonous gastritis include severe pain in the upper part of the abdomen, nausea, vomiting, and fever accompanied by chills. Deiningen’s sign, which is upper abdominal pain associated with acute pancreatitis that decreases or increases in the supine position or sitting position, respectively, was observed in this case. Generally, peritoneal irritation signs such as abdominal tenderness, guarding, and the Blumberg sign are present, and blood test results indicate inflammation. All of these findings are nonspecific, and thus, it is necessary to differentiate phlegmonous gastritis from an acute abdomen (e.g. perforation peritonitis, acute cholecystitis, and acute pancreatitis).
Phlegmonous gastritis is a rare disease characterized by nonspecific clinical symptoms and blood test results, and thus, a comprehensive approach, including various imaging modalities, is used for its diagnosis. Upper gastrointestinal endoscopy often shows erosion, ulcers with a cloudy coating, and diffuse edema in patients with phlegmonous gastritis. Biopsy is also important for its diagnosis. Identifying the microbial species by gastric mucosal culture is also useful for diagnosis and is absolutely necessary for selection of appropriate antibiotics. Abdominal ultrasound and abdominal CT typically reveal the following characteristic findings of phlegmonous gastritis: thickening of the gastric wall, low-intensity areas within the gastric wall (indicative of an abscess), accumulation of gas, and localized fluid accumulation. However, the diffuse and localized types are often misdiagnosed as scirrhous stomach cancer and gastric submucosal tumor, respectively, and thus, it is important to exclude these diseases. Because the main site of inflammation is the submucosa, 8 endosonography is useful for diagnosis of phlegmonous gastritis 7 that has characteristic findings of submucosal thickening and a hypoechoic area within the submucosa. 9 Prominent edema mainly in the submucosa is characteristic of phlegmonous gastritis. Infiltration of neutrophils can be observed at the early stage, while infiltration of monocytes, lymphocytes, and plasma cells become prominent and fibrosis occurs when the disease progresses toward the chronic stage. 2 Indeed, we found marked infiltration of inflammatory cells and gastric wall thickening associated with fibrosis in our patient.
Conservative approaches (e.g. antibiotic therapy, continuous gastric aspiration) and invasive approaches (e.g. gastrectomy and endoscopic drainage) are currently used for treatment of phlegmonous gastritis. Previously, gastrectomy was often the preferred treatment option.3,10 However, recent studies showed that conservative therapy led to improvement in many cases, leading to the understanding that phlegmonous gastritis can be treated mainly by antibiotics when complications such as bleeding, perforation, and strictures are absent. In this study, conservative therapy reduced inflammation as evidenced by blood tests, but fibrosis-associated hardening of the entire thickness of the gastric wall and strictures persisted, requiring surgical therapy.
We searched the Pubmed and the Igaku Chuo Zasshi database for phlegmonous gastritis cases reported after 2000, and a review of 77 relevant cases (including the present case) revealed that conservative therapy was used in 52 cases (67.5%), surgical resection in 21 cases (27.3%), drainage in 3 cases, and other approaches in 1 case. In recent reports, many cases occurring after biopsy or endoscopic therapy for the primary disease were successfully treated conservatively.11–13 The overall cure rate (87.0%, 67/77) was good. There were 10 fatal cases including 2 autopsy cases and one death due to the primary disease. The cure rates of conservative therapy, surgical resection, and drainage were 84.6% (44/52), 95.2% (20/21), and 100% (3/3), respectively. Meanwhile, 5 cases including the present case were phlegmonous gastritis secondary to SMA syndrome.14–17 Although conservative therapy was the preferred approach in many cases, phlegmonous gastritis secondary to SMA syndrome appeared to be severe, requiring surgery in 3 cases,14,15 and worsening of the patient’s condition resulted in death in 2 cases (40%).16,17 In our case, initial conservative therapy and proactive nutritional care was effective in the acute stage. However, we opted for surgical resection with the aim of achieving a complete cure because of persistent inflammation and the presence of strictures. There were no major postoperative complications, and the patient’s condition is satisfactory approximately 2 years after hospital discharge.
Conclusion
We encountered a case of phlegmonous gastritis secondary to SMA syndrome. Marked gastric enlargement due to SMA syndrome caused ischemia in a wide area of the gastric wall, which might have caused ulceration followed by a bacterial infection, leading to phlegmonous gastritis. Conservative therapy successfully reduced inflammation, but surgical therapy was required to treat strictures that had formed after fibrosis of the gastric wall. Although antibiotic therapy is the standard therapy, phlegmonous gastritis following SMA syndrome tended to be severe. Surgical therapy in the acute phase can be avoided by combining antibiotic therapy with proactive nutritional intervention, as shown in the present case.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
