Abstract
Fifty canine gastrointestinal (GI) mesenchymal tumors were examined to determine the occurrence of leiomyomas (LM) and GI stromal tumors and to compare their clinicopathologic features. Twenty-one tumors (42%) were histologically reclassified as gastrointestinal stromal tumors (GISTs) and 29 tumors (58%) as LMs on the basis of their histologic similarity with homologous human tumors. The GISTs occurred equally in males and females, with a mean age of 11 years (range 5–14 years). Five GISTs (24%) were associated with clinical signs and six (29%) had metastasis in liver or abdominal cavity. The GISTs occurred in large intestine (10, 48%), small bowel (six, 29%), stomach (four, 19%), and mesentery of small intestine (one, 5%). Histologically, they were highly cellular spindle, or less commonly epithelioid tumors with mitotic rates ranging from 0 to 19 per 10 HPF. Eleven tumors (52%) were positive for CD117 (KIT); seven (33%) were positive for smooth muscle actin but none for desmin and S-100 protein. Sequences of KIT exon 11, often mutated in human GISTs, were evaluated from four GISTs. Deletion of Try556-Lys557 coexisting with duplication of Gln555 in one case of GIST and T to C transition resulting in substitution of Pro for Leu575 in another were identified. The LMs occurred predominantly in males (82%) with a mean age of 11 years (range 8–17 years). Nine tumors (31%) had associated clinical signs. They occurred in the stomach (22, 76%), esophagus (four, 14%), and intestines (three, 10%); all were paucicellular, had no mitoses, and were composed of mature smooth muscle cells. Twenty-eight (97%) were positive for smooth muscle actin and 18(62%) for desmin but none for CD117 and S-100. Both GISTs and true LMs occur in the GI tract of dogs. Both tumors have distinctive pathologic features.
Gastrointestinal (GI) stromal and smooth muscle tumors have been reported in dogs, 13,29,37 horses, 2,6 and nonhuman primates. 1 But none of the studies have so far separated canine gastrointestinal stromal tumors (GIST) and true smooth muscle tumors and examined their clinicopathologic and molecular genetic features. This would be important because GISTs and leiomyomas (LM) are known to be separate clinicopathogic entities in humans. 23
GI mesenchymal tumors of humans are currently divided into GISTs and LMs on the basis of histologic features and cell differentiation. GISTs constitute the majority of mesenchymal tumors of the GI tract. They are most common in the stomach and small intestine but can occur anywhere in the GI-tract. Clinically, the human GISTs have a spectrum from small benign tumors to frankly malignant sarcomas. 23
GISTs typically express KIT (CD117), similar to hematopoietic stem cells and the interstitial cells of Cajal. 10,32 Many GISTs have activating KIT mutations, which likely represent a driving force in GIST oncogenesis. 7,11,14,15,20,34 The majority of KIT mutations have been identified in the juxtamembrane domain (exon 11) of the gene. These mutations lead to spontaneous, ligand-independent tyrosine kinase activation 7 and may affect regulatory function of the kinase molecule. 18 The KIT juxtamembrane domain protein sequence is highly preserved among different species and shows 100% homology between dog and human. Detection of similar KIT mutations in canine GISTs, as seen in human GISTs, would strongly suggest that tumorigenesis of dog and human GISTs follows the same molecular pathways.
In humans, the GI LMs typically occur in the esophagus and are very rare in the stomach and intestines, with the exception of small polypoid LMs in the colon and rectum. 22,23 LMs are composed of well-differentiated smooth muscle cells that are typically positive for smooth muscle actin and desmin, markers for smooth muscle differentiation. These tumors are negative for CD117 and typically have an indolent clinical course. 23
The purpose of this study was to explore the histomorphologic, immunohistochemical, and molecular genetic profile of GI mesenchymal tumors in dogs, especially the typical LMs and specific stromal tumors (GISTs), in analogy with similar tumors in humans.
Materials and Methods
Case material
Case searches on GI LMs and stromal tumors in dogs were performed in the files of the Department of Veterinary Pathology of the Armed Forces Institute of Pathology between years 1943 and 2000. The following key words for esophagus, stomach, duodenum, ileum, jejunum, colon, cecum, rectum, and mesentery were used: LM, leiomyosarcoma (LMS), smooth muscle sarcoma, sarcoma, neurofibroma, schwannoma, peripheral nerve sheath tumor, and malignant peripheral nerve sheath tumor. Representative material was available from a total of 57 dogs. Seven cases were eliminated because on reexamination they represented other tumors (five unclassified sarcomas, two angiosarcomas). The remaining 50 cases were included in the study and classified as GISTs or LMs on the basis of similarity with analogous human tumors. Twenty-four cases were military working dogs (MWD). Clinical data were reviewed from the records.
Histology and immunohistochemistry
Representative tissue samples were sectioned at 5 μm, and stained with hematoxylin and eosin (HE). Immunohistochemical staining was performed on deparaffinized sections with linked streptavidin-biotin (LSAB) complex technique using an LSAB detection kit (Dako, Carpinteria, CA) with diaminobenzidine as the chromogen. Antibodies to the following antigens were used: KIT (CD117), a polyclonal antibody at 1:400; Santa Cruz Biotechnology, Santa Cruz, CA; catalog no. se-168, α-SMA (1A4 monoclonal at 1:8,000; Sigma Chemicals, St. Louis, MO), desmin D33, monoclonal at 1:100; Dako), S-100 protein (polyclonal antibody at 1:1,600; Dako). Microwave epitope retrieval was performed prior to the immunostaining for KIT. CD34 (QBEND-10, monoclonal, diluted at 1:80; Biogenex Laboratories, San Ramon, CA) was also evaluated but could not be included in the study because the antibody did not exhibit any reactivity in dog tissues. Appropriate negative and positive controls were included.
Kit mutation analysis
In five cases (four GISTs and one LM), partly degraded genomic DNA was obtained from formalin-fixed, paraffin-embedded tissues using previously described methods. 14 Polymerase chain reaction (PCR) assay to screen degraded DNA samples for the mutation in the KIT juxtamembrane domain was developed. High molecular weight DNA was obtained from the fresh tissue of a healthy Chihuahua dog using standard proteinase K and phenol-chloroform extraction. Canine KIT intron 10 was PCR amplified using forward primer, dck10F (5′ ATCCATCCCCACACCCTGTT 3′) and reverse primer, dck11R (5′ CCTTCCACTGTACTTCATAC 3′). Canine KIT intron 11 was PCR amplified using forward primer, dck11F (5′ GGAGTTTCCCAGAAACAGGC 3′) and reverse primer, dck12R (5′ TCATGGCCGCATCCGACTTA 3′). Primers were designed on the basis of canine KIT cDNA sequences (AF044249, AF099030) deposited in the GeneBank database (GBD). Reaction conditions were standard as recommended by Perkin-Elmer (Norwalk, CT) with the annealing temperature of 55 C. PCR amplification products were size fractionated on agarose gels, purified, and sequenced directly as previously described. 16 The sequences were analyzed using the Lasergene software (DNASTAR, Inc., Madison, WI) and aligned with normal human and canine KIT sequences deposited in the GDB. One forward (d1F) and two reverse primers (d2R, d3R), flanking closely canine KIT juxtamembrane domain and allowing PCR amplification of the small products, were designed. Primer sequences and their positions are shown in Fig. 1. DNA templates were PCR amplified for 30 cycles with d1F and d2R, and then aliquots of the PCR products were reamplified for 40 cycles using d1F and nested reverse d3R primers. Expected size of the amplification products was 240 and 190 base pair (bp) for the first and nested reaction, respectively. Reaction conditions were standard as recommended by Perkin-Elmer (Norwalk, CT) with the annealing temperature of 50 C. The amplification products were size fractionated, purified, and sequenced directly. Recently published canine KIT cDNA sequences show differences in the length and codon numbering. 16,21 We have aligned canine sequences AF044249, AF099030, and human KIT cDNA. Predicted protein sequences of canine and human KIT juxtamembrane domains show 100% homology. Deletion of Gln255 and insertion of Gly-Asn-Ser-Lys between Lys511and Glu512 generated the differences in codon numbering between AF099030 and AF04429 canine sequences. The four amino acids insertion was similar to that previously reported in an alternatively spliced variant of KIT, KIT A. 32 To avoid confusion we have decided to number the codons of the amplified KIT juxtamembrane domains according to GBD submission AF044249. In addition, we are providing the numbers of the corresponding human KIT codons in parentheses.

PCR amplified sequence of canine KIT aligned with human KIT exon 11. Canine KIT intron 10 and intron 11 sequences are shown in small characters. Human KIT sequence is shown below. Predicted amino acid sequence and codon numbers are shown above. Gray boxes mark nucleotide differences between human and canine sequences. Black lines indicate primer sequences.
Transmission electron microscopy
Ultrastructural studies were performed on two GISTs (dog case Nos. 10 and 11) and two LMs (dog case Nos. 10 and 13). Tissue for electron microscopy was recovered from formalin-fixed tissues that were deparaffinized, hydrated, postfixed in 1% osmium tetroxide, dehydrated, cleared, and embedded in epoxy resin. One-micrometer sections were cut and stained with toluidine blue for preliminary light microscopic examination. Thin sections (80–90 nm) were cut, stained with uranyl acetate and lead citrate, and examined with a Zeiss EM 10 transmission electron microscope.
Results
GIST
Clinical features
The signalment, clinical features, tumor histomorphomology, and immunohistochemistry analyses are summarized in Table 1. There were 10 males and 10 females ranging in age from 5 to 14 years with an average age of 10.5 years (median = 11.5); the sex of one dog and the ages of two dogs were unknown. Eight tumors occurred in German Shepherd Dogs, two in mixed breeds, and one each in Belgian Malinois, Black Labrador, Collie, Dachshund, Irish Setter, Standard Poodle, Scottish White Terrier, and Terrier Cross; three were of unknown breed.
GIST signalment, clinical signs, histologic characteristics, and immunohistochemical results.
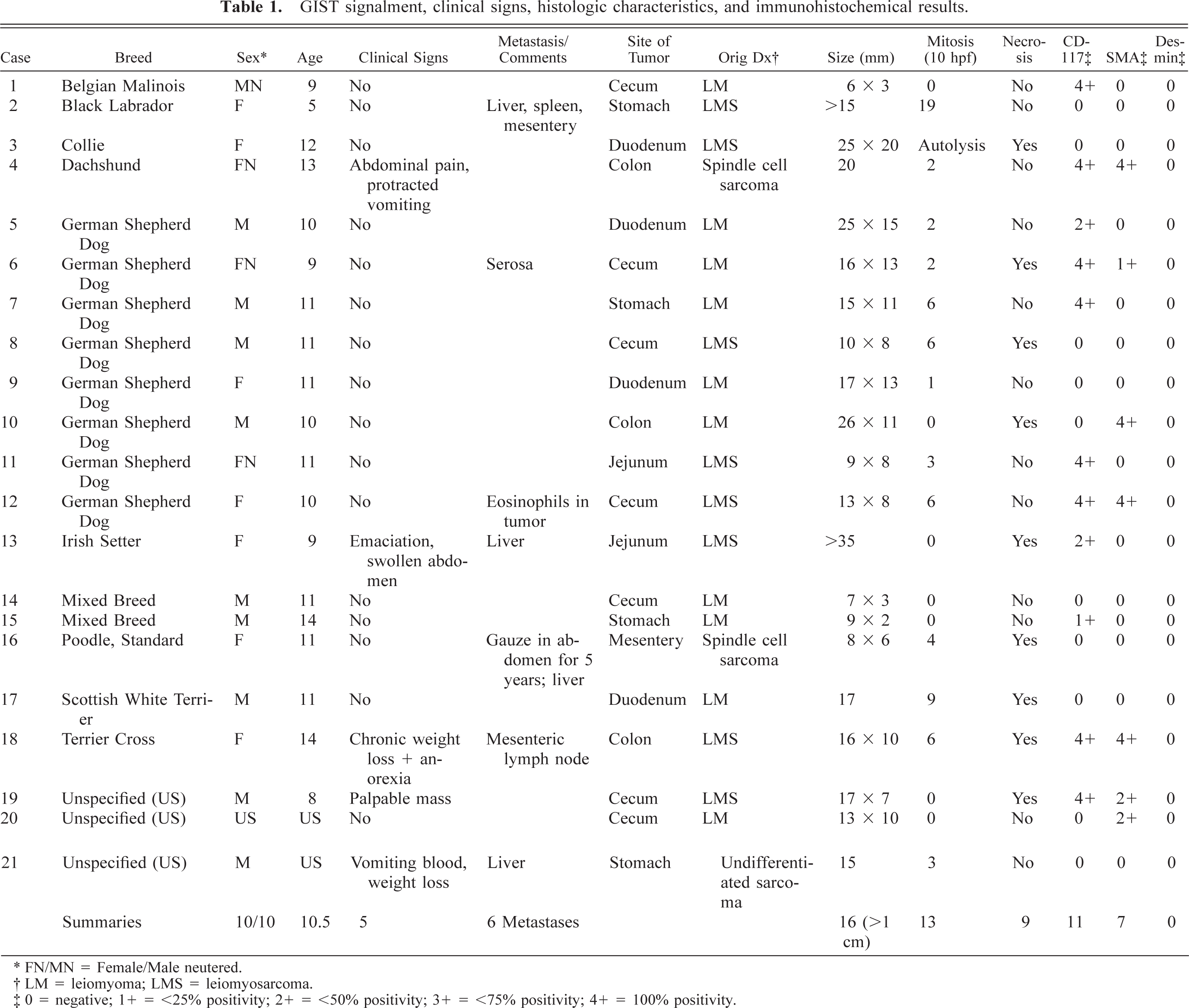
∗ FN/MN = Female/Male neutered.
† LM = leiomyoma; LMS = leiomyosarcoma.
‡ 0 = negative; 1+ = <25% positivity; 2+ = <50% positivity; 3+ = <75% positivity; 4+ = 100% positivity.
Clinical signs attributable to neoplasia were observed in five cases (24%), the other 16 tumors were reported as incidental findings at necropsy and not considered causal factors in the animals' deterioration or death. Weight loss and vomiting each presented in two dogs; abdominal pain, swollen abdomen, anorexia, and palpable mass each presented in one dog.
The most common site for GIST was large intestine (10), seven of which occurred in the cecum. Six tumors were from the small intestine (including four from the duodenum), four from the stomach, and one from the small bowel mesentery. Metastases were seen in the liver (four), and one each in the spleen, mesentery, serosa, and mesenteric lymph node.
Pathology
The tumors primarily involved the tunica muscularis and often extended transmurally, sometimes in an exophytic manner (Fig. 2). The median diameter from glass slide measurements was 15 mm (range 6–35 mm). Sixteen tumors measured at least 1 cm in greatest diameter. Overlying mucosa, when present, was not ulcerated.
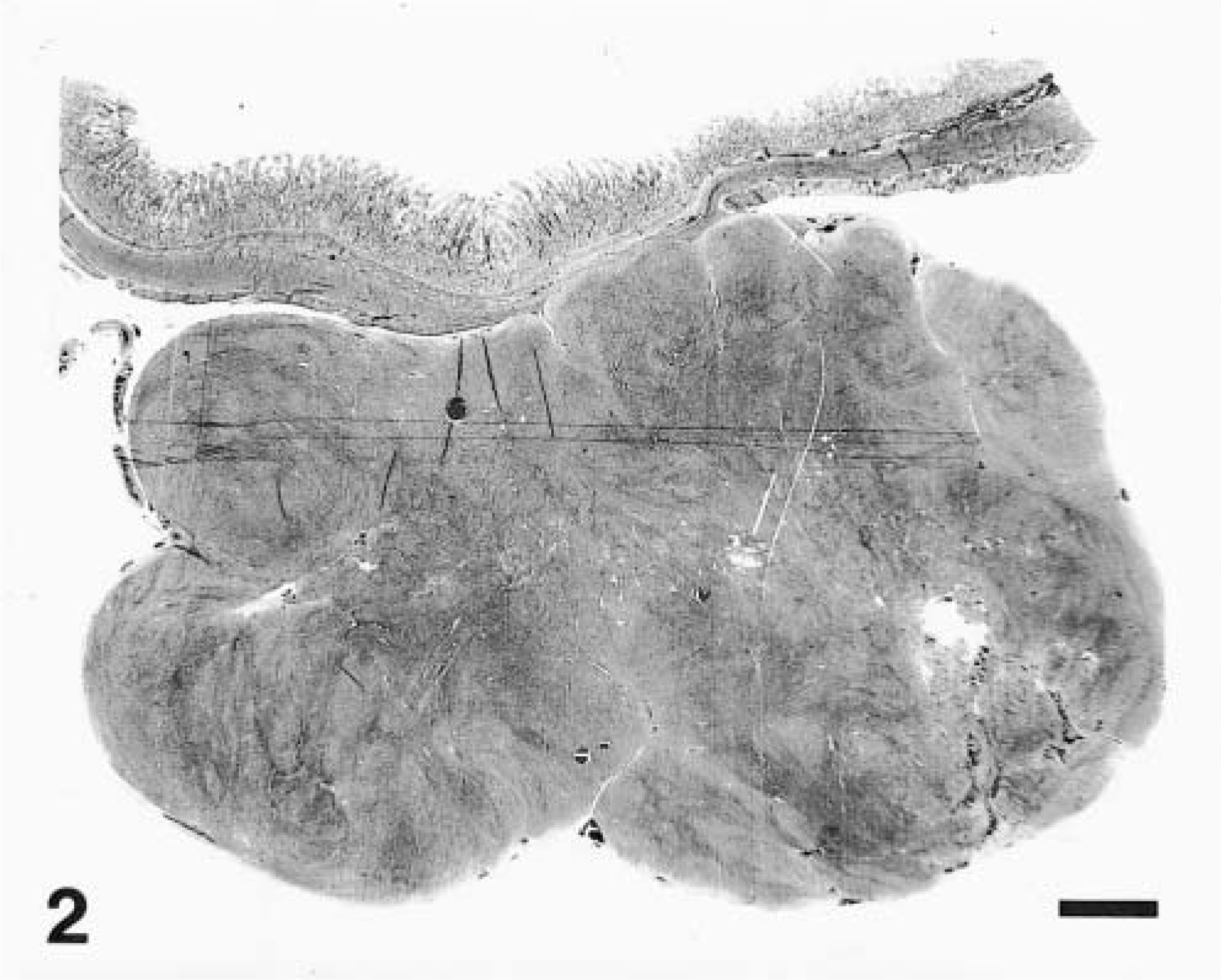
Duodenum; dog No. 9. A low power micrograph of a GIST shows an exophytic tumor bulging outward from tunica muscularis, with overlying intact mucosa. HE. Bar = 0.2 cm.
All tumors were highly cellular with an overall basophilic appearance on HE stain. The majority was composed of spindle cells often arranged in streams and short interlacing bundles (Fig. 3). Infiltration between the normal smooth muscle fibers was often seen (Fig. 4). A few tumors had a more epithelioid morphology, sometimes with perinuclear cytoplasmic vacuolization (Fig. 5). Nine tumors (43%) had foci of coagulation necrosis (Fig. 6). There was only minimal nuclear pleomorphism. Mitotic activity varied from 1 to 19 per 10 HPF; seven tumors (33%) had no mitotic activity. The latter finding may partly result from delay of fixation and autolysis. The histologic features were essentially similar at all tumor sites, although all metastases were not available for evaluation. Moderate numbers of eosinophils were observed in one tumor (dog No. 12).
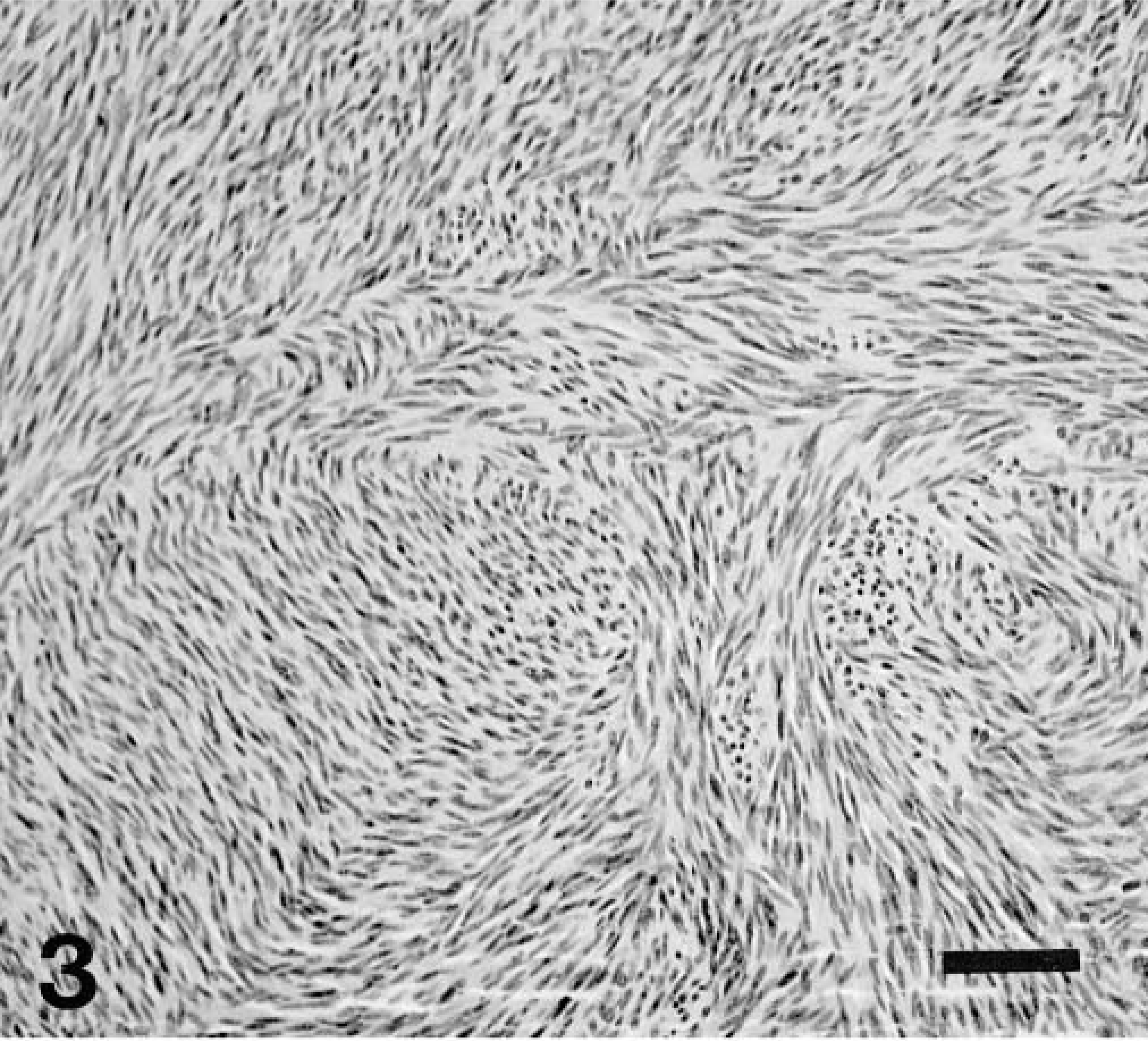
Jejunum, GIST; dog No. 11. Highly cellular spindle cell neoplasm composed of fusiform cells arranged in streams and bundles. HE. Bar = 50 μm.

Cecum, GIST; dog No. 1. Spindle cell neoplasm that intermingles with surrounding smooth muscle fibers. HE. Bar = 33.3 μm.
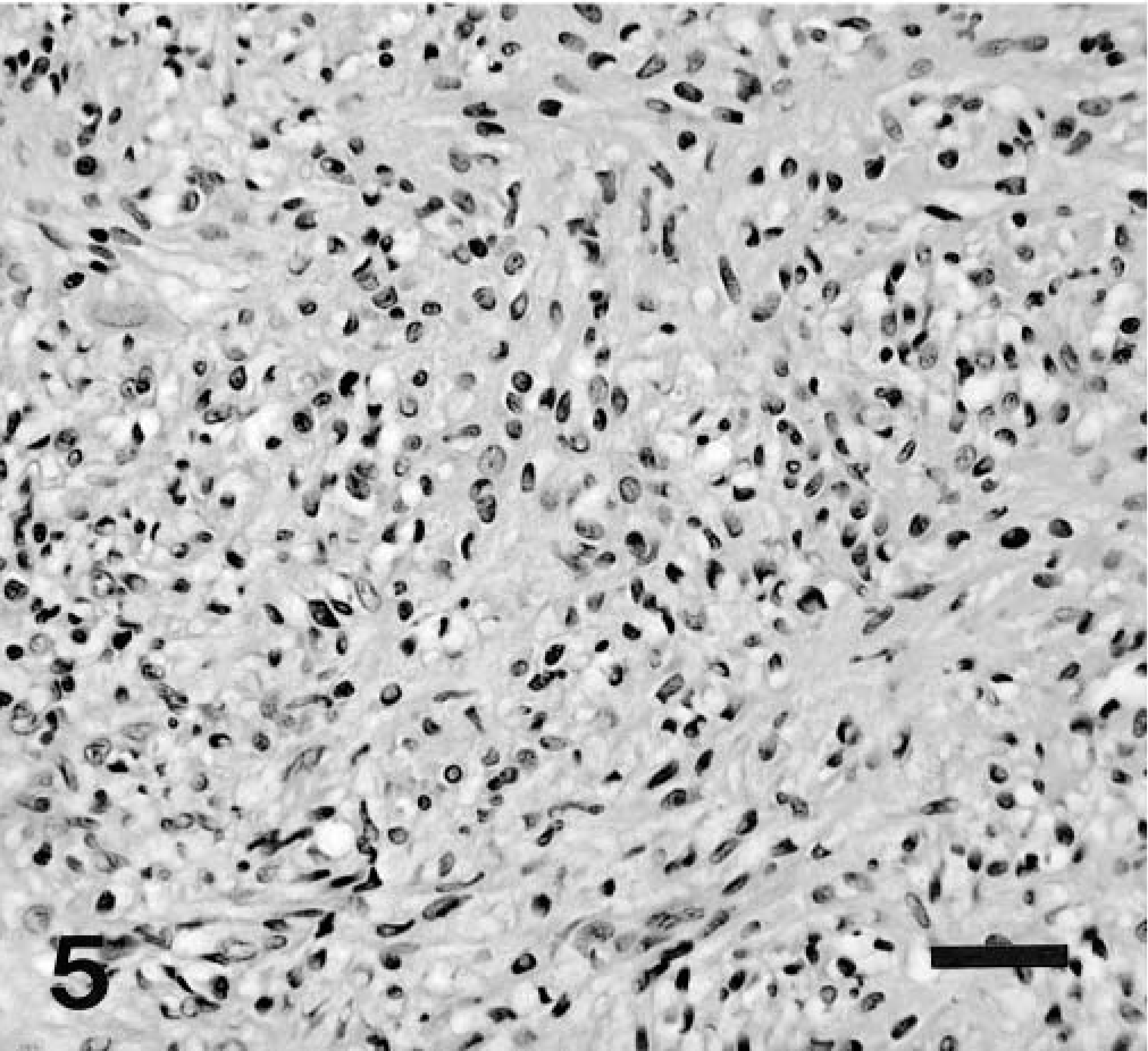
Colon, GIST; dog No. 20. Predominantly polygonal, epithelioid cells with prominent perinuclear vacuoles. HE. Bar = 50 μm.
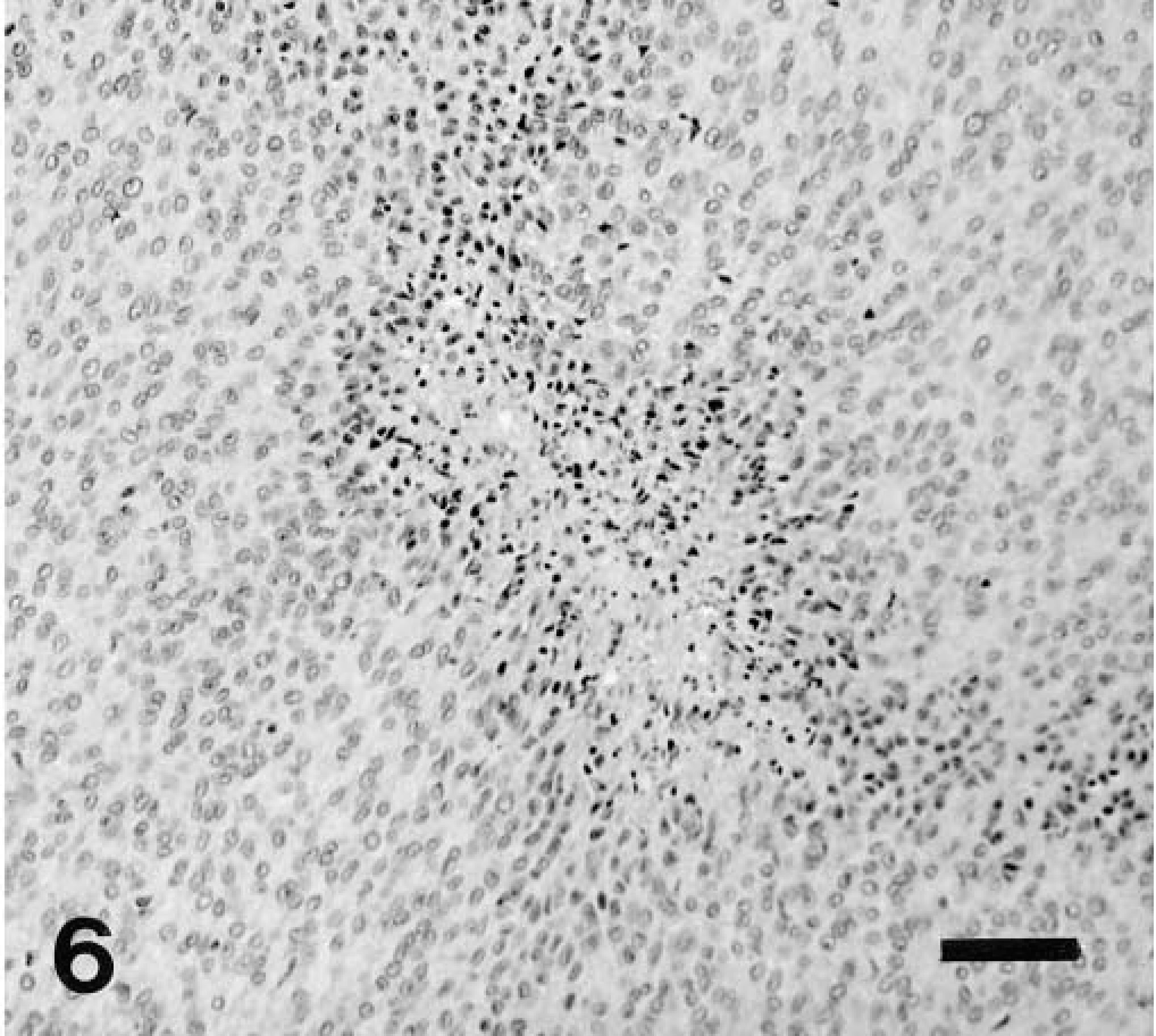
Colon, GIST; dog No. 18. Highly cellular neoplasm composed of polygonal cells with foci of coagulative necrosis. HE. Bar = 50 μm.
Eleven tumors were positive for KIT, most of them showing global positivity in all tumor cells (Fig. 7A, B). In several cases, KIT positive internal controls, such as mast cells, were not evident, which was attributed to inadequate antigenic preservation. Seven tumors were positive for α-smooth muscle actin (SMA; Fig. 8). All were negative for desmin and S-100 protein.
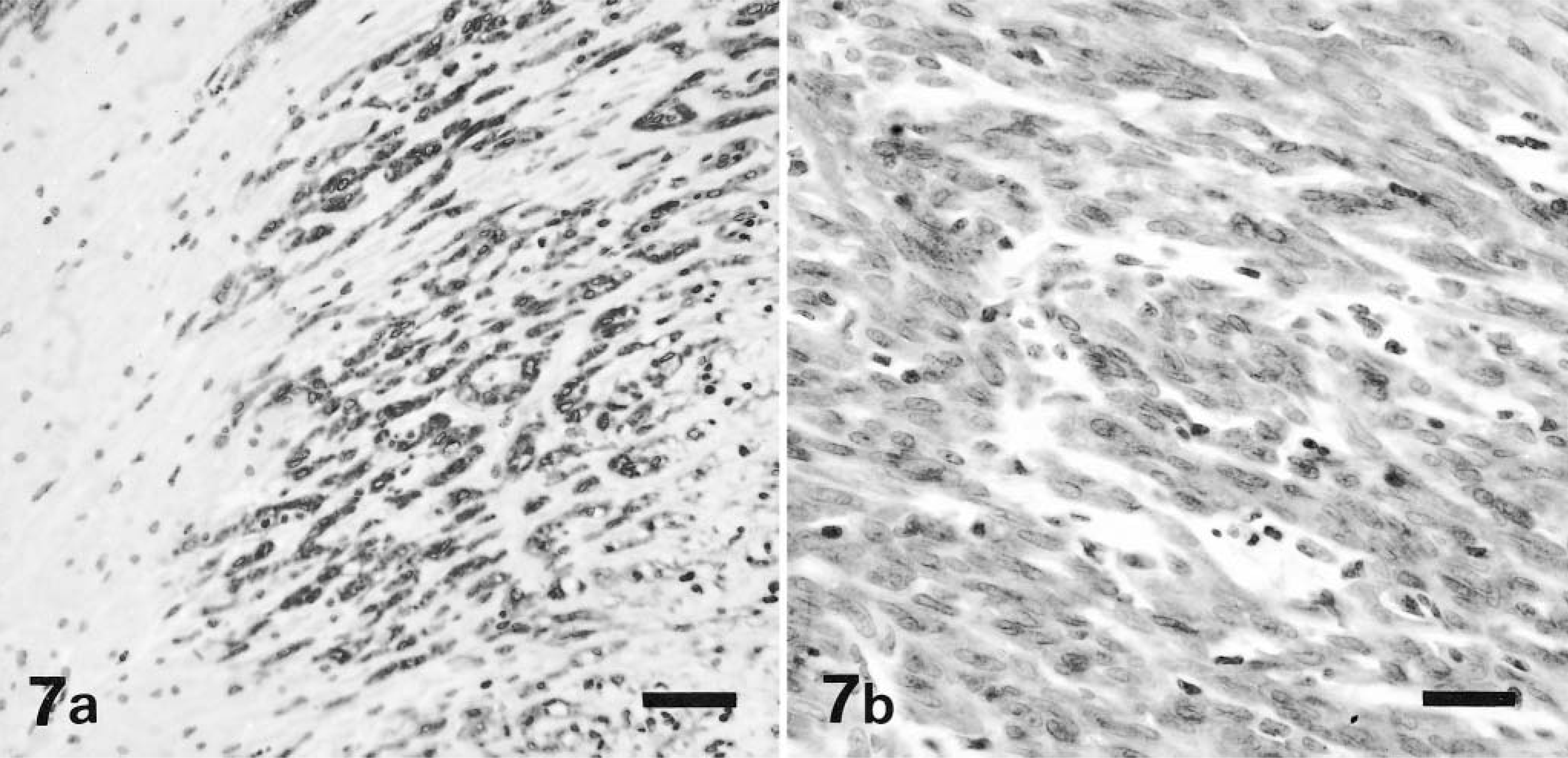
Cecum, GIST; dog No. 1. Majority of neoplastic cells demonstrate strong cytoplasmic staining for KIT (CD 117) antigen. Immunohistochemical stain for KIT, also stained with hematoxylin. Bar = 50 μm. Fig. 7b. Cecum, GIST; dog No. 19. Majority of neoplastic cells demonstrate strong cytoplasmic staining for KIT (CD 117) antigen. Immunohistochemical stain for KIT, also stained with hematoxylin. Bar = 25 μm.
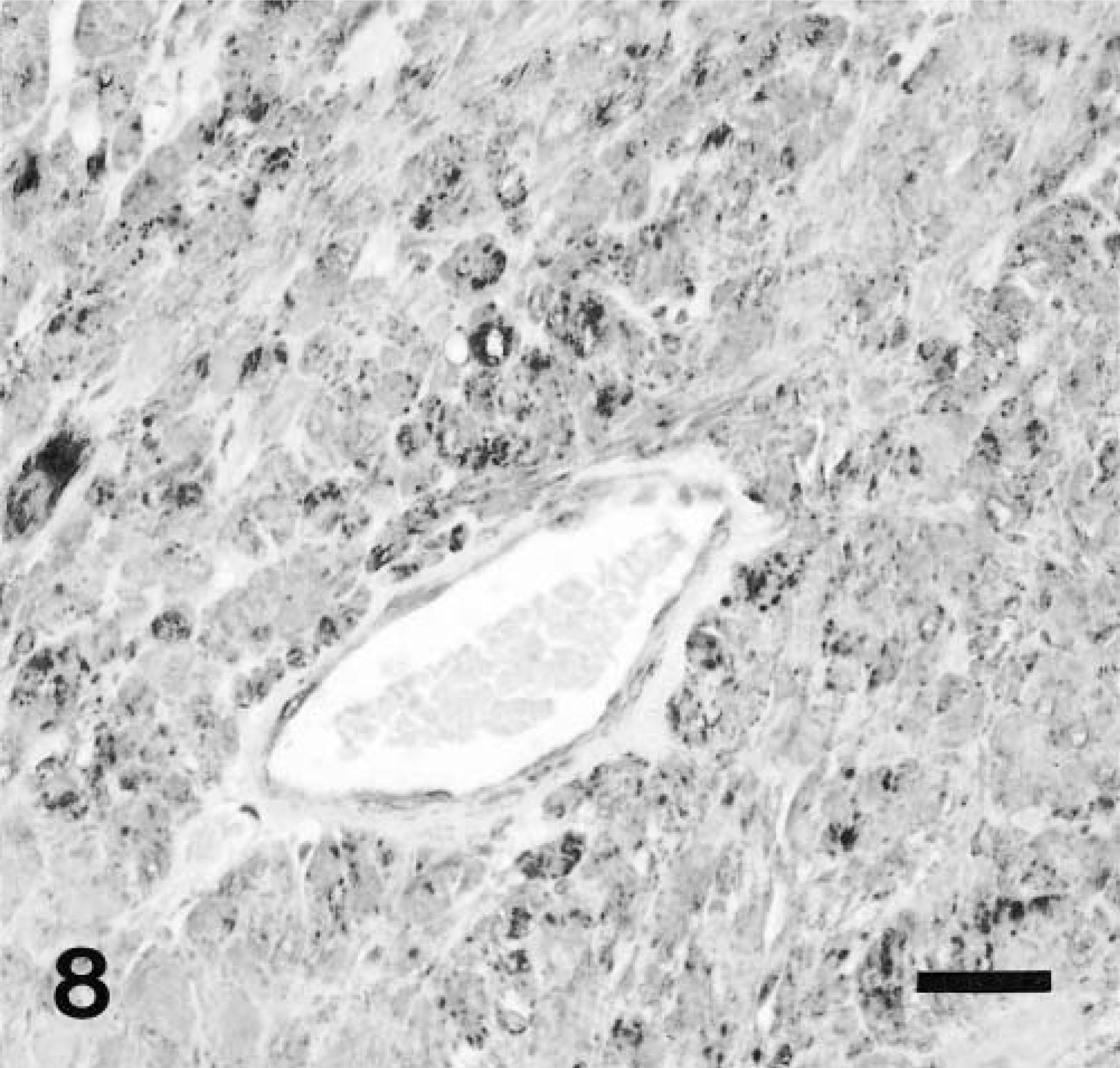
Colon, GIST; dog No. 18. Majority of neoplastic cells demonstrate strong cytoplasmic staining for SMA antigen. The vascular pericytes are also positive. Immunohistochemical stain for SMA, also stained with hematoxylin. Bar = 25 μm.
Electron microscopy examination of two tumors (dog Nos. 10 and 11) revealed no evidence of smooth muscle or neural differentiation.
Kit mutation status
The seminested PCR strategy was successfully used to amplify the KIT juxtamembrane domain in four GISTs. Mutations were identified in two cases (Fig. 9). Dog No. 12 showed 6-bp deletions eliminating Try556 (h 557)-Lys557 (h 558) coexisting with duplication of Gln555 (h 556); in dog No. 18, T → C transition was found at codon 575, resulting in substitution of Pro for Leu575 (h 576).
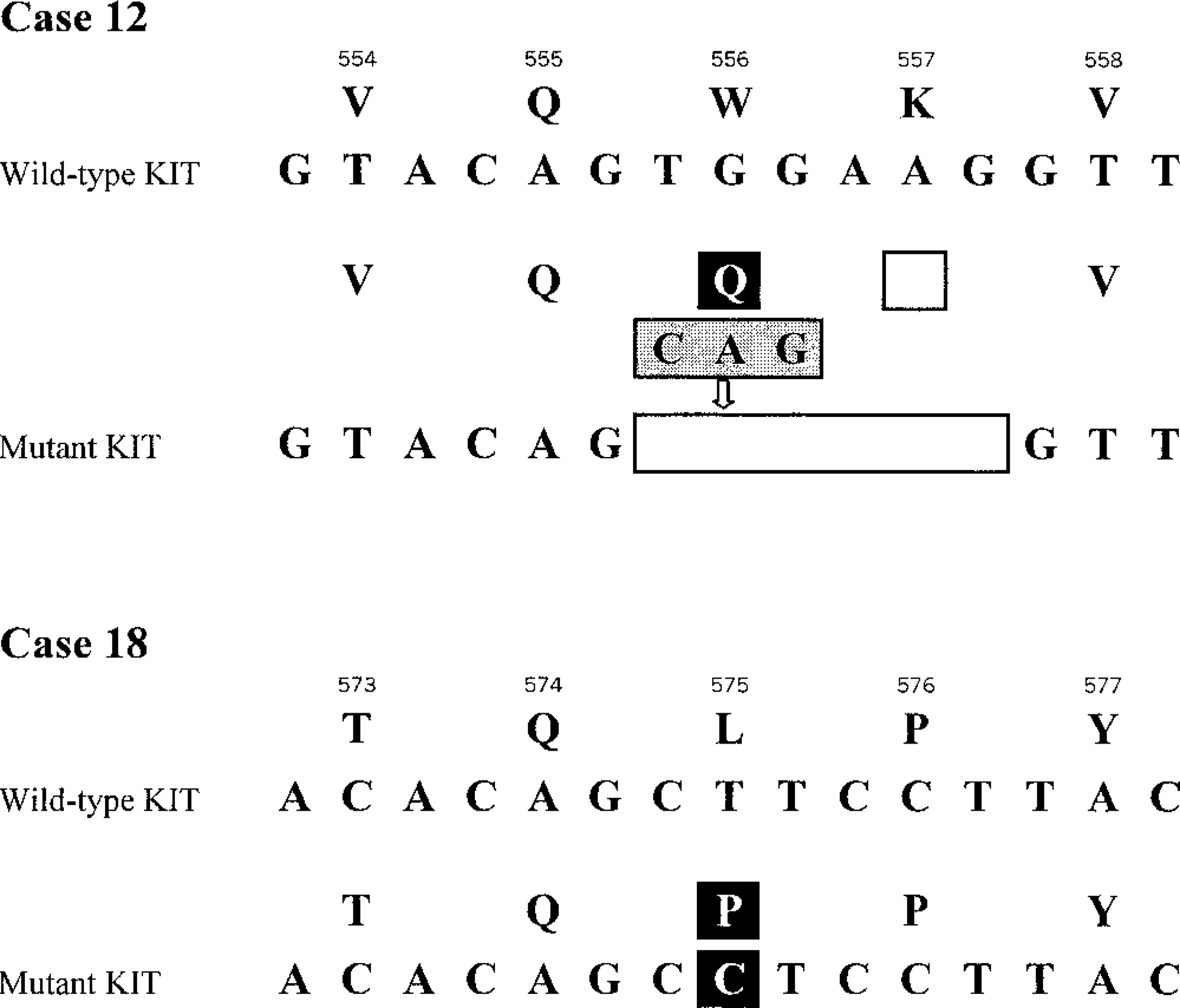
KIT juxtamembrane domain mutations identified in dog GISTs. Wild-type and mutant KIT sequences are aligned. Predicted amino acids and codon numbers are shown above. Black, gray, and clear boxes indicate missense mutations, insertion/duplication, and deletion, respectively.
LM
Clinical features
The signalment, clinical features, tumor histomorphomology, and immunohistochemistry analyses are summarized in Table 2. There were 23 males (82%) and five females ranging in age from 8 to 17 years, with an average age of 10.8 years (median = 11); the sex of one dog was unknown. Thirteen cases were German Shepherd Dogs, and one each of Airedale, Australian Shepherd, Belgian Malinois, Boston Terrier, Boxer, Bull Terrier, Chow Chow, Cocker Spaniel, Kerry Blue Terrier, mixed Terrier, Pomeranian, Poodle, small haired Terrier, West Highland Fox Terrier, and one of unknown breed. Nine tumors (29%) had associated clinical signs: vomiting in four dogs, palpable masses and diarrhea in two dogs, weight loss, radiographic signs, abdominal hemorrhage, gastric-dilatation volvulus, intussusception, anorexia, and rectal occlusion, each in one dog. The remaining 20 tumors were reported as incidental findings at necropsy.
Leiomyoma signalment, clinical signs, histologic characteristics, and immunohistochemistry results.
∗ FN/MN = Female/Male neutered.
† LM = leiomyoma; LMS = leiomyosarcoma.
‡ 0 = negative; 1+ = <25% positivity; 2+ = <50% positivity; 3+ = <75% positivity; 4+ = 100% positivity.
A great majority of the LMs (22/29, 76%) occurred in the stomach. Four tumors involved the esophagus, one of them at the esophagogastric junction. One LM each was located in the small bowel (ileum), colon, and rectum.
Pathology
The LMs were typically sharply circumscribed intramural tumors that were located beneath a normal mucosa in the tunica muscularis (Fig. 10). Although gross measurements of the tumor size were not systematically available, the median diameter measured from the slides was 15 mm (range 5–27 mm). All but five tumors measured at least 1 cm in greatest diameter. There was ulceration on the overlying mucosa in dog No. 17.
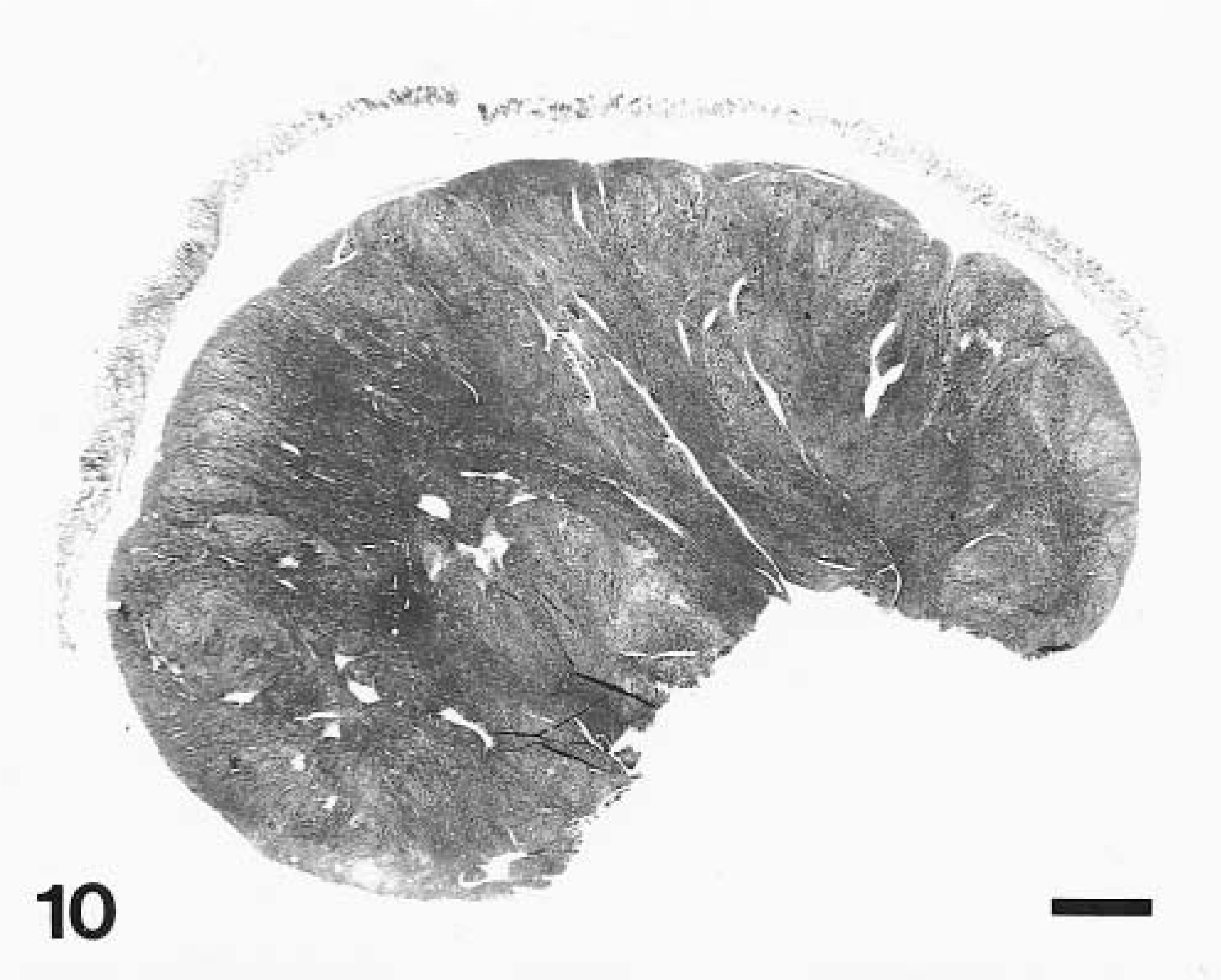
Stomach, LM; dog No. 1. Neoplasm diffusely expands the tunica muscularis with intact overlying mucosa. HE. Bar = 0.2 cm.
Histologically, the LMs were composed of well-differentiated smooth muscle cells with a strongly eosinophilic overall appearance, a consistent feature at all tumor locations. These tumors had low or moderate cellular density, and the nuclei were elongate and small (Fig. 11). Mitotic activity was negligible in all cases. But coagulation necrosis was present in eight tumors; calcification was present in three cases.

Stomach, LM; dog No. 14. Paucicellular spindle cell tumor composed of well-differentiated smooth muscle cells with fibrillary, often clumped cytoplasm and elongate nuclei with blunt ends. HE. Bar = 50 μm.
Immunohistochemically, the tumor cells were positive for α-smooth muscle actin (Fig. 12) in 28 of 29 cases (97%) and for desmin in 18 of 29 cases (62%). All LMs were negative for KIT.
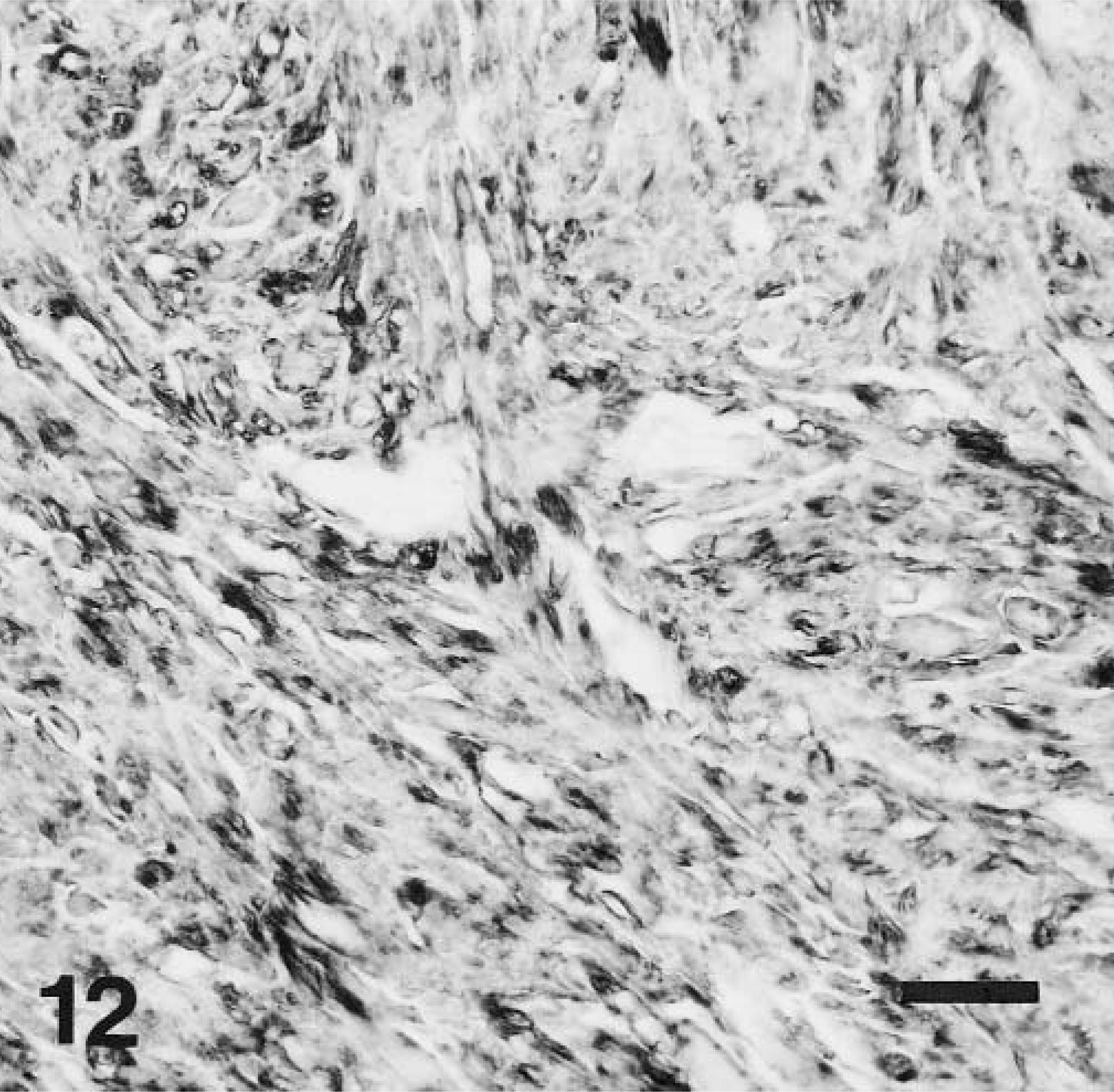
Stomach, LM; dog No. 12. Neoplastic cells are strongly positive for SMA antigen. Immunohistochemical stain for SMA, also stained with hematoxylin. Bar = 25 μm.
Electron microscopy revealed well-differentiated smooth muscle cells with abundant cytoplasmic bundles of actin-filaments in both cases that were studied.
DNA for molecular study was available in one LM. In dog No. 2, KIT was PCR amplified and sequenced. No mutation in KIT juxtamembrane domain was detected.
Discussion
In this study, we evaluated a series of 50 GI mesenchymal tumors of the dog to improve the understanding of these tumors and their relationship with the corresponding tumors in humans. GI neoplasia in the dog has a reported incidence of 12–120 cases per 10,000 cases, in which LMs and LMS comprise 10–30% of all intestinal tumors. 29 The present series documents the occurrence of both typical GISTs and LMs in dogs and demonstrates numerous distinct differences between these tumor groups.
Because of the referral patterns of the institute, nearly half of the GISTs in this study occurred in MWDs, which are, in general, either Belgian Malinois or German Shepherd Dogs. These breeds are more likely to experience pathologic conditions typically associated with larger breed dogs. A recent study for cause of death in the national canine population revealed 71% of deaths were due to euthanasia subcategorized as geriatric dog or poor quality of life. 5 Many geriatric MWDs are euthanatized because of age-related musculoskeletal abnormalities. 26 In addition, because of their unique lifestyle, MWDs are also less likely to suffer from vehicular trauma, poisonings, and other ailments commonly associated with a free-roaming or house pet lifestyle. Because of these multiple confounding factors, conclusive breed-specific tumor incidence in this study cannot be established.
Compared with the clinicopathologic features of human GISTs, the canine GISTs also predominantly occurred in older subjects. 23 The GISTs in dogs occurred most commonly in the colon and small intestine (together 76%), whereas only 19% of them were located in the stomach. The human GISTs, in contrast, are most common in the stomach and are very rare in the colon. 23 The canine GISTs appeared to have a clinical spectrum from apparently indolent tumors to those that behave as sarcomas. The propensity of GISTs to metastasize in the abdominal cavity and liver, typical of human malignant GIST, was also documented in this study.
No malignant tumors were composed of well-differentiated smooth muscle cells and reclassified as LMS in this study, indicating that such tumors must be rare in dogs. In earlier literature, canine LMS were reported to be most common in the jejunum and cecum. 37 The similar location patterns of the LMS in earlier literature to our GISTs and the fact that all LMSs were reclassified as GISTs in this study suggest that most canine GI LMSs probably represent GISTs. In humans, true LMSs are rare in the GI tract, and seem to have unique clinicopathologic features, such as a polypoid intraluminal presentation. 24,25
A majority of the GISTs diagnosed in this study were documented as positive for KIT (CD117). We believe that many tumors gave false negative results due to inoptimal antigen preservation, and as with for human GISTs, would expect most if not all be KIT-positive, which in fact is becoming a definitional feature of GISTs. 23 Co-expression of CD117 and smooth muscle actin, previously documented in 20–30% of human GISTs, was seen in 33% of GISTs in this study. Similar to their human counterparts, the GISTs in dogs were also consistently negative for desmin and S-100 protein. 23
The KIT gene (proto-oncogene c-kit) encodes for a transmembrane type III tyrosine kinase receptor whose ligand is the stem-cell factor. KIT is expressed in hematopoietic stem cells, mast cells, interstitial cells of Cajal, germ cells, melanocytes, and certain epithelial cells, 7,10,11,23,31 and has been described as a critical factor in the survival of stromal cells derived from canine bone marrow. 9 In addition to GISTs, KIT is expressed in canine mastocytoma 17,30 and has also been reported in certain tumors of the breast. 12
Mutations causing activation of KIT have been found in human mastocytosis, 3,19 acute myelogenous leukemia, 4 myeloproliferative disorders, 28 seminoma, 36 sinonasal lymphomas, 8 and GISTs. 7 In GISTs, the majority of mutations are clustered in the juxtamembrane domain of the gene. 7,11 Recently, similar type KIT juxtamembrane domain mutations have been reported in canine mastocytomas. 16,21 These mutations lead to the ligand-independent tyrosine kinase activity, autophosphorylation of the kinase domain of the KIT protein, and subsequently downstream activation of the KIT signaling pathways. 7,18,35 KIT mutations also induce malignant transformation in transfected cell lines. 7 The KIT juxtamembrane domain mutations identified in this study are similar to those found in human GISTs. Deletions of TGG AAG coding for Try557 and Lys558 are commonly seen KIT alterations in human GISTs. 23,33 T → C transition resulting in a substitution of Pro for Leu576 has also been reported in a few human GISTs. 14 Molecular alteration, duplication of Gln555 (h 556) followed by deletion of Try556 (h 557)-Lys557 (h 558) reported in this study has been found in one of the 300 analyzed human GISTs (J. Lasota, unpublished). Biological significance of these mutations is probably similar to that of comparable mutations found in human GISTs 7 and more recently in canine mastocytomas. 21 In two cases, no mutations were found in the juxtamembrane domain. Other KIT domains may be mutated in these tumors as well. Some human GISTs lacking mutation in the juxtamembrane domain show specific Ala502-Tyr503 duplication in exon 9 (extracellular domain) or A → G transition in exon 13 (kinase domain) resulting in substitution of Lys for Glu642. 15,20 Large insertions of duplicated codons have been found in a small subset of human GISTs 22,27 (J. Lasota, unpublished observations) and in canine mastocytomas. 16,17,21 PCR amplification of the mutated allele with a large insertion may be impossible from the severely degraded DNA. Finally, human GISTs without detectable mutation in KIT-coding sequences have also been reported. 23
Detection of KIT mutations similar to those seen in human GISTs strongly suggests that tumorigenesis of dog and human GISTs follows the same molecular pathways. Our study provides, for the first time, evidence that similar KIT mutations may be involved in the pathogenesis of GISTs in dogs and humans.
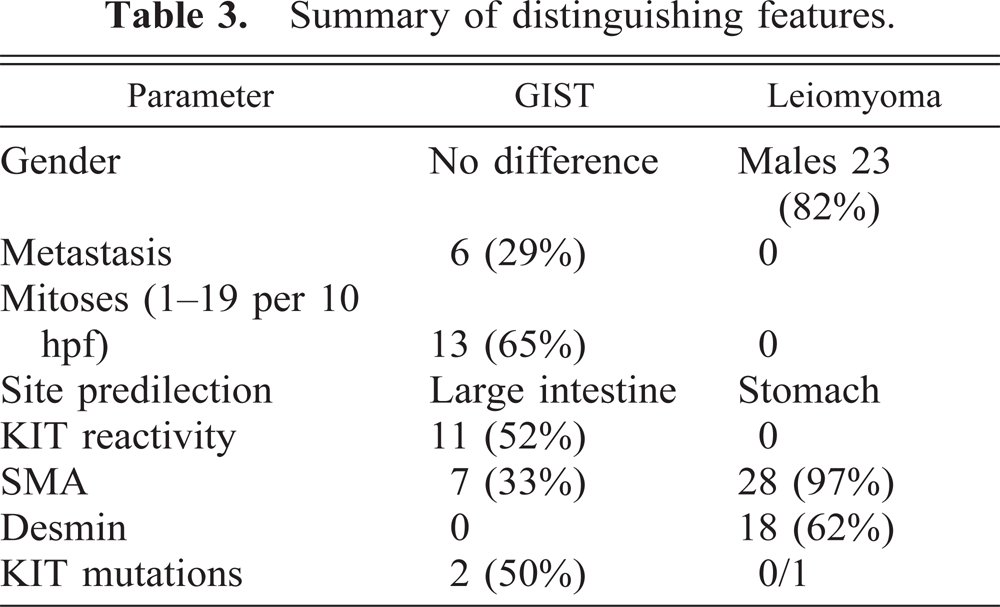
GI LMs that show phenotypic features of typical smooth muscle cells occur, by far, most commonly in the esophagus in humans; published series show median ages of 30–35 years. 22 In humans, these tumors are very rare in the stomach and intestines. In dogs, by contrast, the LMs occur most commonly in the stomach of geriatric dogs, with a male sex predilection. 29 The absence of KIT mutations in both canine and human 14 LMs adds further distinction in support of recognizing GISTs as a separate tumor category, although further study in the dog is required to confirm the one observation reported here. The key features of GISTs and leiomyomas are compared in Table 3.
Summary of distinguishing features.
In conclusion, our findings on canine GI mesenchymal tumors, and comparison with the data from those of humans, reveals similar dichotomy, yielding two groups: GISTs and LMs. Re-evaluation of GI mesenchymal neoplasms in the dog is necessary to thoroughly understand the incidence, natural history, biology, and etiology of GISTs and true smooth muscle tumors.
Footnotes
Acknowledgements
We thank J. Jenkins for electron microscopy, R.-A. V. Ferris and A. Morataya for photomicroscopy, and W. Inskeep, D. Dunn, and M. Sullivan for editorial comments. D. Frost is a lieutenant colonel in the US Army. The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army or the Department of Defense.
