Abstract
Gastrointestinal stromal tumors (GISTs) have been rarely reported in guinea pigs. We aimed to characterize the clinical and pathological features of GISTs in 4 guinea pigs and investigate the presence of mutations in exon 11 of the KIT proto-oncogene receptor tyrosine kinase (
Mesenchymal tumors, including gastrointestinal stromal tumors (GISTs), leiomyomas, leiomyosarcomas, malignant schwannomas, and benign schwannomas, can originate in the gastrointestinal tract. GISTs develop in the submucosa and tunica muscularis, originating from the interstitial cells of Cajal, which are the pacemakers of peristaltic action and express the tyrosine kinase receptor KIT. Upon binding to KIT, the stem cell factor (ligand) activates tyrosine kinase and induces cell differentiation and proliferation. 15
It can be difficult to determine if GISTs are benign or malignant. In humans, they are classified as having a high or low risk of recurrence based on tumor size and mitotic count.7,18 Histologically, GISTs include spindle cells, epithelioid, and mixed types.7,18 The spindle-cell type resembles other mesenchymal tumors, whereas the epithelioid-type resembles epithelial tumors. Immunohistochemistry helps reach a definitive diagnosis, with immunohistochemical positivity for KIT supporting the diagnosis of GISTs.8,22 Numerous studies have reported on the use of immunolabeling in humans, dogs, and mice for GIST diagnosis. Few studies have reported its use in other animals, such as chimpanzees, horses, pigs, cats, and guinea pigs.6,8,12,17,20-27,31 Some reports of GISTs in horses and monkeys have not been confirmed by immunohistochemistry.1,4,11 The anatomic location of GISTs varies according to the species, with the stomach and small intestine and the cecum and small intestine being the most common locations in humans and dogs, respectively.6,8,22
Some human GISTs do not express KIT antigens.
Materials and Methods
Cases
The study was based on the retrospective selection of GISTs in 59 necropsy and 74 biopsy samples from 133 guinea pigs (
Histological Analysis
All specimens were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin wax. The 4-µm-thick sections were stained with hematoxylin and eosin. The mitotic count was obtained by counting the number of mitotic figures in 50 consecutive microscopic high power fields (HPFs), which covered an area of 7.95 mm2, as prescribed by the Miettinen Classification of human GISTs. 18 We performed labeled-polymer immunohistochemistry using N-Histofine MAX PO (M) (Nichirei Biosciences, Tokyo, Japan), with anti-KIT (1:200; mouse monoclonal antibody; DAKO, Glostrup, Denmark), anti-DOG1 (prediluted; rabbit monoclonal antibody; Nichirei), anti-smooth muscle actin (SMA; 1:400; mouse monoclonal antibody; DAKO), and anti-SOX10 (1:500; mouse monoclonal antibody; Santa Cruz Biotechnology, Dallas, TX, USA) as the primary antibodies. 19 Normal small intestinal tissues were used as a positive control (interstitial cells of Cajal for KIT and DOG1, tunica muscularis for SMA, and peripheral nerves for SOX10). Mouse immunoglobulin, of the same isotype and diluted to similar concentrations, was used as the negative control for the primary antibody. As a positive control, normal guinea pig interstitial cells of Cajal of the small intestine displayed co-positivity with fluorescent staining using mirror sections for DOG1 and KIT (Supplemental Figures S1–S2). Immunolabeling intensity was subjectively classified as negative (–), weak (+), moderate (++), or strong (+++), based on the degree of cytoplasmic and membranous labeling in neoplastic cells. We obtained the percentage of immunolabeled cells from 5 random HPFs (0.795 mm2) per section.
Analysis of Kit Exon 11 Mutations
Following deparaffinization, tumor tissues were scraped from each glass slide, and the genomic DNA was extracted using a High Pure PCR Template Preparation Kit (Roche Diagnostics GmbH, Mannheim, Germany). We used the following primers: forward primer (5′-TTGTTCTCTCCCCTGAGTGC-3′) and reverse primer (5′-CGGAAAGCCCCTGTTTCATA-3′). The genomic sequence of guinea pig
Results
The study cases included 2 males and females. Two were 6 years old, 1 was 7 years old, and the other was of an unknown age. All cases underwent necropsy (Table 1). The clinical signs varied as follows. Case 1 was emaciated after 6 months of diarrhea and died of an unknown cause. Case 2 had edema with pleural and pericardial effusions for 1 year and died of circulatory failure. Case 3 had decreased appetite, diarrhea, hypoglycemia, and tonic convulsions. A radiopaque mass in its abdomen (Fig. 1) was initially suspected to be an insulinoma because glucose administration reduced the clinical signs. Following 2 months of emaciation, this case (case 3) died of myocardial degeneration and renal failure, and the mass was histologically identified as a GIST, not an insulinoma. Case 4 had dyspnea, ascites, and emaciation for 2 months and died of circulatory failure owing to myocardial degeneration.
Clinical and Pathologic Findings and Mutation Status of Gastrointestinal Stromal Tumors in 4 Guinea Pigs.

Abbreviation: KIT, KIT proto-oncogene receptor tyrosine kinase.
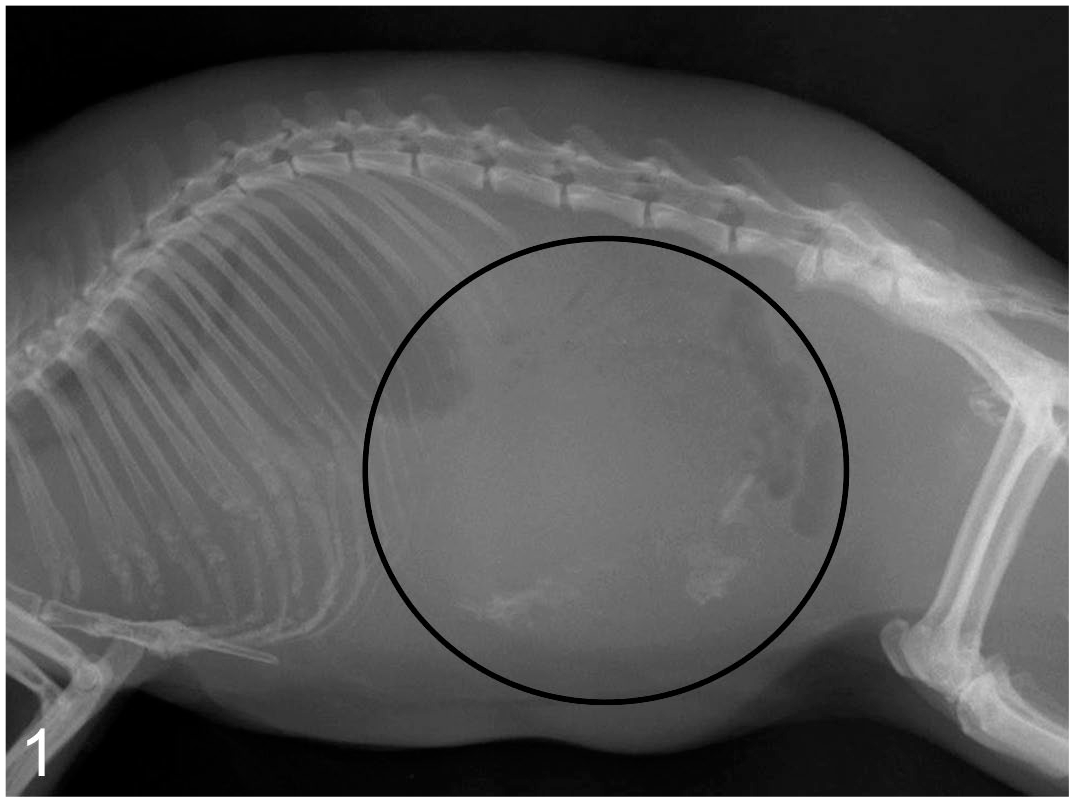
Gastrointestinal stromal tumor, stomach, guinea pig, Case 3. The radiograph shows an abdominal mass (black circle) with scattered calcification.
Cases 1–3 had large masses (4–6 cm diameter) (Figs. 2 and 3) that abluminally expanded the gastric wall at the cardia (case 1) or pylorus (cases 2 and 3). None had luminal obstruction, despite 1 (case 2) with metastases in the mesentery of the duodenum and cecum. Case 4 had a large mass (1.5 × 2.0 × 1.5 cm) abluminally extending from the small intestinal wall, without luminal obstruction. In case 1, an additional 5 × 4 × 5 cm tumor was in the mesentery near the cecum, which was diagnosed as sarcoma not otherwise specified. In addition to the tumors of the gastrointestinal tract, there was a 3 × 3 × 3 mm insulinoma in the pancreas and a thyroid follicular adenoma (6 × 5 × 5 mm) in cases 2 and 4, respectively.
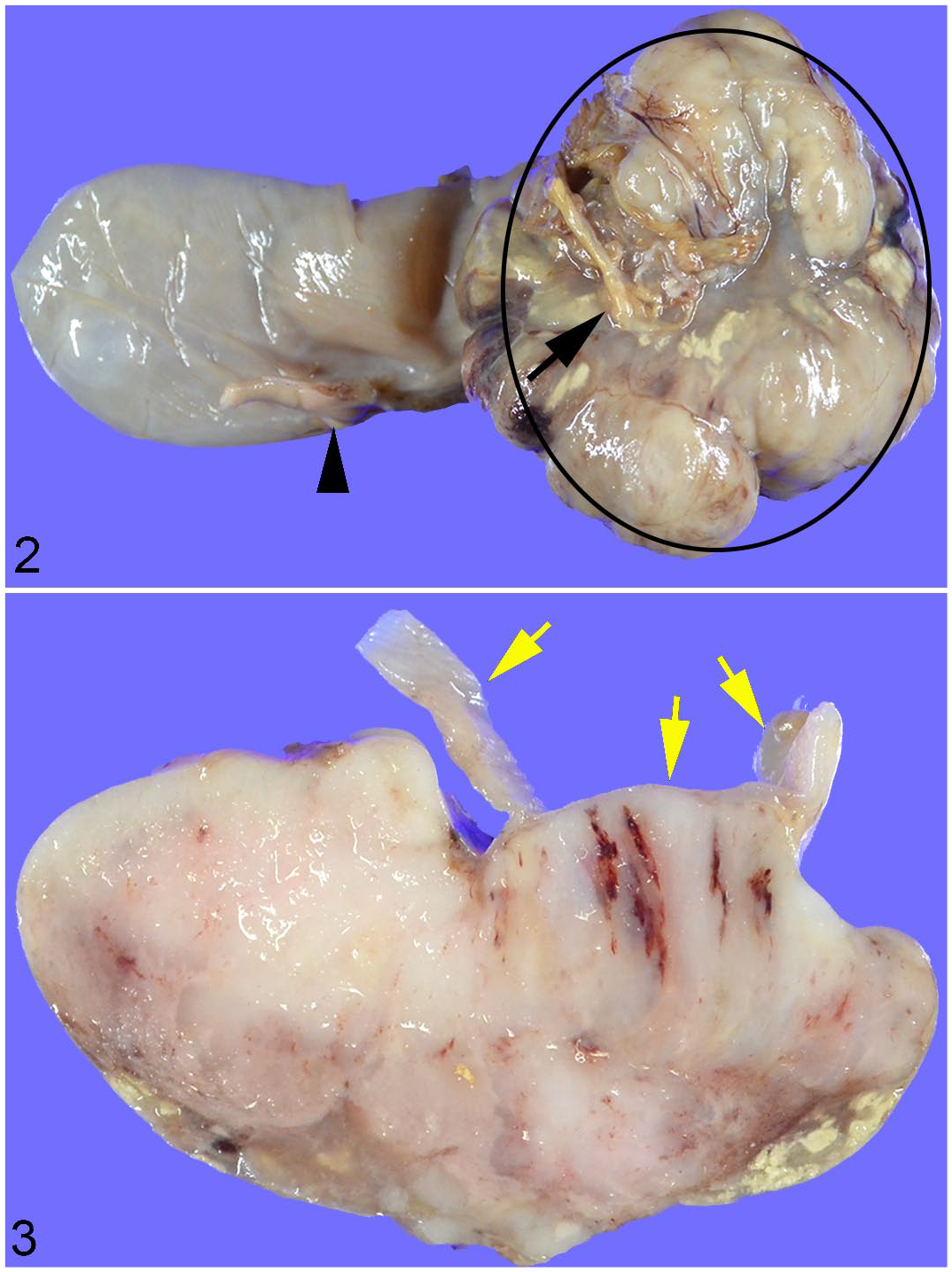
Gastrointestinal stromal tumor, stomach, guinea pig, Case 3.
Histologically, 2 of the 3 gastric cases (cases 2 and 3) had extraluminal masses extending from the tunica muscularis to the serosa of the pylorus. One case (case 1) had a well-demarcated extraluminal mass at the cardia; however, the gastric layers involved could not be precisely determined owing to partial resection. In case 2, a small (3 × 5 × 3 mm) mass was present under the serosa, adjacent to the well-demarcated and partially invasive, grossly identified large (3 × 3 × 3 cm) mass; this case also had multiple small (1–5 mm diameter) masses in the duodenal mesentery and a submucosal 2 × 1 × 1 cm mass in the cecum. The mass in case 3 was well-demarcated and solitary. In case 4, a well-demarcated mass extended from the lamina propria to the serosa of the small intestine, forming an expansile and extraluminal growth.
Tumors in all 4 cases were composed of densely arranged spindle cells in bundles intermixed with small amounts of collagenous stroma (Figs. 4–7). Two pyloric tumors had nuclear palisading with multifocal necrosis and hemorrhage. In all cases, neoplastic cells displayed a moderate amount of eosinophilic cytoplasm, frequently oriented into bipolar extensions, in addition to round to oval nuclei with small nucleoli, mild anisocytosis, and anisokaryosis (Fig. 8). The mitotic count ranged from 0/50 HPF (7.95 mm2; case 3) to 5/50 HPF (7.95 mm2; cases 1, 2, and 4). Immunohistochemically, the 3 gastric tumors were diffusely to multifocally positive for c-KIT, DOG1, and SMA but negative for SOX10. The small intestinal tumor was diffusely positive for c-KIT and DOG1 but negative for SMA and SOX10 (Figs. 9–11, Table 2). The plasma membrane had a stronger labeling intensity than the cytoplasm for both KIT and DOG1; the labeling intensity was stronger for DOG1 than for KIT (Table 2).
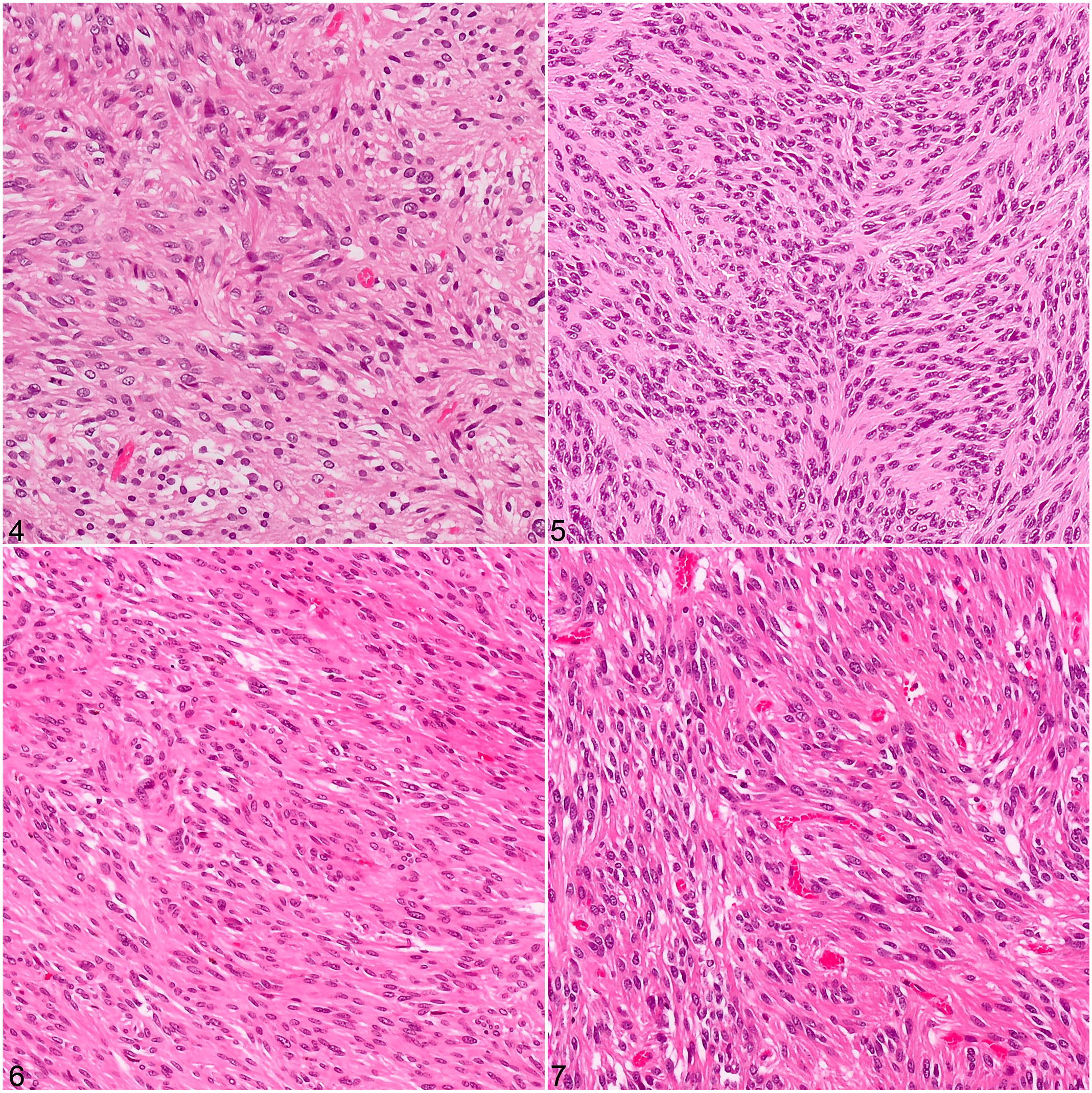
Gastrointestinal stromal tumors, guinea pigs. Neoplastic spindle-shaped tumor cells are arranged in bundles and intermixed with small amounts of collagenous stroma. Hematoxylin and eosin.
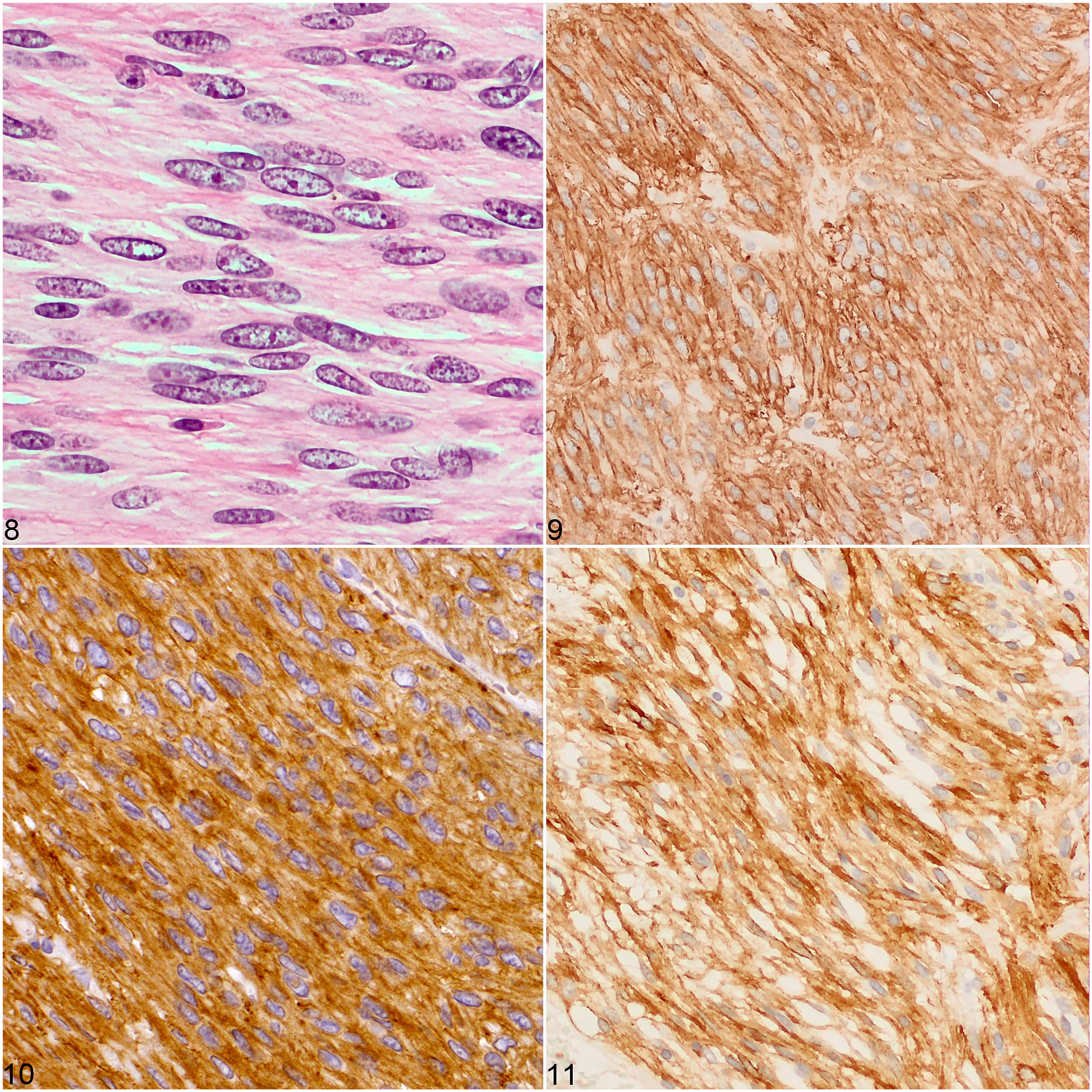
Gastrointestinal stromal tumors, guinea pigs.
Immunohistochemical Findings in Gastrointestinal Stromal Tumors in 4 Guinea Pigs.
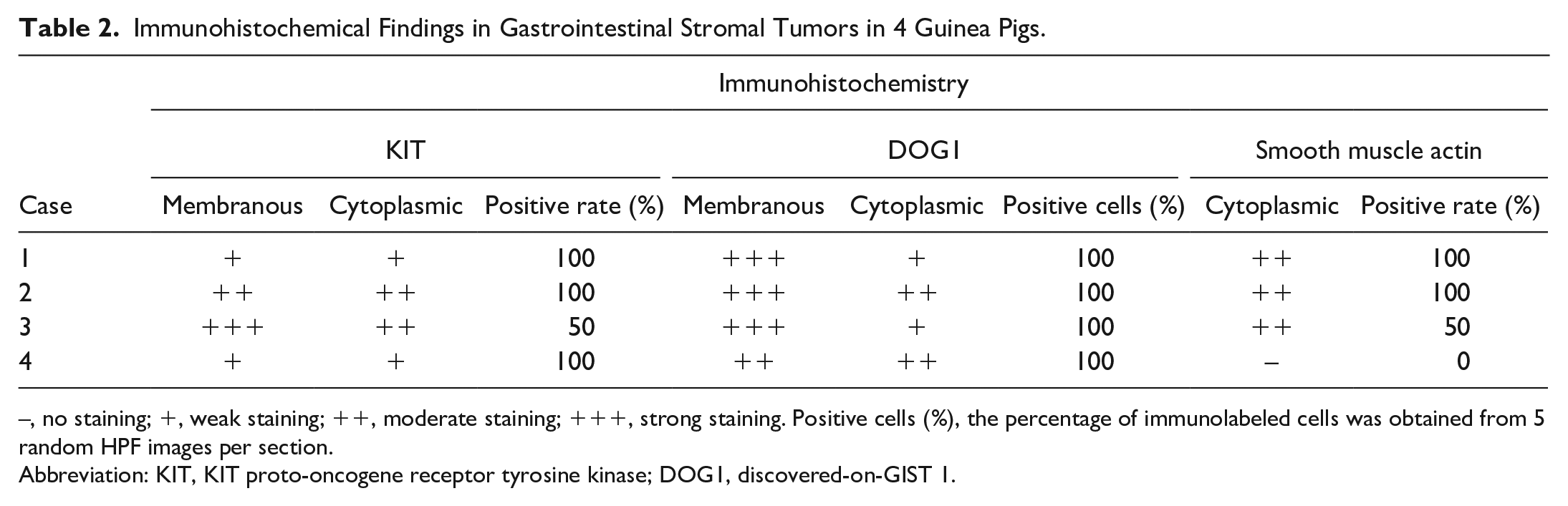
–, no staining; +, weak staining; ++, moderate staining; +++, strong staining. Positive cells (%), the percentage of immunolabeled cells was obtained from 5 random HPF images per section.
Abbreviation: KIT, KIT proto-oncogene receptor tyrosine kinase; DOG1, discovered-on-GIST 1.
Sequencing chromatograms displayed normal and mutated sequences in all cases. We aligned the sequence of each PCR product with the wild-type
Discussion
This study characterized the clinical and histopathological features and
GISTs in humans and other animals are frequently positive for mesenchymal markers other than KIT or DOG1.7,8,16,22 Moreover, 25% and 35% of the gastric and small intestinal cases in humans, respectively, and 33% of canine cases are positive for SMA.8,18 All 3 gastric cases in this study were positive for SMA, whereas the small intestinal case was negative. This suggests that gastric GISTs in guinea pigs have evidence of smooth muscle or myofibroblastic differentiation. SMA-positive GISTs should be differentiated from leiomyosarcomas and myofibroblastic tumors, as the morphological characteristics of the spindle cell type of GISTs, leiomyosarcomas, and myofibroblastic tumors are similar on hematoxylin and eosin staining. GISTs of this study were differentiated based on KIT- and DOG1-immunolabeling.
In a previous report of a small intestinal GIST in a guinea pig, 17 the cells were highly atypical with a mitotic count of approximately 19/10 HPFs. They were positive for KIT and SMA immunohistochemically. Unlike the previous report, cellular atypia was mild, and mitoses were rarely observed in this study. 17 Furthermore, the absence of SMA expression also distinguished the present case from the aforementioned case.
Mutations in exon 11 of the
The relative age, anatomic location, growth pattern, and metastatic potential of these cases were similar to those of human GISTs. 22 The mean age of occurrence is relatively high: 65 and 11 years in humans and dogs, respectively. 22 Guinea pigs have a lifespan of 4–8 years; therefore, they were at an advanced age (6–7 years) at presentation. In humans, the most common anatomic location of GISTs is the stomach, followed by the small and large intestines. 22 Contrarily, the large intestine (particularly the cecum) is the most common location in dogs, followed by the small intestine and stomach.6,8 In this study, GISTs were most common in the stomach, similar to those in humans. The growth patterns of GISTs can be extraluminal (abluminal expansion from the gastrointestinal wall), intramural (expansion within the gastrointestinal wall), or intraluminal (protrusion into the gastrointestinal lumen), with the extraluminal pattern being the most common in humans (79%) and dogs (70%).6,8,22 All of these guinea pig tumors had an extraluminal growth pattern. GIST metastasis occurs in 23% of humans 22 and 29%–45% of dogs6,4. One guinea pig GIST in this study metastasized.
The risk assessment for metastasis in humans is performed using the tumor size and mitotic count.7,18 Large tumors and those with mitotic counts >5/50 HPFs or >10/50 HPFs (where 50 HPFs = 7.95 mm2) are considered high risk in humans.7,18 The tumor in case 2, which had invasive growth and metastasis, had low mitotic activity (5/50 HPFs, 7.95 mm2), whereas the remaining 3 cases without tumor invasion or metastasis had no or low mitotic activity (0–5/50 HPFs, 7.95 mm2). The previously reported case of small intestinal GIST in a guinea pig had a high mitotic count (19/10 HPFs, 2.37 mm2); however, metastasis was absent. 17 Therefore, the metastatic risk of GISTs in guinea pigs may be better correlated with invasive growth, as the mitotic count was not predictive of the biological behavior in these cases.
Clinical signs are often absent in humans and dogs until the tumor becomes sufficiently large to compromise organ function and metastasize to other organs.6,8,22 In this study, gastrointestinal signs included diarrhea in 2 cases (cases 1 and 3). Case 3 had a large tumor, decreased feeding, hypoglycemia, and tonic convulsions. An islet cell adenoma (insulinoma) was initially suspected because the intravenous administration of glucose reduced the signs, and islet cell adenoma is a sporadic tumor in guinea pigs. 30 However, the mass was identified as a GIST rather than an insulinoma on necropsy. The gastric GIST may have led to functional compromise and decreased food intake, resulting in hypoglycemia. Furthermore, hypoglycemia can be a paraneoplastic phenomenon. Its possible mechanisms include excessive consumption of glucose by the tumor and decreased hepatic gluconeogenesis secondary to neoplasia or IGF-2 secretion by the tumor. 3 The small size of the pancrease compared with the enlarged gastrointestinal lumen in guinea pigs makes it difficult to identify pancreatic lesions using ultrasonography. In addition, there is no established method to measure blood insulin levels in guinea pigs, thus necessitating a differential diagnosis.
In conclusion, GISTs are uncommon in guinea pigs. Immunohistological and
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221087630 – Supplemental material for Gastrointestinal stromal tumors with Kit gene mutation in 4 guinea pigs (Cavia porcellus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221087630 for Gastrointestinal stromal tumors with Kit gene mutation in 4 guinea pigs (Cavia porcellus) by Kengo Ueda, Masamine Takanosu, Yumiko Kagawa, Akiko Ueda, Naoko Ano, Kohji Nomura and Kiyokazu Ozaki in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
