Abstract
Ten cecal tumors were identified during the postmortem examination of seven horse carcasses at slaughter (one horse had three tumors). The multinodular and hemorrhagic tumors ranged from 1 to 10 cm in diameter and consisted of spindle cells arranged in thin, interconnected trabeculae that were often separated by sinuses filled with mucinous fluid, erythrocytes, and siderophages. Spindle cells of all tumors were immuno-positive for vimentin, neuron-specific enolase, and c-kit protein but lacked reactivity with antibodies to glial fibrillary acidic protein, S100 protein, and desmin. In one tumor, spindle cells diffusely bound antibodies to synaptophysin. Most tumors contained focal reactivity to smooth muscle actin antibodies; one tumor reacted diffusely. Ultrastructurally, tumor cells were connected by desmosome-like structures and exhibited extended cell processes; some contained dense core neurosecretory granules. These equine stromal tumors appeared to share some characteristics with human gastrointestinal stromal tumors.
Lipomas, lymphomas, leiomyosarcomas, and adenocarcinomas are among the most common tumors of equine intestines, but primary tumors of the equine cecum are rarely described. 3 The most commonly reported equine cecal tumors are adenocarcinomas, myxosarcomas, and gastrointestinal stromal tumors (GISTs). 2–4 However, there are also individual reports of a leiomyoma, hemangiosarcoma, and papillary adenoma. 1 The term gastrointestinal stromal tumor has been used to describe a specific group of mesenchymal tumors of the gastrointestinal tract of humans 10 and similar tumors in the gastrointestinal tract of dogs, macaques, and horses. 2 8 In this study, we describe the morphologic and immunohistochemical features of a group of equine cecal stromal tumors of similar derivation.
Tumors were detected during routine postmortem inspection at slaughter. Formalin-fixed tissues and some frozen specimens were submitted to the US Department of Agriculture, Food Safety and Inspection Service, Eastern Laboratory, for examination. These distinctive tumors were detected in the ceca of seven horses during the course of postmortem examination of the visceral organs of approximately 15,000 horse carcasses. In this postmortem examination, visceral organs, including small intestines, colon, and ceca, were observed. Because of the nature of this examination, enteric lesions were most visible in the cecum and colon. When tumors were detected, a more detailed gross examination of the intestines and mesenteric lymph nodes was performed. A single jejunal tumor similar to the described cecal tumors was also detected during the examination of these carcasses.
Grossly, tumors were discrete, subserosal to intramural, multinodular masses that protruded from the serosal surface of the cecal wall (Fig. 1). There was never ulceration of the adjacent cecal mucosa. Masses usually affected the distal (apical) cecum (Table 1) and ranged in size from 1 to 10 cm in diameter (average 6 cm). Cut surfaces of tumors were tan to red and sectioned tumors often oozed viscous fluid. Most tumors were solitary, but one horse's cecum contained three distinct tumors, one of which was only 1 cm in diameter. This small tumor was diffusely tan without gross evidence of hemorrhage (Fig. 1, inset). Metastases were not detected by gross examination.
Location, size, and character of equine cecal tumors.
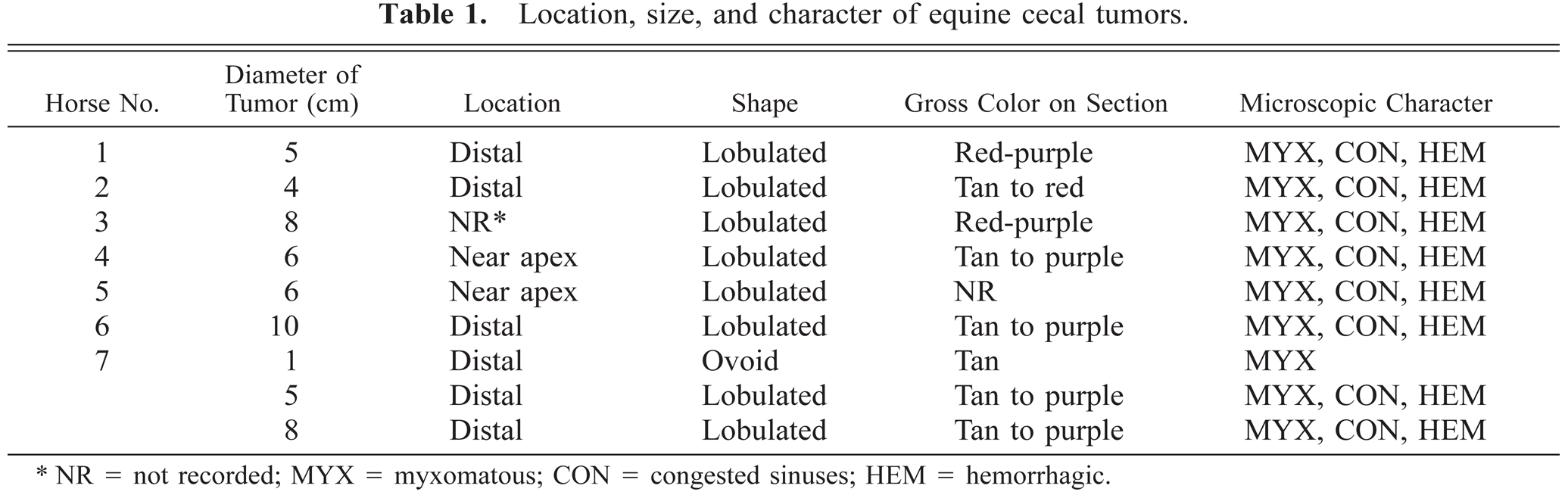
NR = not recorded; MYX = myxomatous; CON = congested sinuses; HEM = hemorrhagic.
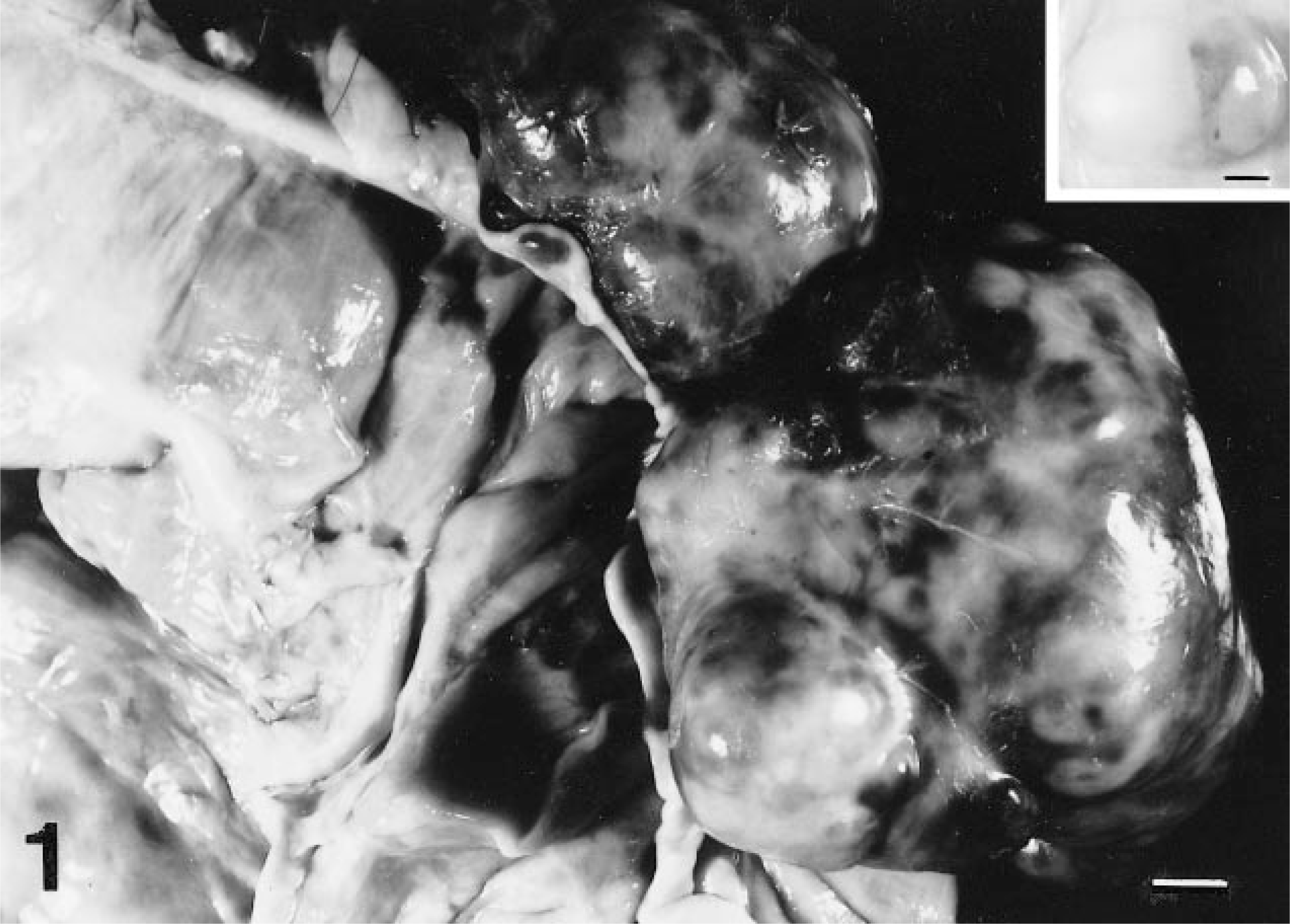
Cecal tumors; horse 7. Larger tumors that are multilobulated and hemorrhagic protrude from the serosal surfaces of the distal cecal wall. Bar = 1 cm. Inset: The smallest tumor lacks evidence of hemorrhage and appears to originate within the muscularis. Bar = 2 mm.
All tissues were fixed in 10% neutral buffered formalin (total fixation times of 48–96 hours), routinely processed, embedded in paraffin wax, sectioned at 4 µm, stained with hematoxylin and eosin (HE), and examined by light microscopy. Some replicate sections were stained with Alcian blue (pH 2.5).
The microscopic appearance of all tumors was similar, but the extent of hemorrhage and the amount of mucinous fluid present varied. Tumors appeared encapsulated and were subdivided by coarse fibrovascular septa, some of which were partially mineralized. Tumors appeared to be growing by expansion rather than invasion, and there was no mucosal ulceration. The peripheral fibrous capsules were covered by muscularis or compressed serosal layers. These capsules often contained areas of hemorrhage or aggregates of siderophages.
Tumors consisted of spindle cells that were most often arranged in 1–10 cell-thick, interconnected, and branching trabeculae (Fig. 2). These trabeculae were separated by sinuses filled with clusters of erythrocytes and siderophages or accumulations of mucinous (stained by Alcian blue) to proteinic fluid that contained scattered erythrocytes (Fig. 3). In some areas, spindle cells were more compactly arranged in storiform patterns. Spindle cells contained abundant amounts of vacuolated, eosinophilic cytoplasm with indistinct cell boundaries. Nuclei were relatively uniform, elongated oval with rounded poles and contained evenly dispersed, finely granular heterochromatin and 1–3 small, basophilic nucleoli. The mitotic rate was markedly low, and individual cell necrosis was common.
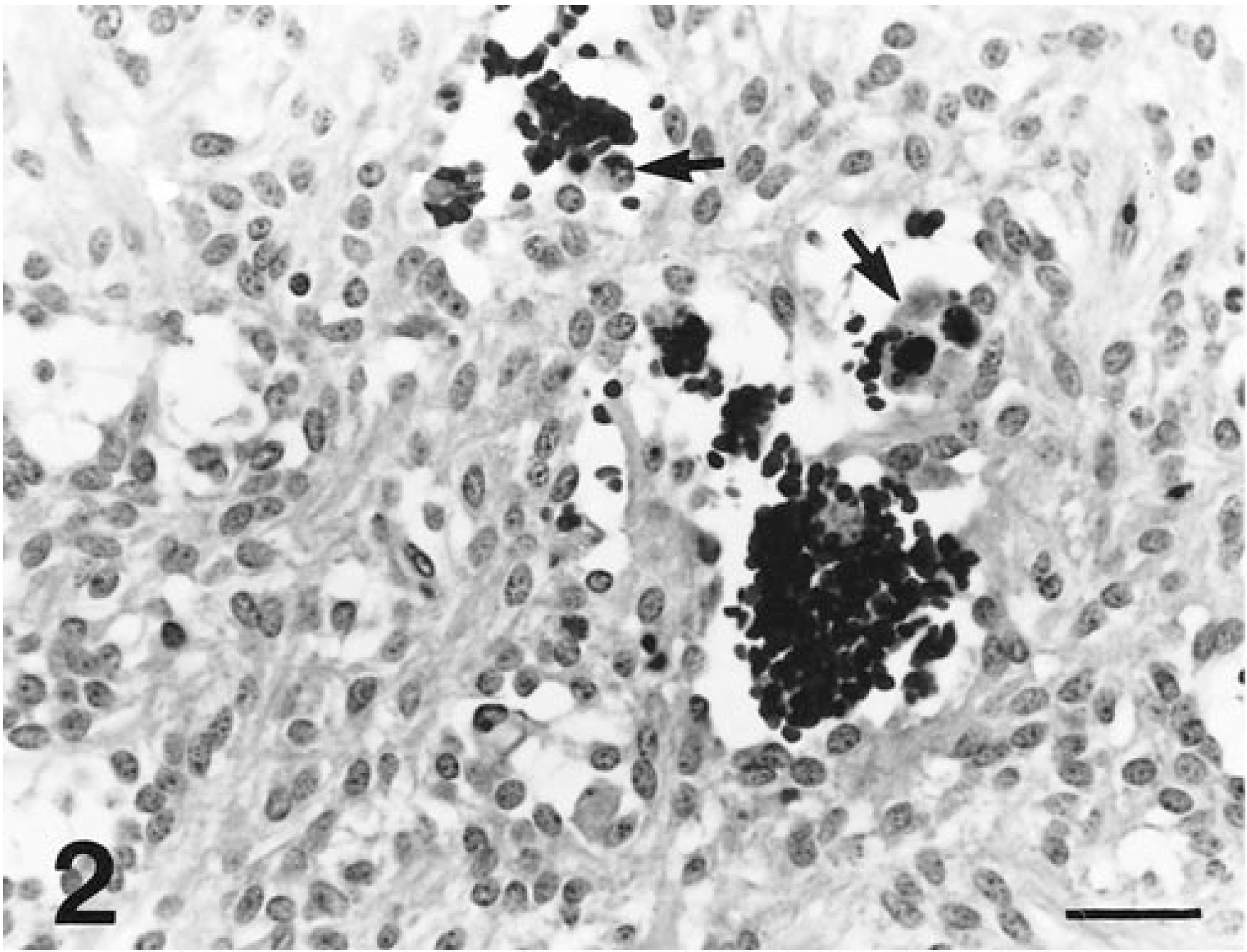
Cecal tumor; horse 7. Sinuses are congested with erythrocytes and contain siderophages (arrows). Tumor cell nuclei are relatively uniform. HE. Bar = 30 µm.
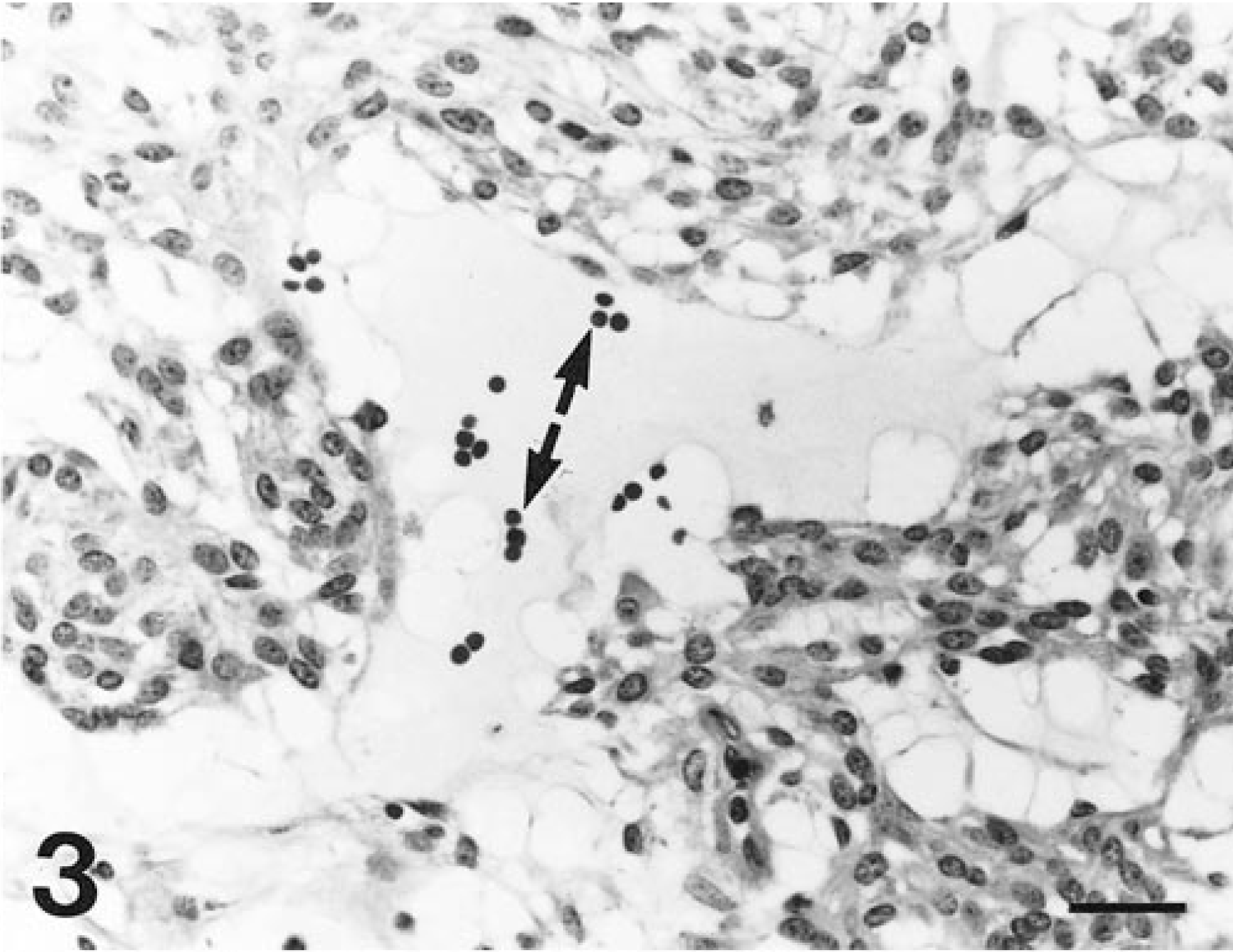
Cecal tumor; horse 6. Spaces between tumor cell trabeculae in some areas are widely dilated by mucinous fluid that contains erythrocytes (arrows). HE. Bar = 30 µm.
Replicate sections were used for immunohistochemical staining by a streptavidin-biotin-alkaline phosphatase method (BioGenex Supersensitive Kit, San Ramon, CA). Sections of tumors were incubated with antibodies to the alpha and beta subunits of S-100 protein (S100), vimentin (VIM), neuron-specific enolase (NSE), desmin (DES), glial fibrillary acidic protein (GFAP), alpha smooth muscle actin (SMA), synaptophysin (SYN), and c-kit protein (KIT). VIM was obtained from DAKO Corporation, Carpinteria, California; other antibodies were from BioGenex Laboratories, San Ramon, California. Sections used for some incubations were pretreated with a citrated (pH 6) buffer (Antigen Retrieval Citra solution, BioGenex) by heating in a microwave oven (boil 6 minutes, cool 30 minutes). Positive controls included equine peripheral nerve, brain, bone marrow, and cecum, and negative controls lacking specific antibodies were incubated in parallel.
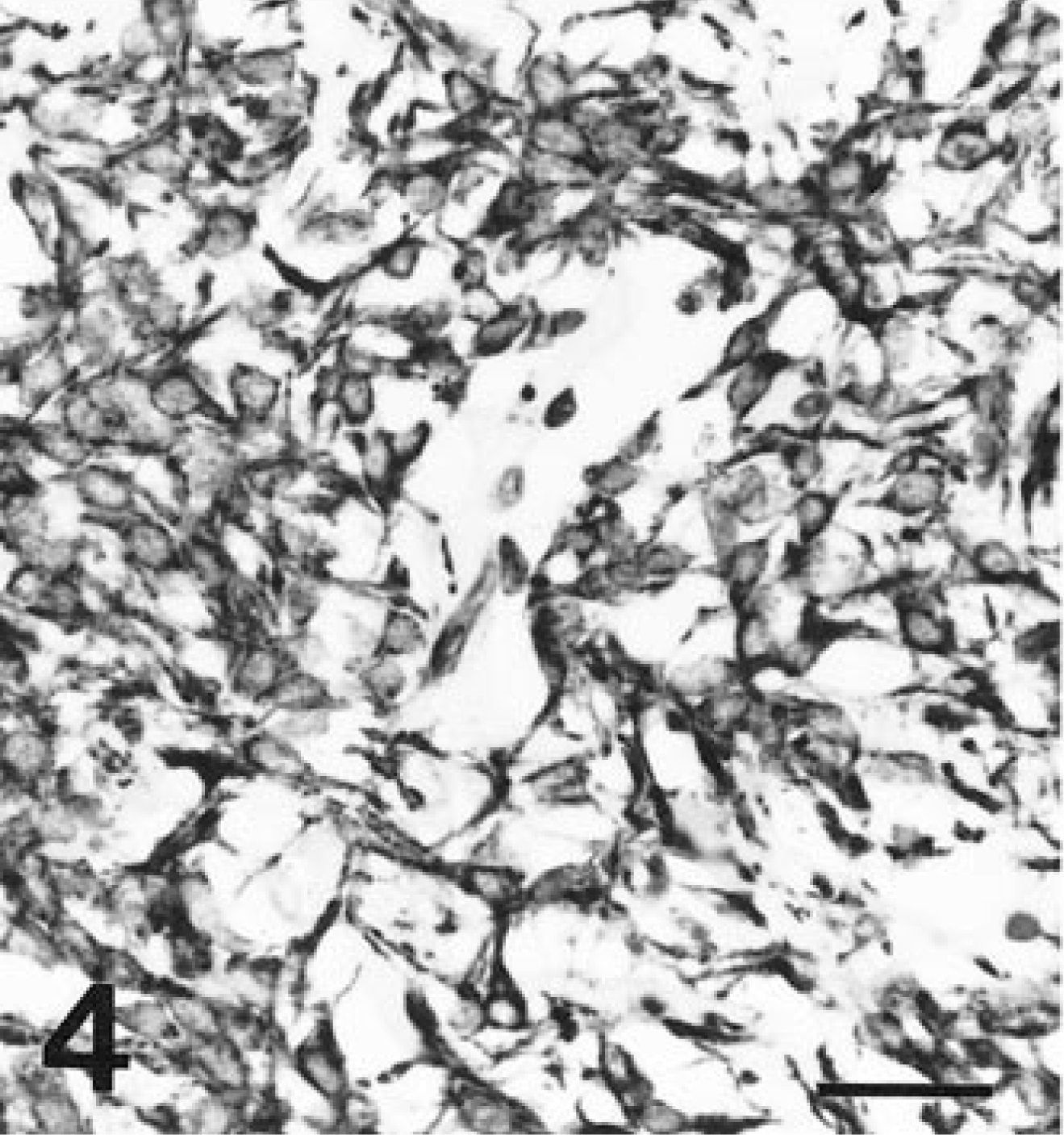
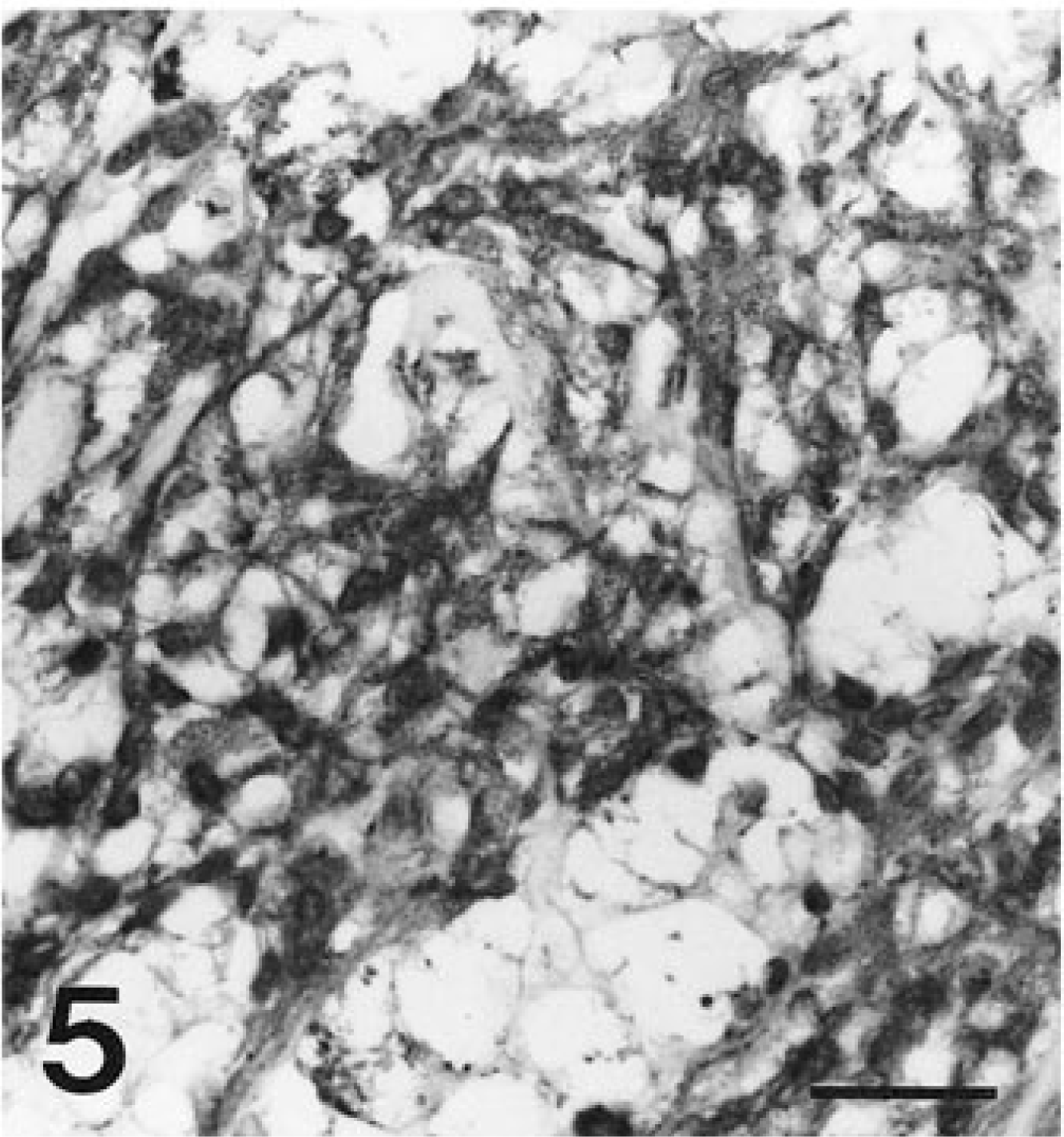
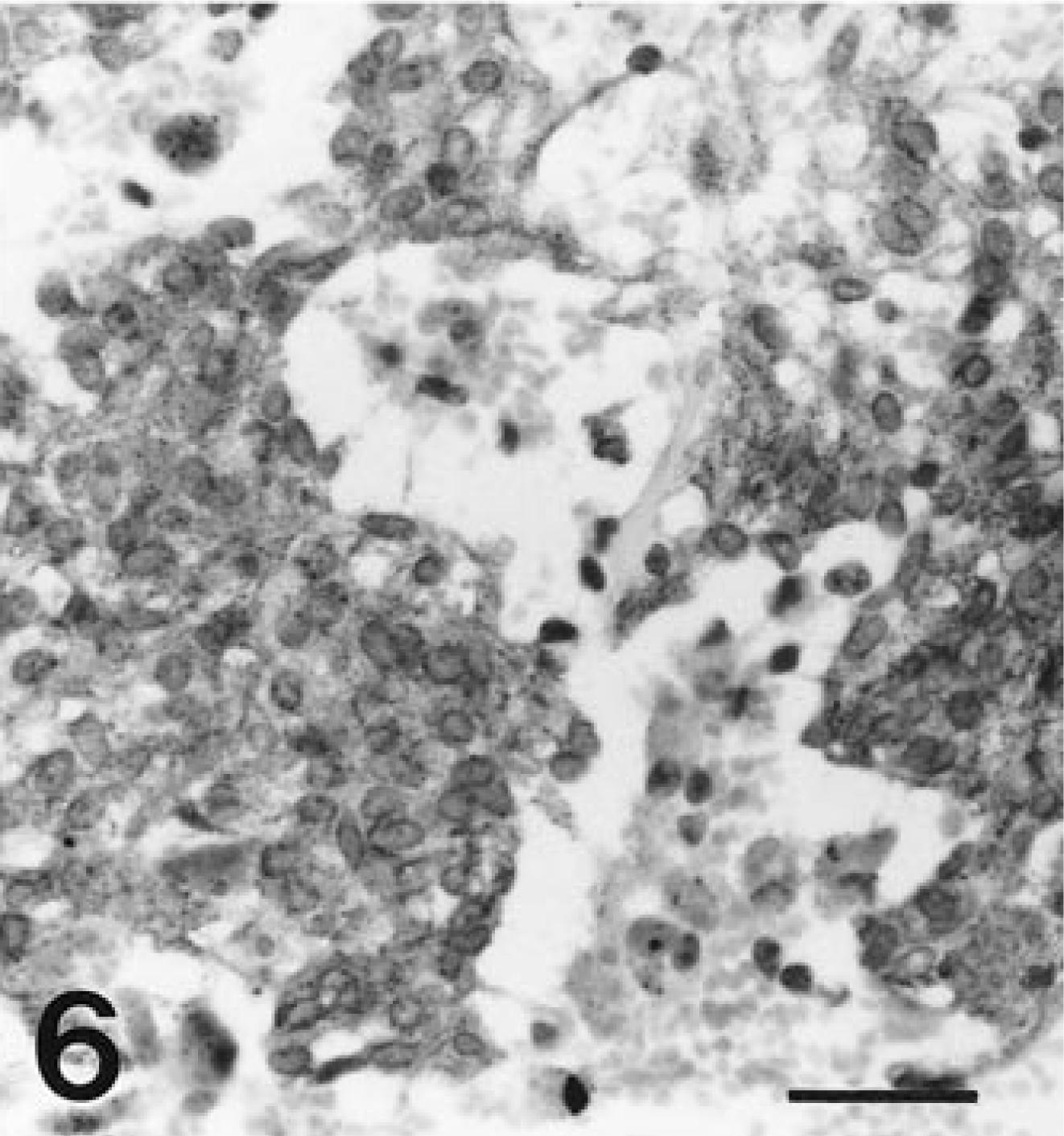
Spindle cells in all tumors exhibited diffuse, strong affinity for antibodies to VIM (Fig. 4) and NSE (Fig. 5). Spindle cells from all tumors had diffuse granular cytoplasmic staining after reaction with antibodies to KIT (Fig. 6). Most tumors had only limited, focal staining after application of antibodies to SMA, but the tumor from horse 5 had mild diffuse staining. The tumor from horse 4 had a diffuse reaction to SYN. No reaction was identified with antibodies to DES, S100, or GFAP in any tumor (Table 2).
Immunohistochemical staining of tumor cells in seven equine cecal gastrointestinal stromal tumors.∗
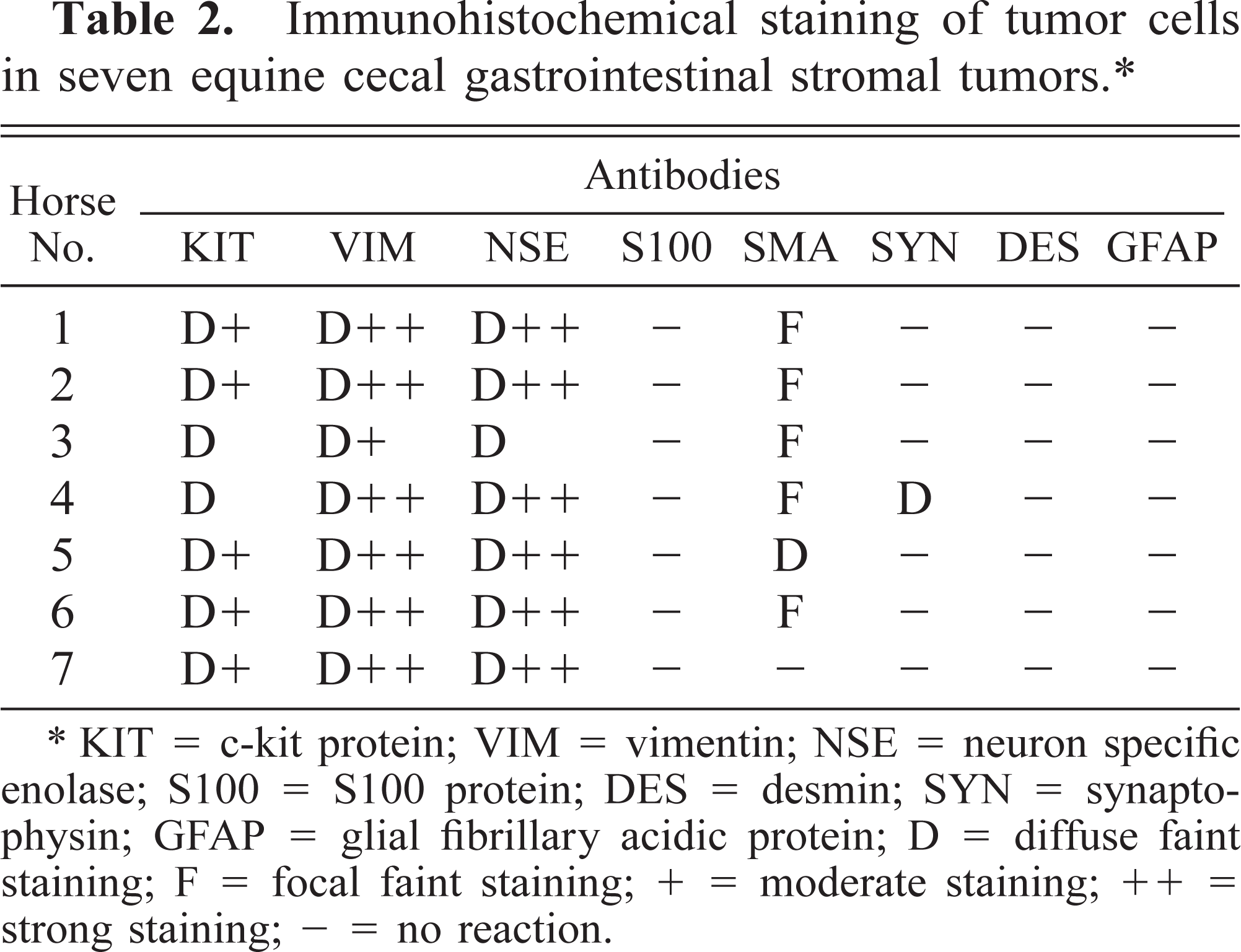
KIT = c-kit protein; VIM = vimentin; NSE = neuron specific enolase; S100 = S100 protein; DES = desmin; SYN = synaptophysin; GFAP = glial fibrillary acidic protein; D = diffuse faint staining; F = focal faint staining; + = moderate staining; ++ = strong staining; - = no reaction.
Cecal tumor; horse 7. Tumor cells diffusely react with antibodies to vimentin. Streptavidin-biotin-alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 30 µm.
Cecal tumor; horse 7. Tumor cells diffusely react with antibodies to neuron-specific enolase. Streptavidin-biotin-alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 30 µm.
Cecal tumor; Cecal tumor; horse 7. Antibodies to c-kit protein stain the cells of all tumors with a diffuse cytoplasmic granularity. Streptavidin-biotin-alkaline phosphatase method, Mayer's hematoxylin counterstain. Bar = 30 µm.
For ultrastructural examination, segments of paraffin-embedded tumors from horses 4 and 7 were cut into 1-mm cubes, cleared with xylene, and immersed in a solution of 2% glutaraldehyde, 2% paraformaldehyde, and 0.2% picric acid in a cacodylate buffer (pH 7.3). These tissues were routinely processed and embedded in Epon, and ultrathin sections were stained with lead citrate and uranyl acetate for electron microscopy.
Ultrastructurally, tumor cells contained relatively uniformly oval nuclei with evenly dispersed to marginated clumps of heterochromatin. Tumor cells contained moderate numbers of mitochondria with lamellar cristae. Cells extended processes connected by desmosome-like junctions (Fig. 7) that interdigitated with cell processes of neighboring cells. Intermediate filaments were evident in cell cytoplasm, and occasional dense core neurosecretory granules were present. Basal laminae were not seen. Some cells contained modified Golgi apparatuses, and there were numerous profiles of endoplasmic reticulum.
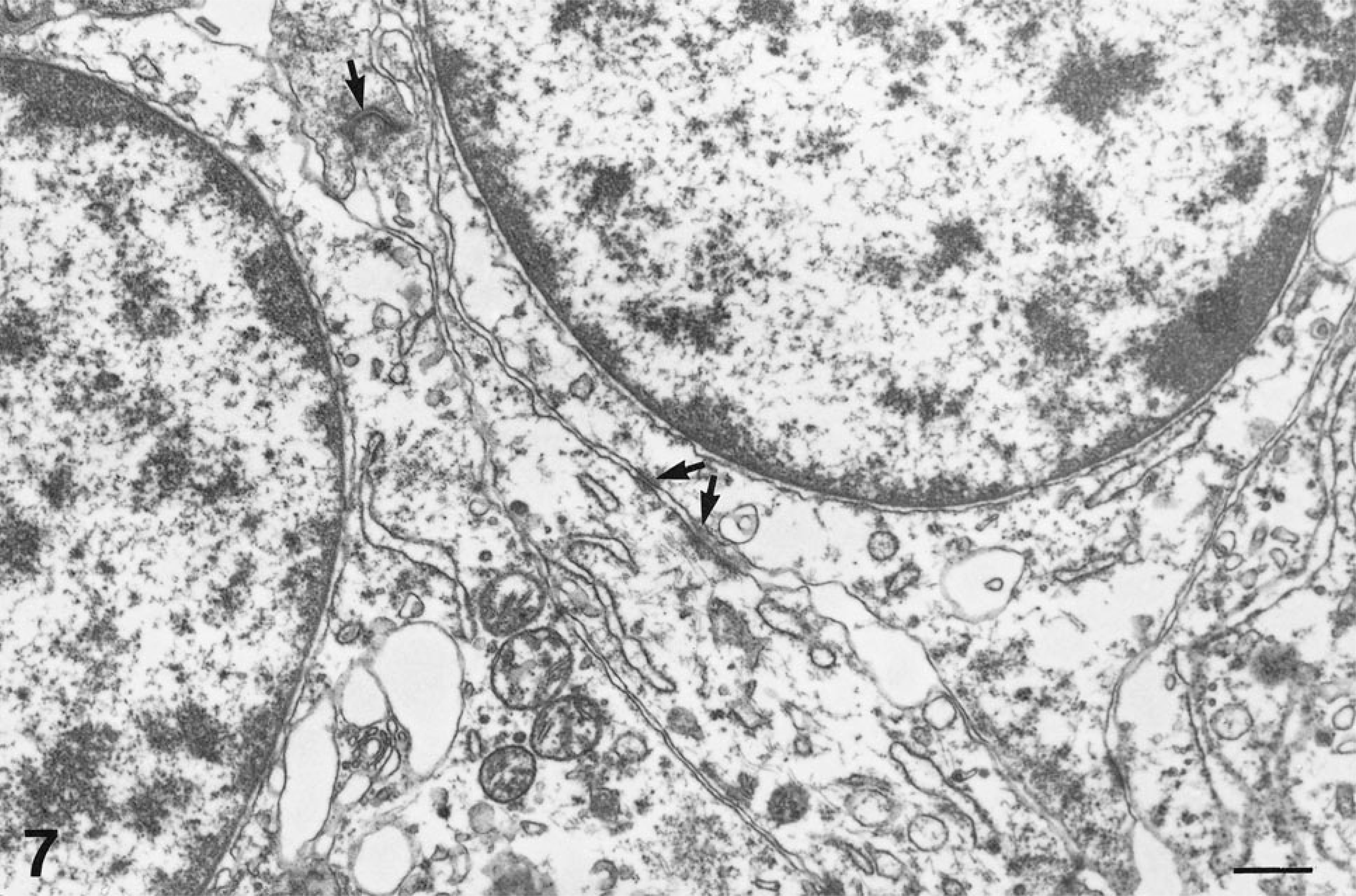
Cecal tumor; horse 7. Extended cell processes that contain intermediate filaments are interconnected by desmosome-like junctions (arrows). Uranyl nitrate and lead citrate. Bar = 500 nm.
In a previous report of seven equine GISTs, the most consistent immunohistochemical staining was for S100 (three of five examined) with only one tumor immunopositive for SMA. In that report, GISTs were identified in stomach, jejunum, cecum, and colon, and some had myxoid changes. 2 In our study of cecal tumors, the most consistent cell markers were VIM, NSE, and KIT. Most tumors were also immunopositive for SMA, with no expression of desmin, S100, or GFAP. This is in contrast with a study of canine GISTs, where most tumors were immunopositive for both VIM and DES and many tumors were also immunopositive for SMA and S100. 8 The immunohistochemical staining pattern of our series of equine cecal GISTs suggests tumor cells could have originated from myofibroblasts. The ultrastructural identification of dense core neurosecretory granules and one tumor diffusely immunopositive for SYN could suggest some neural or neuroendocrine differentiation.
In humans, gastrointestinal stromal cell tumors usually originate in the stomach or small intestine and are commonly stained by antibodies to KIT, CD34, and VIM; some also express SMA, but tumors do not usually express DES or S100. 6 Some human GISTs have been suggested to originate from precursors of KIT-positive enteric pacemaker cells, the interstitial cells of Cajal (ICC). 6 10 These myofibroblasts are intimately associated with the enteric autonomic nerve plexus, and normally generate rhythmic slow wave contractions in enteric smooth muscle. 6 The equine cecal tumors of this report were not examined with antibodies to human CD34 since the antibodies tested did not stain equine control tissues (S. Hafner, personal observation).
The c-kit proto-oncogene encodes the tyrosine kinase receptor KIT, a transmembrane growth factor receptor that normally requires the binding of stem cell factor for its stimulation. 6 10 The expression of KIT is required by ICC, mast cells, melanocytes, and hematopoietic cells for normal development. 10
Some authors identify a subtype of human GISTs with neural differentiation as gastrointestinal autonomic nerve tumors (GANTs). 10 11 These tumors are immunopositive for CD34, NSE, and S100 but lack muscular differentiation. 11 GANTs and GISTs in humans may be both multiple and familial. 10 11 Multiple tumors similar to some human GANTs have been identified in the small intestine of a Pinto gelding. These tumors were classified as multiple benign nerve sheath tumors due to GFAP and S100 immunopositivity. 7 Multiple tumors were present in the cecum of one of the horses of this report; these tumors were thought to represent multiple sites of tumor development rather than metastasis.
Mutations in the gene coding for the KIT receptor may provide a reason for multiple tumor formation. In humans, analyses of the c-kit gene from some GISTs have demonstrated mutations in the juxtamembrane domain that result in a constituitively activated KIT receptor, 5 similar to the constituitively activated KIT receptors that have been found in the neoplastic mast cells of rats, mice, humans, and dogs. 10
The equine cecal stromal tumors described in this report represent a distinctive group of mesenchymal tumors. Tumors were often hemorrhagic and myxomatous. Tumor cells were uniformly immunopositive for vimentin, neuron-specific enolase, and c-kit protein, and most exhibited at least focal immunopositivity for smooth muscle actin, but these cells appeared to lack expression of S100 protein or desmin. These tumors differed from most human GISTs in their myxomatous appearance and location in the cecum but were similar in their expression of KIT and VIM and in the lack of immunopositivity for DES and S100.
Footnotes
Acknowledgements
We thank Kathy Wortham and Alice Self for immunohistochemical and histochemical stains and Mary Ard for assistance with transmission electron microscopy.
