Abstract
Classical swine fever (CSF) virus (CSFV) nucleic acid and antigen were detected in 15 pigs with naturally occurring chronic CSF by in situ hybridization and immunohistochemistry. The most consistent and prominent microscopic lesions were perivascular mononuclear cell infiltration and gliosis in the central nervous system of pigs with chronic CSF. Positive cells typically exhibited a dark brown (in situ hybridization) or red (immunohistochemistry) reaction product in the cytoplasm without background staining. A positive signal for both in situ hybridization and immunohistochemistry was detected in mononuclear cells and lymphocytes of lymphoid tissues. Viral nucleic acid was detected in some tissue sections in the absence of viral antigen. The in situ hybridization technique developed in this study was useful for the detection of CSFV RNA in tissues taken from chronically infected pigs and may be a valuable technique for studying the pathogenesis of chronic CSFV infection.
Classical swine fever (CSF) is an important disease in pigs caused by a small enveloped RNA virus, the CSF virus (CSFV), which belongs to the genus Pestivirus within the family Flaviviridae. 9 The disease is characterized by a peracute, acute, subacute, chronic, atypical, or inapparent course. 27,30,31 Chronic CSF is a lethal disease with a duration of at least 30 days. 18 There is often little evidence of CSF in terms of petechial hemorrhages in most organ systems, but a single organ system (lung, gastrointestinal tract, and central nervous system) may be predominantly affected. 30,31 The clinical signs may often be uncharacteristic and misleading; therefore, chronic CSF was erroneously called atypical CSF. 6,27
Chronic CSF has been observed under both natural and experimental conditions. 5,19 Although the pathogenesis of chronic CSF is poorly understood, it is usually associated with low–virulent strain infection or host factors. 19,30 Low virulent strains generally induced chronic CSF, eventually leading to death or recovery, 3,19,20,24 and may usually be disseminated during a longer period than are high virulent CSFV strains. 29 Outbreak of chronic CSF caused by low virulent strains is more difficult to recognize, and this favors the spread of these strains. These chronically infected pigs are important in the epidemiology of the disease because they act as sources of infection in susceptible pigs.
Classical techniques used to diagnose CSFV infections include the detection of viral antigen using fluorescent- or peroxidase-labeled conjugates, either in cryostat sections of tissues or following up to three serial passages in a cell monolayer. 22 But low virulent CSFV is difficult to isolate in PK-15 cell culture and detect in frozen tissues by fluorescent antibody test. 11,14,20 Laboratory diagnostic techniques must be used to detect chronic CSFV infection if control or eradication is required. One objective of this study was to detect CSFV nucleic acids and antigens in formalin-fixed, paraffin-embedded tissues by using digoxigenin-labeled, 300-base, single-stranded cDNA probes and monoclonal antibody, respectively. The second objective was to determine the location of CSFV in pigs to understand better the pathogenesis of naturally occurring chronic CSF.
Fifteen pigs with chronic CSF were selected from diagnostic cases submitted to the Department of Veterinary Pathology, Seoul National University by the definition of chronic CSF as previously described. 29,30 All selected pigs were found seropositive for CSFV infection but negative for porcine respiratory syndrome virus (PRRSV) by enzyme-linked immunosorbent assay (IDEXX Laboratories, Westbrook, ME). Six out of eight pigs had had an outbreak of acute CSF in the previous 6–8 months. Three pigs (Nos. 1–3) from different herds were submitted because of retardation of growth accompanied by diarrhea. Two pigs (Nos. 4, 5) from the same herd were presented with slight incoordination and pyrexia (40.5–41.7 C). Three pigs (Nos. 6–8) from the same herd had intermitteant diarrhea. Three pigs (Nos. 9–11) from the same herd were presented for severe respiratory disease. Four pigs (Nos. 11–15) from two herds were submitted for necropsy because of retardation of growth accompanied by dyspnea. All pigs were submitted alive, and immediately upon receipt they were humanely killed for necropsy. Seronegative and virus isolation–negative conventional 70-day-old and 90-day-old pigs were used as negative controls. Negative tissue controls were collected from each of the two calves that had been naturally infected with bovine viral diarrhea virus (BVDV). Two 83-day-old pigs experimentally infected with CSFV were used as positive controls. Two positive control pigs were inoculated intranasally with 3 ml of the CSFV strain SNUVR2345 (second passage) at a concentration of 1055, which is the 50% tissue culture infective dose (TCID50) per milliliter. Two positive control pigs were humanely killed 7 days after inoculation. Strain SNUVR2345 was isolated from a 58-day-old pig with chronic CSF.
Samples of brain, lung, liver, kidney, tonsil, lymph node, and small and large intestines were collected from infected and noninfected animals, fixed in 10% (w/v) neutral buffered formaldehyde for 24–48 hours, and embedded in paraffin according to standard laboratory procedures. Five-millimeter slices of brain were taken for histopathology from three areas at the following reference points: frontal cerebrum (genu of corpus collasum), parietal cerebrum (interthalamic adhesion), and cerebellum with brain stem (fastigial recess of fourth ventricle). Four-micrometer-thick sections were stained with hematoxylin and eosin (HE).
For virus isolation, the lymph node, tonsil, and spleen from each pig were pooled, diluted 1:5 in phosphate-buffered saline (PBS, 0.01 M, pH 7.2), ground by homogenization, and centrifuged for 20 minutes at 9,000 × g. Before inoculation, the cell growth medium of monolayered PK-15 cells grown in a 25-cm2 flask was removed, and the monolayers were washed twice with minimal essential medium plus 4% fetal bovine serum. Then, the cells were inoculated with 1 ml of the homogenate per flask and incubated for 5 days. The cultures were then frozen and thawed after 3 days of incubation, and 1 ml of medium and cells were transferred to a new monolayer of PK-15 cells, this time containing minimal essential medium plus 4% fetal bovine serum. Second-passage cultures were incubated for 5 days. The cultures were fixed in 80% acetone and tested for CSFV antigen by indirect immunofluorescent antibody test. After acetone fixation, wells were stained with monoclonal mouse anti-CSFV antibody (WH303, Central Veterinary Laboratory, New Haw, Addlestone, UK) 8 and fluoroscein isothiocyanate (FITC)–conjugated anti-mouse immunoglobulin G and then viewed with a fluorescence microscope for evidence of specific viral antigen. Wells were also stained with immunohistochemistry. If a detectable virus-specific fluorescent and immunohistochemical signal was present in any cells of the monolayer, the sample was considered positive for CSFV by virus isolation.
The brain, lung, liver, spleen, kidney, and small and large intestines from all 15 pigs were cultured for Actinobacillus pleuropneumoniae, A. suis, Pasteurella multocida, Streptococcus suis, Erysipelothrix rhusiopathiae, and Salmonella sp. Cryostat sections were prepared from the brain, tonsil, and liver samples and processed for fluorescent antibody test using a FITC-conjugated porcine anti-pseudorabies fluorescence antibody (National Veterinary Services Laboratories, Ames, IA). The formalin-fixed, paraffin-embedded lung and lymph node were used for detection of Mycoplasma hyopneumoniae, PRRSV, swine influenza virus (SIV), and porcine circovirus (PCV)–2 by in situ hybridization and immunohistochemistry as described previously. 4,10,12,13
CSFV-infected and noninfected PK-15 cell cultures were used as the starting material for optimization of in situ hybridization and immunohistochemistry. The 100 μl of tissue culture fluid containing 105 TCID50 per milliliter of CSFV strain SNUVR2345 was grown in PK-15 cells in four-well chamber slides (Nunc, Inc., Naperville, IL). After 4 days of incubation at 37 C, the medium was discarded, the wells were rinsed with PBS (0.1 M, pH 7.4), and the cells were fixed by adding PLP (4% paraformaldehyde, 100 mM
The primers from the genome of the Alfort strain sequence were used in this study. The forward and reverse primers were 5′-AGTGACAACGGCACTAATGG-3′ (nucleotides 1198–1217) and 5′-CTTATCTGGAGGGCCTTCTG-3′ (nucleotides 1497–1478), respectively. 16 The primer set resulted in amplified fragments of 300 base pairs (bp). PCR products were purified using a 30-kD cut-off membrane ultrafiltration filter. The nucleotide sequences of the purified PCR products were determined by using BigDye chemistry with the ABI Prism Sequencer (Applied Biosystems, Foster City, CA). Sequencing was performed on the purified PCR products before they were labeled by random priming with digoxigenin–deoxyuridine triphosphate (Boehringer Mannheim, Indianapolis, IN) according to the manufacturer's instructions. In situ hybridization was carried out as previously described. 10
For immunohistochemistry, sections were deparaffinized in xylene, rehydrated through graded alcohols, and air-dried. Endogenous alkaline phosphatase was quenched with 20% glacial acetic acid solution for 2 minutes at 4 C. All slides were then incubated with normal mouse serum (Sigma Chemical Company, St. Louis, MO) in PBS (0.1 M, pH 7.4) for 30 minutes at room temperature to saturate the nonspecific, protein-binding sites. A commercially available monoclonal mouse anti-CSFV antibody (WH303) was used at a dilution of 1:50 in PBS containing 0.1% Tween 20. Monoclonal antibodies were coated on the slides and incubated for 1 hour at room temperature.
After three washes with 0.1% Tween 20 in PBS (0.01 M, pH 7.4), sections were flooded and incubated for 1 hour at 37 C with alkaline phosphatase–conjugated goat anti-mouse immunoglobulin G antibody (Dako, Glostrup, Denmark) diluted 1:200 in PBS (0.01 M, pH 7.4) containing 0.1% Tween 20. The slides were washed three times with 0.1% Tween 20 in PBS. Then, sections were equilibrated with Tris-buffer (0.1 M, pH 8.2) for 5 minutes at room temperature. The final reaction was produced by immersing the sections in a solution of red substrate (Vector Laboratories, Burlingame, CA) for 20 minutes at room temperature.
A. pleuropneumoniae was isolated from lungs of two pigs (Nos. 9, 10). P. multocida was isolated from lungs of four pigs (Nos. 11, 12, 14, 15). M. hyopneumoniae DNA was detected in lungs from all 15 pigs by in situ hybridization. PRRSV, SIV, and PCV-2 were not detected in lungs from all 15 pigs by in situ hybridization and immunohistochemistry. CSFV was isolated from five pigs (Nos. 1, 3, 5, 6, 11). All six viruses isolated from pigs reacted with primers of CSFV (Fig. 1). PCR products from each isolate were sequenced, and their identity was confirmed as CSFV (data not shown).
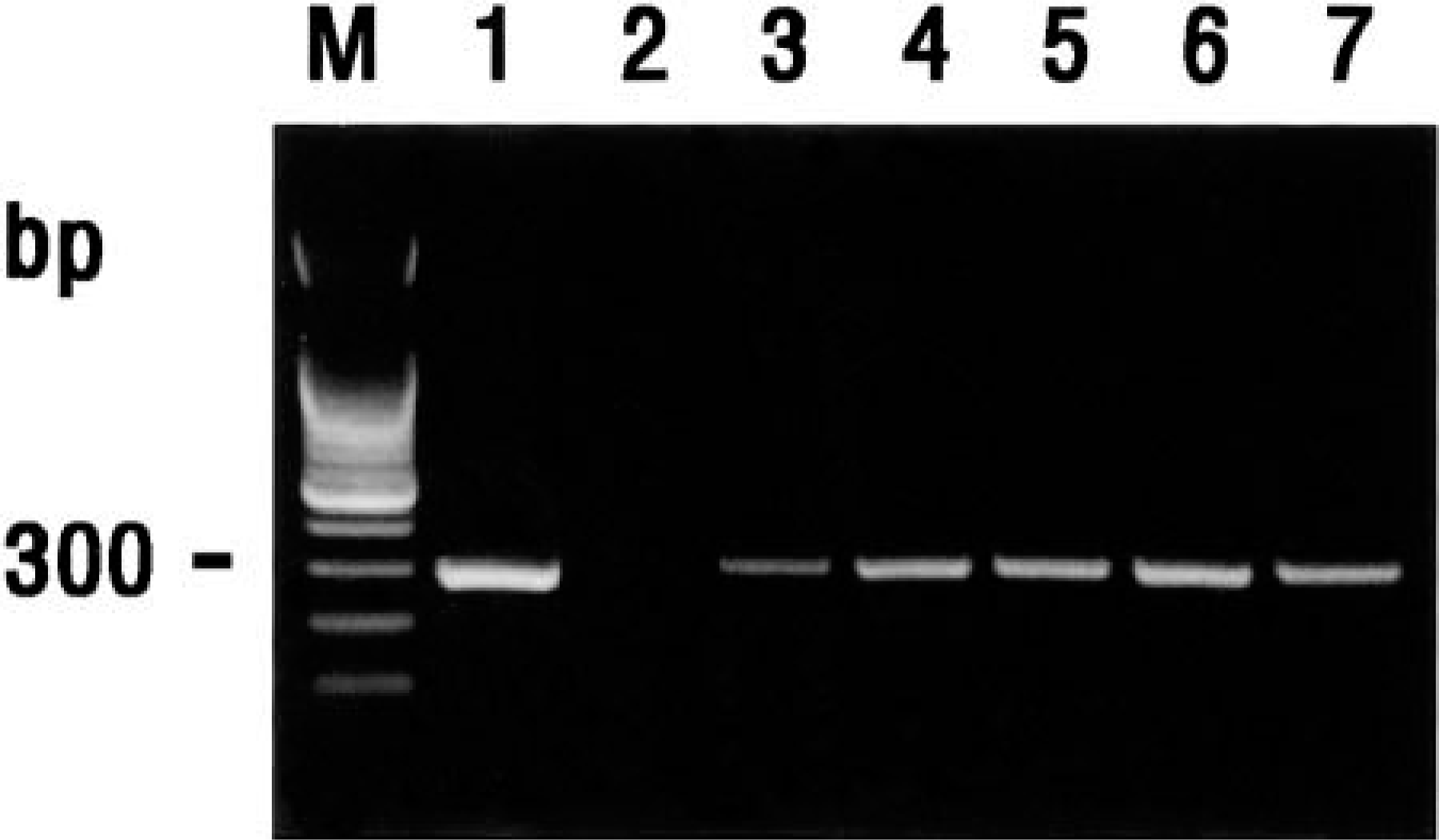
Agarose gel electrophoresis of reverse transcription–polymerase chain reaction–amplified CSFV cDNA products. From left to right: M = 100-bp DNA ladder; lane 1 = positive CSFV strain SNUVR2345; lane 2 = negative control; lane 3 = CSFV isolated from pig No. 1; lane 4 = CSFV isolated from pig No. 3; lane 5 = CSFV isolated from pig No. 5; lane 6 = CSFV isolated from pig No. 6; lane 7 = CSFV isolated from pig No. 11.
The most consistent and significant microscopic changes occurred within the central nervous system. Perivascular mononuclear cell infiltration involving both gray and white matter was the most striking light microscopic change observed in brain from pigs with chronic CSFV. The distribution of lesions was consistently angiocentric and multifocal and involved the cerebrum to the greatest extent, with an apparent predilection for gray matter. In cerebral sections, subependymal and subpial infiltrates were present, but deep parenchymal infiltrates predominated. The Virchow-Robin spaces were markedly distended (10–15 cells thick) with mononuclear cells, specially monocytes, lymphocytes, and plasma cells. In subcortical white matter, perivascular mononuclear cell infiltrates extended into white matter. In cortical gray matter, infiltration of mononuclear cells was accompanied by gliosis. Perivascular mononuclear cell infiltration was also seen in cerebellum.
The predominant microscopic lesions of lymph node were follicular hyperplasia or necrosis in the center of prominent lymphoid follicles. Necrotic foci of 1–10 cells with pyknosis and karryorrhexis were commonly observed in the center of prominent follicles and less often in the surrounding lymphoreticular tissues. A mild tonsillitis with necrosis was observed. Mild perivascular mononuclear cell infiltration involving both gray and white matter was observed in brain from two positive control pigs experimentally infected with CSFV.
CSFV nucleic acids were detected by use of an anti-digoxigenin–alkaline phosphatase conjugate when CSFV-infected PK-15 cells were hybridized with a specific digoxigenin-labeled CSFV cDNA (300 bp) probe. The CSFV nucleic acid signal was specific because this was completely absent from mock-infected PK-15 cells. CSFV antigen was consistently detected in the CSFV-infected PK-15 cells. Distinct cell outlines were visualized, and the nucleus was distinguishable because of its lack of staining. No positive staining cells were detected in the mock-infected PK-15 cells.
The results of in situ hybridization and immunohistochemistry are summarized in Table 1. The morphology of host cells was preserved despite the relatively high temperature required in parts of the incubation procedure. Positive cells typically exhibited a dark brown (in situ hybridization) or red (immunohistochemistry) reaction product in the cytoplasm without background staining. The signal intensity varied within and between histological structures in any one section and between pigs. Viral nucleic acid was detected in some tissue sections in the absence of viral antigen (Figs. 2, 3).
Virus isolation, in situ hybridization (ISH), and immunohistochemistry (IHC) results in 15 pigs naturally infected with classical swine fever virus.
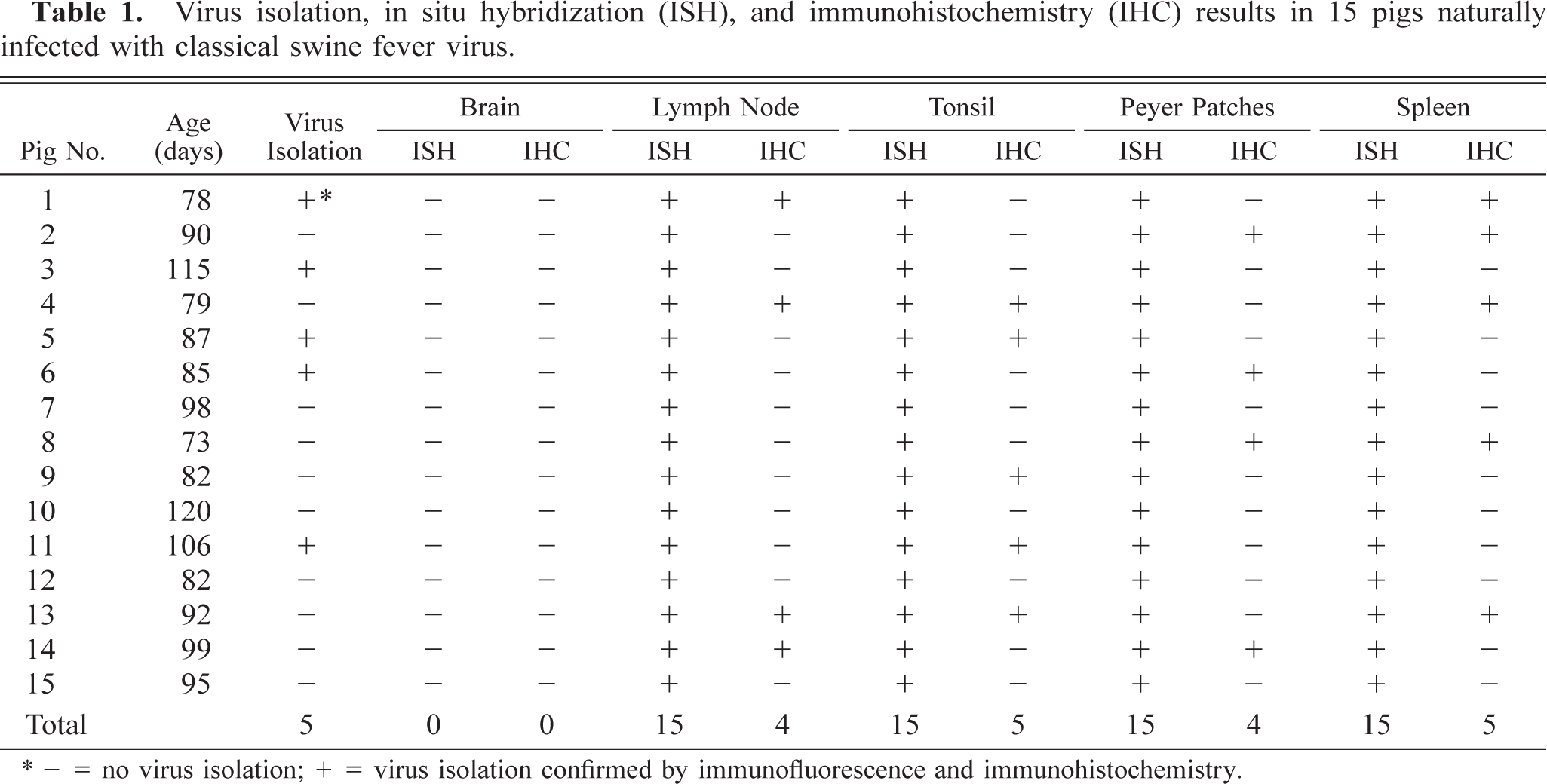
∗- = no virus isolation; + = virus isolation confirmed by immunofluorescence and immunohistochemistry.
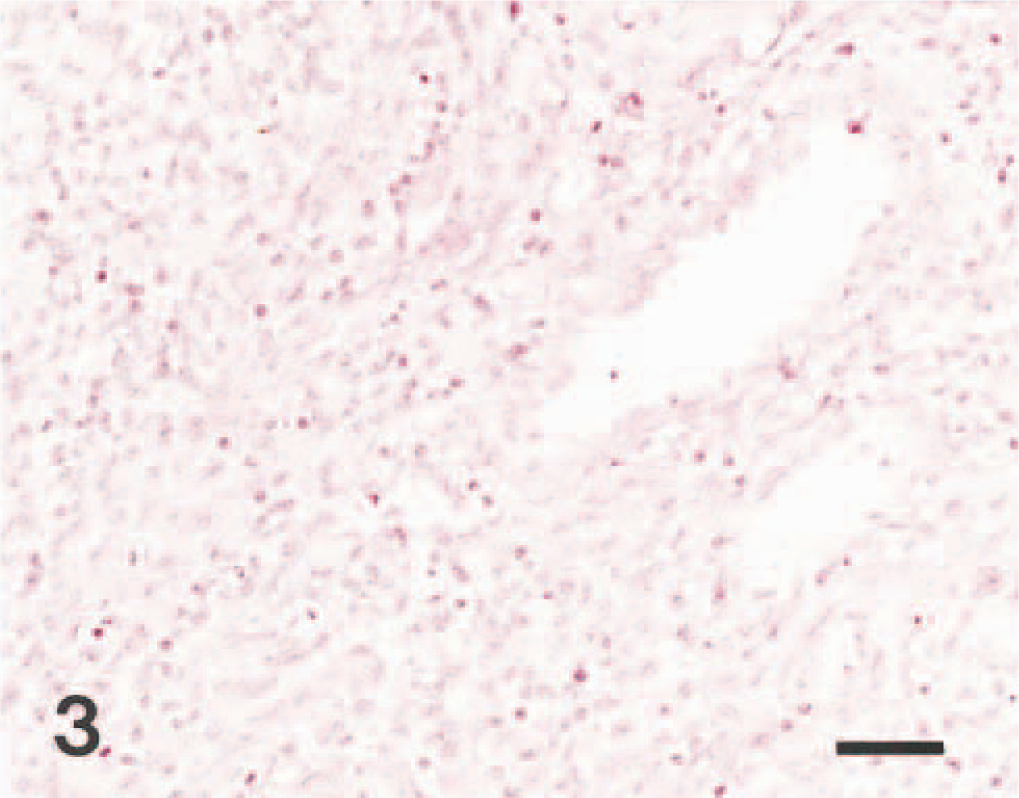
Lymph node; pig No. 2. CSFV RNA (dark brown reaction) is detected in the lymphocytes. In situ hybridization; nitroblue tetrazolium and 5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 55 μm.
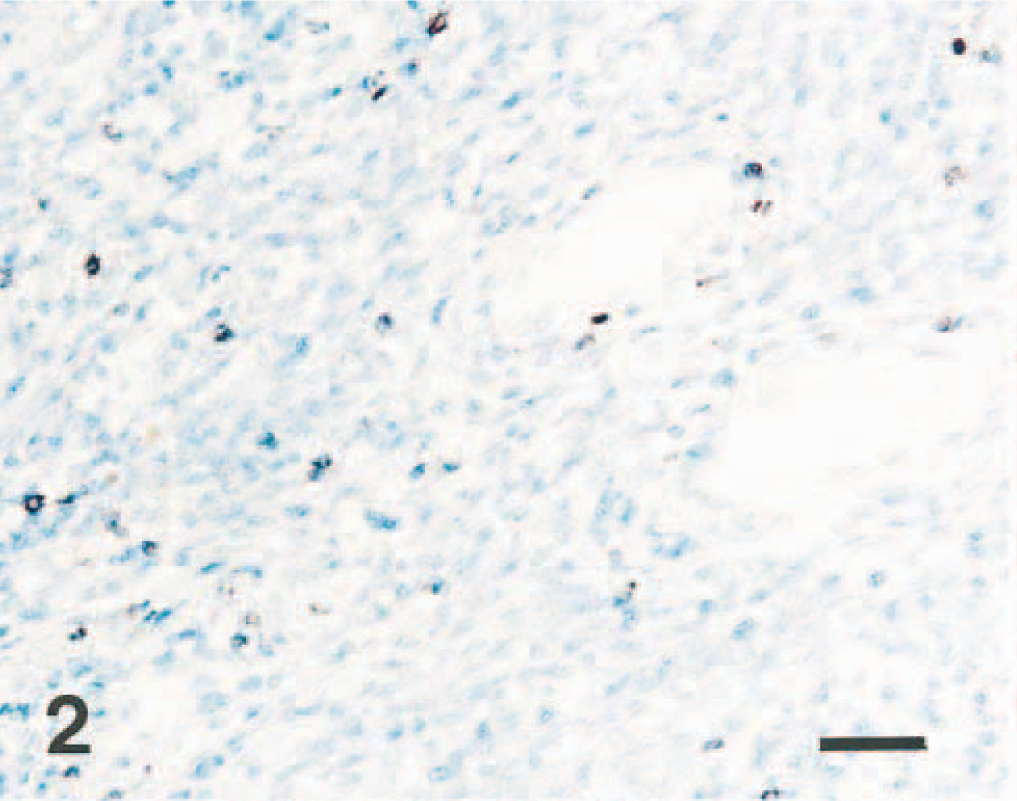
Lymph node; pig No. 2. CSFV antigen (red reaction) is not detected in the lymphocytes of lymph node. Immunohistochemistry; Vector red, hematoxylin counterstain. Bar = 55 μm.
CSFV nucleic acids were consistently found in the cytoplasm of mononuclear cells and lymphocytes in the medullary sinuses and lymphoid follicles of mesenteric and linguinal lymph nodes (Fig. 4). Within mesenteric and other lymph nodes, CSFV nucleic acid was occasionally found in lymphoid follicles within the cortices. CSFV antigens were found in mononuclear cells and lymphocytes randomly scattered in splenic red and white pulp in three pigs (Nos. 1–3) (Fig. 5). In tonsils, CSFV nucleic acid was occasionally detected within cells in the crypt epithelium. No hybridization signals were detected within cells in the center of follicles and in the surrounding lymphocytes. CSFV nucleic acid was occasionally detected within cells in the follicular area of Peyer's patches. No hybridization signal was consistently seen in tissue sections treated with RNase A before in situ hybridization. Sections from negative control pigs and BVDV-infected calves showed no hybridization and immunohistochemical signal for CSFV.
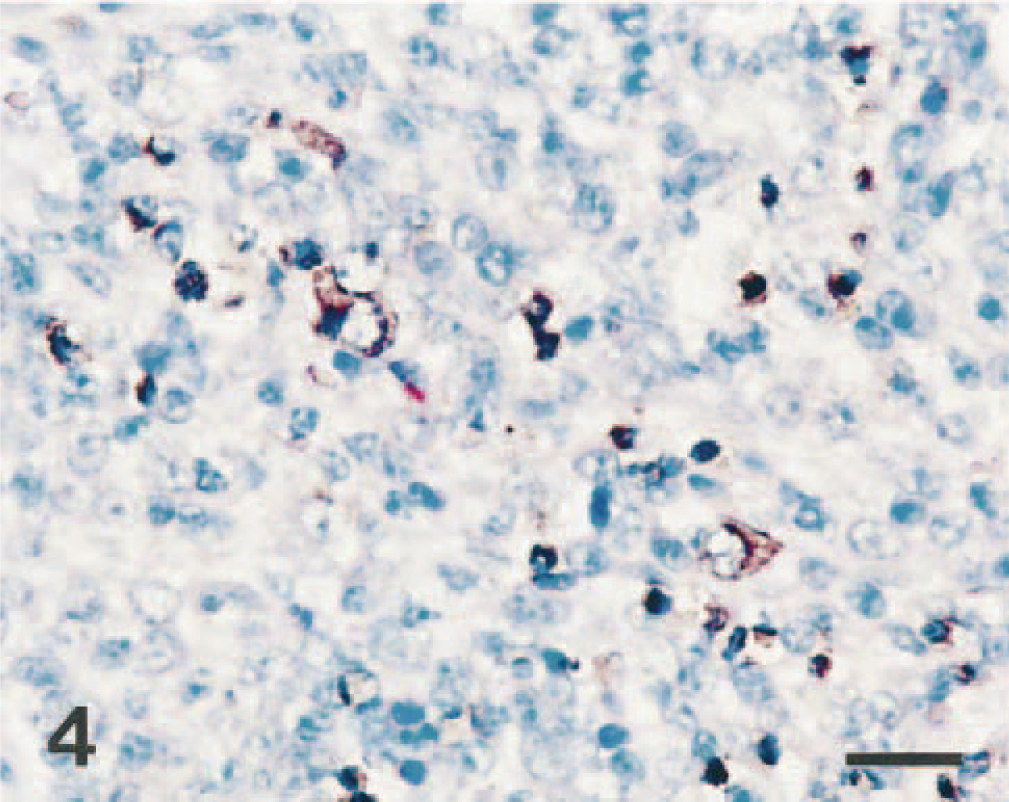
Lymph node; pig No. 4. CSFV RNA (dark brown reaction) is detected in the cytoplasm of mononuclear cells and lymphocytes. In situ hybridization; nitroblue tetrazolium and 5-bromocresyl-3-indolylphosphate, methyl green counterstain. Bar = 30 μm.
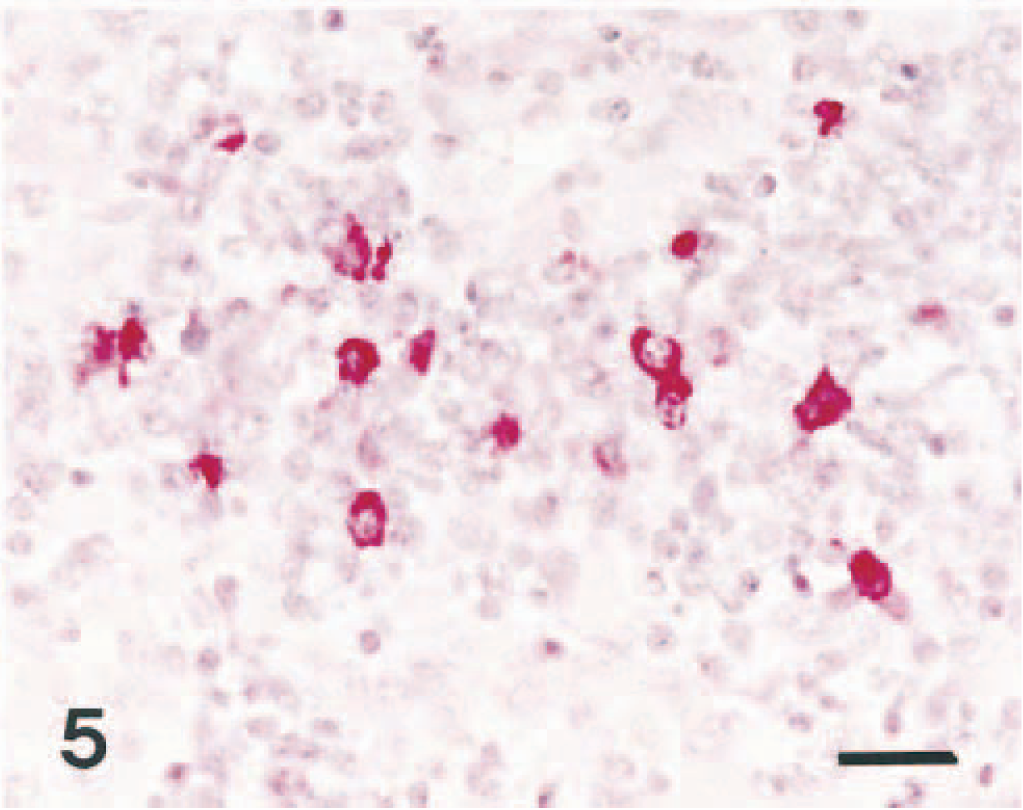
Spleen; pig No. 13. CSFV antigen (red reaction) is detected in the cytoplasm of mononuclear cells. Immunohistochemistry; Vector red, hematoxylin counterstain. Bar = 30 μm.
The results of this study indicate that CSFV nucleic acid and antigen can be detected in formalin-fixed, paraffin-embedded tissue specimens from pigs with chronic CSFV infection by using a nonradioactive labeled cDNA probe and monoclonal antibody, respectively. The microscopic lesions seen in the brains of pigs with chronic CSFV infection are similar to those seen in pigs experimentally inoculated with a low-virulence CSFV strain. 23 In regions where CSF is enzootic, chronic CSFV infection is more frequent, and these cases are often associated with low-virulence CSFV. 30 The mechanisms responsible for the evolution of CSFV strains of low virulence are poorly understood, but this phenomenon may be explained by selection events. In pigs chronically infected with CSFV, the virus replicates in the presence of antibody. This may result in a selection of variants that are antigenically less related to the original virus population or refractory to neutralization. 19 Data collected during the last 10 years of eradication in the USA indicate that 55% of the 135 field isolates collected were found to be strains of low-virulence CSF, which was eradicated in the USA in 1978. 2 These data suggest that chronic CSFV infection is caused by viruses of low virulence.
The distribution of CSFV antigen in infected tissues over time is well documented. 7,21,25 In pigs acutely infected with highly virulent strains of CSFV, viral antigen was detected in tonsillar cryptic epithelial cells and macrophages, reticular and endothelial cells in spleen and lymph nodes, and smooth muscle cells of blood vessels. 7,21 By comparison, CSF viral nucleic acid and antigen were found predominantly within mononuclear cells and lymphocytes of lymph node and spleen from pigs with chronic CSFV infection, 20 and these data were in general agreement with those of a previous study. The difference in the distribution of CSFV in pigs with chronic CSFV infection when compared with pigs that are acutely infected may be related to different immune responses; pigs with acute CSF mount neutralizing antibody response to the virus, 17 whereas the neutralizing antibody response can be absent, impaired, delayed, or only transiently detectable in pigs with chronic CSF. 2,15,24 Differences in the distribution of CSFV could also be due to differences among strains of virus, in the age of the pigs, in the duration of infection, or in the susceptibility of the breed.
Thymic atrophy and lymphoid depletion in lymphoid tissues indicate impaired immunity in pigs with chronic CSF. 3 The hybridization and immunohistochemical signals detected in the present study indicate that CSFV infected the macrophages in lymphoid tissues. Infection of macrophages by CSFV could adversely affect the host defense mechanisms. BVDV, which also belongs to the genus Pestivirus, has been reported to modulate functions of immune cells after infection in vitro, with decreased phagocytic activity of infected alveolar macrophages. 32 Secondary bacterial infections are commonly reported in pigs with chronic CSF, 3 and it might explain why six pigs (Nos. 9–12, 14, 15) had secondary bacterial infections in this study.
Clinical diagnoses of chronic CSFV infection are difficult to make because of the wide variability of clinical signs and gross lesions. 17 The histopathologic changes in the lymphoid tissues of pigs with chronic CSFV infection are also nonspecific, and viral inclusions are not seen. 1,27,28 In this study viral nucleic acid and antigen were detected in formalin-fixed, paraffin-embedded lymphoid tissue samples by in situ hybridization and immunohistochemistry. But viral nucleic acid was detected in some tissue sections in the absence of viral antigen. The reason for this discrepancy is not known. One possibility is that CSFV infection is nonproductive. The previous study suggested that CSFV-infected cells may express no or very little viral antigen on the cellular surface, especially low-virulent CSFV-infected tissues, 31 and this could result in failure to detect viral antigen by immunohistochemistry. Another possibility is that formalin fixation can denature antigens. 26 This ultimately can lead to false-negative results. The CSFV from pigs with chronic CSF was infrequently identified by virus isolation compared with in situ hybridization. Some samples that tested positive for CSFV with in situ hybridization tested negative with virus isolation in the present study. It is possible that CSFV is not well adapted to PK-15 cells or low virus titer in tissue samples. The in situ hybridization technique developed in this study was useful for detection of CSFV RNA in tissues taken from naturally infected pigs and may be a valuable technique for studying the pathogenesis of chronic CSFV infection.
Footnotes
Acknowledgements
The research reported here was supported by the Ministry of Agriculture, Forestry and Fisheries-Special Grants Research Program (MAFF-SGRP) and Brain Korea 21 Project, Republic of Korea.
