Abstract
The objective of this study was to determine if skin punch biopsies are appropriate for the diagnosis of classical swine fever. For this purpose, 6 wild boars and 2 domestic pigs were experimentally infected with the highly virulent classical swine fever virus (CSFV) Koslov and 5 domestic pigs with a CSFV field isolate (genotype 2.3 Uelzen) derived from wild boar. Skin biopsy specimens were tested using virus isolation, real-time reverse transcription polymerase chain reaction (rtRT-PCR), and fluorescent antibody test (FAT) on cryosections. Whereas CSFV Koslov was first detected at 4 days postinfection (DPI) by rtRT-PCR and virus isolation, FAT failed to detect CSFV antigen until 9 DPI. In domestic pigs infected with CSFV 2.3 Uelzen, viral RNA and CSFV were detected at 7 DPI. FAT was negative until 11 DPI. CSFV antigen was detected in endothelial cells of the vascular plexus in the upper dermis as shown by confocal laser-scanning microscopy and double labeling with von Willebrand factor. At 18 DPI, CSFV antigen was present diffusely in capillaries and spindle shaped cells of the dermis, multifocally within keratinocytes of the epidermis and in numerous cells of the inner and outer root sheath epithelium, hair bulb, and intravascular leukocytes. The rtRT-PCR proved to be the test with the highest sensitivity followed by virus isolation and FAT. Taken together, this study demonstrates that skin is easy to sample antemortem and is also suitable as postmortem tissue, and suggests that rtRT-PCR of skin should be included for CSF diagnosis in the acute period of disease.
Classical swine fever (CSF, hog cholera) is a highly contagious and frequently fatal viral disease of domestic pigs and wild boars (feral pigs). The causative agent is a small enveloped virus with a 12.5 kb single-stranded RNA genome of positive polarity. CSF virus (CSFV) belongs to the genus Pestivirus in the family Flaviviridae. 22
CSF currently has a worldwide distribution and sporadic outbreaks have commonly been recorded in the European Union (EU) during the last decade, not only in domestic pigs but also in wild boar. 9,12,14 The latter have often been the source of CSF outbreaks in domestic pig farms in Germany. 7,14 An early, rapid, highly sensitive and precise laboratory diagnosis is very important for detection of infected herds in the context of CSF control. The quality and diagnostic reliability of the detection method used for determination of infected animals is therefore crucial. According to the Diagnostic Manual of the EU 1 and the Manual of Diagnostic Tests and Vaccines for Terrestrial Animals: Mammals, Birds and Bees of the World Organization for Animal Health (OIE) 15 the virological methods used for confirmation of CSF are 1) virus isolation in susceptible cell cultures (which still is the “gold standard”), 2) immunohistochemical detection of virus antigen by fluorescent antibody test (FAT) on cryosections of tissue samples, 3) antigen ELISA, and (4) reverse transcription-polymerase chain reaction (RT-PCR) for detection of viral RNA.
The primary replication of CSFV takes place in the tonsils after natural oronasal infection. From here virus spreads to the mandibular and retropharyngeal lymph nodes via peripheral blood to bone marrow, visceral lymph nodes, and lymphatic tissues in spleen, kidneys, and small intestine (Peyer's patches). 13,18,19 The virus replicates in a broad range of tissues but affects primarily the immune system, resulting in a generalized leukopenia. It was demonstrated that skin epithelial cells are also targeted by CSFV in the early period after experimental infection. 3 Regarding infection of the skin, the presence and replication of related pestiviruses is proven in ruminants, as shown for bovine viral diarrhea virus (BVDV) in cattle or border disease virus (BDV) in sheep. 2,17,21 Therefore, the objective of this study was to evaluate the skin as an additional sample for CSF diagnosis.
To this end, skin biopsy specimens (8-mm-diameter biopsy punch) a from wild boars (n = 6) and domestic pigs (n = 2), which were infected oronasally with 1-ml whole blood of the highly virulent CSFV strain Koslov (106 TCID50/ml), were investigated on day 4 and 7 postinfection (wild boar no. 73 and domestic pig no. 64) and 9 days postinfection (DPI) when clinical signs were present and domestic pig no. 64 died (Table 1). Subsequently, 5 domestic pigs were challenged oronasally with 1-ml cell culture supernatant of the CSFV field isolate “935” (genotype 2.3 Uelzen) derived from wild boar shot in Lower Saxony. The infectious dose contained 106.25 TCID50. The skin samples were collected in a series on different DPI and on the day of death (details see table 2). For all animals, skin punch biopsies were taken from the dorsolateral region of the neck near the base of the ear.
Isolation of CSFV from skin specimens, EDTA blood (buffy coat), and tonsils was carried out on confluent porcine kidney (PK-15) cells b using a mouse anti-CSFV-E2 monoclonal antibody c for detection of virus according to an accredited method. 10
For detection of CSFV RNA, a real-time RT-PCR (rtRT-PCR) protocol established for routine intra vitam diagnosis and validated in the National Reference Laboratory for CSF was used as described earlier. 5,8 Briefly, RNA was extracted from homogenized skin biopsies using RNeasy® Mini Kit d according to the manufacturer's instructions and modified by addition of an internal RNA control after the lysis. For rtRT-PCR, the commercially available QuantiTect Probe RT-PCR Kit d was used. Per single well 3.25 μl RNase-free water, 12.5 μl 2× QuantiTect(tm) Probe RT-PCR master mix, 0.25 μl QuantiTect(tm) Probe RT mix, 2 μl Enhanced Green Fluorescent Protein (EGFP) mix (limit 5), and 2 μl CSF mix 1 were pooled. The EGFP mix and the CSF mix represent primerprobe mixes with the following sequences: CSF mix 1: CSF100F (100 pmol/μl) 5′-ATG CCC AYA GTA GGA CTA GCA-3′, CSF192R (100 pmol/μl) 5′-CTA CTG ACG ACT GTC CTG TAC-3′, CSF Probe 1 (249 pmol/μl) 5′-FAM-TGG CGA GCT CCC TGG GTG GTC TAA GT-TAMRA-3′; EGFP mix 1: EGFP1-F (100 pmol/μl) 5′-GAC CAC TAC CAG CAG AAC AC-3′, EGFP2-R (100 pmol/μl) 5′-GAA CTC CAG CAG GAC CAT G-3′, EGFP Probe 1(282 pmoyμl) 5′-HEX-AGC ACC CAG TCC GCC CTG AGC A-BHQ1–3′. Finally, 5 μl RNA template of the sample were added. The rtRT-PCR was carried out in the iCycler Mx3000P® QPCR System. e A sample was assessed as positive when a threshold cycle (ct) value of less than 40 was recorded.
For detection of virus antigen by direct FAT, skin biopsies were snap-frozen in n-heptane. After washing at room-temperature with phosphate-buffered saline (PBS) for 5 minutes, sections were incubated for 60 minutes with 1:50 diluted fluorescein-isothiocyanate (FITC)-labeled anti-CSF monoclonal antibody BioX f mixed at a ratio of 3:1 with 0.005% Evans blue g to reduce background autofluorescence. For three-color-immunofiuorescence, polyclonal rabbit anti-human von Willebrand factor h (1:500 in PBS) and the anti-CSF BioX were applied. Indocarbocyanin (Cy3)-conjugated goat anti-rabbit antibody i (1:1,000 in PBS) was used as secondary antibody. To stain nuclear chromatin, TO-PRO-3 iodid j (1:2,000 in PBS) was added during secondary antibody incubation. To preserve fluorescence, sections were sealed in glycerol buffer containing 1,4-diazobicyclo(2,2,2)-octane (DABCO) g 25 mg/ml and analyzed with a confocal laser-scanning microscope (LSM510).
As positive control for virus isolation, rtRT-PCR and FAT lymphatic tissues from a domestic pig, experimentally infected with CSFV “Alfort 187” and accredited-as-positive reference material were used. 20 Skin samples from healthy animals, which were tested free as from pestivirus and related antibodies, served as negative controls.
Detection of the CSFV strain Koslov in skin biopsy samples of wild boars (n = 6) and domestic pigs (n = 2) by virus isolation (VI), real-time RT-PCR (rtRT-PCR), and fluorescent antibody test (FAT) on cryosections.∗
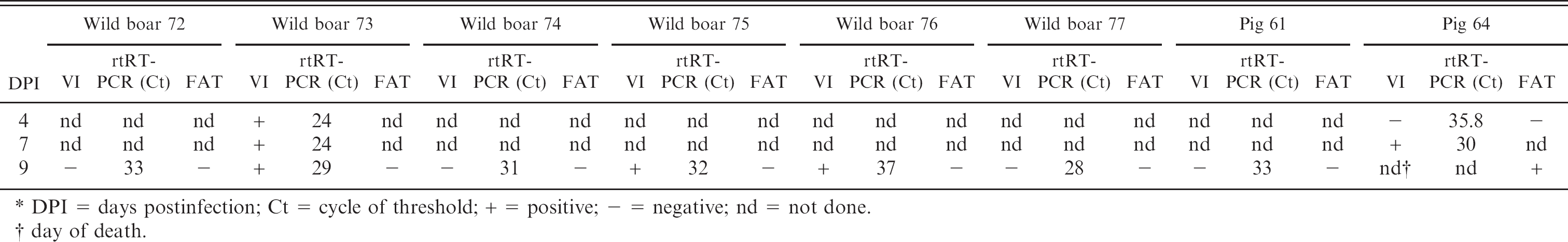
DPI = days postinfection; Ct = cycle of threshold; + = positive; – negative; nd = not done.
day of death.
Sera were tested for antibodies to CSFV by means of a virus neutralization test as indirect neutralizing peroxi-dase-linked antibody assay as earlier described. 11 In an outbreak scenario, it is not only necessary to use highly sensitive, specific, and fast diagnostic methods but also tissues that are representative targets for CSFV and easy to sample. The investigation of skin punch biopsies derived from animals challenged with the Koslov strain revealed that at 4 DPI the rtRT-PCR was already positive for 2 tested animals (ct 28 and 35.8), and 1 wild boar of these was also positive by vims isolation. Six wild boars and 1 domestic pig were tested positive by rtRT-PCR at 9 DPI. In contrast, CSFV was detected in the skin of only 3 wild boars by vims isolation. Remarkably, FAT failed to detect CSFV antigen until this time-point. Although all animals showed clinical signs specific for CSF, distinct skin lesions were not present until 9 DPI. At necropsy 11 DPI, all animals had lesions characteristic for CSF (Table 1).
Detection of the CSFV 2.3 Uelzen in skin biopsy samples of domestic pigs (n = 5) by virus isolation (VI), real-time RT-PCR (rtRT-PCR) and fluorescent antibody test (FAT) on cryosections (n = 9).∗
DPI = days postinfection; Ct = cycle of threshold; + = positive; – = negative; A = neutralizing antibody titer: 40 ND50.
= day of death.
end of the experiment.
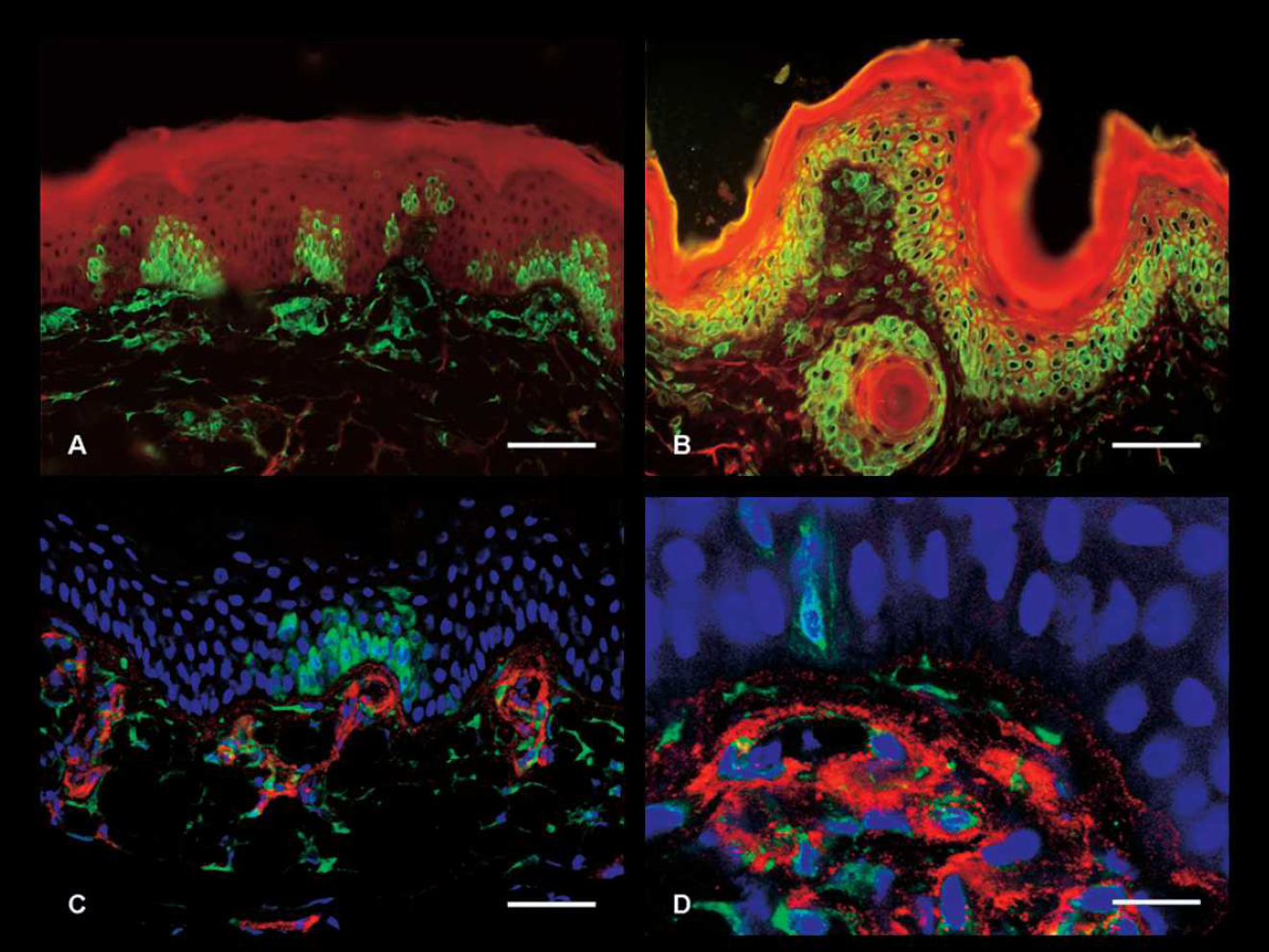
As CSFV genotype 2.3 Uelzen has been the source of several outbreaks of CSF in domestic pigs and wild boars in Germany during the last 2 decades, skin biopsy specimens were also evaluated for this virus type. In principle, the results are consistent with the observations in the group infected with CSFV Koslov but the course of disease and detection of CSFV was slightly delayed (Table 2). The first virologically positive results were obtained at 7 DPI. The domestic pigs were scored RNA positive compared to 3 pigs that were evaluated positive by virus isolation. All animals were diagnosed positive at 9 DPI in both tests. In contrast, immunostaining on cryosections of skin revealed later detection of CSFV antigen at 11 DPI (1 of 5 pigs, animal no. 22). Staining was present in the vascular plexus of the upper dermis, within spindle shaped mesenchymal cells, and at this time point in a small number of basal keratinocytes only (Fig. 1A). At 9 DPI after infection with CSFV Koslov (Fig. 1B) and in a similar extent at 18 DPI after infection with CSFV genotype 2.3 Uelzen, viral antigen was detected as brilliant homogeneous green fluorescence in the cytoplasm of numerous keratinocytes, cells of the inner and outer root sheath epithelium, hair bulb, vascular endothelium, and intravascular leukocytes. By detection of von Willebrand factor, confocal laser-scanning microscopy showed unambiguously that endothelial cells were infected (Fig. 1C, 1D). This is consistent with histopathological findings that showed endothelial cell swelling and degeneration, and microthrombi in dermal capillaries in this location (data not shown). It is plausible that infection of the epidermis takes place via singular keratinocytes in close proximity of infected capillaries in the upper dermis. In the skin of 4 animals, CSFV antigen was detectable between 15 and 22 DPI. However, animal no. 25 showed no immunostaining, which is in contrast to rtRT-PCR and virus isolation. Except for pig no. 21 all other animals developed a low viremia at 4 DPI. Three days later, the content of CSFV in the blood of the infected domestic pigs reached values between 101.75 and 104.0 TCID50/ml (data not shown). Notably, vims isolation was negative in 2 pigs at 15 DPI (pigs nos. 24 and 25) and at 16 DPI (pig no. 25), which had tested positive for CSFV and RNA before. Probably, the moderate neutralizing antibody titers (40 ND50) detected at 15 DPI in these 2 animals were the reason for this negative outcome of vims isolation.
This study demonstrates on the one hand that skin is diffusely infected tissue during the acute course of CSF and diagnosis is possible with skin punch biopsies. The prerequisite for the detection of vims in skin samples is the presence of viremia, which also results in the widespread infection of the dermis and epidermis with endothelium, dermal fibroblasts, and surface or hair follicle epithelium affected. On the other hand, the skin is obviously affected later than other tissues. 6,18 As expected, rtRT-PCR showed the highest sensitivity with the possibility to detect infected pigs beginning at 4 DPI. This corresponds with the early detection of RNA in other organs or blood using rtRT-PCR. 4,5,16 If skin punch biopsy specimens are involved in the diagnostics of CSF, rtRT-PCR should be the method of choice. Vims isolation proved to be less sensitive than rtRT-PCR especially in the early stage of a CSFV infection and within the late infection period when neutralizing antibodies are present. Although FAT is a rapid diagnostic method for detection of viral antigen in cryosections of tonsils and other lymphatic tissues, it is less sensitive than virus isolation, RT-PCR, or rtRT-PCR. 14,20 The efficacy of FAT on skin sections crucially depends on the amount of accumulated CSFV antigen, which obviously increases with time. As a morphologically based method, FAT explains the pathogenesis of multifocal cutaneous infarction and helps to understand the findings of rtRT-PCR in skin biopsies of CSFV infected pigs. The question if skin is a location of CSFV persistence can not be answered by these investigations. However, in the presence of neutralizing antibodies in the chronic or subclinical course of disease it is hypothesized that virus isolation or FAT with cutaneous tissues will yield negative results whereas viral genome could be still detectable here. Further studies are needed to clarify this query.
In conclusion, the diagnostic investigations of skin punch biopsies by virus isolation, rtRT-PCR, and FAT strongly suggest that skin tissue is a suitable target for detection of CSFV infected animals. The ease of collection antemortem compared to blood sampling makes cutaneous punching an interesting alternative for intra vitam CSF diagnostics. As the diagnostic sensitivity of the CSFV rtRT-PCR assay was higher than that of virus isolation and immunostaining of cryosections, RNA detection by rtRT-PCR should be used for diagnosis of CSF based on skin biopsies. Depending on the virulence of CSFV and the stage of the viremia, a reliable positive diagnosis usually cannot be expected within the first days after infection but is consistent towards the end of the first week postinfection.
Acknowledgements. We thank Brigitte Dannenfeld, Gabriele Czerwinski, and Silvia Schuparis for excellent technical support. We are deeply grateful to PD Dr. Stefan Finke for help with confocal laser-scanning microscopy, and U. Polster, DVM, for bacteriological investigation. This work was supported, in part, by funds from the Federal Ministry of Nutrition, Agriculture and Consumer Protection and by the EC project “CSF Vaccine and Wild Boar” (contract number SSP 1–501599).
Footnotes
a.
Tierärztebedarf Lehnecke, Schortens, Germany.
b.
Collection of Cell Lines in Veterinary Medicine, FLI, Riems Island, Germany.
c.
Dr. B. Köllner, FLI, Riems Island, Germany.
d.
Qiagen, Hilden, Germany.
e.
Stratagene, Heidelberg, Germany.
f.
Bio-X Diagnostics, Jemelle, Belgium.
g.
Sigma-Aldrich, Taufkirchen, Germany.
h.
DakoCytomation, Hamburg, Germany.
i.
Dianova, Hamburg, Germany.
j.
Molecular Probes, Invitrogen, Karlsruhe, Germany.
