Abstract
Cerebellar abiotrophies have a nonsex-linked, autosomal, recessively inherited basis in a number of species, and lesions typically reflect profound and progressive loss of Purkinje cells. In this report, an unusual form of abiotrophy is described for two sibling Border Collies. Extensive loss of the cerebellar granular cell layer was present with relative sparing of Purkinje cells of two female pups. The biochemical basis for this form of cerebellar abiotrophy is unknown, but the lack of disease in other siblings supports an autosomal recessive mode of inheritance.
Abiotrophy in the central nervous system results from spontaneous, premature and progressive neuronal degeneration and death, usually attributable to an unidentified intrinsic metabolic defect, and is distinct from programmed cell death that occurs as part of “normal” developmental processes in many tissues. Cerebellar abiotrophies represent a collection of postnatal syndromes that are progressive and incurable, and they occur in a wide range of species. Histologically, cerebellar abiotrophies typically involve a primary loss of Purkinje neurons,1 but a stringent and complex relationship exists between Purkinje cells and granule neurons. Migration of Purkinje cells is dependent on the secretion of reelin by granule neurons, and lack of reelin leads to ectopic Purkinje cells.7 This in turn leads to a loss of granule cells because the number of Purkinje cells largely determines the number of granule neurons, probably because of the production of trophic factor(s) by Purkinje cells.
A purebred Border Collie bitch produced a litter of seven puppies, five female and two male. The bitch and all pups had been vaccinated against parvovirus, distemper virus, canine adenovirus 1, canine parainfluenza virus, and Bordetella bronchiseptica using C5 Virbac (CSL), with the pups vaccinated at 6, 12, and 16 weeks of age. Two female littermates presented with sudden onset of neurologic signs at 4 months of age. Clinical signs were progressive for both puppies. Puppy No. 1 was euthanatized at 5 months and puppy No. 2 at 9 months. The bitch had previously produced several litters with no affected pups, but a new dog sired the current litter.
Puppy No. 1 first presented with an intention tremor, dysmetria and a wide based stance. There was a progressive loss of menace reflex, but no loss of vision or pupillary light reflexes. In puppy No. 2, the owner first noticed trembling and wobbling of the pelvic limbs, which progressed over a 3-month period to periodic falling, difficulty in climbing stairs, and head nodding. At 7 months of age, the puppy was observed to be bright and alert and stood with a wide-based, crouching stance. The puppy exhibited a hypermetric gait that was more severe in the pelvic limbs and was exacerbated at faster gaits. The puppy would fall with sudden turns or stops. A slight side-to-side head nod was present and an intention tremor was observed at times.
For both the puppies, results of a complete blood count, routine serum biochemical analysis, and cerebrospinal fluid tap were unremarkable. Serology for toxoplasmosis was negative for both pups. Puppy No. 2 was also negative for Neospora on serology and exhibited a normal nerve conduction velocity (approximately 50 m/s). A magnetic resonance imaging performed on puppy No. 2 revealed the cerebellum to be mildly reduced in size.
Postmortem evaluation of both the puppies revealed abnormalities confined to the cerebelli, which were symmetrically reduced in size. Histologically, for both the puppies, there was diffuse and profound loss of granular cells (Fig. 1). Within depleted granular layers, scattered nuclear dust, suggestive of apoptotic remnants, was present (Fig. 2). There was slight thinning of the molecular layer and preservation of Purkinje neurons with a subjective increase in Bergmann glia. The depth of white matter in cerebellar folia was reduced, and low numbers of axonal spheroids were present in white matter. For puppy No. 2, the parafloccular lobes exhibited only a moderate loss of granular neurons (Fig. 3). Luxol fast blue and periodic acid-Schiff stains revealed normal myelination within the cerebellar white matter.
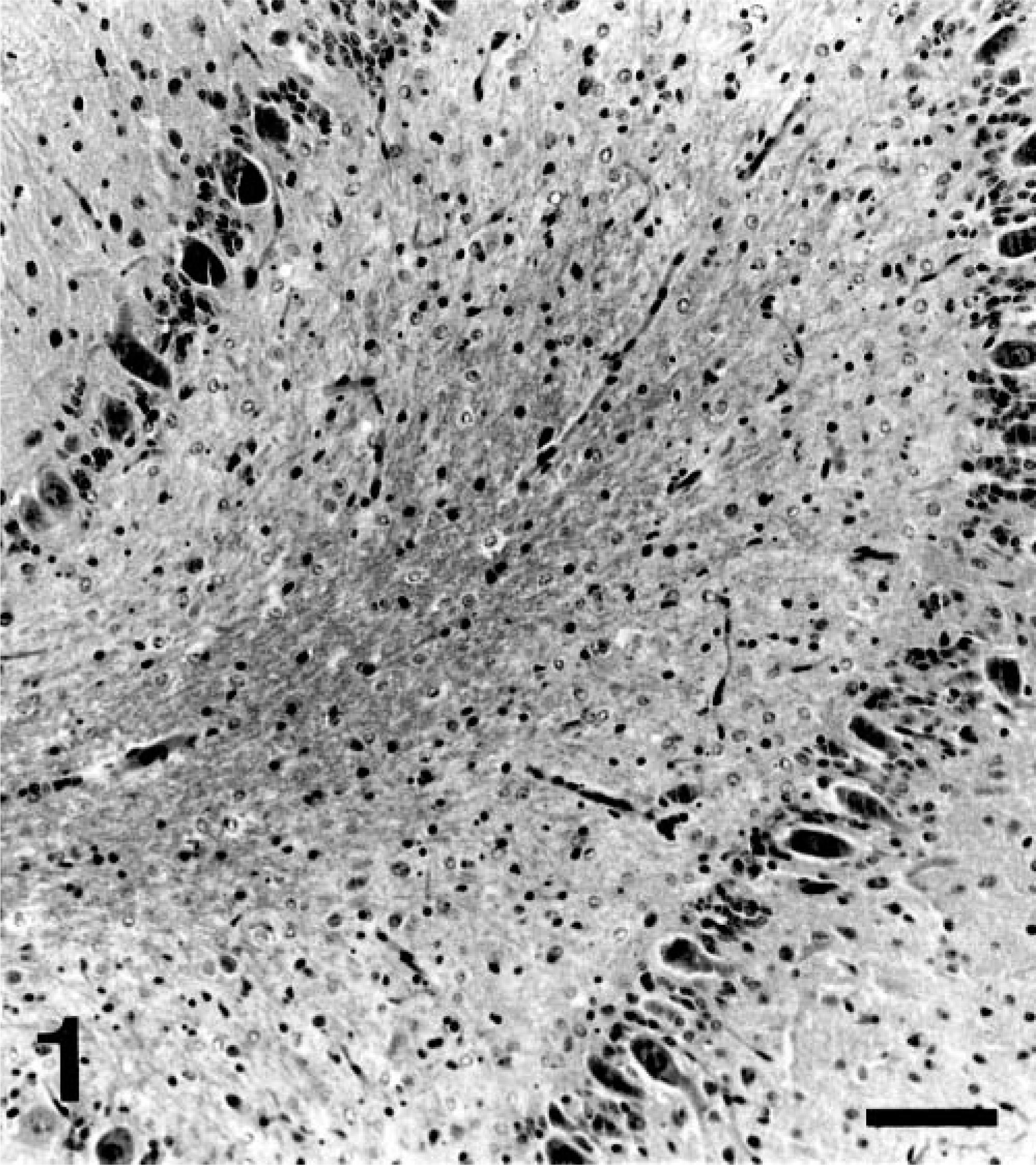
Cerebellar vermis; Border Collie, puppy No. 2. Severe paucity of granular cell neurons, with preservation of Purkinje cell neurons. HE. Bar = 100 μm.
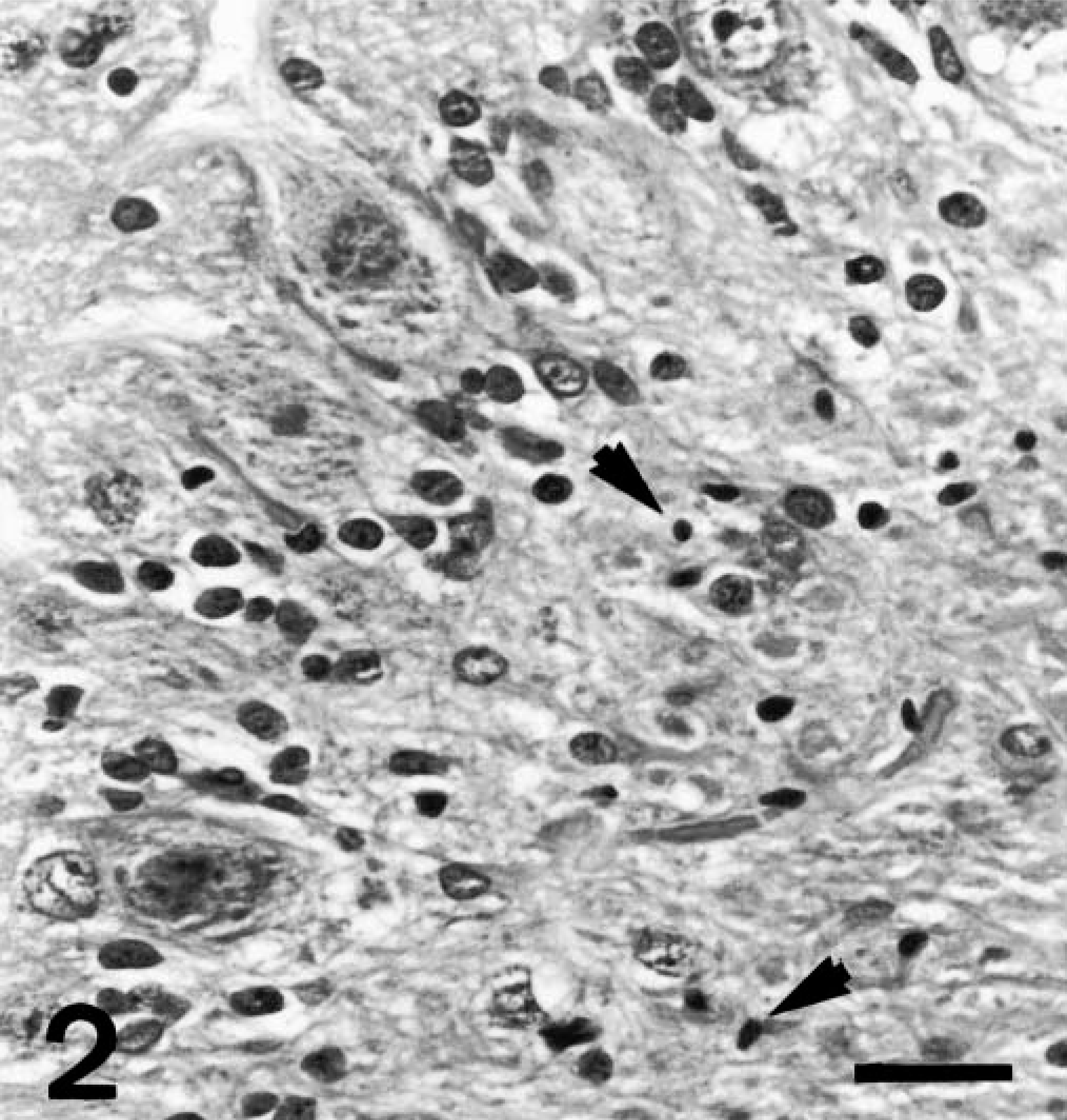
Cerebellar vermis; Border Collie, puppy No. 2. Scattered pyknotic nuclei (arrows), suggestive of apoptotic remnants within depleted granular neuron layer. Normal Purkinje cells. HE. Bar = 30 μm.
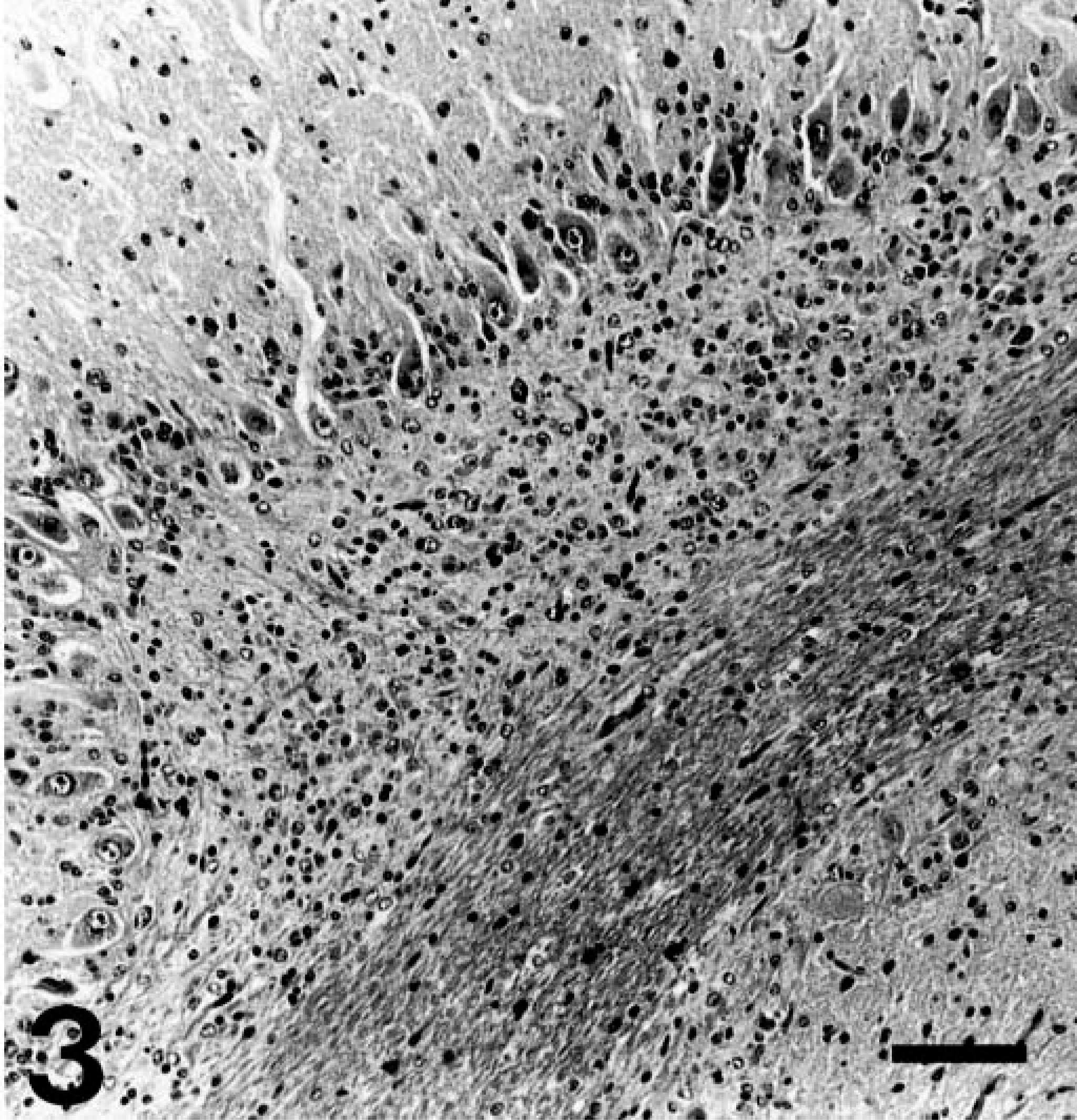
Cerebellar parafloccular lobule; Border Collie, puppy No. 2. Moderate loss of granular neurons. Compare with Fig. 1. HE. Bar = 100 μm.
The pathogenetic mechanisms leading to cerebellar abiotrophies are obscure. Abiotrophies in general include a wide range of conditions in domestic animals including numerous storage diseases.2 In many of these conditions, the specific enzyme defect is well known as well as the mechanism of inheritance. For many dog breeds, cerebellar abiotrophy has been demonstrated to have a nonsex linked, autosomal recessive mode of inheritance. Cerebellar abiotrophy has been previously reported only once in Border Collies in New Zealand;3 therefore, too few animals have currently been reported to confirm such a genetic mechanism, although a similar mode of inheritance is suspected.
In the current case, despite the loss of granule neurons, the architecture of the cerebellar cortex was intact. This indicates that neuronal migration of the different cell types comprising cerebellar grey matter occurred normally. The nonuniform pattern of granule cell loss is typical of cerebellar cortical abiotrophies where lesions are not uniformly distributed, and typically become more severe and widespread in cases where affected littermates have been allowed to progress.1,4 The parafloccular lobules are derived embryologically from the archicerebellum,5 which may contribute to the differences in lesion severity, although the mechanism remains unknown.
A mechanism to explain the primary degeneration of Purkinje cells in cerebellar abiotrophies has been attributable to excitotoxic degeneration.8 The cause for the selective granule neuronal degeneration is less certain, but channelopathies (disorders of ion channels) may be involved. Homozygous weaver (wv/wv) mice exhibit severe loss of cerebellar granule cells. Granule cell precursors proliferate normally in the external granular layer (EGL), exit the cell cycle, and die just before their migration from the EGL.10 It is proposed that a defect in chromosome 16, which encodes for a potassium ion channel (G-protein–coupled inward-rectifying K+ [GIRK-2]),9 leads to failure of GIRK-2 channels to moderate NMDA receptor–based depolarization, resulting in excitotoxicity and massive apoptosis of granule cells.6 But in these two dogs, at least some granule cells migrated appropriately because nuclear dusting was present in the deeper layers of the cerebellar gray matter and was not just restricted to the EGL. Therefore some differences appear to exist between weaver mice and these two dogs.
