Abstract
A pair of rabbits gave birth to a set of littermates (F1) with symptoms of early-onset ataxia. Microscopic examination revealed cerebellar degenerative disease in 5 of 6 littermates. Light microscopy was used to compare the thickness of each cerebellar layer in affected animals in contrast to a normal control. Affected animals showed narrowing of the molecular layer of the vermis, reduced density of Purkinje cell dendrites and irregular thickness in their branchlets, and reduced density of granular cells and scattered pyknotic cells in the granular layer. Pyknotic cells were apoptotic granular cells, confirmed by positive staining using the TUNEL method. Electron microscopy confirmed the thinning of the molecular layer seen by light microscopy and also showed a reduced number of parallel fibers, which indicate granular cells axons, and a reduced number of synaptic junctions between Purkinje and granular cells. Purkinje cells had electron-dense, irregularly shaped cytoplasm with irregularly shaped nuclei, and some of these cells had a central chromatolysis-like region. These findings support a diagnosis of cerebellar cortical abiotrophy, a hereditary condition that causes nerve function impairment leading to early-onset progressive degeneration of the cerebellar cortex.
Dysplastic and degenerative diseases of the cerebellum include hypoplasia, degenerative conditions due to ischemia, infectious agents, autoimmunity, and abiotrophy, a hereditary condition, among others. When endogenous or exogenous factors cause a defect or anomaly in fetal cerebellar components, the cerebellum is generally found to be immature at birth (hypoplasia). For degenerative diseases like abiotrophy, no abnormalities are observed in cerebellar components at birth, but degeneration occurs either soon after birth or upon reaching adulthood. Degenerative diseases of the cerebellum occurring spontaneously after birth in humans are diagnosed as postnatal cerebellar degenerative disease, even in the absence of hereditary evidence. The term cerebellar cortical abiotrophy is used in veterinary medicine for hereditary, early-onset, progressive cerebellar disease occurring postnatally and covers degenerative diseases of the cerebellar cortex, including those with a reduced number of granular cells and Purkinje cells, even when the underlying pathogenesis is unknown. Cerebellar cortical abiotrophy and other degenerative cortical diseases have been reported to occur most frequently in dogs,2–5,7–9,11–13,15,17,20,23,25 with additional reports in cats,3,4,9,23,24 cattle,1,23 sheep, horses, swans, monkeys, 23 alpaca, 14 and many strains of mutant mice.6,10,22,23 All reports fit the definition of abiotrophy in terms of basic morphology, although some differences have been noted in regard to the progression of nerve degeneration, ratio of degenerative Purkinje cells to granular cells, changes in axons or myelin sheaths that indicate a secondary progressive lesion, and possible differences in the pathogenesis.
Histopathological, immunohistochemical, and electron microscopy investigations support a diagnosis of cerebellar degenerative disease with clinical evidence of early-onset ataxia in littermates (F1) delivered from a single pair of rabbits. Repeated mating of the pair confirmed reproducibility of the disease, indicating a hereditary pathogenesis. To our knowledge, hereditary cerebellar degenerative disease and cortical abiotrophy have not been reported in rabbits.
Materials and Methods
Six white rabbit littermates (Wbl:JW) SPF (3 males, 3 females) were examined (Table 1 ). Five of the littermates (rabbit Nos. 1–5) showed ataxia and became emaciated between postnatal days 25 and 31, resulting in the need for intraperitoneal euthanasia with pentobarbital. All 5 were compared with an age-matched euthanatized control rabbit born from a different litter. Central nervous system (CNS) tissues were fixed by intracardiac perfusion for about 30 minutes with approximately 500 ml of 4% paraformaldehyde at the time of euthanasia, followed by exsanguination. After perfusion, the CNS tissues (cerebrum, cerebellum, medulla oblongata, and spinal cord) of 3 affected rabbits (rabbit Nos. 2–4) and the control rabbit were fixed in phosphate-buffered 10% formalin solution for 48 hours. The tissues were transected, embedded in paraffin, and then serially sectioned at 4 μm for light microscopic examination. Representative sections were stained with hematoxylin and eosin (HE); cerebellar sections were prepared with additional stains, including Kluver-Barrera, Bodian, and Holzer. Immunohistochemical stains, including CNP (MAB326, Chemicon International, Inc, Billerica, MA), myelin basic protein (MBP) (as24567, Abcam pls, Cambridge, UK), synaptophysin (No. 61012, Progen Biotechnik GmbH, Heidelberg, Germany), and calbindin (Mob295, DBS, Pleasanton, CA), were applied to cerebellar sections using the LSAB kit (Dako Japan Co, Kyoto, Japan). Sections of paraformaldehyde-fixed cerebellum from 2 affected rabbits (rabbit Nos. 1 and 5) and the control rabbit were processed for electron microscope examination by postfixation in 1% osmium tetroxide (OsO4), dehydration, and embedding in Epon-Araldite. Semithin sections were stained with toluidine blue. Thin sections, approximately 90 nm thick, were stained with uranyl acetate and lead citrate and examined at 80 kV with a Hitachi electron microscope.
Animal Information
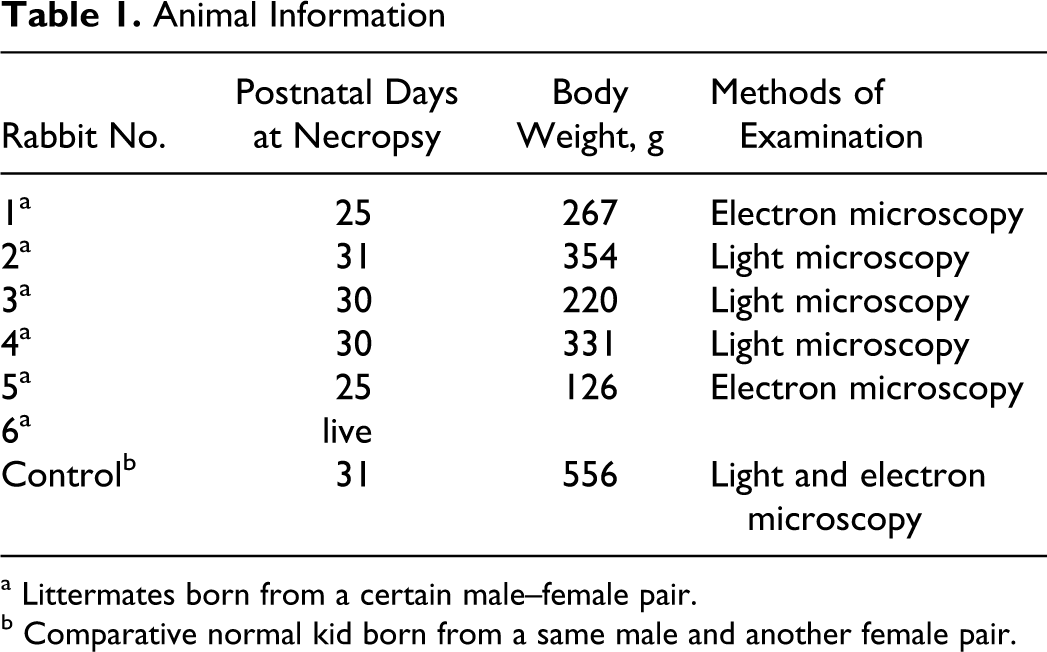
a Littermates born from a certain male–female pair.
b Comparative normal kid born from a same male and another female pair.
Results
Clinical Features
In 5 (rabbit Nos. 1–5) of the 6 rabbit littermates (F1) born from a specific rabbit pair, dystasia and dysbasia started to occur on postnatal day 10. Rabbits were nonambulatory and ataxic. From postnatal day 20, they were recumbent, but not paralyzed, often lifting their heads or thrashing all 4 limbs. Other symptoms included occasional opisthotonus and hypertonia (opisthotonus of the trunk), seizures characterized by thrashing forelimbs, and tension in the hindlimbs and rare nystagmus. The affected rabbits (rabbit Nos. 1–5) showed poor development and lower body weights (<23–64%) than the control rabbit, likely due to an inability to eat by themselves, and were euthanatized between postnatal days 25 and 31 (Table 1). The remaining rabbit littermate (rabbit No. 6) is currently alive and healthy. There was no evidence of a sex predisposition to disease. The same parent rabbit pair mated again, delivering a litter of 6 (F1) (3 males, 3 females). One of the females from this second litter also developed dystasia and ataxia. None of the animals from the second litter were used for this report.
Gross Findings 888
There was no significant difference in gross cerebellar size between affected rabbits and the control at postnatal days 25–31 (Fig. 1 ). No abnormalities were noted in the cerebra, medulla oblongata, or spinal cords of affected rabbits. Gross examination of CNS tissue in cross-section revealed no abnormalities in cerebellar size, lobe formation, or proportion of white and gray matter in the affected rabbits or control.
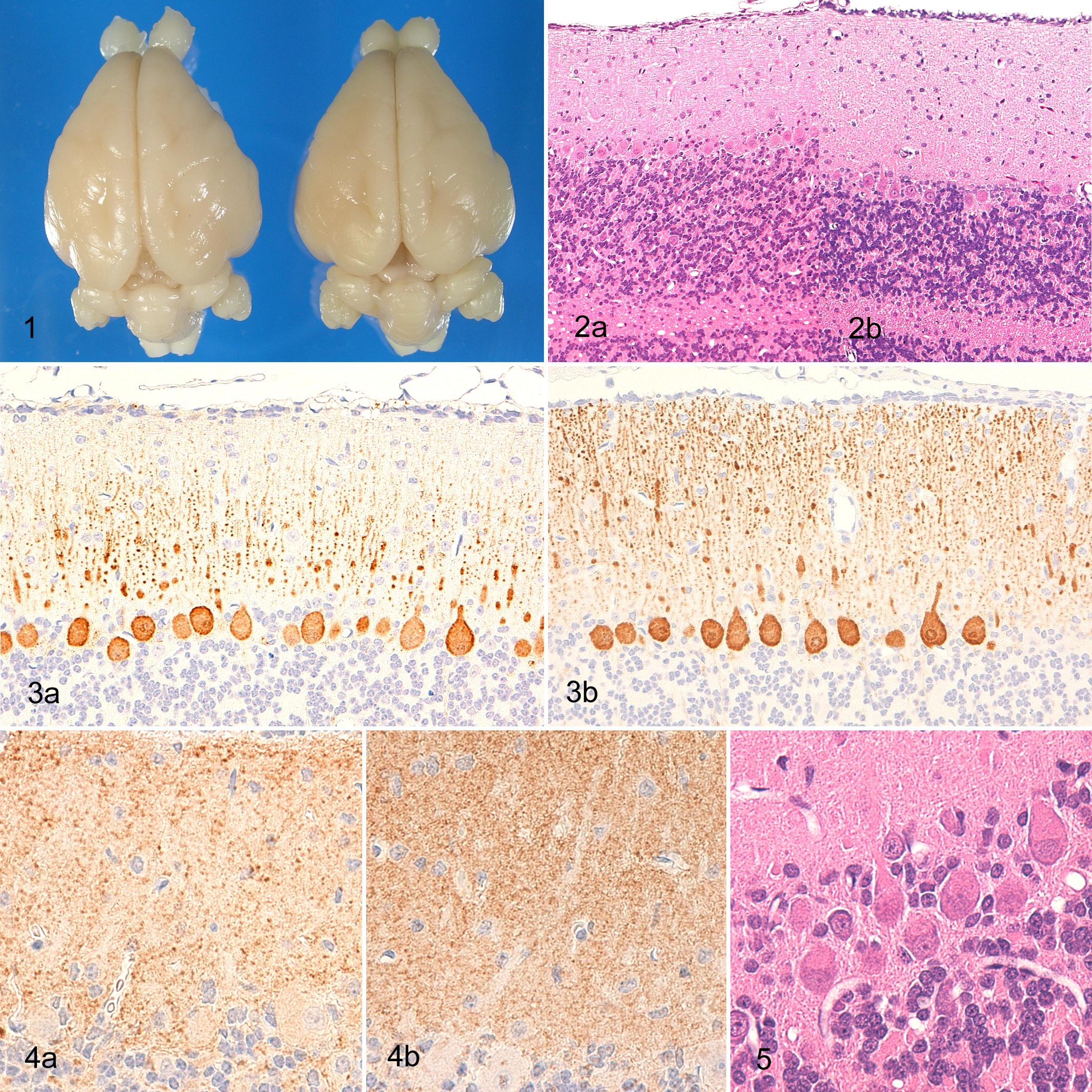
Gross photograph of brain fixed in 10% phosphate-buffered formalin solution; rabbits, right (control) and left (rabbit No. 4). There is no obvious difference in size between the affected and normal brain.
Histological Findings
Microscopically, the width of the molecular layer in the cerebellar vermis was thinner in affected rabbits than in the control (Fig. 2). There was no change in the width of the molecular layer in the remainder of the cerebellum and no change at all in the width of the external granular layer, granular layer, and medulla.
The external granular layer consisted of 1 or 2 flat layers in both the affected rabbits and the control, with no distinct differences between the 2 groups. The external granular layer showed minimal mitosis or apoptosis.
The number of Purkinje cells and their dendrites was easily observed using calbindin-stained sections. The cerebellum was cut in a transverse plane to eliminate sequential observation of dendritic trees from Purkinje cells that branch sagittally to the molecular layer. In affected rabbits, small, positively staining dots representing dendritic shafts extending from Purkinje cells toward the outside of the molecular layer and dendritic branchlets were only present in fragments (Fig. 3), confirming the reduced density of Purkinje cell dendrites in the molecular layer and the irregular thickness of the branchlets in affected rabbits. Synaptophysin-stained sections showed positive staining in the synaptic vesicles. Fewer positive vesicles were observed in the molecular layer of affected rabbits compared with the control (Fig. 4).
In both affected and control rabbits, Purkinje cell bodies formed an incomplete line between the inside of the molecular layer and the granular layer. A similar number of Purkinje cells were observed in both affected and control rabbits. There was no evidence of delayed Purkinje cell migration from the granular layer (ectopic Purkinje cells). Central chromatolysis, indicated by round, clearly demarcated, pale eosinophilic regions that are equal to or larger in size than corresponding Purkinje cell nuclei, was noted in the Purkinje cell bodies (Fig. 5) of the affected rabbits. In rabbit No. 2, small, dark-stained Purkinje cells were observed.
In the granular layer of affected animals, the density of granular cells was reduced (Fig. 6). Pyknotic cells were scattered among the granular cells and showed positive staining by the TUNEL method, indicating that these pyknotic cells are apoptotic granular cells (Fig. 7).
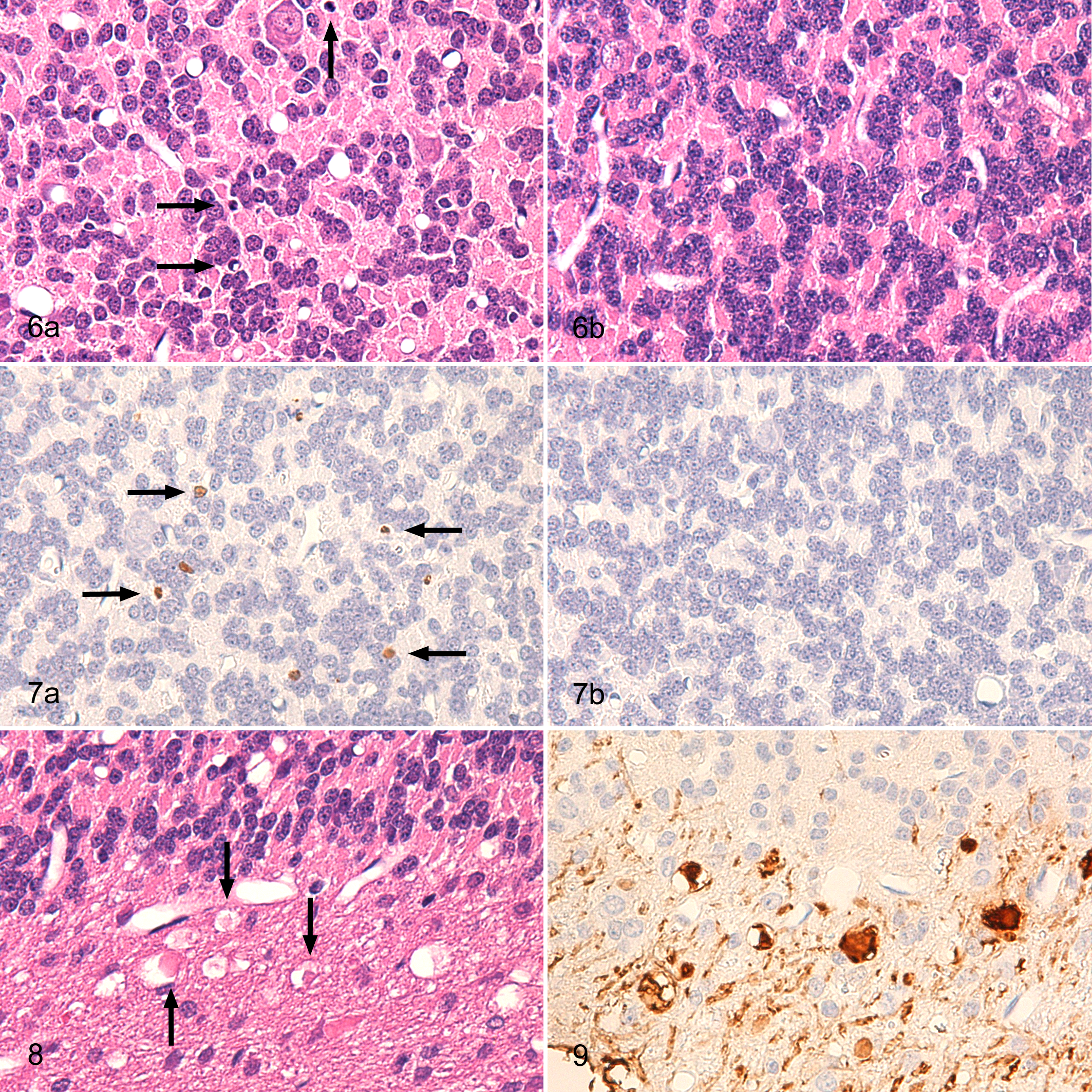
A cross-section of cerebellum; rabbit. (a) Rabbit No. 4; the density of granular cells was reduced in comparison with normal granular layer (b). Pyknotic cells were scattered in the granular layer (arrows). HE.
Round, empty vacuoles and vacuoles containing eosinophilic globules or eosinophilic debris (Fig. 8), ranging in size from 5 to 50 µm, were scattered along the border between the granular layer and medulla and extended into the center of the medulla. Calbindin-stained sections showed positive staining for the eosinophilic globules and debris in the vacuoles (Fig. 9) and were judged to represent degenerated or swollen axons or vacuolated myelin sheaths. Kluver-Barrera–stained and MBP-stained sections showed no demyelination in the medulla, whereas Holzer-stained sections showed no marked gliosis.
Observation of CNP-stained sections showed a slight reduction in oligodendroglia in the granular and molecular layers of affected rabbits compared with the control. Bodian staining showed no significant reduction in nerve fibers in any layer.
There were no degenerative lesions or glial reactions in cerebellar nuclei or the surrounding neuropil in any stained sections.
Ultrastructural Findings
Thinning of molecular layers was represented by a reduced number of parallel fibers representing granular cell axons. Synaptic vesicles and mitochondria were observed at the terminals of the parallel fibers and formed a junction with a spine branching off from a Purkinje dendrite. A high-density “fuzz,” representing postmembranous thickening, was observed on the cytoplasmic side of the cell membrane of the Purkinje dendrite spine. The affected rabbits showed a reduced number of synaptic junctions between Purkinje and granular cells (Fig. 10). In place of the decreased number of parallel fibers, numerous small spines without synaptic junctions filled the gaps in the molecular layer.
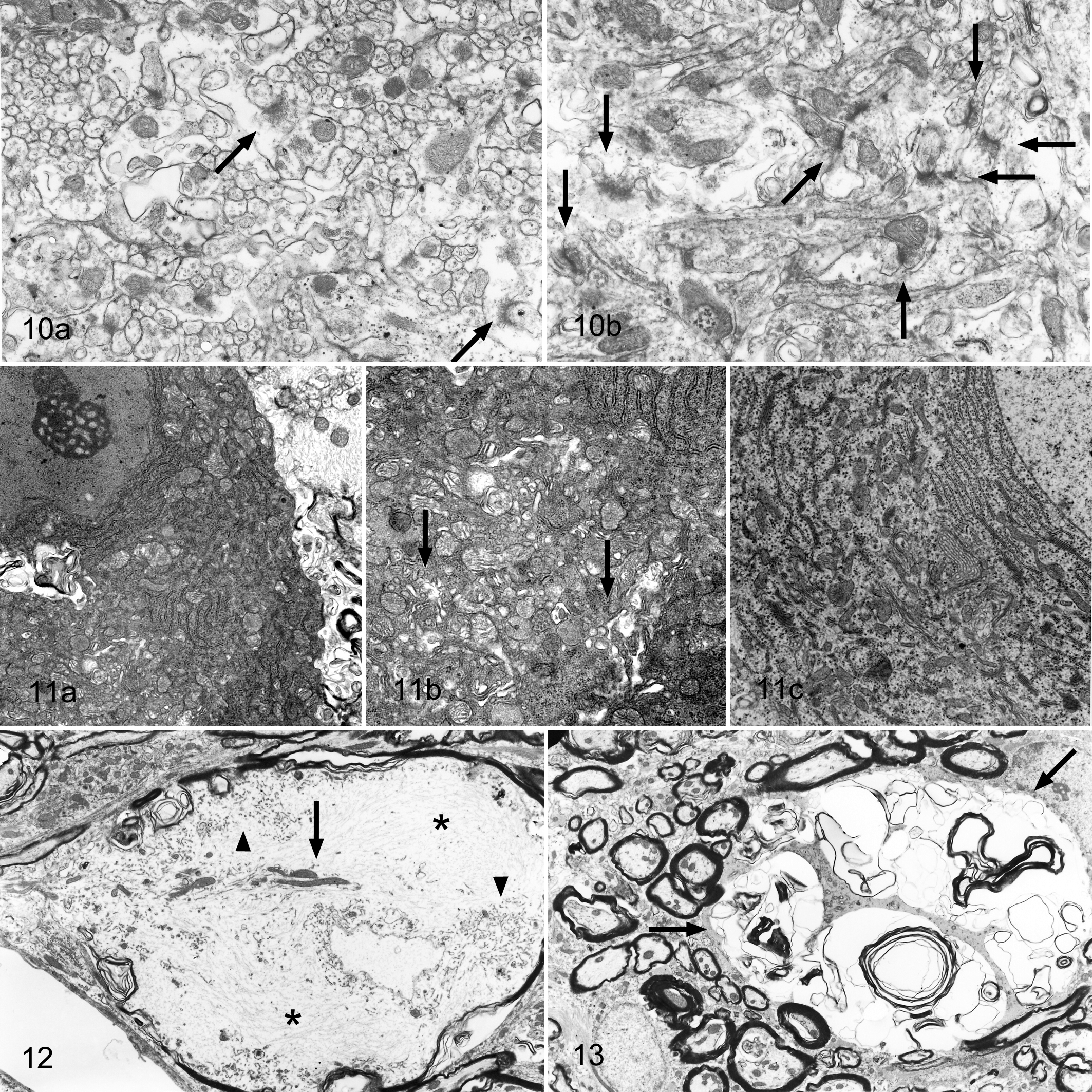
Electron micrograph of cerebellar molecular layer; rabbit. Junctions between Purkinje’s spines and parallel fibers of granular cells (arrows). (a) Rabbit No. 5; the number of synaptic junctions between Purkinje cell spines and parallel fibers was less than in the control rabbit (b).
There were some dark neurons in the Purkinje cell layer of the affected rabbits. The cytoplasm of these Purkinje cells showed high electron density consistent with the aggregation of cytoplasmic organelles, whereas the nuclei were irregular, slender, and marginally arranged in comparison with normal Purkinje cells. Some of these cells had central chromatolysis-like regions, characterized by the loss of rough endoplasmic reticula, expansion of Golgi vesicles, and an increased number of mitochondria (Fig. 11). The granular cells retained normal morphology, even in the affected rabbits, but sometimes showed pyknosis.
In the medulla of affected rabbits, swollen axons were often observed (Fig. 12), characterized by many neurofilaments, mitochondria, and membranous materials. In the granular layer and medulla, vesicles corresponding to those observed by light microscopy contained membranous materials, myelin structures, vesicles, and cytoplasmic debris containing mitochondria (Fig. 13). Electron microscopy appears to support the presumption that these vesicles were degenerated axons and myelin sheaths. Viral particles were not observed in nerve cell bodies or glial cells of the cerebellum by electron microscopy.
Discussion
The cerebellar ataxia in the present study is suspected to be hereditary because it occurred in littermates soon after birth and was reproduced in a subsequent litter born from the same parents. The lesion was characterized by a lack of cerebellar atrophy, an apparent reduction the size of the molecular layer in the cerebellar vermis, abnormalities in Purkinje cell bodies and dendrites, a decreased number of granular cells and their parallel fibers, and occasional degeneration of axons and myelin sheaths. These findings appear to fit with the diagnosis of cerebellar cortical abiotrophy used in the veterinary field, which is a hereditary condition causing nerve function impairment leading to early-onset, progressive degeneration of the cerebellar cortex. 20 Many reported cases of abiotrophy show remarkable degeneration and depletion of Purkinje and granular cells,2,3,5,6,9,12,14,15,17,20,23,25 although some cases show only slight histological changes in spite of drastic clinical signs, as seen in these affected rabbits.1,8,23,24
A single instance of abiotrophy may be caused by a variety of pathogeneses. In these affected rabbits, HE-stained sections revealed no marked loss or remarkable reduction in the number of nerve cells, but immunostaining and electron microscopy identified the presence of abnormalities either in the nerve cells themselves or in the connections between them. The reduced density of Purkinje cell dendrites as well as the irregularity in the thickness of branchlets revealed by calbindin staining suggests that Purkinje cells produced less dendritic branching than normal and that this branching occurred in abnormal directions. The TUNEL-stained sections confirmed the apoptosis of granular cells, suggesting that granular cell numbers slowly decreased without any corresponding glial reactions or other relevant effects. Electron microscopy revealed that Purkinje cell bodies were irregular in shape and stained darkly and that some of these cells showed central chromatolysis. In addition, the number of Purkinje cell spines and synapses at the terminals of granular cells was reduced, as was the number of parallel fibers. These reductions suggest the presence of abnormalities in the connections between Purkinje and granular cells. The synaptophysin-stained samples revealed that positive spots in the molecular layer were narrower in the affected rabbits than in the control, reflecting the reduced number of parallel fiber–Purkinje cell synapses noted by electron microscopy. These findings for Purkinje and granular cells are considered sufficient to explain the drastic clinical signs of cerebellar nerve function disorder noted in the affected rabbits. If cerebellar cortical abiotrophy is defined as a hereditary and progressive disease causing abnormalities in cerebellar cortical nerve cells soon after birth, then the present case appears to represent a form of cerebellar cortical abiotrophy in rabbits.
Hereditary ataxia has been previously reported in rabbits, with clinical signs manifesting at 2–3 months of age and the main lesions situated in the vestibular and cerebellar nuclei and the vestibulocerebellar fasciculus.18,19,21 The present case differs because the main lesions were situated in the cerebellar cortex rather than the cerebellar nuclei and the clinical signs occurred soon after birth.
Diseases resulting in congenital cerebellar ataxia include hypoplasia, neuroaxonal dystrophy, and lysosomal storage diseases as well as cerebellar cortical abiotrophy. Hypoplasia is caused by an exogenous (eg, intrauterine infection) or hereditary defect where certain factors inhibit normal differentiation during fetal cerebellar development. This leads to a loss or reduction of cerebellar cell components or displacement from their normal position. Clinical signs occur when animals begin to walk but are not progressive. Although clinical signs may improve in some cases because of adaptation by other senses, hypoplasia is a dysfunction in which clinical signs remain basically unchanged. 9 Affected rabbits described in the present case had clinical features that occurred early but were progressive with an increasing severity of hypertonia and opisthotonos around postnatal day 30. Such progressive neurological signs are inconsistent with previously described cases of hypoplasia. In addition, the cerebellum of affected rabbits did not show any abnormal gross appearance, suggesting that there was no abnormal cerebellar differentiation before birth, which also suggests that hypoplasia is not the cause of clinical signs in this case.
Neuroaxonal dystrophy, which has been frequently reported in dogs, is a degenerative, early-onset, progressive disease characterized by spheroid formations that indicate nerve fiber degeneration.15,16 Axonal degeneration (spheroid formation) can also occur in abiotrophy,1,20,23,24 suggesting that neuroaxonal dystrophy and abiotrophy share certain common features. In affected rabbits in the present case, eosinophilic globules were observed at the border of the granular layer and medulla. Eosinophilic globules showed positive staining for calbindin, indicating they are of axonal origin, which is consistent with cerebellar spheroids. 16 Electron microscopy confirmed the presence of swollen axons and vacuolated myelin sheaths, although this finding was considered mild because the number of spheroids in affected rabbits appeared lower than that seen in previously reported cases of neuroaxonal dystrophy.15,16 In these affected rabbits, degeneration of axons and myelin sheaths in the medulla may have been a secondary effect caused by the degeneration of Purkinje and granular cells.
Lysosomal storage diseases occur when a lack of specific lysosomal enzymes causes various materials to be deposited in nerve cells and macrophages. They have been frequently reported in dogs and cats. 4 In affected rabbits in the present study, the round, pale eosinophilic regions within the Purkinje cells noted in HE-stained sections were shown to be central chromatolysis by electron microscopy, ruling out the possibility of lysosomal storage disease.
Rabbits typically begin to walk at postnatal day 10, but these affected rabbits were unable to. This allowed for detection of the onset of abiotrophy at an earlier time point than in previous studies. The earliest onset of cerebellar cortical abiotrophy reported to date occurred in Angus cattle that developed convulsions immediately after birth.1,23 In that case, no corresponding morphological changes were noted; obvious changes in Purkinje cells and axons are typically noted in Angus cattle that develop clinical signs several days after birth. In affected rabbits, pathological examinations were conducted at postnatal days 25–31. As ataxia developed, they were unable to compete with normal littermates in suckling during lactation and had difficulty eating by themselves after weaning, which likely explain their subsequent emaciation and early death around postnatal day 30. If they had been able to feed, they may have lived longer and a more obvious depletion of Purkinje and granular cells may have been observed at examination.
The present report is a single-point study of rabbits suspected of having cerebellar cortical abiotrophy around postnatal day 30. The reduced number of parallel fibers and synapses associated with Purkinje cells may merely reflect a reduction in the total number of granular cells. However, the number of granular cells decreased only slightly, whereas the reduction in synapses was remarkable. This finding has important implications for pathogenesis, because there does not appear to be a correlation between the reductions of granular cells and synapses. Abnormalities in the molecular layer may have induced a mild degeneration in Purkinje and granular cells rather than the degeneration of Purkinje and granular cell bodies inducing changes in nerve fibers or synapses in the molecular layer. Considering the progressive nature of abiotrophy, investigations of this condition both before and after the time of observation in the present study will likely aid in the identification of specific pathogenic factors for cerebellar cortical atrophy in rabbits and in other species.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
