Abstract
Cutaneous mast cell tumors (MCTs) are one of the most common tumors in dogs. Currently, prognostic and therapeutic determinations for MCTs are primarily based on the histologic grade of the tumor, but a vast majority of MCTs are of an intermediate grade, and the prognostic relevance is highly questioned. A more detailed prognostic evaluation, especially of grade 2 canine MCTs, is greatly needed. To evaluate the prognostic significance of KIT and tryptase expression patterns in canine cutaneous MCTs, we studied 100 cutaneous MCTs from 100 dogs that had been treated with surgery only. The total survival and disease-free survival time and the time to local or distant recurrence of MCTs were recorded for all dogs. Using immuno-histochemistry, 98 of these MCTs were stained with anti-KIT and antitryptase antibodies. Three KIT- and three tryptase-staining patterns were identified. The KIT-staining patterns were identified as 1) membrane-associated staining, 2) focal to stippled cytoplasmic staining with decreased membrane-associated staining, and 3) diffuse cytoplasmic staining. The tryptase-staining patterns were identified as 1) diffuse cytoplasmic staining, 2) stippled cytoplasmic staining, and 3) little to no cytoplasmic staining. Based on univariate and multivariate survival analysis, increased cytoplasmic KIT staining was significantly associated with an increased rate of local recurrence and a decreased survival rate. The tryptase-staining patterns were not significantly associated with any survival parameter. On the basis of these results, we propose a new prognostic classification of canine cutaneous MCTs, according to their KIT-staining pattern, that can be used for the routine prognostic evaluation of canine cutaneous MCTs.
Introduction
Canine cutaneous mast cell tumors (MCT) are one of the most common neoplasms in dogs, accounting for 7–21% of all cutaneous tumors in dogs. 2,6,15,18 The biologic behavior of canine MCTs is extremely variable. Clinically, MCTs can range from a single benign mass that may be cured with complete surgical excision to fatal metastatic disease. 1,14,29 In one study of 114 dogs, 38% of the dogs died or were destroyed because of the presence of MCT within 2.5 years of diagnosis. 1
Currently, prognostic and therapeutic determinations of MCTs are based primarily on their histologic grade. Several histologic grading systems have been developed in an attempt to correlate the cellular morphology of the neoplasm with the overall survival of the dog. 1,14 The most commonly used grading system defines grade I MCTs as being the most differentiated, whereas grade III MCTs are the least differentiated. Grade II MCTs are of intermediate differentiation. 14 These grading systems have shown a significant difference in survival time between well-differentiated tumors and poorly differentiated tumors, 1,14 but there is still much debate over their relevance, especially when dealing with intermediate-grade MCTs, 11,21,25 which account for more than 40% of all MCTs. 14 The goal of this study was to evaluate the prognostic significance of KIT and tryptase immunohistochemical staining patterns in canine cutaneous mast cell tumors.
The KIT protein is a tyrosine kinase receptor that is a product of the c-kit proto-oncogene, 27 which is expressed in numerous tissues including glioblastoma cells, term placenta, brain, erythroid precursors, melanocytes, basophils, and mast cells. 3,13,27 The ligand for the KIT receptor, stem cell factor (SCF), also called mast cell growth factor, has multiple effects on mast cells, including proliferation, maturation, migration, degranulation, suppression of apoptosis, and adhesion to fibronectin. 3,4,10,12,23,28
Mutations in the juxtamembrane coding region of the c-kit proto-oncogene have been identified in several canine cutaneous MCTs. 5,7,9,16,30 Tandem duplication mutations in this region have been shown to constitutively activate the KIT tyrosine kinase despite the absence of the SCF ligand. 7,9 It has been proposed that mutations in the juxtamembrane domain may play a critical role in the neoplastic transformation of MCTs in dogs. 7,9,30 In one study mutations in the juxtamembrane domain were more prevalent in histologic grades II and III MCTs than in grade I MCTs. 30 In another study MCTs with tandem duplication mutations were twice as likely to recur and twice as likely to metastasize as those without the mutation, although the association between recurrence and metastasis, and the presence of the mutation was not statistically significant. 5
Expression of the KIT receptor in MCTs, and detection of the KIT receptor using immunohistochemistry, has been well established. 8,17 Different patterns of KIT expression have been described in normal mast cells and in neoplastic mast cells. Normal mast cells and some neoplastic mast cells express KIT mainly on the cell membrane, whereas in many neoplastic mast cells KIT accumulates in the cytoplasm, primarily adjacent to the nucleus. 17 A correlation between the expression of the KIT receptor and the histologic grade of MCTs has been made, with well-differentiated tumors weakly expressing KIT and poorly differentiated tumors having a high expression of KIT. 17 We hypothesized that the different patterns of KIT receptor expression in canine cutaneous MCTs correlate with their biologic behavior. We also hypothesized that more benign tumor cells have weak KIT expression limited to the membrane only, and malignant tumor cells have stronger cytoplasmic expression of the KIT receptor.
Tryptase is one of the most common neutral proteases found in mast cells. 19,20 Identification of tryptase using immunohistochemistry has shown excellent specificity and sensitivity to mast cells. 24,26 Poorly differentiated MCTs tend to have fewer granules, and a generally poorer staining pattern when stained with hematoxylin and eosin, toluidine blue, alcian blue, and giemsa, than well-differentiated tumors. 22 It has been assumed that this decrease in the staining pattern is due to the lack of production or storage of cellular components that react with the stain. 22 We hypothesized that well-differentiated MCTs have a stronger cytoplasmic expression of tryptase than poorly differentiated MCTs and that such a difference in expression could be used to predict the biologic behavior of canine cutaneous MCTs. To test these hypotheses a retroprospective study was conducted.
Materials and Methods
Source of MCTs
One hundred cases of canine cutaneous MCTs were selected from over 1,000 cases of MCTs submitted to the Diagnostic Center for Population and Animal Health at Michigan State University between 1998 and 2001. All cases were submitted as routine biopsy specimens from a total of eight different veterinary hospitals. Tissues had been fixed in 10% neutral buffered formalin for an average of 24–30 hours and were routinely dehydrated in graded alcohol and paraffin embedded. Case selection criteria included 1) original diagnosis (by a Michigan State University pathologist) of a cutaneous MCT, 2) surgical excision as the only treatment, 3) availability of sufficient amounts of tissue (formalin-fixed, paraffin-embedded) for additional testing, and 4) complete history and follow-up data. The original diagnosis of canine cutaneous MCT was confirmed independently by two board-certified pathologists and histologically graded based on the Patnaik histologic grading system. A complete history and follow-up information for each case was obtained from the referring veterinarians, including age, sex, breed, weight, number of masses, location of mass, time before excision, medication at the time of surgery, diagnostic tests that were performed, recurrence, tumor margins, metastasis, survival time, and cause of death. For the purpose of the study, recurrence was defined as clinical reappearance of a mass at the initial tumor site. A distant recurrence was defined as the development of an additional mass at a site distant from where the original mass was observed. Ninety-six of the tumors were stained with both antitryptase antibodies and anti-KIT antibodies. Two of the tumors were stained with antitryptase antibodies only, and two of the tumors were stained with anti–c-Kit antibodies only.
Immunohistochemistry
All MCTs had been fixed in 10% neutral buffered formalin. Sections were cut to a thickness of 5 µm, deparaffinized in xylene, rehydrated in graded ethanol, and rinsed in distilled water. Endogenous peroxidases were neutralized with 3% hydrogen peroxide for 5 minutes followed by rinsing for 5 minutes in distilled water. Antigen retrieval was achieved by incubating slides in antigen retrieval solution in a steamer (Black & Decker, Towson, MD) for 20 minutes. Nonspecific immunoglobulin binding was blocked by incubation of slides for 10 minutes with a protein-blocking agent (Dako, Carpinteria, CA) before application of the primary antibody. Using an autostainer, slides were incubated for 30 minutes with a mouse anti-human mast cell tryptase antibody (Dako) and a rabbit anti-human c-kit antibody (Dako) at dilutions of 1 :100. A streptavidin–immunoperoxidase staining procedure (Dako) was used for immunolabeling. The immunoreaction was observed with 3,3′-diaminobenzidine substrate (Dako). Sections were counterstained with Mayer's hematoxylin. Positive immunohistochemical controls included known MCTs. Negative immunohistochemical controls (Fig. 4) were known MCTs treated identically as routine sections, with 20 minutes antigen retrieval and 10 minutes protein blocking, except that the 30-minute incubation with primary antibodies was replaced by a 30-minute incubation with buffer.
Negative control of cutaneous MCT with no visible staining.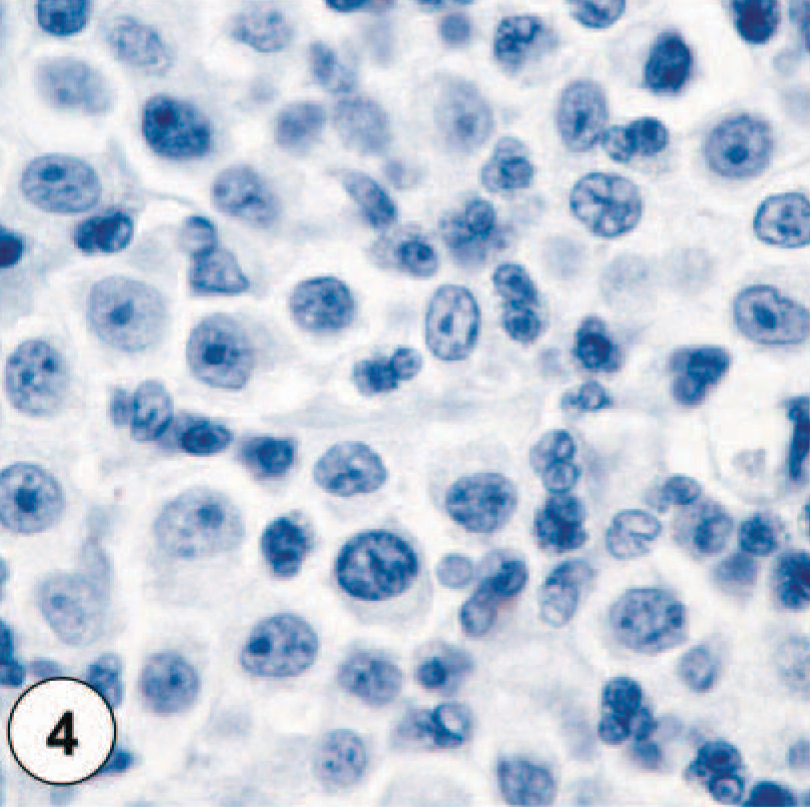
Grading of c-kit–staining pattern
Nonneoplastic mast cells exhibit faint staining of their membrane for KIT and have no cytoplasmic staining. MCTs were divided into three groups based on their KIT-immunostaining pattern (Figs. 1–4). MCTs with KIT-staining pattern I consisted of neoplastic mast cells that stained primarily along the cytoplasmic membrane with only minimal cytoplasmic staining (Fig. 1). The staining intensity for this pattern varied from neoplastic mast cells with extremely faint membrane-associated staining and no cytoplasmic staining to cells with intense membrane-associated staining and small amounts of cytoplasmic staining. KIT-staining pattern II was characterized by neoplastic mast cells with either an intense, focally clustered cytoplasmic KIT staining or a strong positive stippling throughout the cytoplasm (Fig. 2). Membrane-associated staining was not as prominent a feature of these cells. KIT-staining pattern III was defined by neoplastic mast cells with diffuse cytoplasmic staining, obscuring all other cytoplasmic features (Fig. 3). Each MCT was assigned one of these three staining patterns based on the highest staining pattern (staining patterns I versus II versus III) present in at least 10% (estimated based on 100 neoplastic cells in a high-power field) of the neoplastic cell population or present in large clusters of neoplastic cells within the tumor. Cells on the margins of the tissue sections were not considered for classification to avoid possible artifactual staining.
Immunohistochemically stained sections of canine cutaneous mast cell tumors. 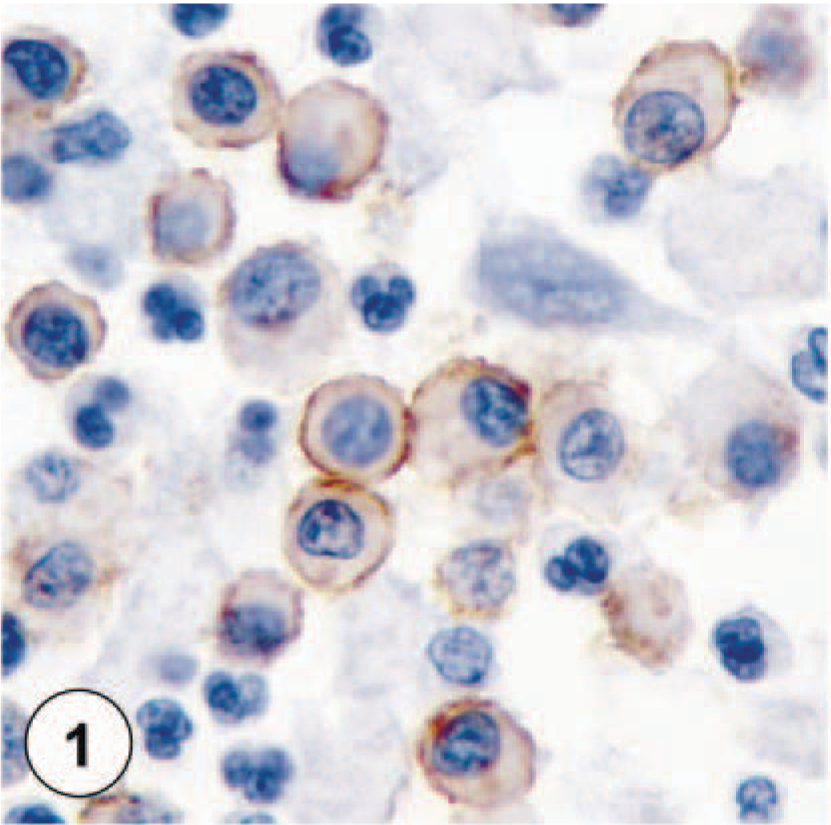
KIT-staining pattern II, characterized by intense focal or stippled cytoplasmic staining of neoplastic mast cells.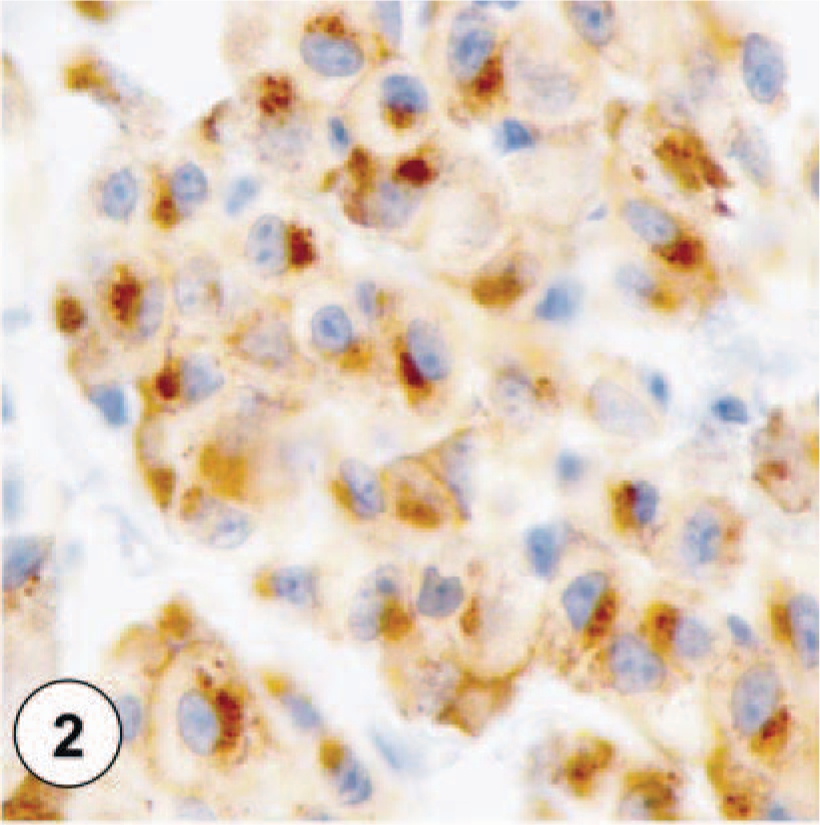
KIT-staining pattern III, characterized by diffuse cytoplasmic staining of neoplastic mast cells.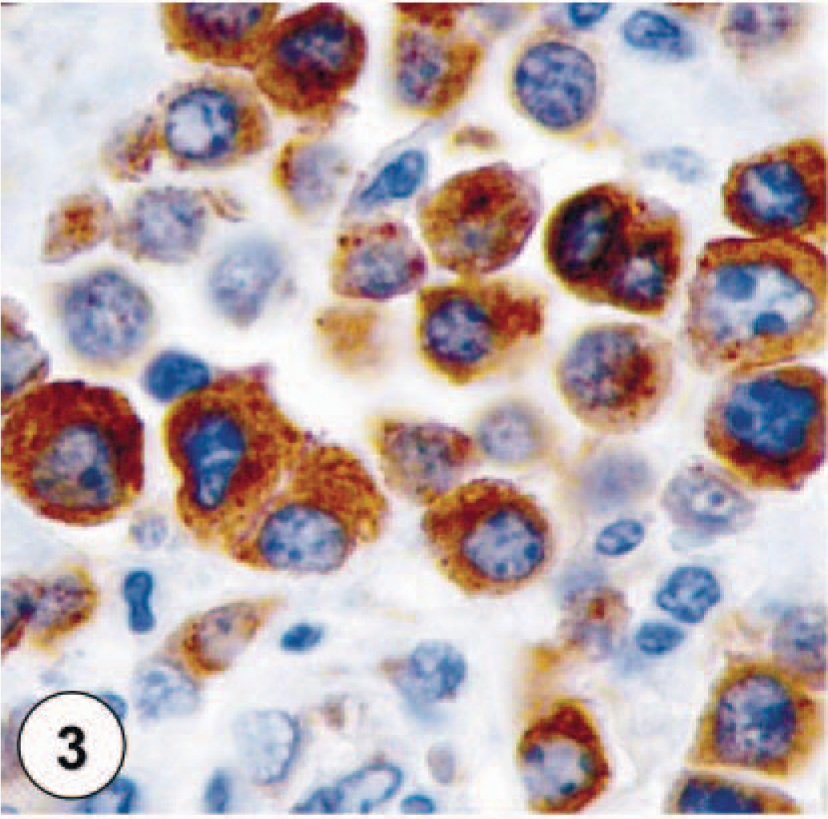
Grading of tryptase-staining pattern
MCTs were classified into three groups based on their different tryptase-staining patterns. Tryptase-staining pattern I was characterized by neoplastic mast cells that were diffusely positive for tryptase throughout the cytoplasm, obscuring all other cytoplasmic features (Fig. 5). MCTs with tryptase-staining pattern II consisted of neoplastic mast cells with moderate amounts of stippling throughout the cytoplasm. In MCTs with tryptase-staining pattern III, neoplastic mast cells had weak cytoplasmic stippling (Fig. 6). Because of the presence of occasional degranulated cells within MCTs, each MCT was assigned to one of these three tryptase-staining patterns based on the predominate staining found in the majority of neoplastic cells throughout the section instead of being classified based on the highest staining pattern present. Cells on the margins of the sections were not considered for classification to avoid possible artifactual staining.
Tryptase-staining pattern I, characterized by diffuse cytoplasmic staining of neoplastic mast cells.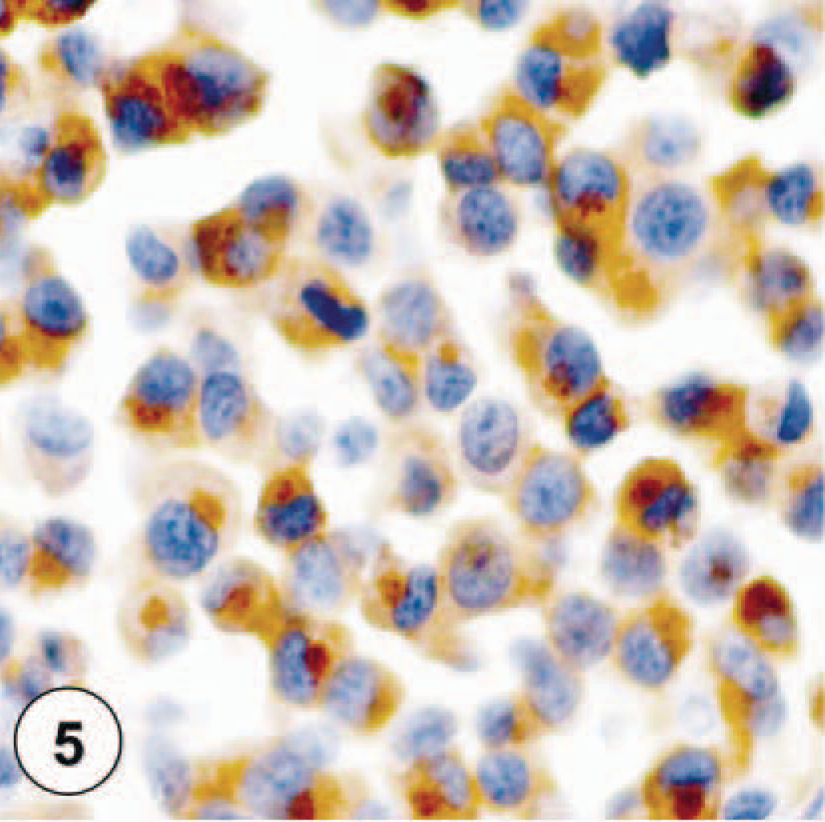
Tryptase-staining pattern III, characterized by weak cytoplasmic stippled staining of neoplastic mast cells.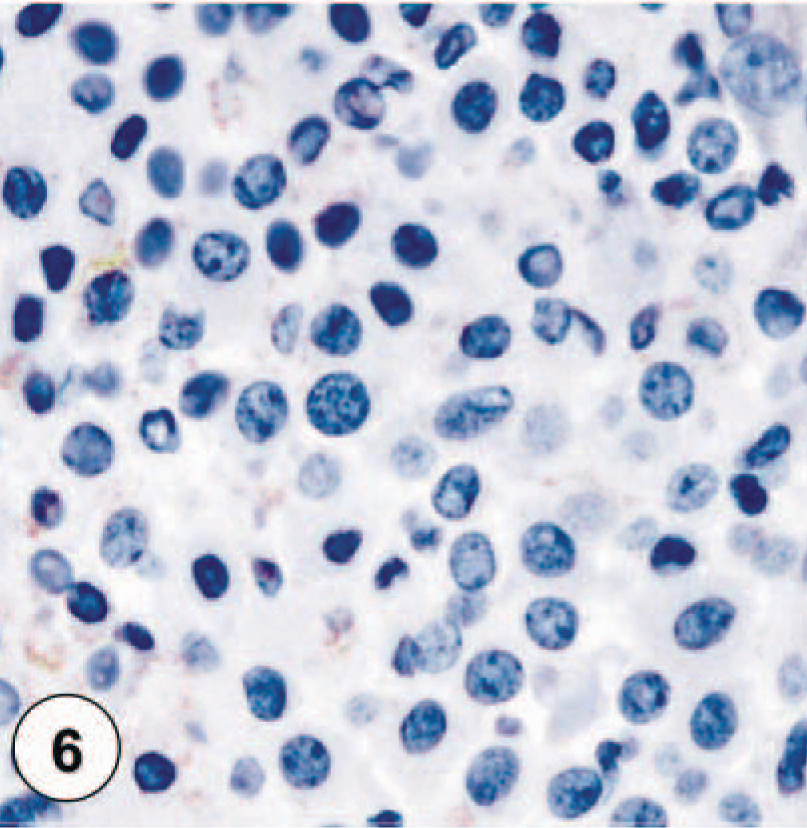
Statistics
Univariable analyses
Before developing multivariable models, each risk factor (prognostic factor) was evaluated for its association with MCT outcomes. Univariable proportional hazards models were developed for each risk factor for each outcome, and the level of association was assessed through the risk factor's
Multivariable survival analysis models
Proportional hazards regression models were developed for survival analysis of different outcomes associated with MCTs. These outcomes included days to local recurrence of MCT, days to distant recurrence, and days to death associated directly with MCT or with complications arising from MCT. Additionally, models were developed for days to death by any cause (including MCT), for comparison with days to death associated with MCT.
Results
A total of 100 cases of canine cutaneous MCTs were included in this study. There was no age or sex predilection in this study. The sex distribution of the cases consisted of 7 intact and 49 spayed female dogs, as well as 5 intact and 38 castrated male dogs and one male dog with an unknown alteration status. A total of 22 dog breeds were represented in this study, including 24 Labrador Retrievers, 12 Boxers, 9 Golden Retrievers, 5 Boston Terriers, 3 Basset Hounds, 3 Cocker Spaniels, and 22 mixed-breed dogs. Fifteen additional breeds were represented by one or two dogs each. The distribution of the cases according to the Patnaik histologic grading system consisted of 17 grade I, 72 grade II, and 11 grade III MCTs.
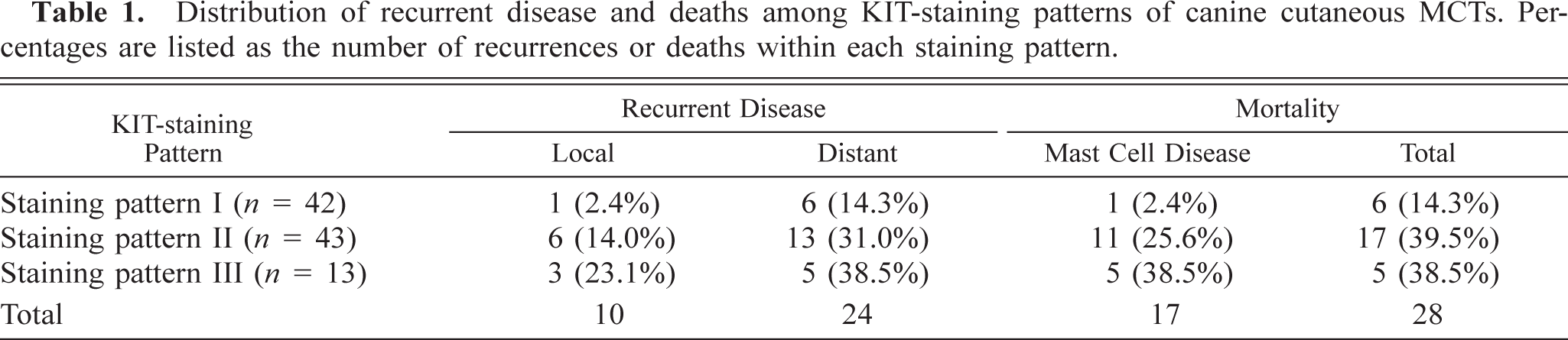
The majority of MCTs in this study had either KIT-staining pattern I (42.9%) or KIT-staining pattern II (43.9%) (Table 1). A total of 28 of 98 (28.6%) dogs died during the follow-up period. Of these, 17 (60.7%) dogs died because of complications associated with their MCTs. Thirty-four (34.7%) of 98 MCTs from 98 dogs that had been stained for KIT had recurrent MCTs.
Distribution of recurrent disease and deaths among KIT-staining patterns of canine cutaneous MCTs. Percentages are listed as the number of recurrences or deaths within each staining pattern.
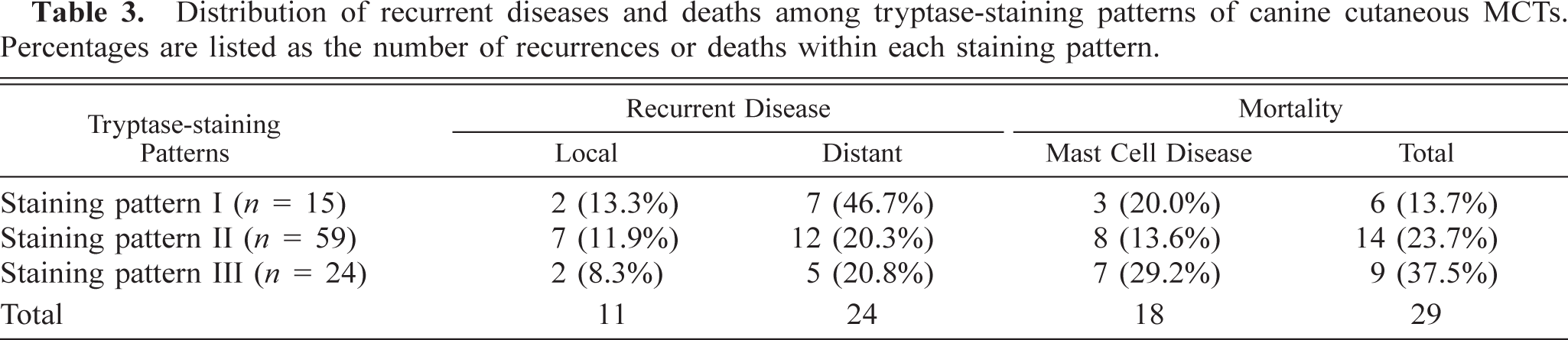
Fifteen of 98 (15.3%) MCTs were classified as having tryptase-staining pattern I, 59 (60.2%) MCTs were classified as having tryptase-staining pattern II, and 24 (24.5%) MCTs were classified as having tryptase-staining pattern III (Table 3). Thirty-five (35.7%) of 98 MCTs from 98 dogs that had been stained for tryptase had recurrent MCTs. Eleven (31.4%) of these dogs had local recurrence at the site of the previous tumor, whereas 24 (68.6%) of these dogs had recurrence at a distant site. A total of 29 of 98 (29.6%) dogs died during the follow-up period. Of these, 18 (62.1%) dogs died because of complications associated with their MCTs.
Distribution of recurrent diseases and deaths among tryptase-staining patterns of canine cutaneous MCTs. Percentages are listed as the number of recurrences or deaths within each staining pattern.
Using uni- and multivariate survival statistics, the KIT-staining patterns II and III were both significantly associated with a shorter overall survival time because of mast cell disease (Table 2, Fig. 7) and a higher risk of local recurrence of MCTs (Table 2, Fig. 8). Of the three classes of tryptase-staining patterns, none were found to be significantly associated with disease-free survival, overall survival of the animal, or local recurrence (Table 3).
Multivariate analysis results of KIT-staining patterns II and III in canine cutaneous MCTs' predictive ability for recurrence, death due to MCT, and death due to any other cause.
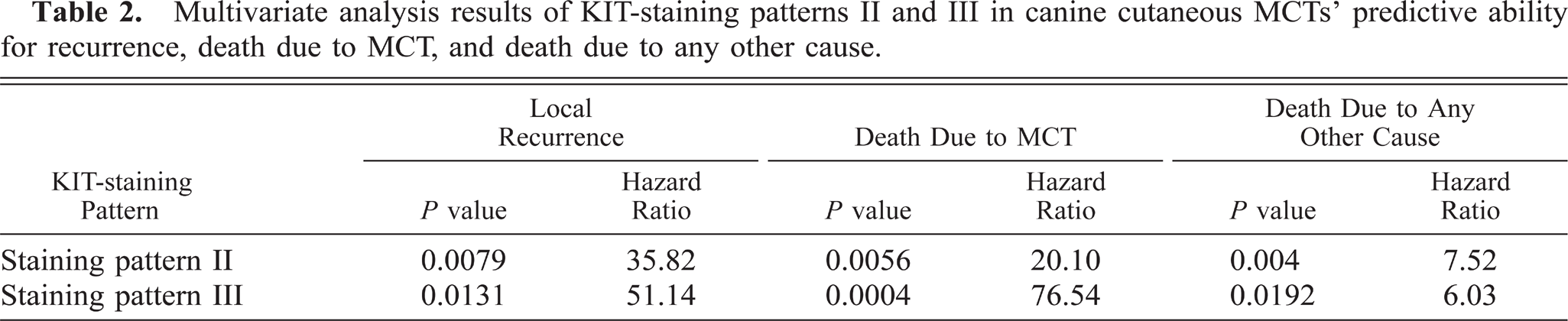
Overall survival curve for dogs with cutaneous MCTs with different KIT-staining patterns. Circles denote point of time at which survival was censored.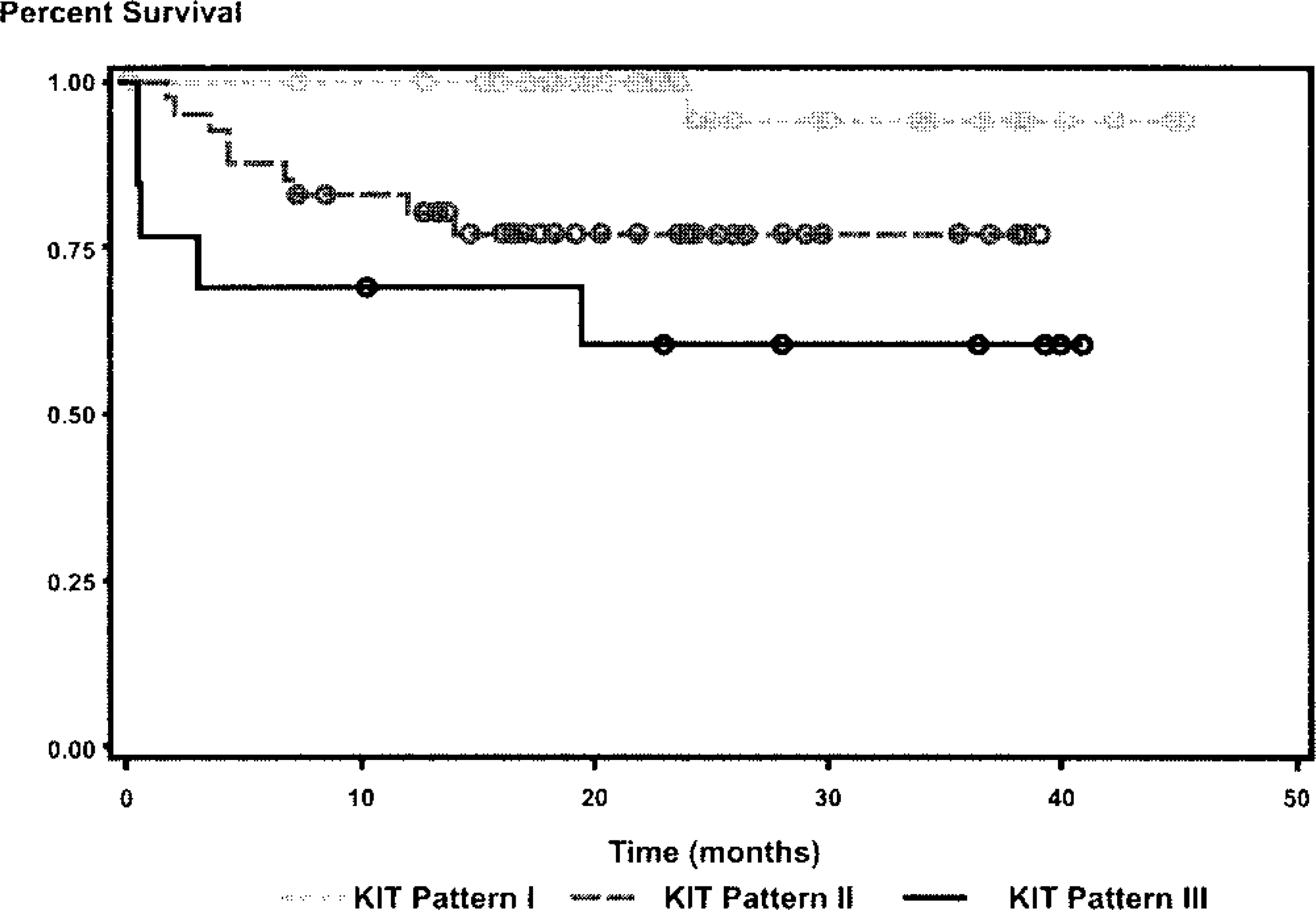
Local recurrence survival curve for dogs with cutaneous MCTs with different KIT-staining patterns. Circles denote point of time at which survival was censored.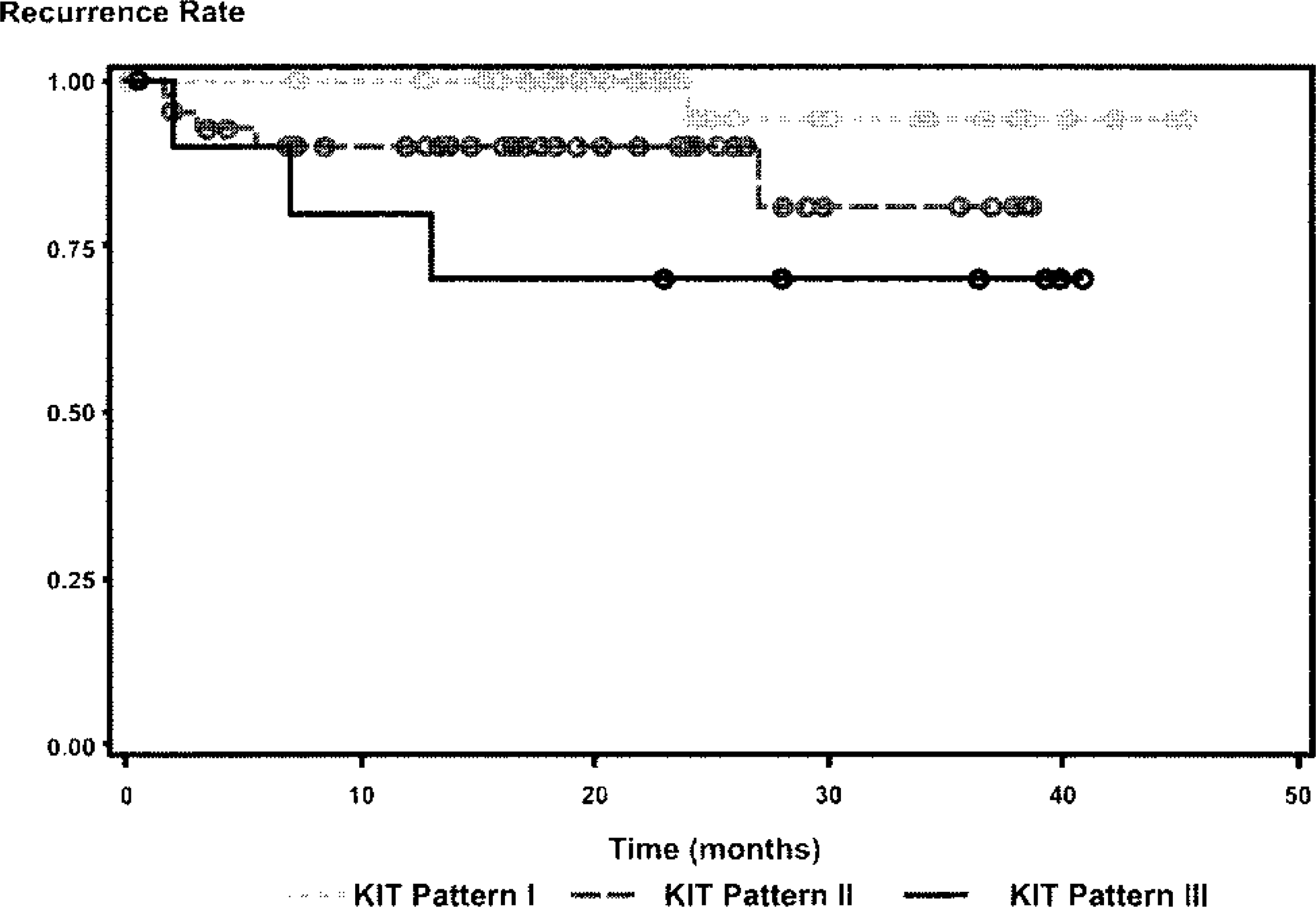
Discussion
Our current results offer a new variable upon which a novel classification of canine cutaneous MCTs can be made. In this study we have shown that perimembrane KIT staining of neoplastic mast cells is not associated with recurrence or a decrease in survival time but that cytoplasmic stippling with decreased perimembrane staining and diffuse cytoplasmic KIT staining are associated with both an increased rate of recurrence and a decreased survival time. With these results we propose a new system for the prognostic grading of canine cutaneous MCTs based on KIT-immunostaining patterns. This system can be used as a valuable tool for the routine prognostic evaluation of canine cutaneous MCTs and to help clarify the ambiguity of the current histologic grading system. Additionally, our results show that tryptase-staining patterns have no prognostic value in the evaluation of canine MCTs.
The KIT receptor is a transmembrane protein, 27 and as such, immunoreactivity of this protein is localized to the cytoplasmic membrane of nonneoplastic mast cells. In dogs, nonneoplastic mast cells have been shown to express KIT exclusively along the cell membrane, whereas MCTs histologically graded as II or III according to Patnaik et al. 14 predominately expressed KIT in their cytoplasm. 17 Our results confirm that a more aggressive biologic behavior of canine cutaneous MCTs is associated with the increase in cytoplasmic staining for KIT. In this study membrane-associated staining for KIT was not associated with local or distant recurrence of MCTs or decreased survival time. This coincides with the idea that the KIT receptor is present along the cytoplasmic membrane of well-differentiated mast cells, and therefore, neoplastic mast cells with predominately membrane-associated KIT expression have a lower degree of malignant transformation and exhibit benign biologic behavior. In contrast, this study demonstrated that canine cutaneous MCTs with increased expression of KIT in the cytoplasm of neoplastic mast cells had an increased risk of local recurrence, and a decreased survival time, both overall and owing to complications of the mast cell disease.
The more aggressive biologic behavior of MCTs with increased KIT expression may be explained by the functional roles that KIT and its ligand, SCF, play in mast cell development. KIT and SCF have been shown to mediate numerous roles in mast cell development, including proliferation, maturation, inhibition of apoptosis, adhesion, and migration. 4,10,12,23,27,28 The exact mechanism by which increased KIT expression causes the malignant transformation of MCTs is unknown. As has been suggested by others, 17 we hypothesize that the cytoplasmic isoform of KIT may be activated by soluble SCF or may contain constitutively activating mutations, which then lead to the inhibition of apoptosis and mast cell adhesion, migration, and proliferation. Further studies are needed to define the exact molecular role that this increase in KIT expression plays in the neoplastic transformation of canine MCTs.
Several studies of canine MCTs have described mutations in the c-Kit proto-oncogene. 5,7,9,16,30 These mutations produce a constitutively activated product 7,9 and have also been suggested to play a role in the malignant transformation of MCTs. 30 Our results suggest that an overexpression of the c-kit gene may be a key factor in the malignant transformation of MCTs. It is currently unknown whether mutations in the c-kit proto-oncogene and the increased expression of the c-kit gene are codependent or independent events. Additionally, if these are independent events, it is unknown whether there is a synergistic relationship between the two events, resulting in a more aggressive biologic behavior.
Cellular differentiation has been commonly used as an indicator of the biologic behavior of neoplasms. In previous studies it has been demonstrated that poorly differentiated MCTs tend to have fewer cytoplasmic granules than well-differentiated tumors. 22 Because tryptase is one of the most common neutral proteases present in mast cell granules, we hypothesized that MCTs with decreased staining with antitryptase antibodies are poorly differentiated and therefore have a more malignant biologic behavior and a worse prognosis. However, this study did not demonstrate a significant association between the tryptase-staining pattern and the biologic behavior of canine cutaneous MCTs after surgical removal.
The biologic behavior of canine cutaneous MCTs is highly variable, and our current ability to give an accurate prognosis for these tumors is hampered by the large number of intermediate-grade MCTs according to the histologic classification. Our studies demonstrate that the evaluation of the immunohistochemical staining pattern of KIT can improve the prognostication of canine cutaneous MCTs. Expression of KIT was prognostically significant for surgically removed canine cutaneous MCTs. Therefore, we propose a new prognostic classification of canine cutaneous MCTs based on their KIT-staining pattern. For canine cutaneous MCTs that have been treated with surgical removal only, increased cytoplasmic KIT staining is a strong indicator for local recurrence and a shorter survival period, therefore supporting a more aggressive therapy of such neoplasms, such as radiation. Further studies are necessary to validate this classification for other treatment protocols and to elucidate the role of increased KIT expression in the tumorigenesis of canine cutaneous MCTs.
Footnotes
Acknowledgements
We thank S. Scot Marsh and Tom Woods for their technical help.
