Abstract
Mast cell tumors (MCTs) are an uncommon primary neoplasm of the nasal cavity in dogs for which there is a paucity of existing literature regarding their clinical behavior and molecular features. The objectives of this retrospective study were to examine the clinical findings, histopathologic and immunohistochemical features, and
Mast cell tumors (MCTs) account for 7% to 21% of all canine neoplasms 27 and exhibit widely variable biologic behavior. The anatomic location of these tumors can be a predictor of their biologic behavior: tumors arising in the oral cavity, mucocutaneous junctions, gastrointestinal tract, or viscera bear the poorest prognoses,6,8,18,21,27 while subcutaneous MCTs are generally regarded as less aggressive than their cutaneous counterparts. 22 MCTs arising in mucosal surfaces or mucocutaneous junctions are associated with a higher incidence of lymph node metastasis at the time of diagnosis.6,8
Previous studies investigating the behavior of mucosal MCTs focused on MCTs arising from the oral or gastrointestinal mucosa, and there remains little information about the behavior and molecular features of MCTs that arise in the nasal mucosa. A previous retrospective study of 4 dogs with primary intranasal MCTs found that survival times ranged from 27 to 134 days, and regional lymph node involvement was reported in 2 cases.
9
No distant metastases were reported, and no investigation into the proliferation indices or
ITDs in the juxtamembrane regions of exon 8, 9, 11, and 12 have been reported in canine cutaneous MCT.4,11,12,25 Of these, exon 11 is the most commonly implicated.4,25 In 1 study of 191 canine cutaneous MCTs, of the 50 tumors that contained
Ki67 is a nuclear protein expressed by cells currently in the cell cycle and not by non-cycling cells, and the proportion of Ki67-positive cells has historically been used as a means of quantifying cell proliferation in a tissue.
20
Argyrophilic nucleolar organizing regions (AgNORs) are nucleolar units involved in ribosomal RNA synthesis
2
that can be identified with a silver-based staining method, and the quantity of silver-stained NOR proteins per nucleus has been positively correlated with the rate of cell replication and tumor mass doubling time.3,16,23,24 Thus, while Ki67 immunostaining allows quantification of the cells currently in the cell cycle, AgNOR histochemical staining measures the rate of cell proliferation, or the generation time. In 1 study of 56 canine cutaneous MCTs, increased Ki67 and AgNOR were associated with decreased survival.
26
In this same study, MCTs tended to have increased expression of cellular proliferation markers if they had aberrant KIT localization or ITDs in the
The aims of our retrospective study were to further evaluate the clinical behavior of canine intranasal MCTs in a larger population, as well as to investigate the
Materials and Methods
Biopsy and necropsy submissions to the Veterinary Diagnostic Laboratories at Colorado State University between 2010 and 2019 were considered for this study. Criteria of inclusion in this study consisted of canine cases with intranasal neoplasms diagnosed as MCTs in patients with no reported history of primary cutaneous, visceral, or oral MCT. All intranasal neoplasms had been fixed in 10% neutral buffered formalin and routinely processed for histopathology. In all cases, slides were routinely stained with hematoxylin & eosin (HE). Toluidine blue stain was utilized in cases with poorly granulated cells to confirm the diagnosis of MCT. Sample dimensions were measured with Phillips Digital Pathology software on scanned slides, and these are reported in Supplemental Table 1.
Cases were reviewed by 2 board certified veterinary pathologists and 1 veterinary pathology resident. Histologic evaluation consisted of assessment of tumor location (i.e., mucosal epithelium or lamina propria) and invasiveness into underlying turbinate bone, mitotic count (as determined by the number of mitoses in a 2.37 mm2 area
15
), degree of cytoplasmic granularity with HE stain, presence or absence of anisocytosis and anisokaryosis, presence or absence of karyomegaly (considered
Patient history obtained from the medical records of the submitting veterinarians included the sex, age, breed, tumor location, comorbidities, and any metastases confirmed with biopsy or fine needle aspiration at the time of diagnosis. Follow-up information obtained from the submitting veterinarians included patient survival times, any treatments the patient received for their MCT-related disease, and reports of metastases (as confirmed by biopsy or fine needle aspiration) following their diagnosis with intranasal MCT.
Immunohistochemistry for CD117 and Ki67 was routinely performed by the histology laboratory at the Colorado State University Veterinary Diagnostic Laboratories as previously described by Webster et al 26 and Kiupel et al, 10 respectively. Immunohistochemistry for AgNOR was routinely performed at a reference laboratory and as previously described in Pich et al. 19 Cases were grouped according to their Ki67 proliferation index and CD117 immunostaining patterns. Ki67 immunopositivity in <30% of neoplastic cells was classified as “low,” in 30% to 60% as “moderate,” and in >60% as “high.” CD117 immunostaining patterns were distinguished based on the peri-membrane (pattern 1), focal (pattern 2), and diffuse cytoplasmic (pattern 3) localization patterns established by Kiupel et al. 10 Because previous studies found no significant difference between focal and diffuse cytoplasmic CD117 immunostaining patterns with regard to histologic grade, cellular proliferation, tumor necrosis, or epidermal ulceration, 7 we chose to combine the groups expressing patterns 2 and 3 into one conglomerate denoted as “atypical” CD117 localization.
Polymerase chain reaction (PCR) for ITDs at exons 8 and 11 of the
Results
A total of 1849 primary intranasal neoplasms were identified between 2010 and 2019 (Supplemental Fig. 1). Twenty (1%) of these neoplasms were primary intranasal MCTs (Supplemental Table 1). Furthermore, MCTs represented 23.8% of all primary round cell neoplasms of the nasal cavity (Supplemental Fig. 2). Males were overrepresented (14/20 cases; 70%), and no breed predilection was observed. In cases with unilateral nasal MCT, the left nasal cavity was more frequently affected than the right (8/14 cases; 57%).
In dogs diagnosed with intranasal MCT, the most frequently reported clinical signs at the time of initial presentation included epistaxis (11/20 cases; 55%) and sneezing (8/20 cases; 40%). Less frequently reported clinical signs included nasal discharge (2/20 cases; 10%), facial swelling (2/20 cases; 10%), snoring (2/20 cases; 10%), coughing (1/20 cases; 5%), vomiting (1/20 cases; 5%), and nasal erythema (1/20 cases; 5%).
Gross tumor descriptions were available for 2 cases that received a necropsy. In 1 necropsy case, the tumor consisted of an irregularly marginated, multilobular, tan mass that filled the left nasal cavity and obliterated the nasal turbinates (Fig. 1). In the second necropsy case, the tumor arose in the left nasal cavity, distorted the left side of the face, and extended through the eroded caudal nasal, maxillary, frontal, and lateral palatine bones. This mass was described as poorly demarcated, mildly firm, and pale tan with multifocal small hemorrhagic foci.
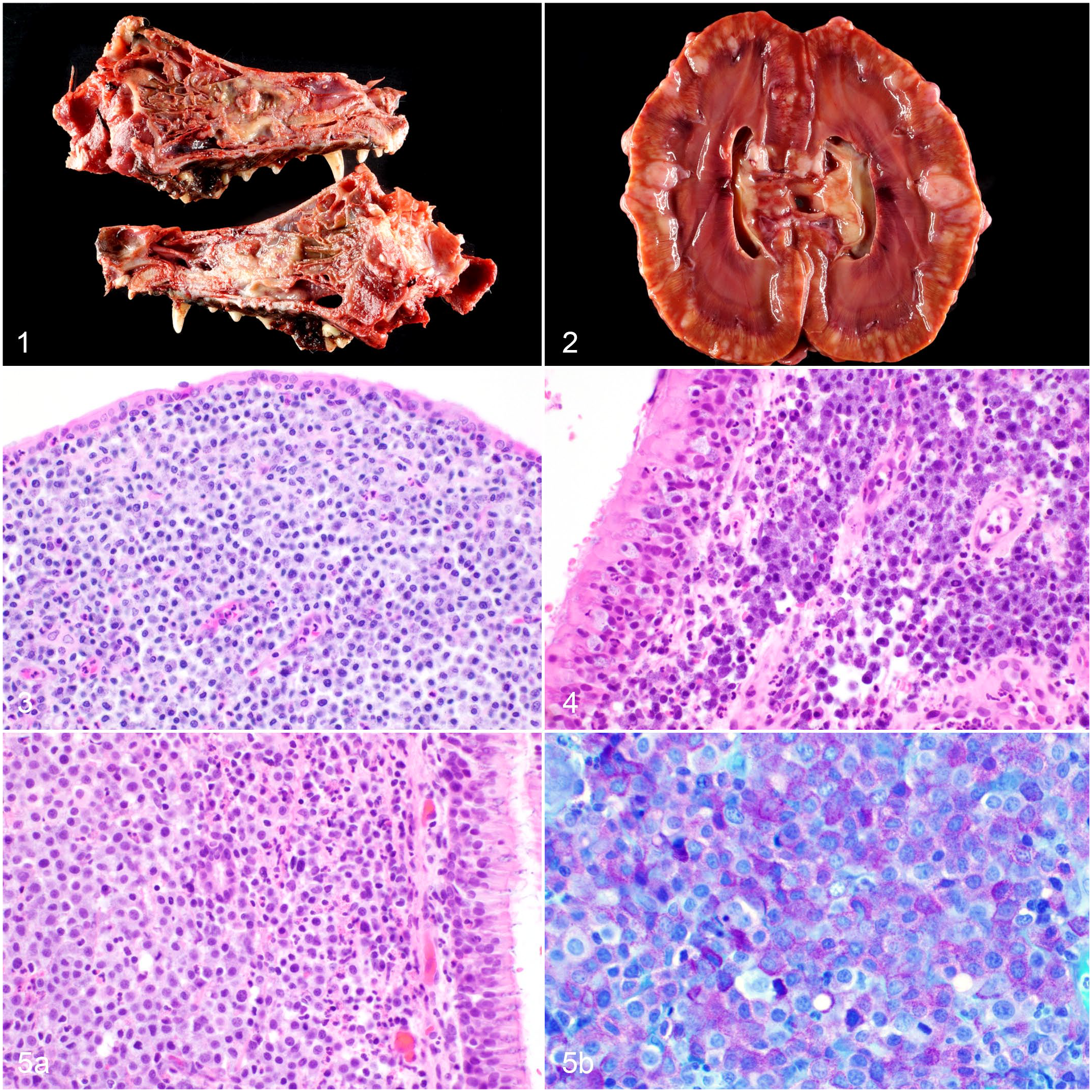
Intranasal mast cell tumor, dog.
On histologic evaluation of all cases, the neoplastic population consisted of discrete round cells that expanded and effaced the lamina propria of the nasal turbinates (18 cases) or nasal vestibule (2 cases) in broad sheets (Figs. 3 and 4). Eight of 20 (40%) cases also demonstrated infiltration of individual and clusters of neoplastic mast cells into the overlying columnar respiratory epithelium, and invasion of the nasal turbinates was observed in 4/20 cases (20%). In all, 3/20 cases (15%) did not include turbinate bone for evaluation of invasiveness.
Neoplastic round cells had an abundant amount of blue-gray cytoplasm and round to oval to reniform nuclei with finely stippled chromatin and 1 to 4 distinct nucleoli. Cytoplasmic granularity varied between cases, with 9/20 (45%) of cases having well-granulated cells with numerous fine, deeply basophilic intracytoplasmic granules (Figs. 3 and 4) and 11/20 cases (55%) having poorly granulated cells (Fig. 5a). A Toluidine blue stain was performed on 12 cases (60%) to confirm a diagnosis of MCT (Fig. 5b). Anisocytosis and anisokaryosis were present in all cases. Karyomegaly was not observed in any of the 20 cases.
Metastasis of the intranasal MCT was confirmed by fine needle aspiration in 11/20 cases (55%). Two out of 20 cases (10%) were reported as having no metastatic disease. In 7/20 cases (35%), information on metastases could not be obtained. Of the 11 cases with confirmed metastases, 10 (91%) had metastasis to the mandibular lymph nodes. Three cases (27%) had metastasis to the retropharyngeal lymph nodes, and 1 case (9%) had metastasis to a cervical lymph node.
One case (Figs. 1 and 2) had distant metastases to the kidneys, lungs, adrenal glands, thyroid gland, tonsils, heart, and omentum. This patient was staged via abdominal ultrasound and thoracic radiographs at the time of diagnosis, at which time only the mass in the nasal cavity was identified (via computed tomography of the cranium). Follow-up thoracic radiographs performed 53 days after this initial imaging revealed a subtle alveolar pattern in the lungs. The extensive metastatic disease was observed at the time of necropsy (and confirmed with histopathology) 58 days after the initial thoracic radiographs and abdominal ultrasound were performed.
Of the 8 cases with epithelial infiltration, 3 were lost to follow-up, but the remaining 5 (63%) lived <1 year. Six of the 8 cases with epithelial infiltration (75%) had confirmed lymph node metastasis. Of the 4 cases with invasion of the turbinate bones, all 4 lived <1 year (20 days, 2 months, 7 months, and 9 months, respectively) and all 4 cases had confirmed lymph node metastasis.
Ki67 immunostaining was high in 9/20 cases (45%), moderate in 3/20 cases (15%), and low in 7/20 cases (35%). One case did not display any detectable immunostaining for Ki67. AgNOR counts ranged from 1.2 to 3.03 per nucleus, with a median of 1.79 AgNORs per nucleus. In 2 cases, AgNOR quantification was not obtained due to insufficient tissue. Atypical CD117 immunostaining patterns were observed in 12/20 (60%) cases, and 6/20 cases (30%) displayed CD117 immunostaining pattern 1. Two cases did not display any detectable immunostaining for CD117.
In the 11 cases with confirmed metastases, 8 cases (73%) had mitotic counts greater than or equal to 8, and 3 cases (27%) had mitotic counts less than 8 (Fig. 6). Atypical CD117 immunostaining was observed in 9/11 cases (82%), and only 1 case (9%) displayed pattern 1 immunostaining for CD117. One case with confirmed metastases did not display any detectable immunostaining for CD117. In cases with confirmed metastases, 7/11 cases (64%), 1/11 cases (9%), and 3/11 cases (27%) had high, moderate, and low Ki67 immunoreactivity, respectively. The median quantification of AgNORs in the 11 cases with confirmed metastases was 1.72 per nucleus. A simple logistic regression did not find a significant correlation between AgNORs per nucleus and the incidence of metastasis. Outcome data were available for 14 cases. The remaining 6 cases were lost to follow-up. Of the cases for which outcome data were available, the survival time ranged from 20 days to 6 years, with only 3/14 (21%) of dogs alive >1 year after the onset of clinical signs (Fig. 7). We chose to report cases as having a <1-year survival or >1-year survival, rather than reporting a median survival time, because of limitations in the follow-up data obtained for some of the patients. For 1 patient, the owners reported that the dog lived less than 1 year after diagnosis but were unable to specify the exact date or age of the patient at the time of death. Two patients had medical records documenting that they lived to at least 1.5 years after their diagnosis but were subsequently lost to follow-up. Given these limitations, we chose to evaluate survival time based on the relative number of patients who lived either less than or greater than 1 year.
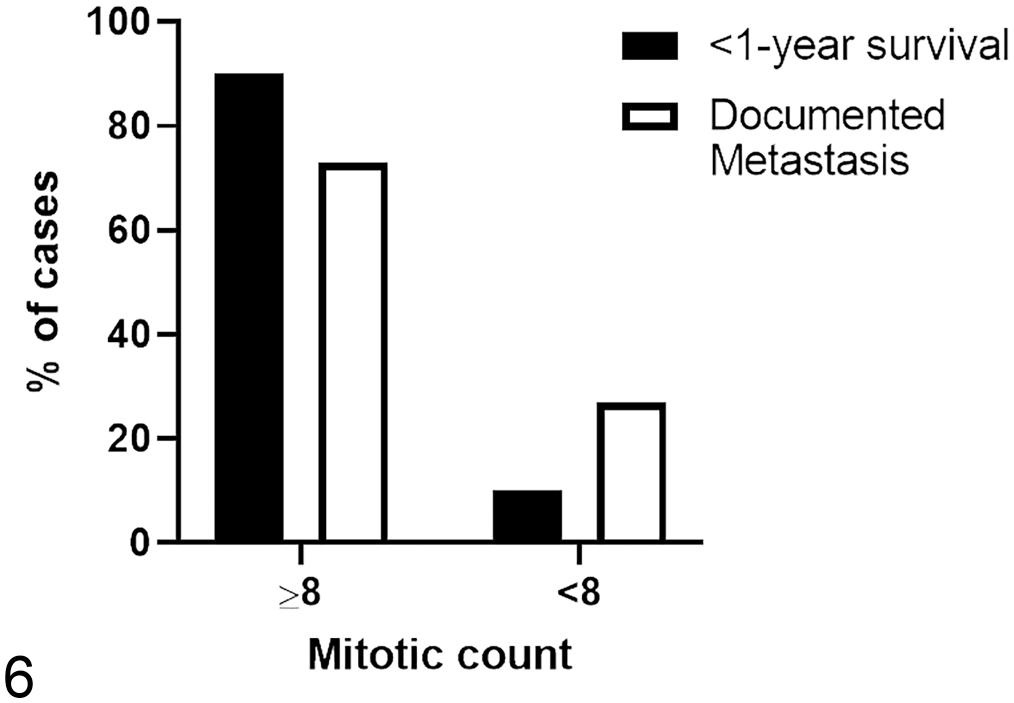
Cases with documented metastases and a <1-year survival time tended to have mitotic counts greater than or equal to 8.
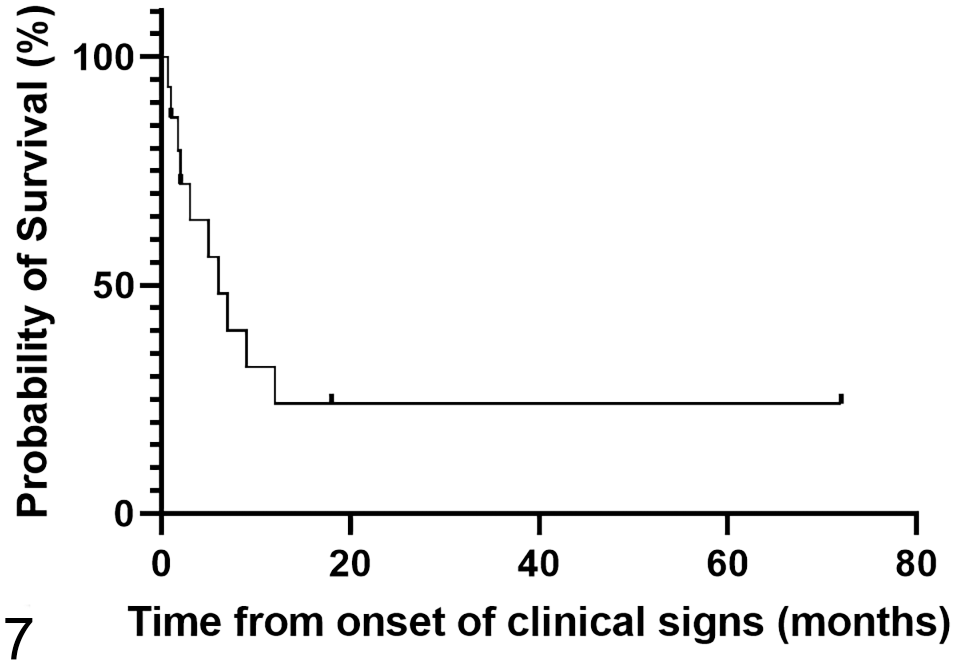
A Kaplan-Meier survival estimate mapping survival time from the reported onset of clinical signs. Of the 14 cases for which outcome data was available, the survival time ranged from 20 days to 6 years, with only 3 (21%) dogs alive 1 year after the onset of clinical signs.
Of the 10 dogs with a survival time <1 year, 4 dogs were euthanized for progression of disease, 1 dog died from respiratory arrest following aspiration of hemorrhagic discharge from the nasal mass (confirmed with necropsy and histopathology), 1 dog presented dead to the primary veterinarian 5 days after starting treatment for a nasal MCT diagnosed 11 days prior, and an explicit reason for death or euthanasia was not provided for 4 dogs that lived <1 year (Supplemental Table 1). In these 10 cases with a <1-year survival, 9 cases (90%) had mitotic counts greater than or equal to 8, and only 1 case (10%) had a mitotic count less than 8 (Fig. 6). Eight cases (80%) displayed atypical CD117 immunostaining patterns. In cases with a <1-year survival, 6/10 cases (60%), 2/10 cases (20%), and 2/10 cases (20%) had high, moderate, and low Ki67 immunoreactivity, respectively. A simple linear regression demonstrated a trend toward lower survival times with increasing mitotic count, but this was not statistically significant (
An ITD in the
Discussion
Intranasal MCTs comprised 1% of the 1849 primary canine intranasal neoplasms evaluated in our study and represented the second most common primary round cell neoplasm arising in the nasal cavity, after lymphoma (23.8% and 44.1%, respectively).
Grossly, the intranasal mast cell tumors in our study were locally destructive, obliterating nasal turbinates and extending through the caudal nasal, maxillary, frontal, and/or lateral palatine bones. Histologically, neoplastic mast cells primarily occupied the lamina propria, although invasion of underlying turbinate bones was appreciated in 20% of cases, and individual and clusters of neoplastic mast cells infiltrated the overlying epithelium in 40% of the cases. Epitheliotropism has been described in rare cases of canine cutaneous mast cell tumors, 17 but the incidence in our study suggests that it may be a more common feature of mast cell tumors arising in the nasal cavity. Over half (55%) of the tumors in our study were composed of poorly granulated mast cells, indicating that Toluidine blue staining may be necessary for confident diagnosis of this entity in the nasal cavity.
In our study, primary intranasal MCTs appeared to be clinically aggressive and prone to metastasis, resembling the behavior reported for other mucosal MCTs. Metastases were present in 55% of intranasal MCT cases, with the mandibular lymph nodes representing the most common site. This is similar to previously reported frequencies of regional lymph node metastasis in 50% to -55% and 58% of MCTs arising from the oral mucosa and oral mucocutaneous junction, respectively.6,8 MCTs arising in the gastrointestinal mucosa exhibit a higher frequency of metastasis: 1 study reported lymph node metastasis in 11/14 dogs (78%) and local visceral metastasis to the liver, kidney, adrenal glands, and other intestinal segments in 2/23 dogs (9%). 18 In contrast, abdominal visceral metastasis was reported in only 1/33 (3%) of dogs in a study of oral mucosal MCTs. 6 In our study, metastasis of intranasal MCT to distant viscera was documented in 1 case (5%), so while rare, intranasal MCTs are similarly capable of distant visceral metastasis. Intranasal MCTs also tended to have a poor prognosis; of the cases in our study with available outcome data, only 21% (3 dogs) lived longer than 1 year, while 71% (10 dogs) died or were euthanized within a year of the onset of clinical signs. An explicit cause of death or rationale for euthanasia was not provided for 4 of these dogs, but progression of nasal MCT was implicated in 6 cases. Epistaxis and sneezing were the most commonly reported clinical signs.
Interestingly, cases with documented metastases or a <1-year survival tended to have mitotic counts greater than or equal to 8, a moderate to high Ki67 proliferation index, and atypical CD117 immunostaining patterns, suggesting that these features could represent potential prognosticators for MCTs arising in the nasal cavity. The presence of lymph node metastasis at the time of diagnosis has been shown to be a useful prognosticator for MCTs arising in the oral mucosa and oral mucocutaneous junctions,6,8 and oral MCTs with mitotic counts >5 correlated with shorter time to tumor progression and overall survival. 6 However, MCTs arising in the gastrointestinal mucosa had poor prognoses in nearly all cases, which precluded relating histomorphologic features to prognosis. 18 Similarly, too few of the dogs in our study had favorable outcomes—no metastases, and survival times >1 year—to allow comparison of these 2 groups with enough statistical power to definitively demonstrate that these histologic and immunohistochemical features predicted the patient’s outcome. A simple linear regression demonstrated a trend toward lower survival times with increasing mitotic count, but this was not statistically significant. AgNOR quantification did not correlate with patient survival or with the incidence of metastasis in the cases in our study. Identifying more cases of dogs with intranasal MCTs that do not develop metastatic disease and live longer than 1 year will be necessary before histologic and immunohistochemical prognosticators can be confidently claimed.
Despite the frequency of atypical CD117 immunostaining patterns in our study, an ITD in the
Treatment protocols varied considerably between cases. Given this variability, the effect of certain treatments on patient outcome and tumor behavior could not be reasonably concluded in this study.
In conclusion, nasal MCTs are uncommon in dogs and account for approximately 1% of all primary intranasal neoplasms in the dog. They appear to be aggressive neoplasms that are prone to metastasis to regional lymph nodes and capable in rare cases of metastasizing to distant viscera. These tumors bear a generally poor prognosis, with survival times of less than 1 year reported for most cases.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221109100 – Supplemental material for Intranasal mast cell tumors: Clinical, immunohistochemical, and molecular features in 20 dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858221109100 for Intranasal mast cell tumors: Clinical, immunohistochemical, and molecular features in 20 dogs by Eileen Larsen, Allison M. Watson and Juan F. Muñoz Gutiérrez in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858221109100 – Supplemental material for Intranasal mast cell tumors: Clinical, immunohistochemical, and molecular features in 20 dogs
Supplemental material, sj-xlsx-2-vet-10.1177_03009858221109100 for Intranasal mast cell tumors: Clinical, immunohistochemical, and molecular features in 20 dogs by Eileen Larsen, Allison M. Watson and Juan F. Muñoz Gutiérrez in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank Erin Napier for her assistance locating archived patient records and Drs. Russell Moore and Zach Weller for their statistical input. Figures were created using GraphPad Prism 8 software.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by the Colorado State University Institutional Start-Up Support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
