Abstract
Neuroendocrine carcinoma of the nasopharynx was diagnosed in a 9-year-old male Golden Retriever. The mass was identified by computed tomography of the nasal cavity and nasopharyngoscopy, and it was surgically excised. Histologic, cytochemical, and electromicroscopic examination of specimens confirmed the type of tumor. The dog was clincally improved for 150 days but was then reexamined because of respiratory difficulty and poor appetite. Thoracic radiographs revealed multiple nodules in all lung lobes, and ultrasonography revealed a mass in the spleen. The dog died the next day.
Neuroendocrine carcinoma was described more than 100 years ago.8 The term carcinoid was coined in 1907 to differentiate a group of intestinal neoplasms, mostly benign with distinct morphologic patterns, from the more aggressive adenocarcinoma.11 Subsequently, neuroendocrine carcinomas were incorporated into the amine precursor uptake and decarboxylation (APUD) system.7 Currently, these neoplasms are grouped under the dispersed neuroendocrine system, which includes organs in which APUD cells are uncommon.7 Neuroendocrine carcinoma has been found in a wide range of organs in humans;7 the gastrointestinal and pulmonary tracts are the most common sites.1,7
In dogs and cats, neuroendocrine carcinoma has been described in the intestines, liver, bile duct, lungs, gallbladder, esophagus, nasal cavity, and skin.2,6,12–17 Most of these tumors are very aggressive.2,12,13,16 A Medline search for a period of 20 years did not reveal any report of neuroendocrine carcinoma in the nasopharynx, which indicates the rarity of this neoplasm.
Immunocytochemical and electron microscopic examination of neuroendocrine carcinoma in dogs has rarely been done.6,8,13,14 Recently, immunohistochemistry has been used to study the biologic behavior and sites of origin of human neuroendocrine carcinoma.1,7
In this report, we describe for the first time a case of neuroendocrine carcinoma in the nasopharynx of a dog including clinicopathologic, histomorphologic, immunocytochemical, and electormicroscopic features of the neoplasm and follow-up information.
A 9-year-old male Golden Retriever was referred to the Animal Medical Center because of upper airway stertor and transient nasal discharge of 3 months duration. Rhinoscopy and biopsy revealed chronic rhinitis, which did not respond to antibiotics.
Results of physical examination were essentially normal. No nasal discharge was seen, and the dog was breathing through the nares. Computed tomography of the nasal cavity and the sinuses revealed soft tissue density in the cavities and a large, discrete mass in the nasopharynx (Fig. 1) The mass appeared to arise from a stalk not attached to the soft palate. Nasopharyngoscopy showed a large mass filling the bony nasopharynx. Thoracic radiography revealed a nodule in the right caudal lung lobe. The owner declined further diagnostic tests to evaluate the nodule in the lung lobe but agreed to surgical removal of the nasopharnygeal mass. A 4 × 2 × 2-cm encapsulated mass arising from a stalk attached to the dorsal nasopharynx was surgically excised, along with the dorsal attachment.
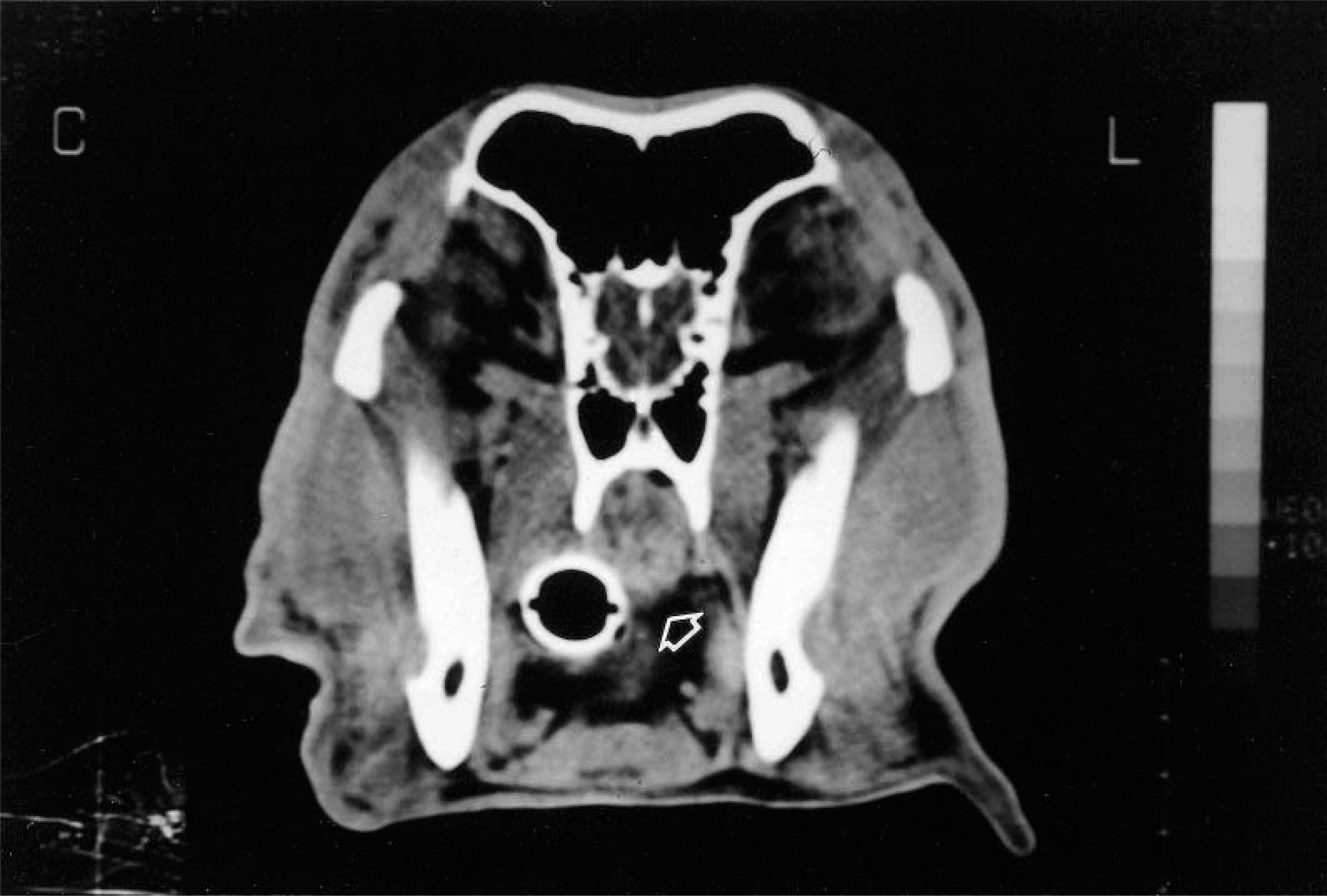
Computed tomography scan. Head; dog. Note Well-outlined nasopharyngeal mass (arrow). An endotracheal tube is located is located below and to the left of the mass.
The dog was clinically improved for 150 days, at which time he was reexamined because of respiratory stertor and poor appetite. Radiographs revealed multiple diffuse nodules in all lung lobes. Ultrasound revealed a 4 × 5-cm mass in the spleen. Mild regenerative anemia (packed cell volume 27%; reference range, 37–55%) and thombocytopenia (50,000 platelets/mm3; reference range, 180,000–500,000 platelets/mm3) were documented. The dog collapsed and died the next day, and permission was not given for necropsy.
Specimens taken at surgery were fixed in 10% buffered formalin, routinely processed, and stained with hematoxylin and eosin and mucicarmine. Sections were evaluated immunocytochemically with a supersensitive multilink immunodetection system following the instructions of the manufacturer (Biogenex, San Ramos, CA). Antiboides against cytokeratin (AE1/AE3), syanaptophysin, neuron-specific enolase (NSE), gastrin, serotonin, somatostatin, and S-100 protein were tested. Small pieces of tissue were fixed in a buffered (pH 6) formaldehyde and glutaraldehyde mixture (3% each) and processed for electron microscopy according to standard procedures.
Histologically, the tumor was ulcerated with superficial necrosis and had small areas of squamous cell lining. The morphology of the tumor was typical of neuroendocrine carcinoma, i.e., groups or sheets of cells were enclosed in fibrovascular or vascular stroma. The round to oval neoplastic cells had vesiculated nuclei and one to three prominent nucleoli. Moderate amounts of eosinophilic cytoplasm were seen. The number of mitotic figures varied in different areas and ranged from zero to five per high-power field (Fig. 2). Areas of perpendicular spindle cells lined the capillaries. A few neoplastic giant cells with large nuclei and nucleoli were seen. Within the neoplastic tissue, highly vascular areas were observed with blood spaces not lined by endothelial cells.
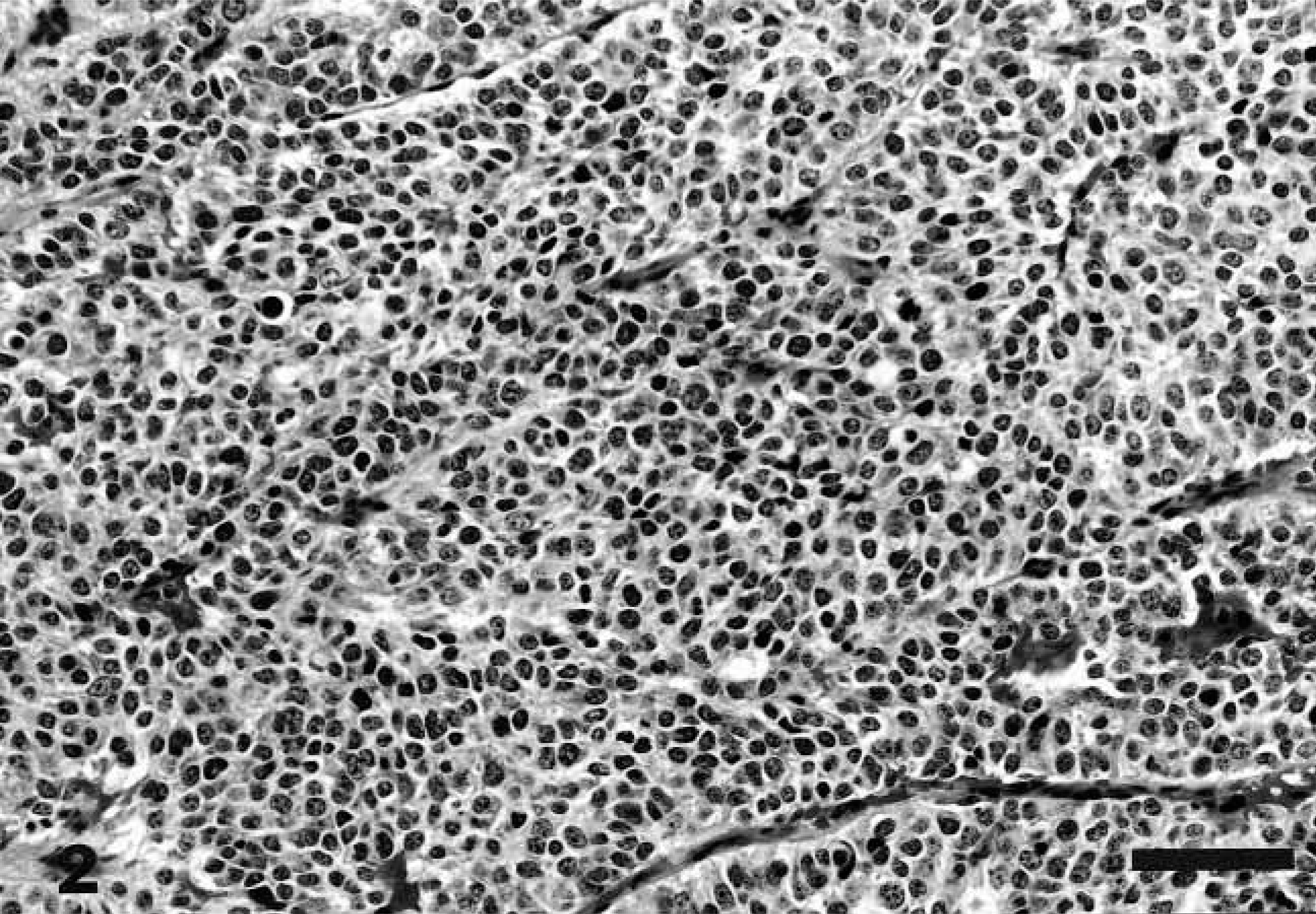
Nasopharyngeal mass; dog. Sheets of hyperchromatic, round to oval neoplastic cells are separated by vascular stroma. 1 cm = 20 μm.
The neoplastic cells did not react to mucine. Immunohistochemically, the neoplastic cells only reacted to synaptophysin and NSE (Figs. 3, 4).

Nasopharyngeal mass; dog. Section immunohistochemically stained for synaptophysin has diffuse, dark intracytoplasmic granules. Supersensitive multilink immunodetection system, diaminobenzidine chromogen, Gill III counterstain. 1 cm = 40 μm.
Nasopharyngeal mass; dog. Section immunohistochemically stained for NSE has pale intracytoplasmic granules. Supersensitive mulitlink immunodetection system, diaminobenzidine chromogen, Gill III counterstain. 1 cm = 40 μm.
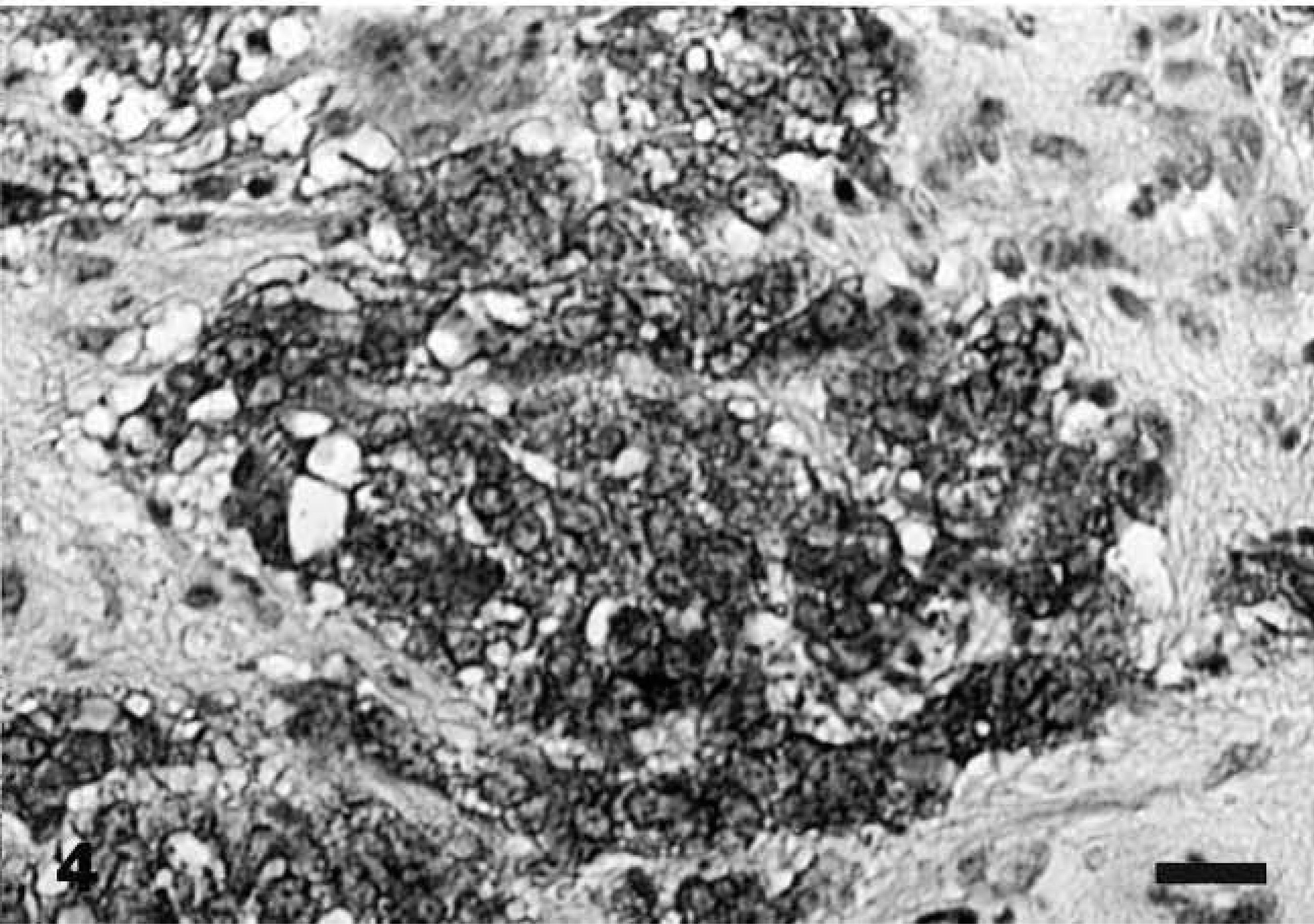
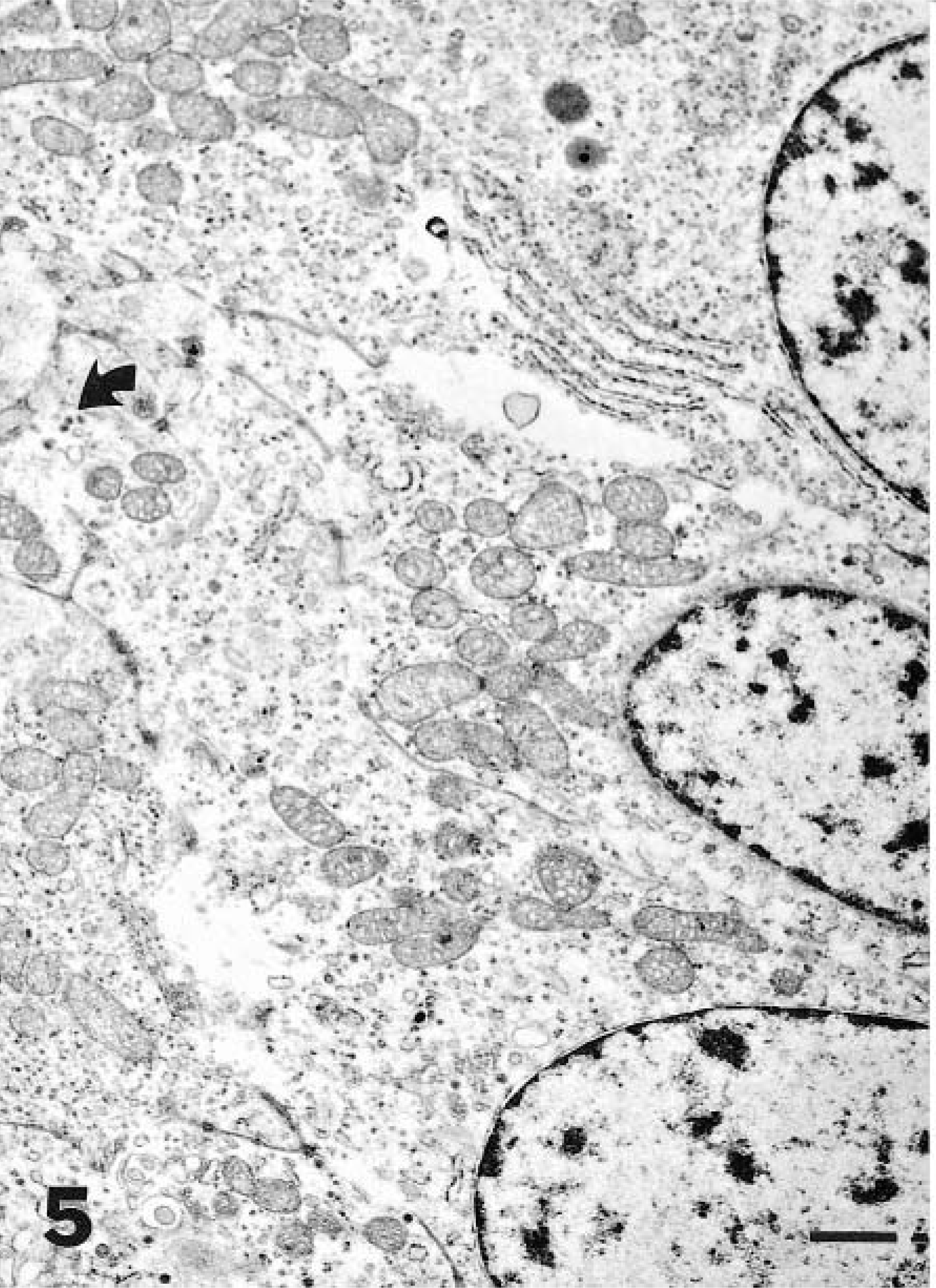
Ultrastructural examination of the tumor revealed clusters of primarily round tumor cells and their processes, which were seen in various planes of the section (Figs. 5, 6). The nuclei were round or oval, and the cell processes were joined by small desmosomes and tight junctions (Fig. 5). The cytoplasm contained stacked cisternae of rough endoplasmic reticulum, clusters of mitochondria with vesicular cristae (Fig. 5), and aggregates of dense-core, neurosecretory-type granules with an average diameter of 120 nm that were primarily concentrated in cytoplasmic processes (Fig. 6).
Nasopharyngeal mass; dog. The abundant cytoplasm contains stacked cisternae of rough endoplasmic reticulum (top), clusters of mitochondria, and sparse, neurosecretory-type granules (arrow). The cells are joined by desmosomes and tight junctions. Bar = 1 μm.
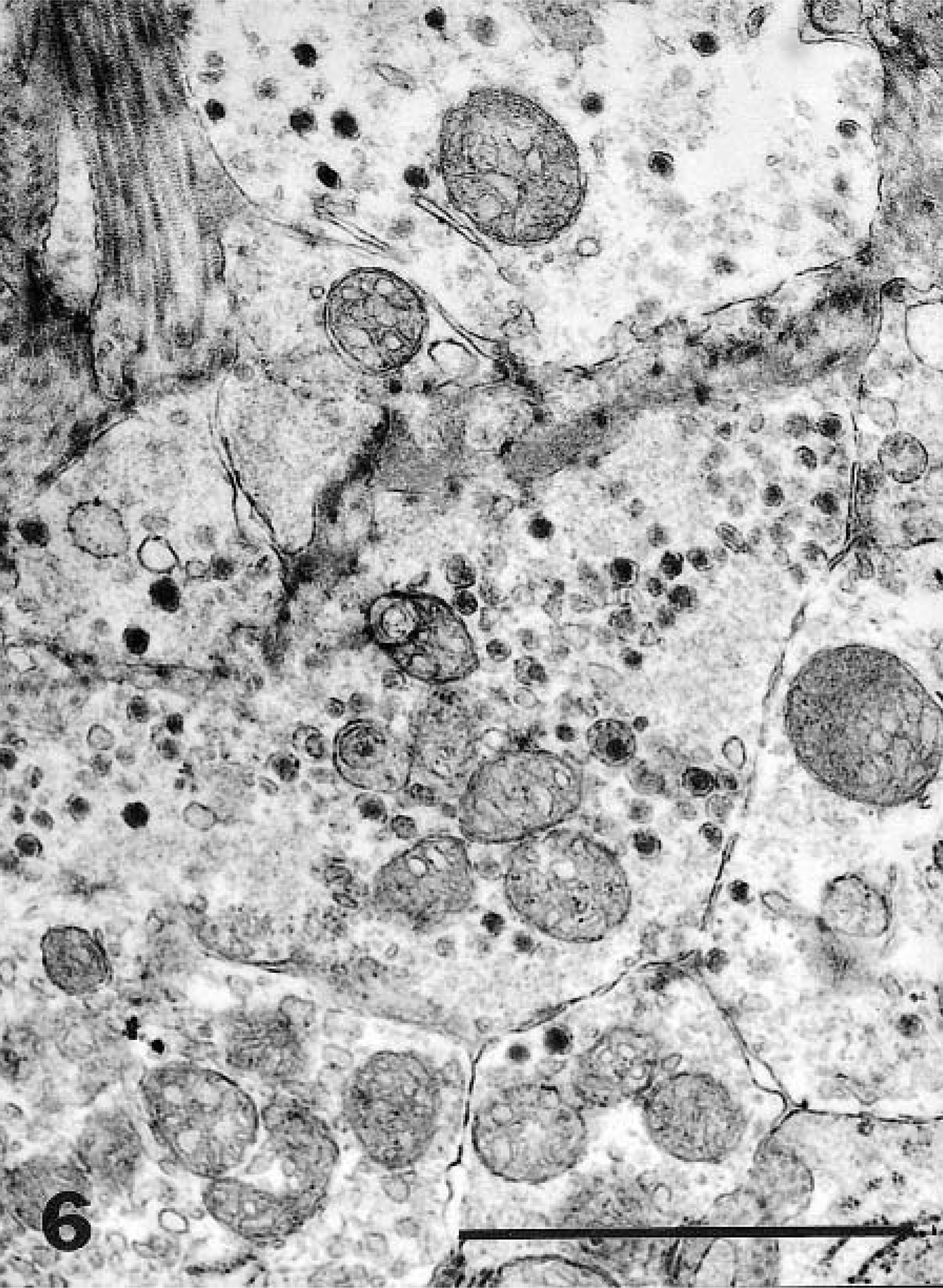
Nasopharyngeal mass; dog. Detail of cytoplasmic processes showing mitochondria and neurosecretory-type granules with an average diameter of 120 nm. Bar = 1 μm.
The nasopharynx is part of the respiratory system that lies behind and above the soft palate and consists of the anterior, posterior, and lateral walls. In adults, most of the nasopharynx is lined by stratified squamous epithelia. The rest is lined by ciliated columnar epithelia. The squamous epithelium lines the interior half of the anterior and posterior walls and the anterior half of the lateral walls. The ciliated epithelium is present in the nasal choanae and over the roof of the posterior wall. The rest of the nasopharynx has alternating islands of squamous and ciliated epithelium. In addition, there may be focal areas of transitional or intermediate epithelium.9,10
The neuroendocrine system of the respiratory tract consists of specialized airway endocrine epithelial cells associated with nerve fibers. These epithelial cells can be solitary or clustered to form neuroepithelial bodies. Solitary neuroendocrine cells are scattered among the respiratory epithelial cells and among the squamous cells in the upper respiratory tract. They are also located in the submucosal gland ducts.4,19,21
Neuroendocrine cells are derived from undifferentiated precursors present in endodermal epithelial cells, which develop into small granule neuroendocrine cells. Later, the small granule neuroendocrine cells proliferate into neuroendocrine cells to form corpuscles of neuroendocrine cells, which become innervated to form the neuroepithelial bodies. Proliferation of these neuroendocrine cells and neuroepithelial bodies contribute to the repair and replacement of the airway epithelial cells and development of neuroendocrine tumors.18,20–22
Neuroendocrine carcinoma has a basic organoid pattern characterized by perivascular arrangement of epithelial cells with various degrees of morphologic differentiation, which makes this tumor distinguishable from other carcinomas and adenocarcinoma irrespective of their origin in different organs.7 Neuroendocrine tumors range from well-differentiated (carcinoid) to poorly differentiated tumors. The pathologic prognostic factors depend on the degree of differentiation. A well-differentiated neuroendocrine tumor could be malignant in the presence of synchronous metastasis and/or invasiveness.
The malignancy of neuroendocrine tumors is based on the following: 1) tumor size (>2 cm), 2) invasion into adjacent tissues, 3) invasion beyond the submucosa and into adjacent tissues, 4) presence of necrosis, 5) overt cell atypia with more than two mitotic cells per 10 high-power fields, 6) hormone expression and loss of chromogen immunoreactivity, and 7) nuclear P53 protein accumulation. It is difficult to predict prognosis from histologic grading.
Other than in specific endocrine organs, such as adrenal medulla, c-cells, and islet cells, neuroendocrine tumors are rarely described in domestic animals. They have been described in the liver, lungs, nasal cavity, skin, and intestines in dogs. Most of these tumors are aggressive and composed of small cell types. In humans, many more sites have been reported, but the basic features are similar to those of neuroendocrine carcinoma seen in dogs.2,7,12–17
Pulmonary neuroendocrine carcinoma in humans is currently divided into small- and large-cell tumors, thus eliminating the atypical carcinoid and oat cell carcinoma. Small-cell carcinoma is highly malignant and carries a very poor prognosis. Atypical neuroendocrine tumor in dogs has been described as having other cell components, such as squamous cells and mucin-secreting cells, and may not fit the pattern of the typical neuroendocarcinoma of the lungs.3,5
Even though progressive lesions were seen radiographically in the lungs and spleen of this dog, the nature of these lesions was not established. However, this tumor and neuroendocrine carcinoma are aggressive tumors in both humans and dogs. The splenic and diffuse pulmonary lesions were probably metastatic lesions.1,3,7,10,13,16
The morphology of this tumor and the absence of S-100 protein and AE1/AE3 eliminated squamous cell carcinoma, adenocarcinoma of the minor salivary glands, lymphoma, and especially melanoma from the differential diagnosis. Direct infiltration by sinonasal neuroendocrine carcinoma or neuroblastoma was eliminated as a possible diagnosis by the lack of lesions in the sinonasal area and by the gross and morphologic characteristics of the tumor.12
