Abstract
A perianal subcutaneous tumor involving the anal sac developed in an 8-year-old male mixed Labrador Retriever dog. Histologically, this tumor showed typical features of the solid-type carcinoma of the apocrine glands of the anal sac. However, neoplastic cells were immunoreactive for cytokeratin 8, chromogranin A, vasoactive intestinal peptide, neuron-specific enolase, and synaptophysin, and negative for S-100 protein, α-smooth muscle actin, vimentin, glucagon, insulin, somatostatin, carcinoembryonic antigen, serotonin, and parathyroid hormone–related protein. Considering the distribution of chromogranin A–positive cells within the anal sac apocrine glands, this tumor was diagnosed as neuroendocrine carcinoma originating from the apocrine glands of the anal sac.
The canine perianal region has different kinds of glands, such as sebaceous glands, perianal (hepatoid) glands, and apocrine glands of the anal sac. The anal sacs are paired cutaneous anal diverticula that are lined by cornified, stratified squamous epithelium, and located within the ventrolateral perianal region of carnivores and many rodents. 2 Carcinomas of the apocrine glands of the anal sac (CAGASs) are uncommon but highly malignant tumors that are believed to arise from the anal sac apocrine secretory epithelium in dogs and cats. 17,23 The masses often invade deeply into the underlying perirectal tissue, and approximately 50% cases appear as a perianal subcutaneous mass. 7 Histologically, the tumor varies from solid, rosette, to tubular types. 6
A possible neuroendocrine (NE) nature of CAGASs is indicated by the characteristic histomorphology 3 and the tumor-associated hypercalcemia that is believed to be caused by increased blood levels of parathyroid hormone–related protein (PTHrP), which is produced by these tumors. 8 One study has shown the presence of a population of NE cells expressing neuron-specific enolase (NSE) both in the anal sac apocrine glands and CAGAS cases. 22 However, limited immunoreactivity in only a small population of neoplastic cells and no immunoreactivity against other NE markers suggested an equivocal NE nature of these tumors.
The current report describes a canine NE carcinoma that developed at the perianal subcutis and was histologically diagnosed as solid-type CAGAS according to the World Health Organization. 6 Histogenesis of this tumor is also discussed.
An 8-year-old, male, neutered mixed Labrador Retriever, weighing 60 kg, was presented to a veterinary clinic with anorexia and vomiting. No remarkable abnormalities were found in the complete blood count (red blood cell count: 6.63 × 106/µl; hemoglobin concentration: 14.9 g/dl; hematocrit: 41.7%; mean corpuscular volume: 62.9 fl; mean corpuscular hemoglobin: 22.5 pg; mean corpuscular hemoglobin concentration: 35.7 g/dl; platelet count: 275 × 103/µl; total white blood cell count: 13.5 × 103/µl; lymphocytes: 3.3 × 103/µl; eosinophils: 0.5 × 103/µl; or other white blood cells: 9.7 × 103/µl), serum biochemistry (glucose: 113 mg/dl; blood urea nitrogen: 12.9 mg/dl; total cholesterol: 242 mg/dl; aspartate aminotransferase: 21 U/l; alanine aminotransferase: 61 U/l; calcium: 8.4 mg/dl), and X-ray examination of the abdominal region, except for a right perianal subcutaneous mass (8 cm × 8 cm × 4 cm) involving the anal sac. Fine-needle aspiration cytology of the tumor portion revealed solid clusters of epithelial cells showing a high nuclear-to-cytoplasmic ratio suggestive of a malignant anal sac apocrine gland neoplasm. After surgical excision of the mass, the animal gradually deteriorated and died 3 months later. Necropsy was not performed. The excised mass was fixed in 10% neutral buffered formalin (pH 7.4), and submitted for routine histological processing.
Macroscopically, the mass showed a well-circumscribed, white, and multinodular appearance accompanied with occasional necrotic foci on cut surface after fixation (Fig. 1). Paraffin sections were stained with hematoxylin and eosin. For immunohistochemistry, the avidin–biotin–peroxidase complex technique was used a with the primary antibodies against cytokeratin 8 (CK8; mouse monoclonal, clone CAM 5.2, 1:1), b chromogranin A (CGA; rabbit polyclonal, 1:3,000), c S-100 protein (rabbit polyclonal, 1:100), d vasoactive intestinal peptide (VIP; rabbit polyclonal, 1:500), e NSE (rabbit polyclonal, 1:200), f synaptophysin (SYP; mouse monoclonal, clone SY38, 1:500), g α-smooth muscle actin (α-SMA; mouse monoclonal, clone 1A4, 1:100), h vimentin (goat polyclonal, 1:200), i glucagon (rabbit polyclonal, 1:75), j insulin (guinea pig polyclonal, 1:300), k somatostatin (rabbit polyclonal, 1:200), l serotonin (rabbit polyclonal, 1:1), m carcinoembryonic antigen (rabbit polyclonal, 1:200), n and PTHrP (rabbit polyclonal, 1:50). o For antigen retrieval, deparaffinized sections were heated in 10 mM citrate buffer (pH 6.0) by autoclaving at 121°C for 10 min with the SYP antibody or by microwaving at 90°C for 10 min for CK8, α-SMA, vimentin, and CEA. No antigen retrieval treatment was performed for CGA, S-100 protein, VIP, NSE, glucagon, insulin, somatostatin, serotonin, and PTHrP. The chromogen was 3,3′-diaminobenzidine; the counterstain was hematoxylin. As positive control tissues, the adrenal medulla, brain, skin, pancreatic islets, and the anal sac of a beagle dog were used, and nonimmunized sera were substituted for the primary antibody as negative controls for immunoreactivity.
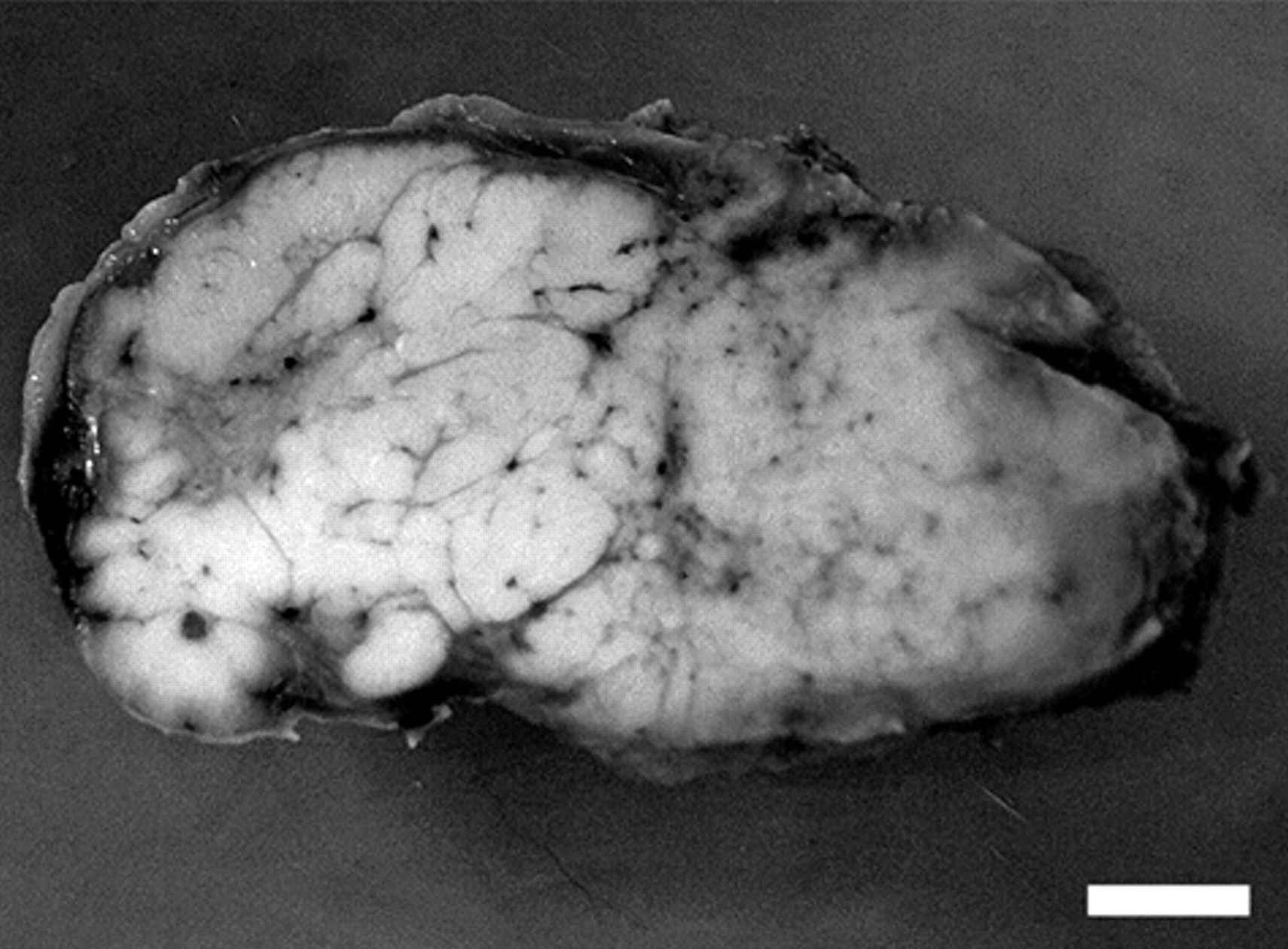
Macroscopic view of perianal subcutaneous mass. The cut surface of the mass after fixation, showing well-circumscribed multinodular appearance accompanied with small foci of necrosis. Bar = 1.0 cm.
Histologically, the mass occupied the subcutaneous tissue surrounding the anal sac epithelium and extended to the sac cavity in some portion exhibiting ulceration (Fig. 2A). The neoplastic tissue showed a multilobulated structure, roughly divided by irregularly shaped trabeculae of connective tissue that were connected with a thick fibrous capsule. Each nodule was incompletely subdivided by a delicate fibrovascular stroma into variably sized nests of neoplastic cells. The neoplastic cells were arranged in solid sheets, appearing polyhedral in shape and with a lightly eosinophilic, finely granular cytoplasm and indistinct cellular borders. Their nuclei were round to ovoid with several apparent nucleoli (Fig. 2B). Perivascular pseudorosettes 24 were scattered, and cyst-like structures were occasionally formed (Fig. 2B). Some areas showed apparent lobular arrangement of neoplastic cells divided by vascular stroma. There were 2–6 mitotic figures per 400× field. Invasive growth of neoplastic cells into the surrounding connective tissues was evident and associated with vascular infiltration.
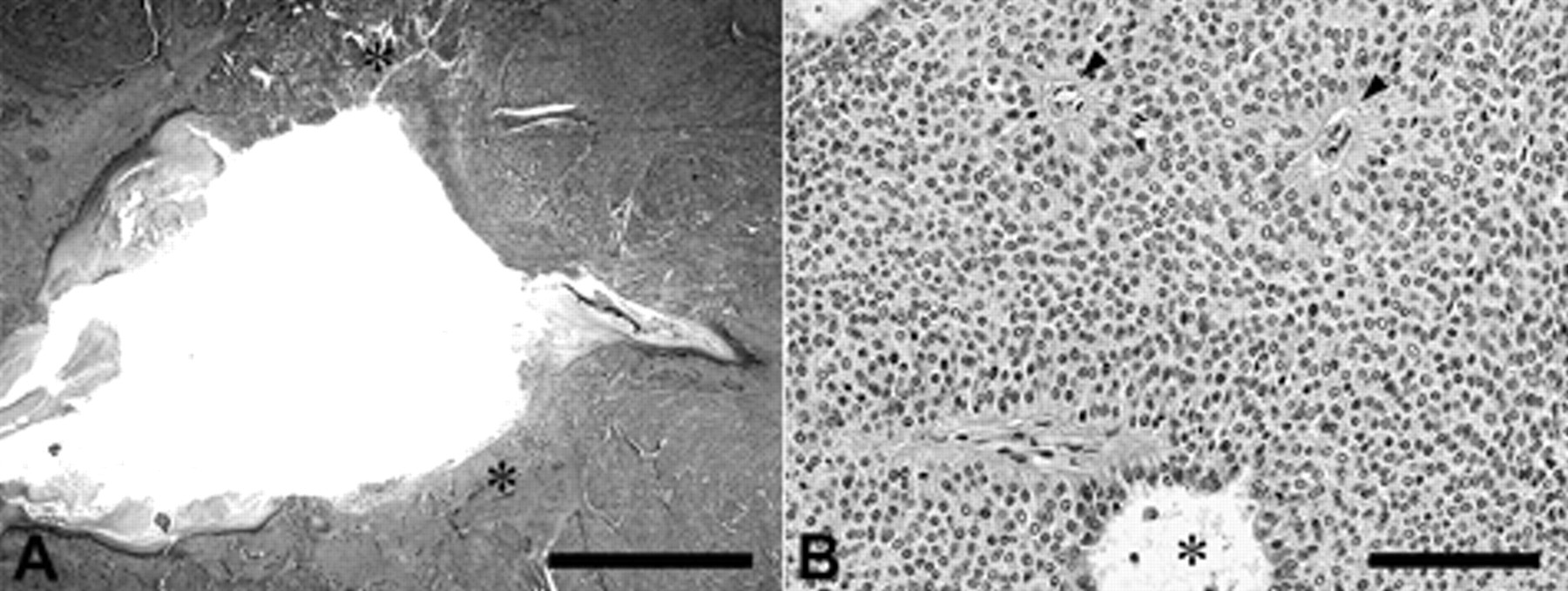
Light micrograph of perianal subcutaneous mass.
Immunohistochemical results for the tumor are listed in Table 1. Weak to strong perinuclear cytoplasmic immunoreactivity for CK8 was diffusely observed in neoplastic cells (Fig. 3A). With regard to CGA, weak to strong finely granular cytoplasmic immunoreactivity was observed in a variable number of neoplastic cells, particularly at the periphery of neoplastic cell nests (Fig. 3B). Although the intensity was low, the immunoreactivity pattern for VIP was similar to that of CGA (Fig. 3C). Cytoplasmic immunoreactivity for NSE was also diffuse, but positive cells were mostly distributed inside the tumor nests, inverse to the peripheral immunoreactivities for CGA and VIP (Fig. 3D). Diffuse, fine, dot-like cytoplasmic immunoreactivity for synaptophysin was also observed (Fig. 3E). While CGA-positive cells were not entirely positive for CK8, distribution pattern of positive cells in the tumor nests was similar between CK8, NSE, and synaptophysin. Neoplastic cells were negative for S-100 protein, α-SMA, glucagon, insulin, somatostatin, carcinoembryonic antigen, serotonin, and PTHrP. Neoplastic cells were also negative for vimentin, although endothelial cells located within the perivascular pseudorosettes were positive.
Immunohistochemical reactivity.

Reactivity: – = no immunoreactivity; + = sparsely distributed; ++ = scattered; +++ = diffusely distributed.
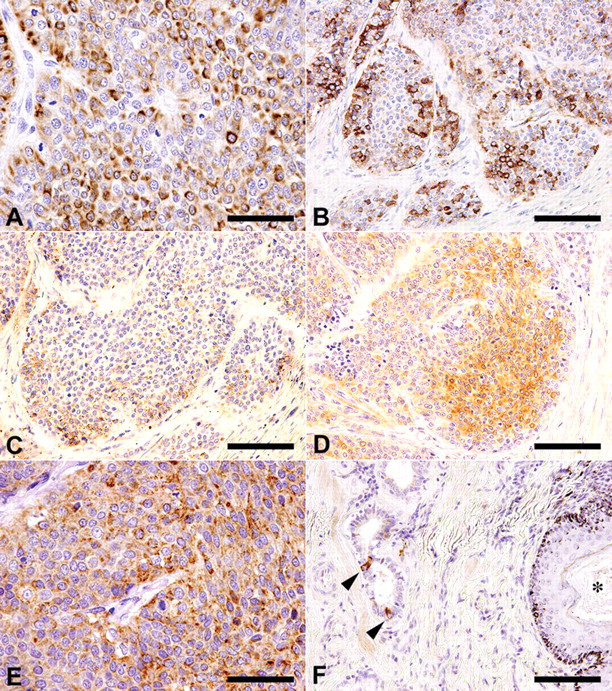
Immunohistochemical staining results of the perianal subcutaneous mass (A–E) and the anal sac (F).
In the normal anal sac, apocrine glands showed weak immunoreactivity for PTHrP. The CGA-positive cells were sparsely distributed within the apocrine gland structure (Fig. 3F). However, there were no immunoreactive cells for NSE, SYP, or VIP in the apocrine glands.
Both the occasional lobular growth pattern and the histological features of the solid-type CAGAS in the present case, such as the solid sheets and perivascular pseudorosettes of neoplastic cells, 6 are in accordance with the solid and/or diffuse growth patterns of NE carcinomas. 21 Neuroendocrine tumors are defined as tumors associated with NE differentiation. Immunohistochemical staining for neuropeptides such as VIP is useful in identifying neoplasms of NE-cell origin in human patients and animals. 10,16,20 Synaptophysin, NSE, and CGA are usually expressed in neoplastic cells of NE-cell tumors. 1,5,16 Cytokeratin-8 is the broad spectrum CK known to be expressed both in NE carcinomas 12,13 and apocrine gland carcinomas such as CAGAS. 17 Neuroendocrine carcinomas typically show perinuclear cytoplasmic localization of CK8, 13,19 in contrast to the diffuse cytoplasmic CK8 immunoreactivity in neoplastic tubular cells in apocrine carcinomas. 15,17 The present case showed a similar pattern to NE carcinomas. Moreover, while CK8-positive cells were not always positive for CGA, the diffuse distribution of NSE- and SYP-positive cells was observed similarly to CK8 immunoreactivity in the present case. The results suggest that the CK8 expression in the present case represents the cellular phenotype of NE-cell origin rather than that of apocrine epithelial cells. Therefore, the positive immunoreactivity for VIP, SYP, NSE, CGA, and CK8, and the histological features of the tumor in the present case fulfill the criteria of NE carcinoma; however, it should be noted that NSE immunoreactivity is often rather nonspecific. 9,11
Because of the perianal location involving anal sac structure and the histological features compatible with solid-type CAGAS, the tumor found in the present case was considered to be a primary tumor derived from the anal sac apocrine glands. In the canine perianal region, the anal canal begins at the anorectal line and is divided into short columnar, intermediate, and cutaneous zones; among multiple types of glands, apocrine sweat glands are distributed within the cutaneous zone in the proximity of the anal sacs. 4 However, apocrine sweat glands are the same as those located in the skin elsewhere on the body. Therefore, malignant tumors that can originate from the cutaneous zone apocrine glands may be histologically distinguishable from CAGASs. Although colorectal carcinoids could develop in the perianal subcutaneous region, the tumor in the present case did not have an anatomical connection with the large intestine. Moreover, gastrointestinal carcinoids often express serotonin or somatostatin, 14 which was not seen in the present case. The tumor in the current case did express VIP as well as CGA. It is known that VIP-producing NE carcinomas can arise from a variety of organs. 14,18 However, no studies have reported VIP-producing NE cells within apocrine glands, and no such cells were detected within the anal sac apocrine glands in the current case. Although detailed analysis of the distribution of VIP-producing NE cells of the anal sac wall may be necessary, sparse distribution of CGA-positive NE cells within the anal sac apocrine glands may suggest that NE carcinomas can arise from these epithelia.
In the present study, positive immunoreactivity of PTHrP was found within the anal sac apocrine glands as well as sparse distribution of CGA-positive NE cells in these epithelia. It is well known that the neoplastic cells of CAGAS secrete PTHrP and cause humoral hypercalcemia of malignancy. 8 Although development of hypercalcemia has been reported in 27% of CAGAS cases, 23 there have been no detailed studies regarding the relationship between hypercalcemia and histological types of CAGAS. In the present case, both hypercalcemia and expression of PTHrP within neoplastic cells were absent. These results suggest that the present case showing typical histological features of solid-type CAGAS could be an NE carcinoma derived from CGA-containing NE cells distributed within apocrine glands that are different from PTHrP-secreting apocrine gland epithelia.
Preliminary analysis of another case that was also diagnosed as solid-type CAGAS also revealed positive CGA immunoreactivity and lack of PTHrP immunoreactivity (data not shown). Of note, a previous report has shown widespread immunolocalization of PTHrP in normal and neoplastic canine tissues; however, one CAGAS tissue examined revealed much higher PTHrP concentration than other normal tissues. 8 Therefore, measurement of PTHrP concentration rather than the analysis of immunolocalization in the neoplastic tissues may be the choice for clarification of the relationship between histological types and the induction potential of hypercalcemia. In summary, the perianal tumor with typical histological features of solid-type CAGAS described herein was considered to be an NE carcinoma arising from NE cells distributed within the anal sac apocrine glands.
Footnotes
Acknowledgements
The authors are deeply grateful to Mrs. Shigeko Suzuki for her excellent technical assistance in preparing the histological specimen.
a.
VECTASTAIN® Elite ABC kit, Vector Laboratories Inc., Burlingame, CA.
b.
BD Biosciences, San Jose, CA.
c.
Yanaihara Institute Inc., Fujinomiya, Japan.
d.
Lab Vision Corp., Fremont, CA.
e.
Millipore Corp., Billerica, MA.
f.
Calbiochem, San Diego, CA.
g.
Chemicon International Inc., Temecula, CA.
h.
Dako Denmark A/S, Glostrup, Denmark.
i.
Santa Cruz Biotechnology Inc., Santa Cruz, CA.
j.
Dako Denmark A/S, Glostrup, Denmark.
k.
Dako Denmark A/S, Glostrup, Denmark.
l.
Dako Denmark A/S, Glostrup, Denmark.
m.
Nichirei Biosciences Inc., Tokyo, Japan.
n.
Dako Denmark A/S, Glostrup, Denmark.
o.
Abcam, Cambridge, United Kingdom.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
