Abstract
Vaginal and vulvar tumors are uncommon in dogs. Knowledge of canine primary clitoral neoplasia is restricted to a few case reports, and only carcinomas have been reported. Cytologic and histologic features reported in the literature seem to overlap with those of canine apocrine gland anal sac adenocarcinoma (AGASA). Clinical features also recall those of canine AGASA, such as locoregional metastases and hypercalcemia of malignancy (HM). In this study, 6 cases of primary canine clitoral carcinomas (CCCs), with and without HM, were investigated by means of cytology, histopathology, electron microscopy, and immunohistochemistry for neuroendocrine markers including chromogranin A (CGA), synaptophysin (SYN), neuron-specific enolase (NSE), and S-100. In all 6 tumors, cytologic findings were consistent with malignant epithelial neoplasia of apocrine gland origin. The tumors examined were classified into 3 different histological patterns representing different degrees of differentiation: tubular, solid, and rosette type. Both CGA and SYN were mildly expressed in 2 of 6 tumors, while NSE was consistently expressed in all 6 cases. None of the tumors were S-100 positive. Transmission electron microscopy revealed electron-dense cytoplasmic granules compatible with neuroendocrine granules in all 6 cases. CCCs presented clinicopathologic features resembling AGASAs with neuroendocrine characteristics, and 2 of 6 neoplasms were considered as carcinomas with neuroendocrine differentiation and were positive for 3 neuroendocrine markers. CCCs can often present with HM, and long-term outcome is likely poor. Our study concludes that CCC seems to be a rare tumor, but it might be underestimated because of the overlapping features with AGASA. Further studies should aim to define the true incidence of this disease.
Keywords
Vaginal and vulvar tumors are uncommon in dogs, accounting for approximately 3% of all canine neoplastic diseases. 4,21 Benign mesenchymal histotypes prevail among others and mainly affect intact dogs. Conversely, malignant tumors seem to be more common in spayed females. 4,7,9,21 The vestibular or vaginal walls are more often affected, whereas the clitoris seems to be rarely involved. 4,12,13,17,21
The clitoris is the female homologue to the male penis and occupies a portion of the anterior region of the vulva. The clitoris represents a relatively large structure in dogs, consisting of three main parts: the crura, the body, and the glans. The crura fan out to support the exterior structures of the clitoris and attach to the underlying tissues. Extending from the crura is the body, the main cylindrical region of the clitoris, which contains 2 thin columns of erectile tissue (
Knowledge of canine primary clitoral tumors is based on a few case reports, and only carcinomas have been reported. 12,13,17 Cytologic and histologic features were similar among cases, resembling those of an apocrine gland anal sac adenocarcinoma (AGASA). Clinical features seem also to recall those of dogs with AGASA, such as locoregional metastases and hypercalcemia of malignancy (HM). 12,13,17 Differently from other tumors, where dogs and humans have been shown to share similar features, canine clitoral carcinoma (CCC) may be unique to dogs. 8 In humans, carcinoma of the clitoris is characterized by a transitional cell pattern or by features of poorly differentiated nonkeratinizing squamous cell carcinoma; although metastatic potential is a concern, a link with paraneoplastic hypercalcemia seems unlikely in humans. 1 The aim of this study was to describe clinical, cytological, histopathologic, immunohistochemical, and ultrastructural features; laboratory findings; and biological behavior of a series of CCCs.
Materials and Methods
Cases
Medical records from 2 university teaching hospitals and 5 histopathology and 2 clinical pathology laboratories were searched to identify cases with a diagnosis of CCC confirmed by histology. Cases were considered eligible for the study only if formalin-fixed paraffin-embedded tissues were available for review and if there was a detailed record of a physical examination, including rectal palpation. If available, cytology slides and clinicopathologic reports were retrieved. The signalment, history, clinical signs, imaging findings, laboratory results, treatment methods, and survival times were either summarized from the medical records or were retrieved via telephone calls with each referring veterinarian.
Cytology, Histopathology, and Immunohistochemistry
All cytology samples were originally analyzed by a board-certified clinical pathologist (A.L.M., F.C., P.M., M.P.). All the histologic slides, from all the institutions, were reviewed by the same board-certified veterinary pathologist (R.V.). Histologic sections were prepared from formalin-fixed paraffin-embedded blocks that had been fixed in 10% neutral buffered formalin and routinely stained with hematoxylin-eosin. Since the histopathologic pattern of clitoral carcinomas in dogs has been reported to overlap with AGASAs, 13,17 the tumors examined were morphologically classified into tubular, solid, rosette, or mixed type: the tubular pattern consisted of neoplastic cells arranged in variably sized lumina lined by cuboidal cells, the solid pattern was defined by nest structures, the rosette type was characterized by nuclei located in the basal aspect of neoplastic cells radially arranged around small amounts of proteinaceous eosinophilic material, and the mixed pattern was defined by a mixture of 2 or 3 of the patterns described above.
Representative sections of the lesions were subsequently selected for immunohistochemistry (IHC). The primary antibodies to synaptophysin (SYN), chromogranin A (CGA), neuron-specific enolase (NSE), S100, and pan-cytokeratin (AE1/AE3) are summarized in Table 1. All tissue sections were processed using the automated staining system BenchMark XT (Ventana Medical Systems, Tucson, AZ), where samples were deparaffinized, rehydrated, and processed for blocking endogenous peroxidase and epitope retrieval. Primary antibodies were incubated according to the protocol suggested by Ventana Medical Systems. As revelation system, ultraView Universal DAB Detection Kit was used (Ventana Medical Systems). Upon completion of the immunolabeling, sections were counterstained with Mayer’s hematoxylin. Positive controls comprised canine pancreas for SYN, CGA, and NSE; canine intestine and brain for AE1/AE3 and S100, respectively; the positive controls were selected according to the guidelines of the manufacturer. Negative controls were performed substituting the primary antibodies commercial unrelated immunoglobulins at the same concentration (Dako Universal Negative Control, Santa Clara, CA). Control sections were treated at the same time as sample sections.
Panel of Antibodies Used for Immunohistochemistry.
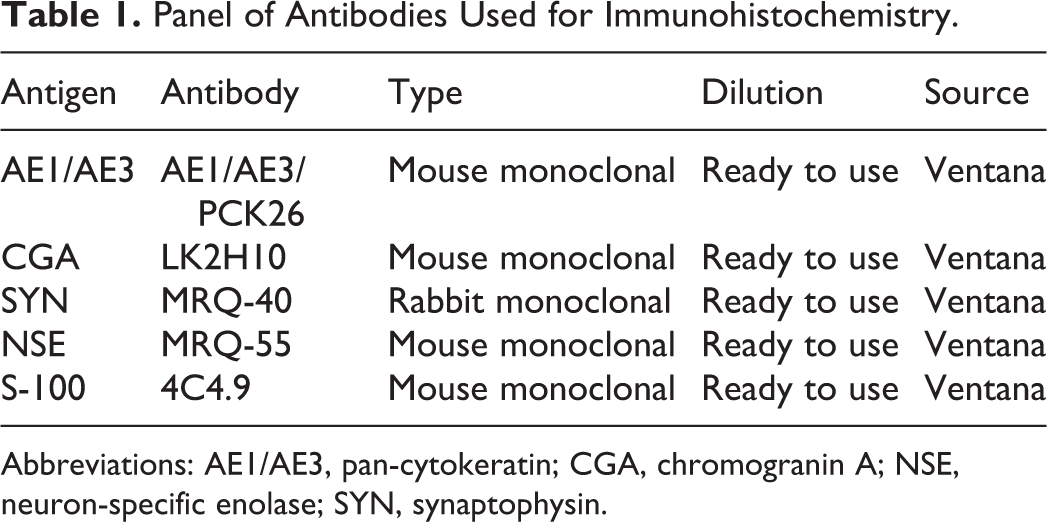
Abbreviations: AE1/AE3, pan-cytokeratin; CGA, chromogranin A; NSE, neuron-specific enolase; SYN, synaptophysin.
The IHC results were quantitatively categorized as follows: neg = no tumor cells stained; (+) = less than 10% or isolated positive tumor cells; + = 10%–50% of positive tumor cells; + + = 50%–90% of positive tumor cells; + + + = greater than 90% of positive tumor cells. Staining intensity was semiquantitatively reported as either light (barely perceptible brown stain), moderate (positive stain but weaker if compared with controls), or high (positive stain comparable with positive controls).
Transmission Electron Microscopy
Ultrastructural analysis was performed on all 6 cases. From formalin-fixed paraffin-embedded sections, the area of interest was identified and excess wax trimmed out. Tissue samples were processed following standard transmission electron microscopy (TEM) procedures and were subsequently embedded in fresh resin in polyethylene molds and polymerized at 60°C overnight. Toluidine blue–stained, 0.5-μm-thick semi-thin sections were used for selecting relevant areas for 75-nm ultrathin sections contrasted with lead citrate and uranyl acetate. Sections were examined under a Phillips EM208 S (FEI UK, Cambridge, UK) at 80 kV equipped with a charge-coupled device camera (ES500 W Erlangshen; Gatan, Pleasanton, CA). Neoplastic cells were assessed for the presence of electron-dense cytoplasmic granules suggestive of neuroendocrine differentiation. 5
Results
Cases
Six cases met the inclusion criteria. Breeds included were Basset Hound, Bullmastiff, English Springer Spaniel, Golden Retriever, and Bernese Mountain Dog; all dogs were female and spayed. The median age was 9.5 years (range, 5–13.5 years). The presenting clinical signs varied, including presence of a vulvar mass (Fig. 1), polyuria-polydipsia, frequent licking of the vulva, and, in 2 cases, hemorrhagic vulvar discharge. In 1 case, the owner did not report any abnormality and the mass was discovered as an incidental finding during a routine health checkup (case 3). The median duration of clinical signs was 30 days (range, 4–300 days).
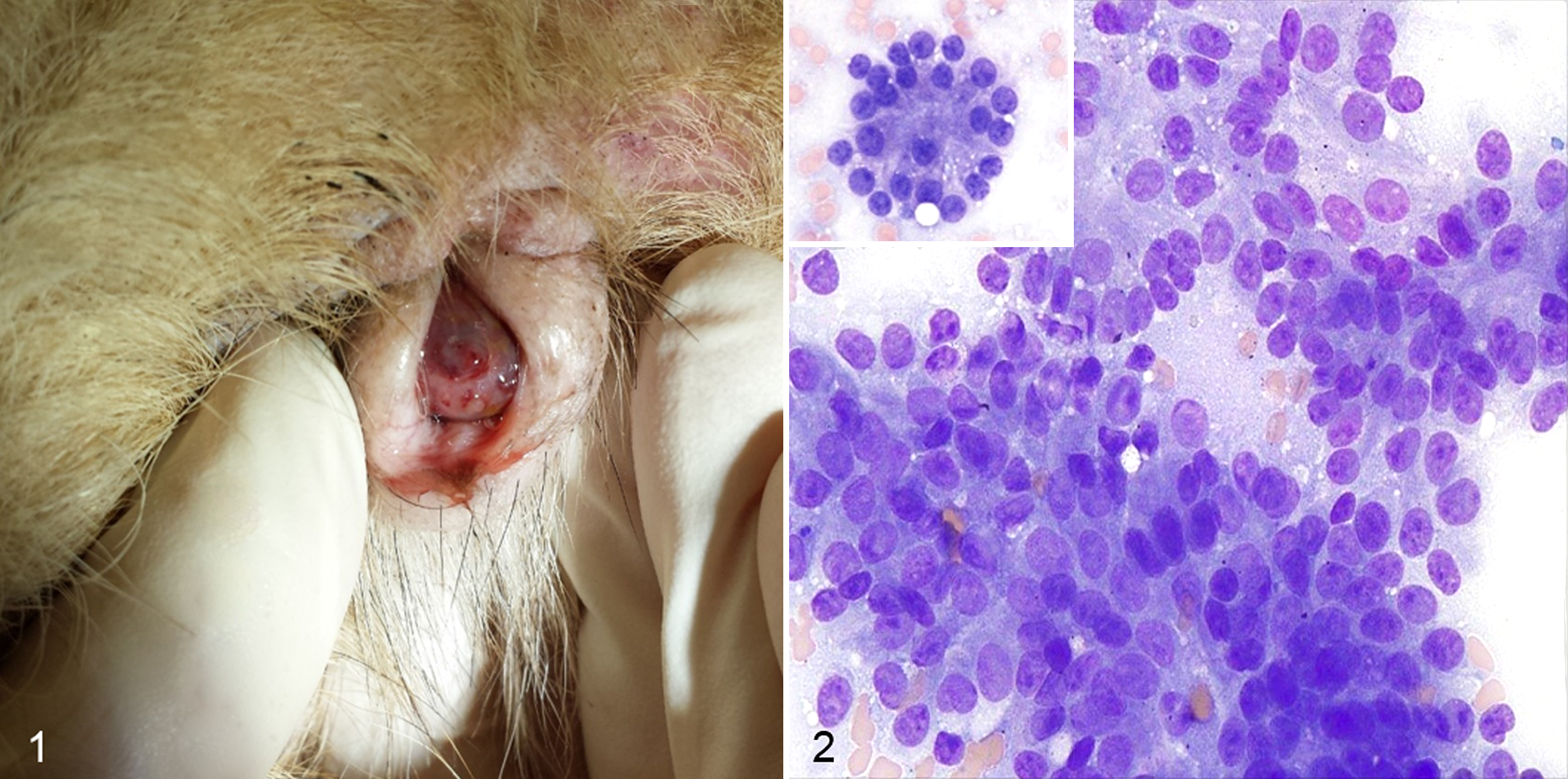
On physical examination, a clitoral mass could be detected in all cases, and the median size was 3 cm (range, 1–4 cm). All clitoral masses were firm, nodular, hyperemic, and effaced the normal clitoral structure extending to the clitoral fossa. The physical examinations were otherwise unremarkable, except for 1 dog (case 4) that had inguinal lymphadenopathy. Rectal examination was recorded in all cases, and anal sacs were easily expressed revealing no abnormalities. Supplemental Table S1 summarizes signalment, preexistent conditions, clinical signs, and physical examination findings.
Laboratory tests, including complete blood count and serum biochemistry profile, were performed in all dogs. Four cases had unremarkable hematology results, 1 had mild normocytic, normochromic nonregenerative anemia with concurrent moderate thrombocytopenia, and 1 had mild lymphopenia. On serum biochemistry, urea was elevated in 4 dogs, including 1 with concurrent creatinine elevation; cholesterol was elevated in 2 dogs, alkaline phosphatase was elevated in 2 dogs, and 1 dog was hyperglycemic. In the hyperglycemic dog, diabetes mellitus was diagnosed years prior to developing the clitoral mass. Hypercalcemia was detected in 2 of 6 cases, and this was confirmed through plasma ionized calcium elevation (case 2 and 4). In case 2, the parathormone-related peptide (PTHrp) was tested, and this was elevated, supporting HM. None of the hypercalcemic dogs had previously documented abnormalities of blood calcium. Supplemental Table S2 summarizes laboratory findings.
A diagnosis of malignant epithelial neoplasia was made by cytologic examination of fine-needle aspirates of the clitoral mass in 5 out of 6 cases, whereas 1 case was diagnosed by means of histopathology after surgical excision (case 5). Abdominal ultrasonography and 3-view thoracic radiographs were performed in 2 cases, 2 had total-body computed tomography, 1 had abdominal ultrasound only, and 1 had no imaging studies because of financial constraints. In the 4 cases in which thoracic imaging was available, no evidence of macroscopic metastatic disease was reported at the time of the initial consultation. Concerning abdominal imaging, regional lymphadenopathy was described in 3 of the 5 dogs: 1 dog had bilateral medial iliac and internal iliac lymphadenopathy, 1 dog had right medial iliac and right inguinal lymphadenopathy, and 1 dog had bilateral inguinal lymph node enlargement (Table 2). Fine-needle aspirates of the abnormal lymph nodes were examined in all 3 dogs, and these were confirmed to contain neoplastic epithelial cells based on cytopathology.
Diagnostic Imaging, Therapy and Case Outcome in Dogs With Clitoral Carcinoma.a

Abbreviations: US, abdominal ultrasound; CT, computed tomography of thorax and abdomen; LN, lymph node; NA, not applicable; NP, not performed; TXR, 3 views, thoracic radiographs.
aIn cases 1, 2, and 4, the detection of metastasis was based on diagnostic imaging and confirmed by cytology.
Treatment modalities varied among cases (Table 2). Surgical excision of the clitoral mass was performed in 4 dogs; surgical margins were not clear in 3 cases and were >8 mm in 1 case. None of the cases underwent excision of metastases. One dog had prednisolone treatment only, and 1 had no treatment. One dog received adjunctive intravenous carboplatin treatment, and objective partial tumor response was documented by reduction in size of the inguinal lymph nodes (>50%) and normalization of plasma ionized calcium concentrations. This dog received 7 cycles of carboplatin (300 mg/m2), remaining in stable disease with no further significant variation in lymph node size and ionized calcium concentration).
At the time of submission of this study, 2 dogs were alive and in complete clinical remission at 500 (case 3) and 100 (case 1) days after diagnosis, and 1 dog was in stable disease at 420 days after diagnosis (case 4). Three cases were euthanized because of tumor-related causes at 7 (case 6), 150 (case 1), and 180 (case 2) days after diagnosis. Postmortem examination was not performed in any of the cases.
Cytology
Cytology of the clitoral mass was performed in 5 of 6 dogs, and samples were considered adequate in all cases. Cells were arranged in dense cohesive clusters and often appeared as bare nuclei embedded in a background of light blue cytoplasm, with infrequent appearance of cytoplasmic borders (Fig. 2). Few cellular clusters formed an acinar or rosette-like arrangement (Fig. 2), and some others were associated with amorphous pink material. Nuclei were round, 10–15 μm in diameter, with finely stippled to granular chromatin and 1–2 small, round distinct nucleoli. Cytoplasm was light blue and occasionally contained small numbers of clear, distinct vacuoles. Anisocytosis and anisokaryosis were mild to moderate. Mitoses were infrequent. Low numbers of inflammatory cells were present and consisted of large vacuolated macrophages with signs of phagocytosis and containing a moderate amount of globular dark green-blue pigment and lymphocytes. Rare neutrophils, likely blood derived, were also noted. Cytological findings of the metastatic sites were consistent with the primary tumors.
Histopathology and Immunohistochemistry
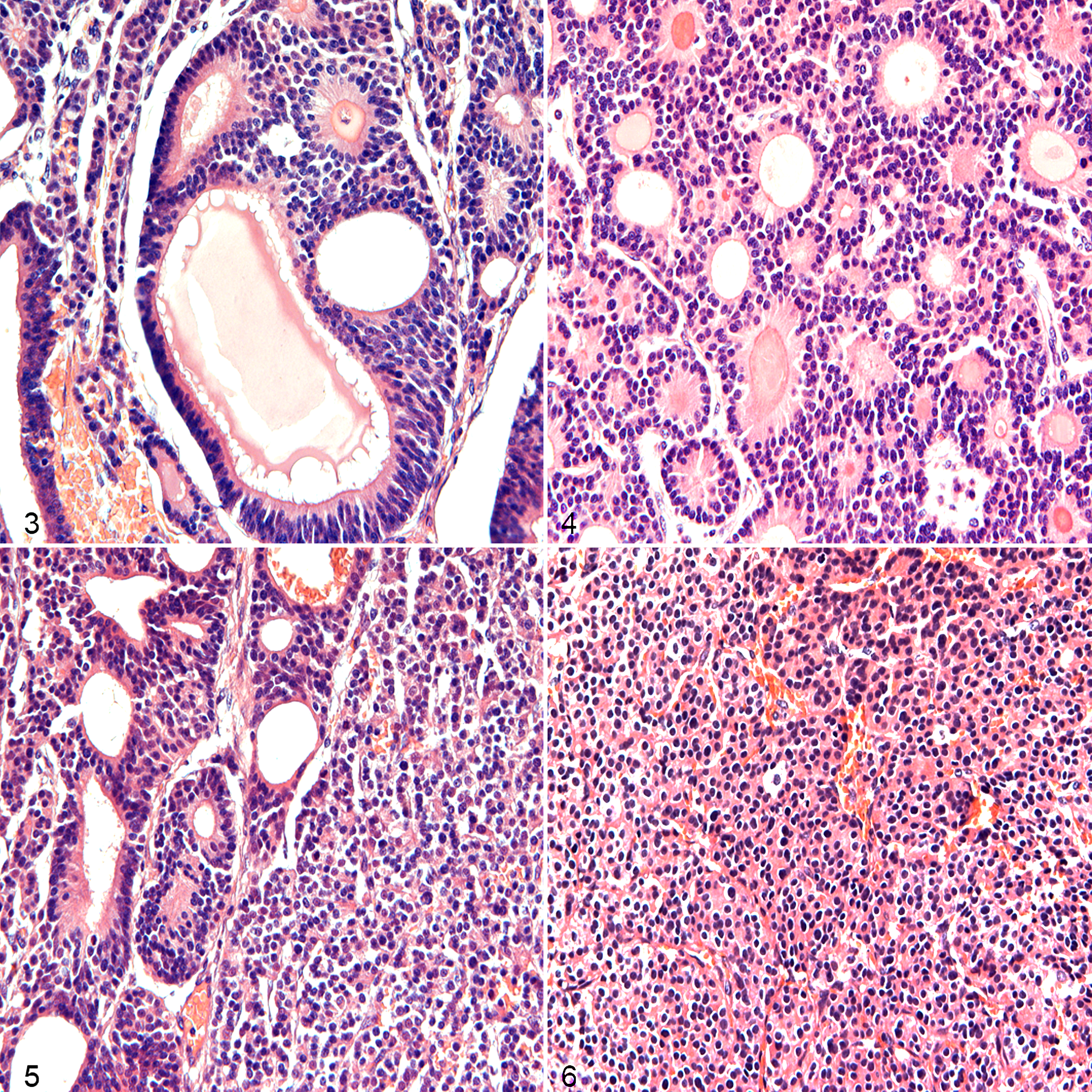
In all 6 cases, the normal clitoral microscopic anatomy was completely effaced and replaced (normal clitoral tissue was not observed) by a multilobulated, moderately demarcated, partially capsulated epithelial neoplasm showing 3 distinct patterns (tubular, solid, and rosette type) admixed with fine fibrovascular stroma (Figs. 3–6). The 3 patterns coexisted (ie, mixed type) in 3 of 6 tumors (cases 3, 4, and 6). Two tumors (cases 1 and 2) showed a mixed arrangement that included only 2 main patterns (tubular + solid, and solid + rosette), while only 1 case was considered as a pure rosette type (case 5). Mitotic index ranged from 1 to 5 mitoses per 400× field (0.237 mm2) among tumors, with occasional bizarre figures observed. Anisocytosis and anisokaryosis were mild to moderate in all the masses. All cases had neoplastic infiltration of lymphatics at the periphery of the masses. Morphological data are presented in Supplemental Table S3.
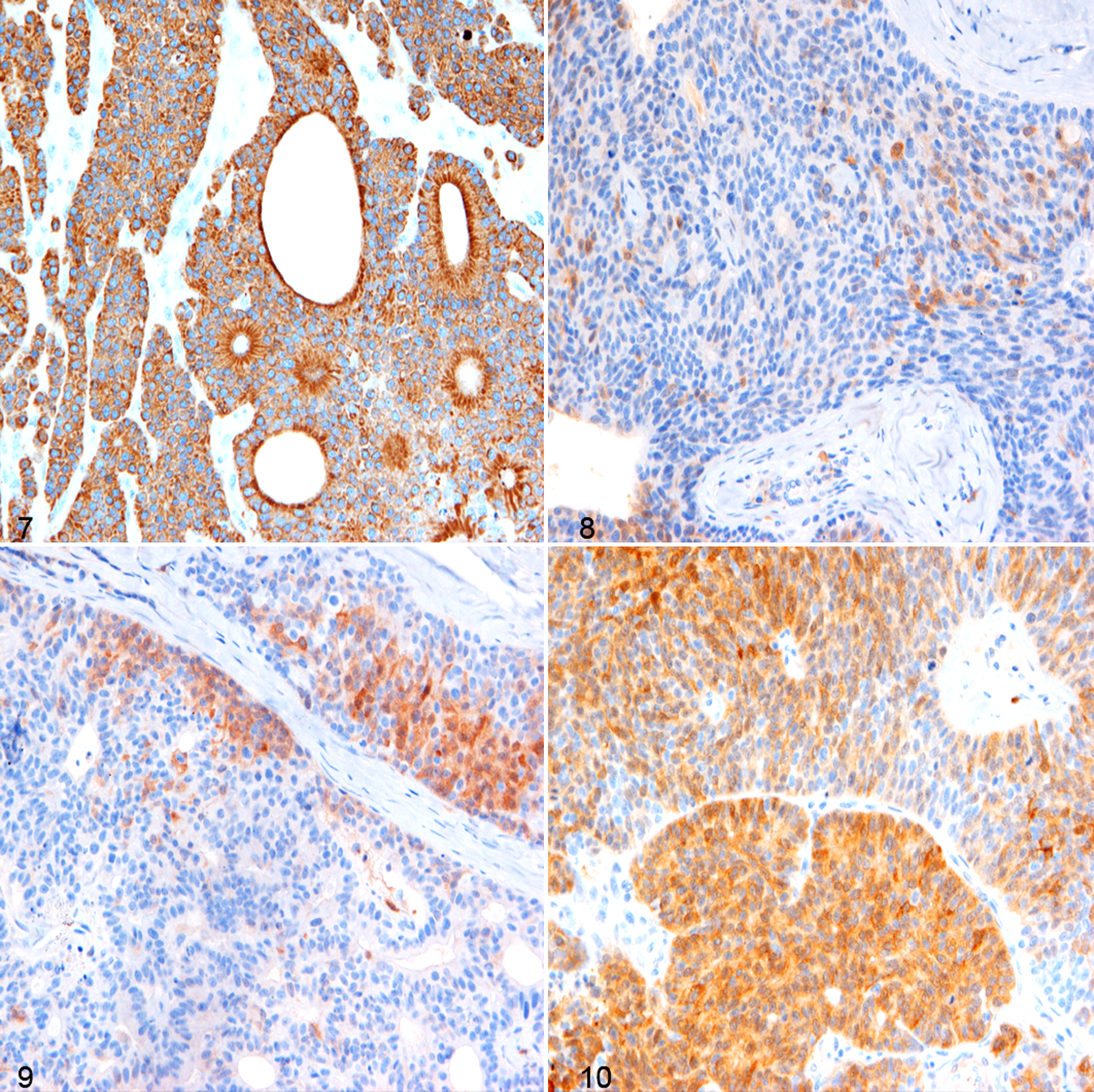
Immunohistochemical expression of AE1/AE3 was high and consistent (+++) in >90% of tumor cells in all the cases (Fig. 7), whereas neuroendocrine markers were variably expressed among the cell population (Table 3). Both CGA and SYN were mildly expressed (+) in 2 of 6 cases, with few positive cells (<10%) localized to marginal areas of neoplastic nests or less frequently as part of rosette-like structures (Figs. 8 and 9). NSE was consistently expressed in all 6 cases with light to moderate staining, and the percentage of positive cells varied among tumors (ranging from 10% to 90%). This was mainly located within tumor nests (Fig. 10). S-100 expression was not observed in any of the neoplasms.
Immunohistochemical and Ultrastructural Findings in Canine Clitoral Carcinoma.
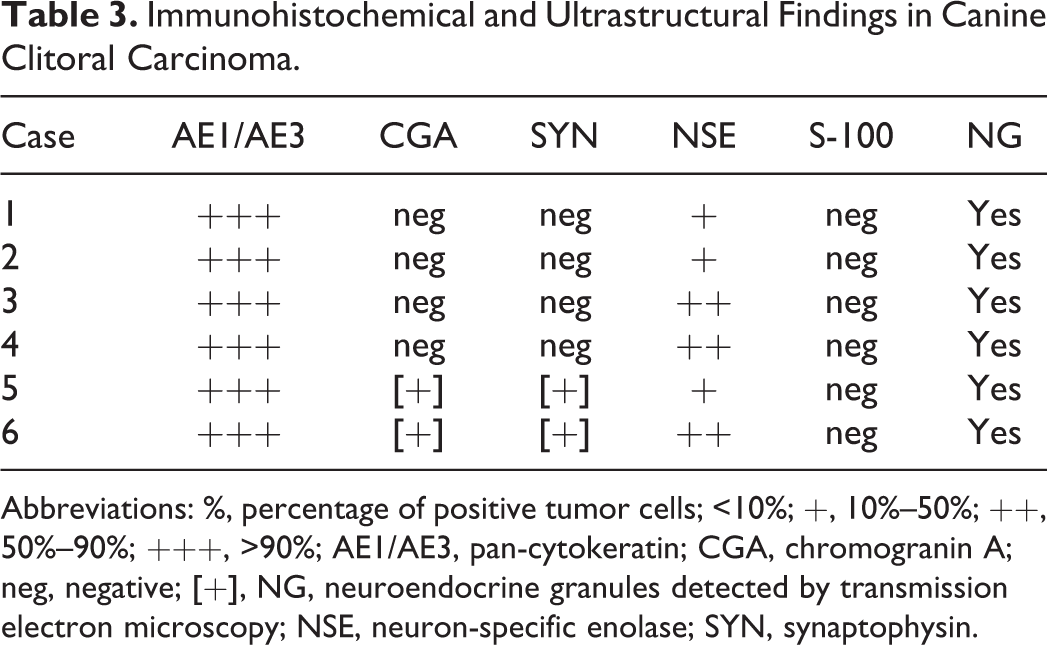
Abbreviations: %, percentage of positive tumor cells; <10%; +, 10%–50%; ++, 50%–90%; +++, >90%; AE1/AE3, pan-cytokeratin; CGA, chromogranin A; neg, negative; [+], NG, neuroendocrine granules detected by transmission electron microscopy; NSE, neuron-specific enolase; SYN, synaptophysin.
TEM
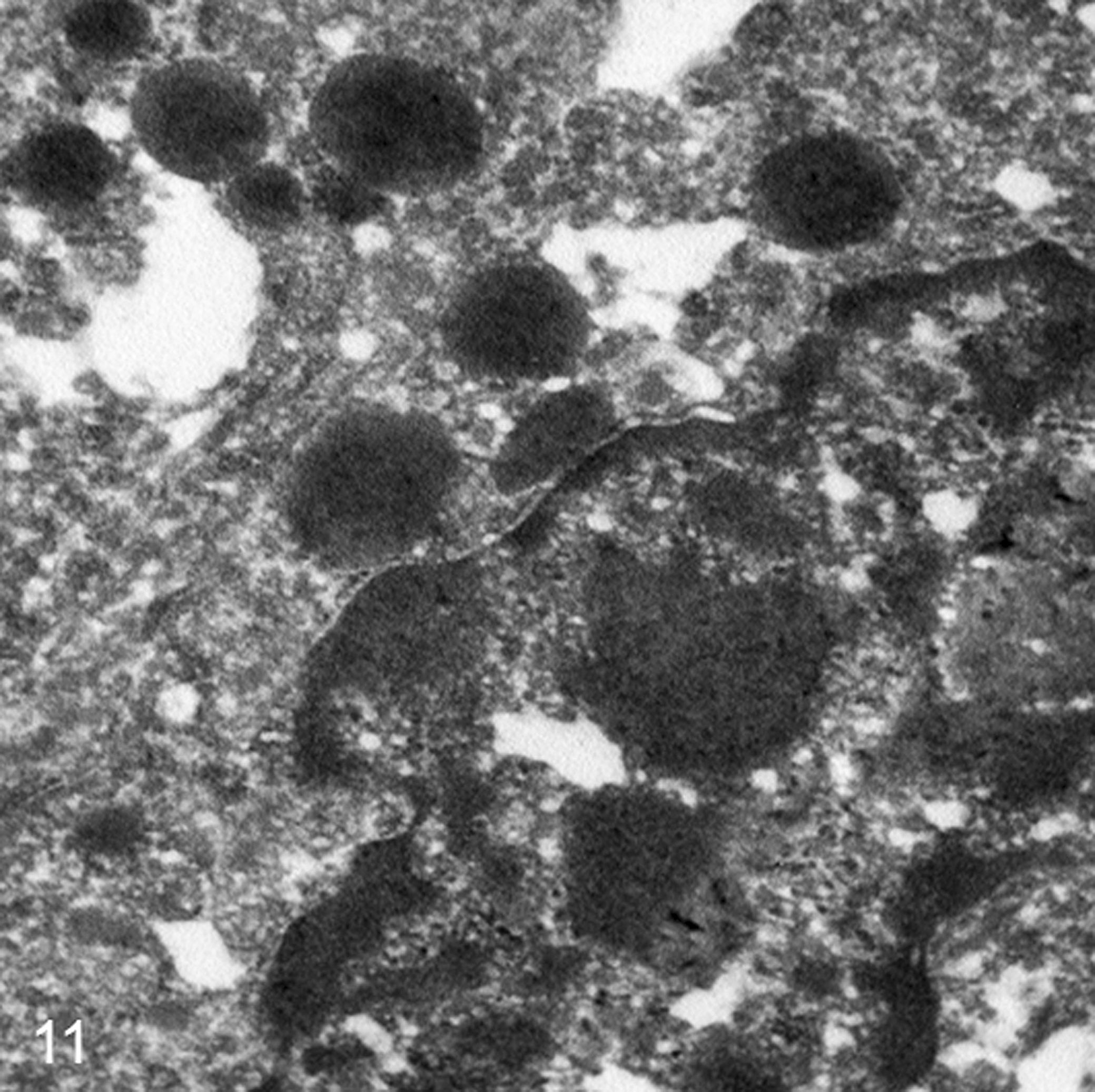
In all samples, tubular, solid, and rosette-like areas were observed on ultrastructural evaluation (Table 3). The quality of all samples was suboptimal because of the formalin fixation, and distinct cellular details were not available for all the fields observed. Tubular structures were lined by cuboidal cells sitting on a prominent basal membrane. Microvilli were present on the luminal surface of neoplastic cells forming tubular structures, and occasional cytoplasmic blebs, devoid of organelles, protruded from the apical surfaces. Desmosomes were constantly present along cell borders, and often plasma membranes of adjacent neoplastic cells showed characteristic invaginations. Nuclei were oval to round and uniform in dimensions and shape with a peripheral rim of heterochromatin and a paler central region (euchromatin) with 1 or 2 evident nucleoli. Small, round to oval osmiophilic granules were present and often associated with Golgi apparatus or near the cellular apex. The granules were present in all 6 cases and measured 200–400 nm, with or without an external limiting membrane and with a central electron-dense core (Fig. 11).
Discussion
Clitoral carcinoma is a rare neoplasm of the female canine genital apparatus, originating from the apocrine glands located at the base of the clitoris and in the clitoral fossa. This tumor was described for the first time in 2010 and subsequently reported in few isolated cases over the past few years. Nevertheless, it has become evident that this neoplastic condition presents microscopic and clinical features resembling those of an AGASA. 12,13,17
Considering the numerous institutions involved in this study, we can postulate that so far, CCC remains a very rare condition with likely a low prevalence among canine female genital tract tumors.
Of the 6 cases matching the inclusion criteria, no breed prevalence was noted, and most of the cases were adult to geriatric, which is in alignment with 2 previously published CCC cases and with the literature on vaginal and vulvar tumors. 4,7,12,13,21 Interestingly, all dogs in this study were spayed years prior to the occurrence of tumor-related clinical signs and/or the detection of a clitoral mass, suggesting that CCC is not a hormone-dependent tumor, which is consistent with the previous literature. 12,13 On the other hand, a protective role of sexual hormones could be hypothesized. Nonetheless, no studies have investigated the long-term effect of spaying on the clitoris’ structure, such as variation of differentiated epithelial to basal cells ratio as reported for canine prostate gland, where basal cells may have a predominant role in carcinogenesis; for this reason, this assumption remains just speculative at this stage. 10,20
In regard to their presentation, most dogs in this study had clinical signs related to lower urogenital tract disease; however, 2 of them had the masses found incidentally, suggesting that the diagnosis of CCC can be less obvious in certain cases.
Concerning hypercalcemia, this initially appeared to be a consistent finding in the case of CCC, but our results suggest that this abnormality may not always be present: only 2 of 6 cases resulted to be hypercalcemic (4 of 8 considering all the documented CCC cases). 12,13 Although no extensive workup was performed to exclude other causes of hypercalcemia other than neoplasia, we presumed CCC was the primary reason in both cases. In case 2, the elevated PTHrp strongly supported HM, and the staging workup did not show presence of other neoplastic processes. In case 4, plasma iCa normalized after the dog had an objective response to intravenous chemotherapy, suggesting a causative effect between CCC and hypercalcemia. Moreover, none of the cases was reported to be hypercalcemic on laboratory tests performed prior to the diagnosis of CCC, further supporting the link between CCC and HM. This would be expected as it is well known that the most common cause of hypercalcemia in the dog is cancer; several tumors have been associated with HM, such as lymphoma (35%), AGASA (27%), and multiple myeloma (20%), among others. 3 Based on our study and the previous literature, a dog with CCC has about 33%–50% chances to present with paraneoplastic HM 12,13 ; CCC should therefore be considered in the differential diagnosis of hypercalcemia in female dogs, aiming for a careful examination of the vulva, especially in spayed cases. HM may be due to ectopic production of parathormone or PTHrp by neoplastic cells, lytic bone metastases, primary hyperparathyroidism, interleukin-1β, and transforming growth factor–β, among other causes. 3 The HM seen in lymphoma and AGASA is mainly due to PTHrp and so seems to be in CCC; however, PTHrp was assessed just in 1 case and, therefore, another pathophysiologic mechanism might also be possible.
Complete staging workup was performed in 4 of 6 cases, and abdominal imaging was available in another case, with results suggesting that locoregional macroscopic metastases may be common at the time of the diagnosis in CCC, which is also in agreement with the literature. 12,13 Based on our data, the inguinal (2 of 5 cases), medial iliac (2 of 5 cases), and internal iliac (1 of 5 cases) lymph nodes should be scrupulously evaluated in the case of CCC. This represents a clinical similarity with AGASA, in which numerous studies have found a metastatic rate of at least 50% at the time of the diagnosis, with the medial iliac, sacral, and internal iliac lymph nodes representing the most common metastatic sites. 16 Given the small sample size, it is not possible to comment on the prognostic significance of the primary tumor size and number/size of metastases; however, the 2 cases presenting with no gross metastatic disease were alive and free of disease at the time of the last staging workup, 500 and 100 days postdiagnosis. Interestingly, despite the presence of metastatic disease, dogs that received palliative treatment, either debulking surgery, chemotherapy, or treatment for management of HM, survived between 5 and 14 months. Concerning chemotherapy, it is pivotal to highlight that the dog receiving intravenous carboplatin had an objective and long-lasting response (>50% reduction in tumor volume and iCa normalization), suggesting that this chemotherapeutic therapy might be useful in the management of CCC cases.
Fine-needle aspiration from all of the masses harvested large numbers of naked nuclei embedded in a background of lightly blue cytoplasm, forming dense clusters, and occasionally showing an acinar or rosette-like arrangement. These cytological features, which are now considered typical for CCC, closely resemble those of AGASA and neuroendocrine neoplasms. 17 Therefore, localization of the primary mass is considered crucial for a correct interpretation. Interestingly, in all of the submitted samples from CCC, small numbers of foamy macrophages and small amounts of amorphous pink material were seen interspersed between the neoplastic cells. The accumulation of an eosinophilic secretory product in the glandular lumina, often mixed with macrophages, is a feature described in apocrine and modified apocrine gland neoplasms, including AGASA. 6
Concerning the histopathologic features, the tumors examined were classified into 3 different histological types representing different degrees of differentiation: tubular, solid, and rosette type. Since rosette-like and solid patterns are commonly found in tumors of neuroendocrine origin, and since CCCs share morphological features with AGASA with potential of neuroendocrine differentiation, we postulated that CCCs may be neuroendocrine in nature. 19 Moreover, CCCs can present with HM, which is a clinical feature often associated with AGASAs, reinforcing the hypothesis of a possible neuroendocrine origin for this neoplasm, which also nicely overlaps morphologically with AGASA. Nevertheless, IHC for neuroendocrine markers has been performed only on 1 previously reported case with no evidence of neuroendocrine marker expression. 17 In the other 2 reported CCCs, only the epithelial origin was demonstrated by means of pan-cytokeratin and E-cadherin immunolabeling. 12,13 In the present study, we submitted all 6 cases to a panel of IHC with antibodies against AE1/AE3 and several neuroendocrine markers (CGA, SYN, NSE, and S-100) to further prove the epithelial origin of CCCs and investigate the relationship between morphological features and neuroendocrine differentiation, respectively. In our study, the distribution and intensity of stain of CGA- and SYN-positive neoplastic cells (in 2 of 6 tumors) was less than that of NSE-positive cells (6 of 6 tumors). Some studies suggest that NSE and SYN could be more useful than CGA in dogs and cats to prove neuroendocrine differentiation. 14,15 We classified our cases as neuroendocrine in origin if positive for at least 2 neuroendocrine markers. Two tumors in our population (1 morphologically classified as rosette type and 1 as mixed type with solid + tubular + rosette pattern) showed positive signals for CGA, SYN, and NSE and therefore were considered carcinomas with neuroendocrine features.
NSE expression was consistently observed in all 6 tumors with different degrees of intensity and percentage of cells stained (see Table 3); despite this, as discussed above, it should be considered a useful marker to investigate the neuroendocrine nature of neoplasia only if coupled with other neuroendocrine markers. AGASAs are associated with HM, and TEM studies have demonstrated a possible link between this clinical feature and the presence of secreting granules (possibly containing PTHrp). 11 All 6 of our cases were examined by means of TEM and showed the presence of cytoplasmic osmiophilic granules that are consistent in dimension (200–400 nm), shape, and location with secretory granules and are very similar to small granules observed in the cytoplasm of a subpopulation of AGASAs associated with HM. 11 Despite this ultrastructural finding, only 2 of 6 tumors examined in this study presented with hypercalcemia of malignancy, and therefore, further studies should focus on the actual content of cytoplasmic granules to demonstrate or rule out the presence of hypercalcemic factors. Larger osmiophilic bodies (measuring 0.6–2.2 μm) that are often observed in AGASAs along small granules were not observed in neoplastic cells in this study despite the hypercalcemic status. 11
To conclude, CCC is a malignant neoplasm of the female external genital tract that seems to mainly present in adult to geriatric spayed females and has an aggressive and infiltrative regional growth with high incidence of locoregional lymph node metastasis. CCC can often present with HM, and long-term outcome is likely poor; however, cases in which surgical excision can be performed and that are free from metastases may have a better prognosis. Cytology, histopathology, and TEM features resemble those of canine AGASA, which represents the main differential. CCC seems to be a rare neoplastic disease, but its presence might have been underestimated because of the overlapping features with AGASA, especially when tissue samples are taken from metastatic sites. Further studies should aim at this point to define the true incidence of this disease.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818759772 - Canine Clitoral Carcinoma: A Clinical, Cytologic, Histopathologic, Immunohistochemical, and Ultrastructural Study
Supplemental Material, DS1_VET_10.1177_0300985818759772 for Canine Clitoral Carcinoma: A Clinical, Cytologic, Histopathologic, Immunohistochemical, and Ultrastructural Study by Ranieri Verin, Francesco Cian, Jennifer Stewart, Diana Binanti, Amy L. MacNeill, Martina Piviani, Paola Monti, Gianna Baroni, Sophie Le Calvez, Timothy J. Scase, and Riccardo Finotello in Veterinary Pathology
Footnotes
Acknowledgments
We thank Marion Pope for the technical assistance with electron microscopy and Monica Pepi for the technical assistance with IHC procedures. We would like to thank all the referring veterinarians and oncology specialists that have contributed by providing their cases.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
