Abstract
An intact, 8-year-old, male Golden Retriever dog was presented for evaluation of a nasal mass and approximately 30 firm, raised, variably ulcerated dermal and subcutaneous masses. Histopathology of both nasal and multiple skin masses revealed multiple nonencapsulated, infiltrative masses comprising clusters, anastomosing trabeculae, and packets of neoplastic, round to ovoid, hyperchromatic cells with marked nuclear molding. Surrounding the neoplastic cells was a marked stromal response in which many of the spindle-shaped cells expressed muscle-specific actin and had ultrastructural features consistent with myofibroblasts. A literature search indicates that this is the first report in a peer-reviewed journal of cutaneous metastasis of a nasal neuroendocrine tumor in any domestic animal species.
Primary neoplasms of the sinonasal region are uncommon tumors in dogs. Of those diagnosed, the majority are carcinomas, followed by sarcomas of cartilage, fibrous tissue, or bone. 5 In addition to epithelial, neuroepithelial, and mesenchymal elements, the sinonasal area also contains cells of the dispersed neuroendocrine system. These cells originate embryologically from the endoderm, specifically the gut, and are distributed widely throughout the respiratory, hepatobiliary, and gastrointestinal systems. Neoplasms of the dispersed neuroendocrine system are rare in domestic animals, but they have been reported in the esophagus, stomach, intestines, liver, gallbladder, bile duct, lungs, pharynx, and nasal cavity. 1,4,9,10,15 –19,21 –24 Despite their widespread and diverse locations, and likely origin from multipotent stem cells rather than fully differentiated neuroendocrine cells, well-differentiated neuroendocrine tumors share a common classic histopathological appearance consisting of sheets, nests, or cords of small to medium-sized cells separated into “packets” by a delicate fibrovascular stroma. 25 Less well-differentiated tumors, on the other hand, can present a diagnostic challenge, and in human pathology they are often grouped under the umbrella of “small cell carcinoma.” In this report, we present the clinicopathologic, histomorphologic, immunocytochemical, and electron microscopic features of a case of a poorly differentiated neuroendocrine carcinoma of the nasal cavity of a dog with marked widespread cutaneous metastases and an unusual histopathological appearance that includes a marked myofibroblastic stromal response.
Differential Diagnoses
Grossly, differential diagnoses for a primary nasal tumor with presumed cutaneous metastases would include nasal adenocarcinoma, transitional carcinoma, neuroendocrine carcinoma, olfactory neuroblastoma, and primitive neuroectodermal tumor; and for multicentric disease, it would also include many carcinomas, sarcomas, and round cell tumors.
Clinical History and Gross Findings
An intact, 8-year-old, male Golden Retriever dog was presented to the Auburn University Small Animal Teaching Hospital (AUSATH) in October 2009 with a 9-month history of sneezing, snorting, discharge, and pawing at the nose, and a 2-month history of nasal deviation and multiple cutaneous and subcutaneous masses. Additional clinical history included previously treated heartworm disease, hypothyroidism treated with thyroxine supplementation, protein-losing nephropathy, and a grade II fibrohistiocytic nodule in his spleen that had previously been treated by splenectomy (slides were reviewed by JWK, and the diagnosis was confirmed). Biopsies of the nasal passages approximately 4 months prior to presentation revealed only a chronic, marked, ulcerative, and pyogranulomatous rhinitis. Upon presentation, the dog was bright, alert, and responsive, and vital signs were within normal limits. Approximately 30 firm, raised, 1- to 3-cm diameter cutaneous and subcutaneous masses were noted over the body, predominantly over the trunk (Fig. 1). There was moderate asymmetry of the rostral muzzle with deviation of the nose to the left, but masses could not be visualized in the nares or external nasal cavity, and no nasal discharge was present at that time. Imaging of the nasal cavity was initially declined by the owner. Biopsies were obtained of 4 of the dermal masses: the right shoulder, the dorsal midline, and 2 from the dorsal neck.
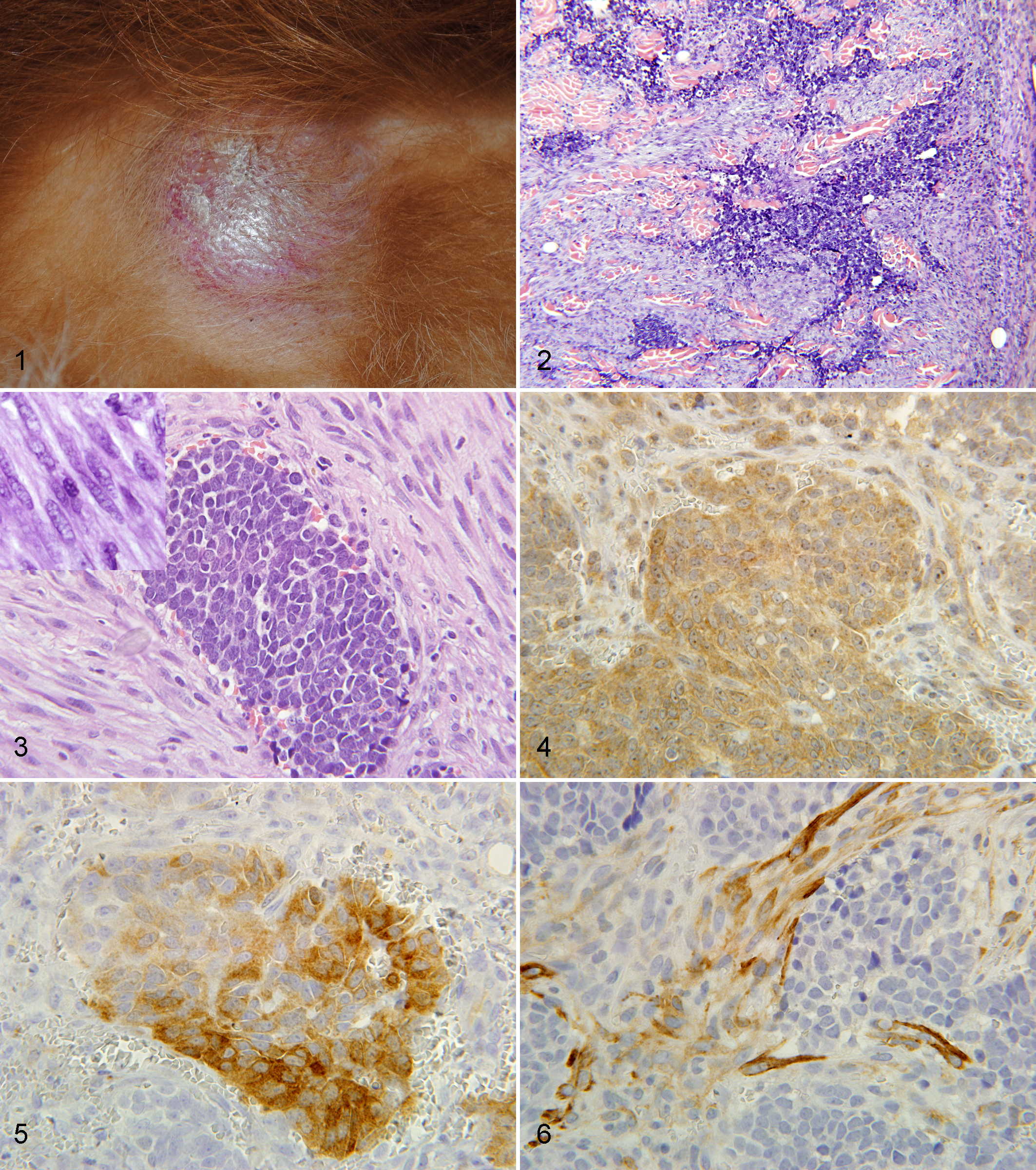
Gross image, cutaneous metastasis of poorly differentiated nasal neuroendocrine carcinoma.
Based on the diagnosis obtained at that time, the dog was initially treated with cyclophosphamide (250 mg/m2) and furosemide (2 mg/kg) intravenously. One week later, the cutaneous masses had increased in size, and many were erythematous with superficial erosion or ulceration. Magnetic resonance imaging of the nasal passages was permitted at this time. On T2-weighted transverse images, an abnormal signal intensity was present in the right nasal passage, beginning in the rostral portion of the nasal passage with extension caudally to the cribriform plate. There was extension into the ventral aspect of the right frontal sinus and laterally just across the midline. On the post-contrast images, there was non-uniform enhancement of the mass within the nasal passage. Fluid was present in the frontal sinus, with small amounts of a soft tissue mass involving the ventral aspect. The cribriform plate appeared intact.
The overall impression of the findings was most consistent with a neoplasm. Incisional biopsies of the nasal mass were submitted, and the histopathological appearance was similar to that of the multiple, previously biopsied cutaneous masses. Despite palliative radiation therapy (4 consecutive daily doses of 5 Gy each for a total of 20 Gy) and a variety of chemotherapeutic agents including carboplatin (300 mg/m2 intravenously), piroxicam (0.3 mg/kg orally once daily), and CCNU (60 mg/m2 orally once), the dog continued to deteriorate clinically, developed neurologic signs, and was euthanized approximately 3.5 months after initial presentation. Necropsy was not permitted.
Microscopic Findings
Sections of all submitted tissues were fixed in 10% neutral buffered formalin, then subsequently paraffin embedded with routine methods, sectioned at 4 to 5 µm, stained with hematoxylin and eosin, and examined via light microscopy. The dermis contained a densely cellular mass composed of clusters, packets, and broad, anastomosing trabeculae of neoplastic round to polygonal cells that often elevated the overlying epidermis and extended into the subcutis (Fig. 2). These cells were moderately pleomorphic and had scant to small amounts of eosinophilic to amphophilic, faintly granular cytoplasm and round to ovoid hyperchromatic nuclei with prominent nuclear molding, coarsely clumped chromatin, and a single paracentral magenta nucleolus (Fig. 3). Cells very rarely formed small, primitive, rosette-like structures, and many had nuclear cytoplasmic invaginations. Mitoses averaged 5 per 400× field. Surrounding the neoplastic cells was a marked stromal response, composed of broad, intersecting, moderately cellular streams of spindle-shaped cells with elongate, tapered, or blunt-ended nuclei, embedded in variable amounts of fibrocollagenous matrix. Multifocally, these cells were strongly reminiscent of smooth muscle and occasionally appeared to abut and blend with the adjacent piloerector muscles (Fig. 3). In some areas at the interface between neoplastic and myofibroblastic cells, there was mild, multifocal hemorrhage and mild infiltration of small lymphocytes, plasma cells, and macrophages.
Paraffin-embedded tissues were sectioned at 3 to 5 µm and placed on charged glass slides, then stained with the following antibodies on a Dako Autostainer (Carpinteria, CA) using 3,3′ diaminobenzidine (DAB) as the chromogen: vimentin, cytokeratin AE1/3, cytokeratin 18, neuron-specific enolase, chromogranin A, synaptophysin, neurofilament protein, type III β-tubulin, CD3, CD79a, CD18, and muscle-specific actin (Table 1). All antibodies were purchased from Dako (Carpinteria, CA), except CD18 (Dr. Peter Moore, University of California–Davis); and cytokeratin 18, type III β-tubulin, and neurofilament protein (Abcam, Cambridge, MA). Immunohistochemically, the neoplastic cells displayed positive reactivity for chromogranin-A (Fig 4, diffuse with areas of increased intensity), neuron-specific enolase (Fig. 5, multifocal), and negative reactivity for vimentin, cytokeratin AE1/AE3, cytokeratin 18, CD3, CD79a, CD18, synaptophysin, S-100, GFAP, type III β-tubulin, and neurofilament. Spindle-shaped cells within the stromal population displayed positive reactivity for vimentin (diffuse) and muscle-specific actin (Fig. 6, multifocal), and negative reactivity for all other previously mentioned markers.
Immunohistochemical Antibodies, Protocol Used, and Reactivity in Metastatic Nasal Neuroendocrine Carcinoma
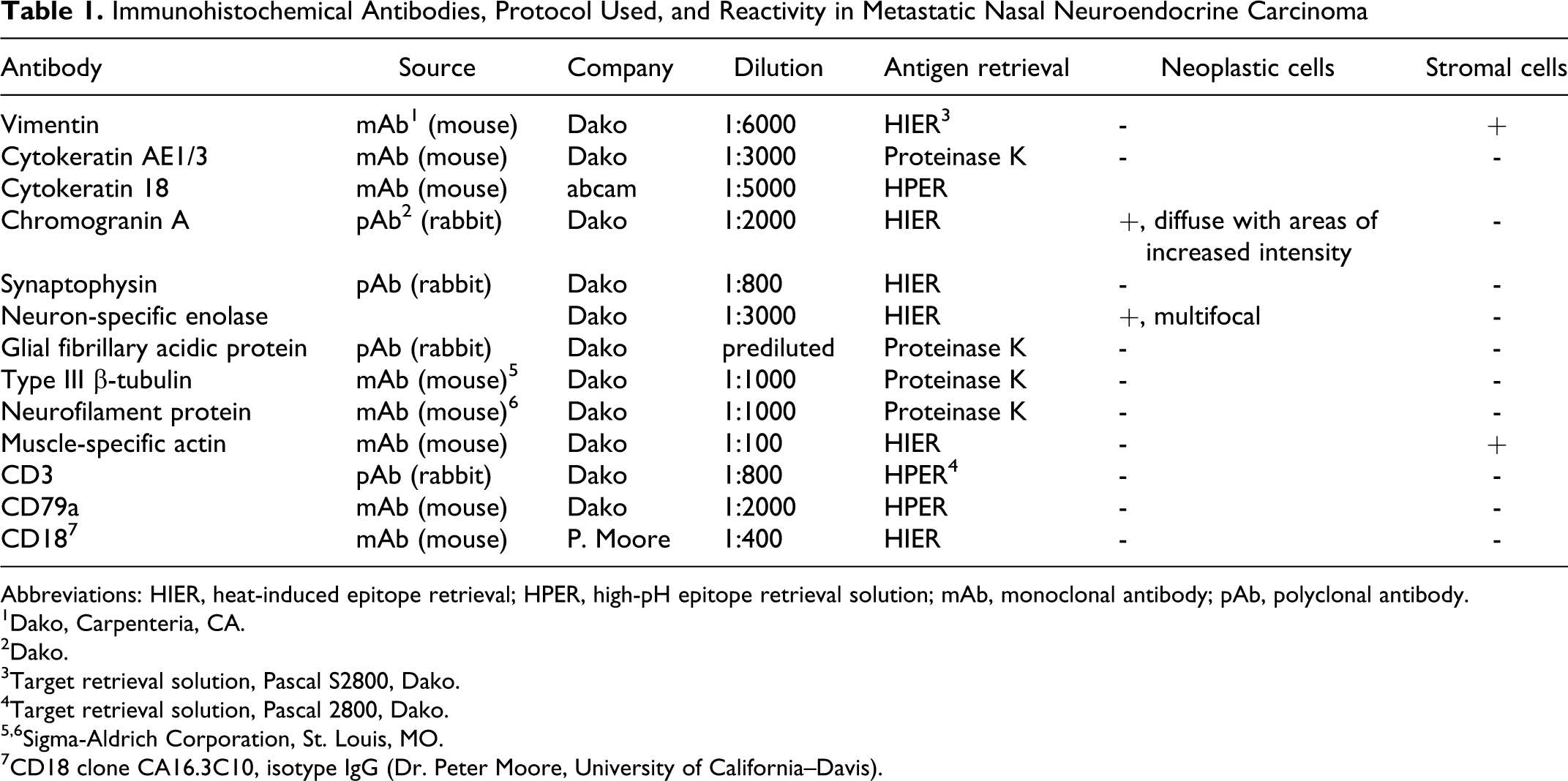
Abbreviations: HIER, heat-induced epitope retrieval; HPER, high-pH epitope retrieval solution; mAb, monoclonal antibody; pAb, polyclonal antibody.
1Dako, Carpenteria, CA.
2Dako.
3Target retrieval solution, Pascal S2800, Dako.
4Target retrieval solution, Pascal 2800, Dako.
5,6Sigma-Aldrich Corporation, St. Louis, MO.
7CD18 clone CA16.3C10, isotype IgG (Dr. Peter Moore, University of California–Davis).
For transmission electron microscopy, sections of tissue were fixed in fresh 5% glutaraldehyde, postfixed with 1% osmium tetroxide, processed routinely, and embedded in plastic. Sections 0.5-µm thick were stained with toluidine blue for selection of appropriately representative microscopic fields, which were then excised, cut at 60- to 90-nm thick, mounted on copper grids, stained with uranyl acetate and lead citrate, and examined with an electron microscope at 60 kV (Phillips EM 301, Phillips Electronic Instruments, Mount Vernon, NY). Ultrastructurally, the neoplastic cells had multiple desmosomal attachments to neighboring neoplastic cells and rare membrane-bound dense core structures consistent with neurosecretory granules (Fig. 7). The adjacent myofibroblastic cells were surrounded by collagen fibrils and had abundant rough endoplasmic reticulum; multiple plasma membrane attachment plaques; intracytoplasmic, peripherally located actin filaments; and focal dense bodies (Fig. 8).
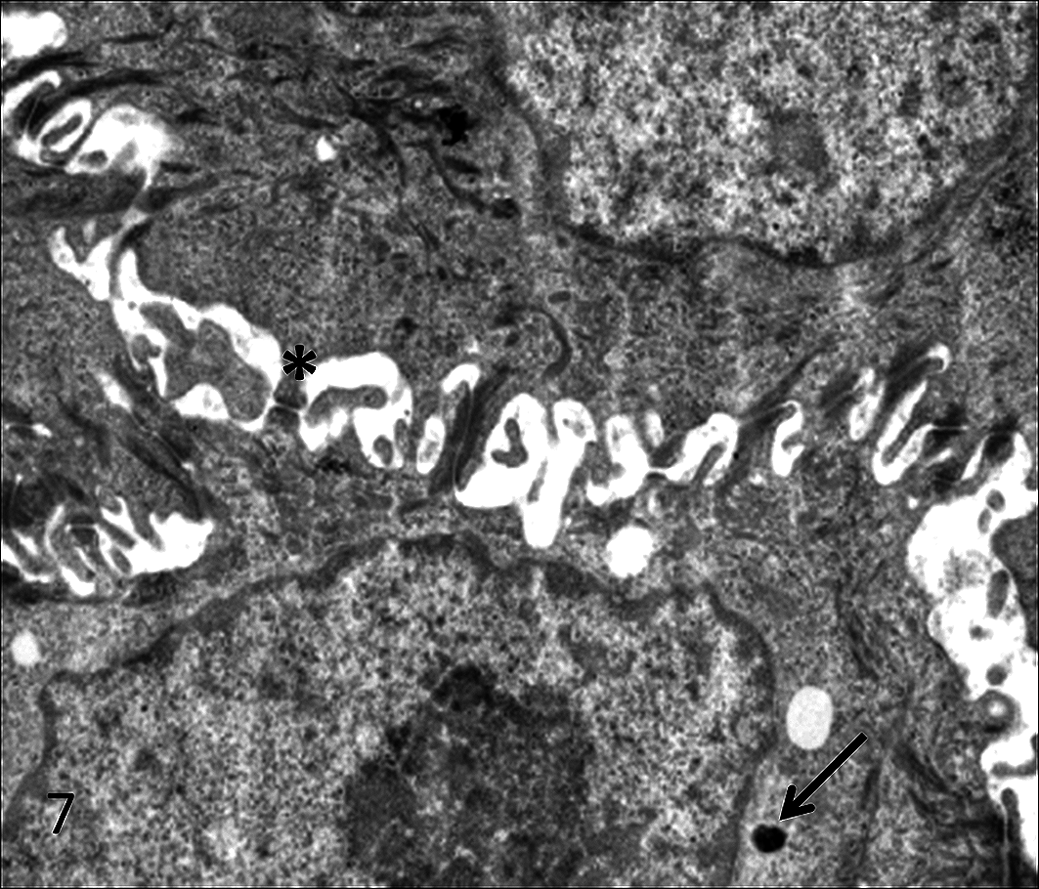
Transmission electron micrograph of poorly differentiated neuroendocrine carcinoma. Neoplastic cells exhibit multiple desmosomal attachments (asterisk) and very rare membrane-bound dense core neurosecretory granules (arrow). Uranyl acetate–lead citrate staining.
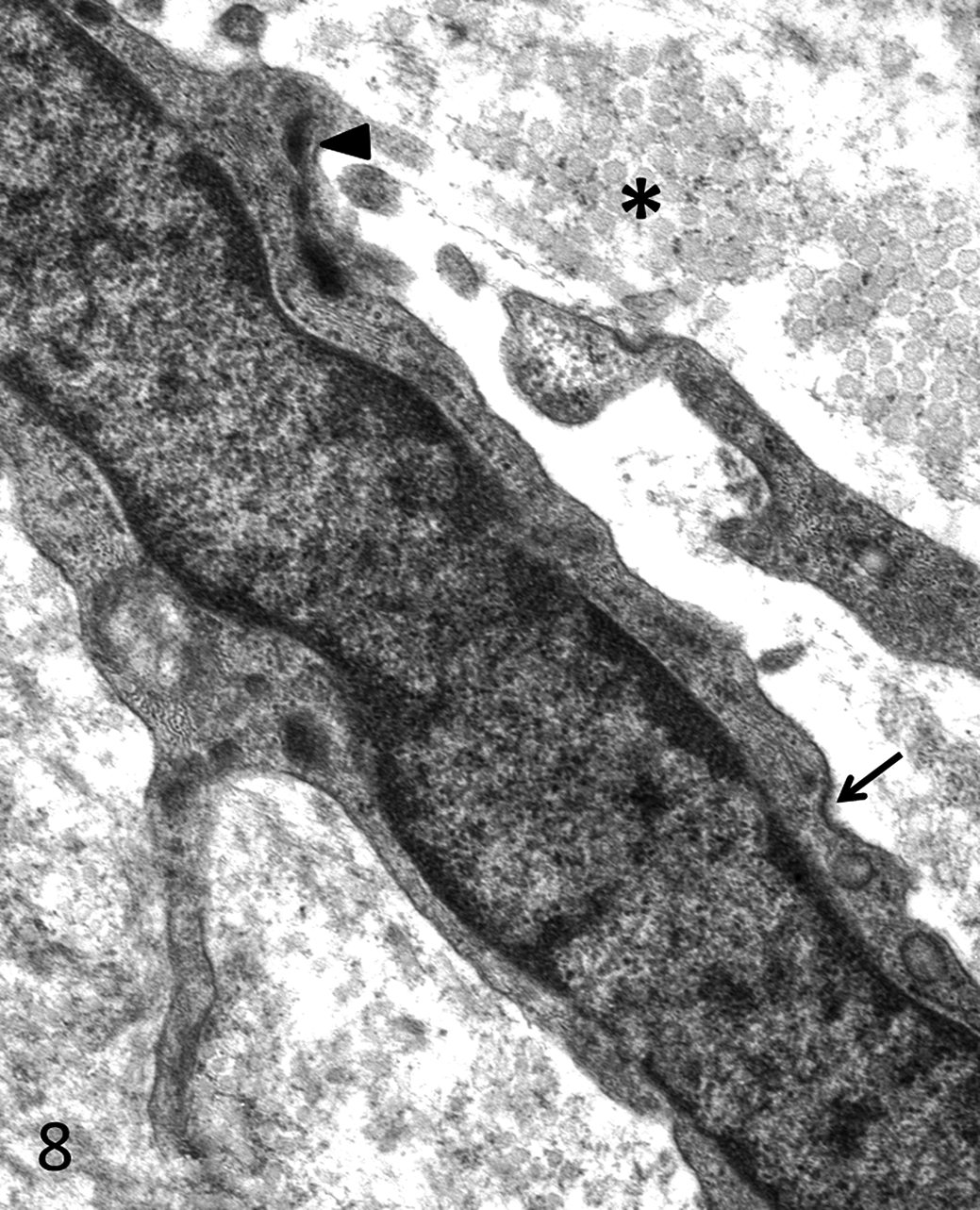
Transmission electron micrograph of stromal myofibroblast. Cells are embedded in a variably abundant collagenous matrix (asterisk) and have abundant rough endoplasmic reticulum, occasional focal dense bodies (arrowhead), and electron dense attachment plaques (arrow). Uranyl acetate–lead citrate staining.
Diagnosis
Based on the light microscopic appearance, immunohistochemical profile, and ultrastructural findings, these masses were diagnosed as a poorly differentiated neuroendocrine carcinoma with marked myofibroblastic stromal response and multiple cutaneous metastases.
Discussion
Tumors of the dispersed neuroendocrine system are rarely reported in the dog and are even more uncommonly reported in sinonasal and nasopharyngeal locations. 8,10,13,21 When these tumors occur in the sinonasal cavity, histopathologic differentiation from olfactory neuroblastoma can be problematic, as light microscopic features such as small-to medium-sized hyperchromatic cells, peripheral palisading, and rosette formation have been described in both tumors. 25 Additionally, whereas the histomorphologic features of cell packeting and granular cytoplasm of well-differentiated neuroendocrine tumors is distinct and often diagnostic, more poorly differentiated neuroendocrine carcinomas may in whole or in part lack these features. There can also be considerable immunohistochemical overlap between neuroendocrine tumors and olfactory neuroblastomas. However, Sako et al recently demonstrated the potential utility of neurofilament immunohistochemical staining in differentiating the 2, whereby all 11 examined well-differentiated neuroendocrine carcinomas in their study uniformly lacked neurofilament immunoreactivity. 21 Neoplastic cells in the present case were uniformly negative for both neurofilament and type III β-tubulin. Cytokeratin staining results in the literature are conflicting. Though Sako et al report cytokeratin AE1/3 staining in the tumors in their case study, 21 at least one well-cited author (Patnaik) reported negative results with cytokeratin AE1/3 immunohistochemical staining of a canine nasopharyngeal neuroendocrine tumor and a feline Merkel cell tumor. 17,19 Additionally, a loss of cytokeratin expression would not be inconsistent with a poorly differentiated tumor.
Determination of malignancy in neuroendocrine tumors is based on tumor size (> 2-cm diameter), evidence of invasion, presence of necrosis, cellular atypia and mitotic rate, and factors such as hormone expression and nuclear p53 protein accumulation. 17 Ultrastructurally, differentiation of neuroendocrine carcinoma from olfactory neuroblastoma is made by observing the presence of cytoplasmic 100- to 400-nm diameter, membrane-bounded, dense-core neurosecretory granules (if well-differentiated). Additional distinguishing features of neuroendocrine tumors include distinct desmosome-like intercellular junctions and an absence of neurofilaments, microtubules, dendritic processes, olfactory vesicles with microvilli, premelanosomes, or sustentacular cells. 21 In the present case, only rare neurosecretory granules were observed ultrastructurally; this finding is not inconsistent with a poorly differentiated tumor, as it has been noted in human respiratory small cell carcinomas that approximately 11% do not have ultrastructurally identifiable, dense-core neurosecretory granules. 20 Several prominent desmosome-like intercellular adhesions were identified in the present case, a feature that is consistent with previously reported neuroendocrine tumors.
Classification of tumors arising from the dispersed neuroendocrine system can be confusing because of the evolving nature of the nomenclature. Previously held views regarding the origin of these tumors as arising from fully differentiated neuroendocrine cells are being reshaped by modern molecular and genetic knowledge of stem cells, cellular plasticity, and genetic switches. 7 Additionally, spontaneous or posttreatment acquisition of a neuroendocrine phenotype is documented in a variety of tumors, most notably in human prostate cancers, in which it is associated with invasion, metastasis, and poorer prognosis. 27 Likewise, a recent case series of 18 dogs with nasal adenocarcinomas showed that these tumors expressed varying amounts of neuroendocrine differentiation. 10 Until further insight into the histogenesis of these tumors emerges, diagnostic pathologists must continue to rely on a combination of immunohistochemical markers and ultrastructural features to identify poorly differentiated tumors.
A unique aspect of this case is the presence of widespread cutaneous metastases. A literature search in PubMed indicates that this is the first report of widespread cutaneous metastasis of a nasal tumor in any domestic animal species. Unfortunately, because a necropsy was not allowed, another primary tumor site cannot be definitively ruled out in this case. Reports of widespread multiple cutaneous metastases of primary neuroendocrine carcinomas, including respiratory and genitourinary, are very rare in the human literature. In people, the identified sites of the primary tumors are the larynx, lung, 2,3 mediastinum, urinary bladder, 26 uterus, or uterine cervix. 6,8,9 Although a major limitation in the present case is the absence of a full necropsy evaluation, the presence of a single large tumor in the nose and multiple smaller, uniformly sized tumors in the dermis is highly suggestive of a primary nasal tumor with cutaneous metastases.
In summary, we present the diagnostic challenge of a presumptive primary poorly differentiated nasal neuroendocrine tumor in a dog associated with widespread cutaneous metastases and a marked myofibroblastic stromal response. A literature review indicates that this is the first such report in a peer-reviewed journal of cutaneous metastases from a nasal neuroendocrine tumor in any domestic animal species. The elimination of other potential differential diagnoses was made based on a combination of clinicopathologic, histomorphologic, immunocytochemical, and ultrastructural features.
Footnotes
Acknowledgements
The authors acknowledge the valuable assistance of the pathology residents and staff pathologists at the Armed Forces Institute of Pathology for confirmation of our initial diagnosis and repeat immunohistochemical staining; Dr Brian Summers for insights on the case; Ms Karen Wolfe for electron microscopy assistance; and Ms Elizabeth Landreth and Ms. Kristina Cammack for histotechnology and immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
