Abstract
Melengestrol acetate (MGA) contraceptives are widely used in zoo felids to regulate fertility and may have deleterious effects on endometrial health. To determine whether MGA exposure was associated with endometrial disease, the genital tracts of 212 zoo felids (99 MGA treated and 113 control) representing 23 species were evaluated. Adenomatous and cystic hyperplasia were prevalent in both MGA-treated (85%) and control (61%) groups, and the risk of developing these lesions increased with age. Treatment with MGA further increased the risk of developing advanced hyperplasia regardless of dose, and treatment for >72 months significantly elevated that risk, whereas parous animals had a lower risk. Endometrial polyps, fibrosis, adenomyosis, and hydrometra occurred in both MGA-treated and control animals. MGA treatment was associated with an increased risk of hydrometra and mineralization but not of adenomyosis, polyps, or fibrosis after adjusting for advanced hyperplasia. Acute or chronic endometritis were associated with advanced hyperplasia but not with MGA treatment. These results indicate that proliferative and inflammatory endometrial lesions are common spontaneous diseases in zoo cats, and MGA contraceptives increase the risk of some diseases. The association of MGA with endometrial lesions that could impair fertility should be considered when using this contraceptive in genetically valuable felids.
Keywords
Melengesterol acetate (MGA) contraceptives have been used extensively in zoologic parks to implement breeding programs for endangered felids. MGA-impregnated silastic implants placed subcutaneously or intramuscularly deliver sufficient concentrations of MGA over a 2-year period to prevent pregnancy in the majority of treated cats.2,21 Although in one study zoo felids treated for 1 year with MGA were subsequently fertile, many cats are treated with MGA continuously for ≥4 years, and the effects of long-term MGA treatment on endometrial health in zoo cats have not been investigated.2,21 Prolonged exposure to megestrol acetate has been associated with endometrial hyperplasia and pyometra in domestic cats,13,17 and progestin contraceptives may have similar effects on zoo felids. To assess whether MGA administration was associated with endometrial disease in zoo felids, we evaluated female reproductive tracts from multiple species through an American Zoo and Aquarium Association collaborative study. Here, we describe the hyperplastic and inflammatory lesions in the endometria of 212 zoo felids and confirm that MGA exposure is a risk factor for development of these lesions.
Materials and Methods
Study population
Reproductive tracts were requested from female felids that died or were ovariohysterectomized in US zoologic parks from 1988 to 1995. Reproductive tracts from 212 felids of known age and contraceptive history were received from 78 zoologic parks. Ninety nine of these felids were on MGA contraceptives for at least 6 months, and 113 felids were never exposed to MGA or other contraceptives (subsequently referred to as MGA-treated and control animals, respectively). Fifty-seven of the 99 treated cats had MGA contraceptive implants in place at the time of uterine removal. Ovaries were submitted from 67 MGA-treated and 73 control felids, but ovaries from the other felids had been removed for in vitro fertilization and embryo cryopreservation studies and were not available for histologic examination. Parity information was available on 92 MGA-treated felids (36 parous and 56 nulliparous) and 102 controls (22 parous and 80 nulliparous). The species represented in the study population and the proportion of each species that was treated with MGA are presented in Table 1. The age distribution for the study population is depicted in Fig. 1.
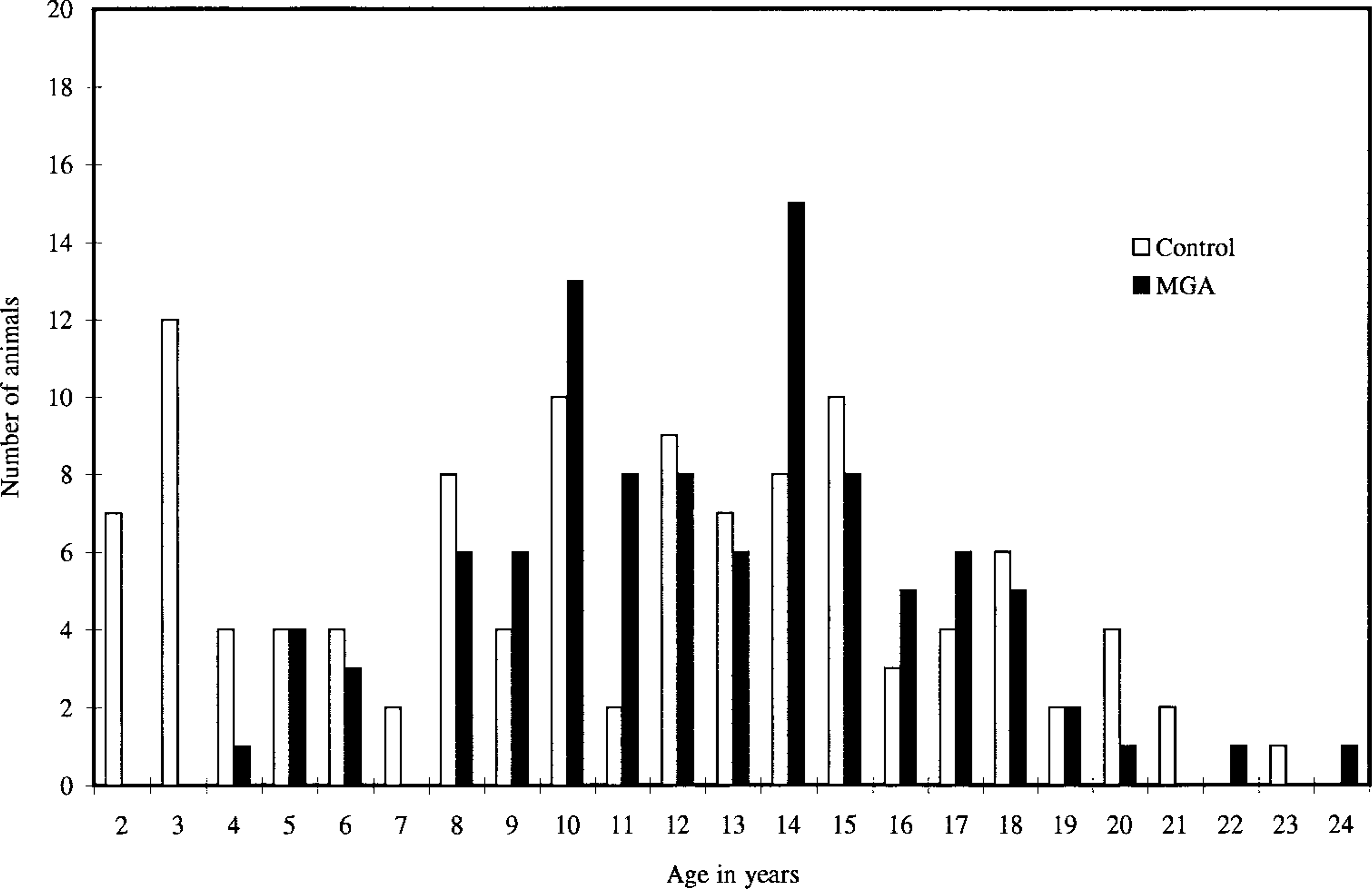
Age distribution of the zoo felid population evaluated for melengestrol acetate (MGA) contraceptive effects on endometrial disease.
The proportion of melengesterol acetate (MGA)-treated and control felids with endometrial hyperplasia (EH) by species.
For each MGA-treated animal, the dose, duration of treatment, and constancy of treatment were calculated to assess whether these variables influenced the severity of lesions. Because MGA was delivered by diffusion from silastic intramuscular implants, doses for these analyses were based on the total amount of MGA incorporated in an implant divided by the body weight of the animal. Study animals were categorized as having a short, medium, long, or very long duration of MGA exposure (6 months–2 years, 2–4 years, 4–6 years, or >6 years). These intervals were selected because MGA implants are recommended for 2-year periods. Constancy of treatment was categorized as either continuous or intermittent (>6 months between implants), and implants that were not removed were considered inactive after 3 years.
Histopathology
Reproductive tracts were fixed in 10% neutral-buffered formalin at the contributing zoologic park before shipment. On receipt of formalin-fixed tracts at the study site, all gross lesions were recorded, and then two transverse sections of each uterine horn, sagittal sections of each ovary, and representative sections from all gross lesions were obtained. Tissues were embedded in paraffin, sectioned at 7 μm, and stained with hematoxylin and eosin (HE). Uteri with endometrial mineralization were first suspended in 15% formic acid until decalcified and then embedded and sectioned as for other tissues.
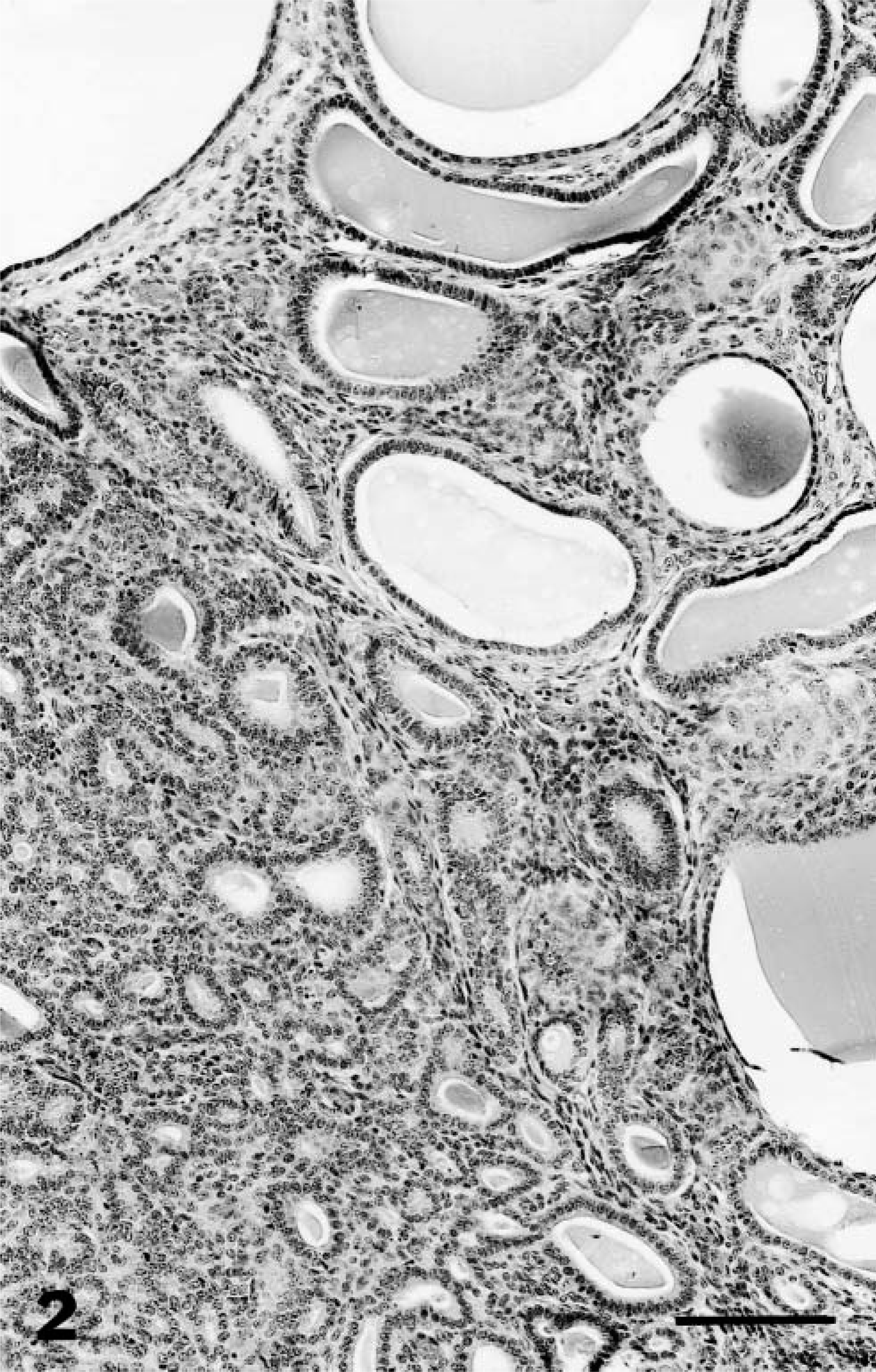
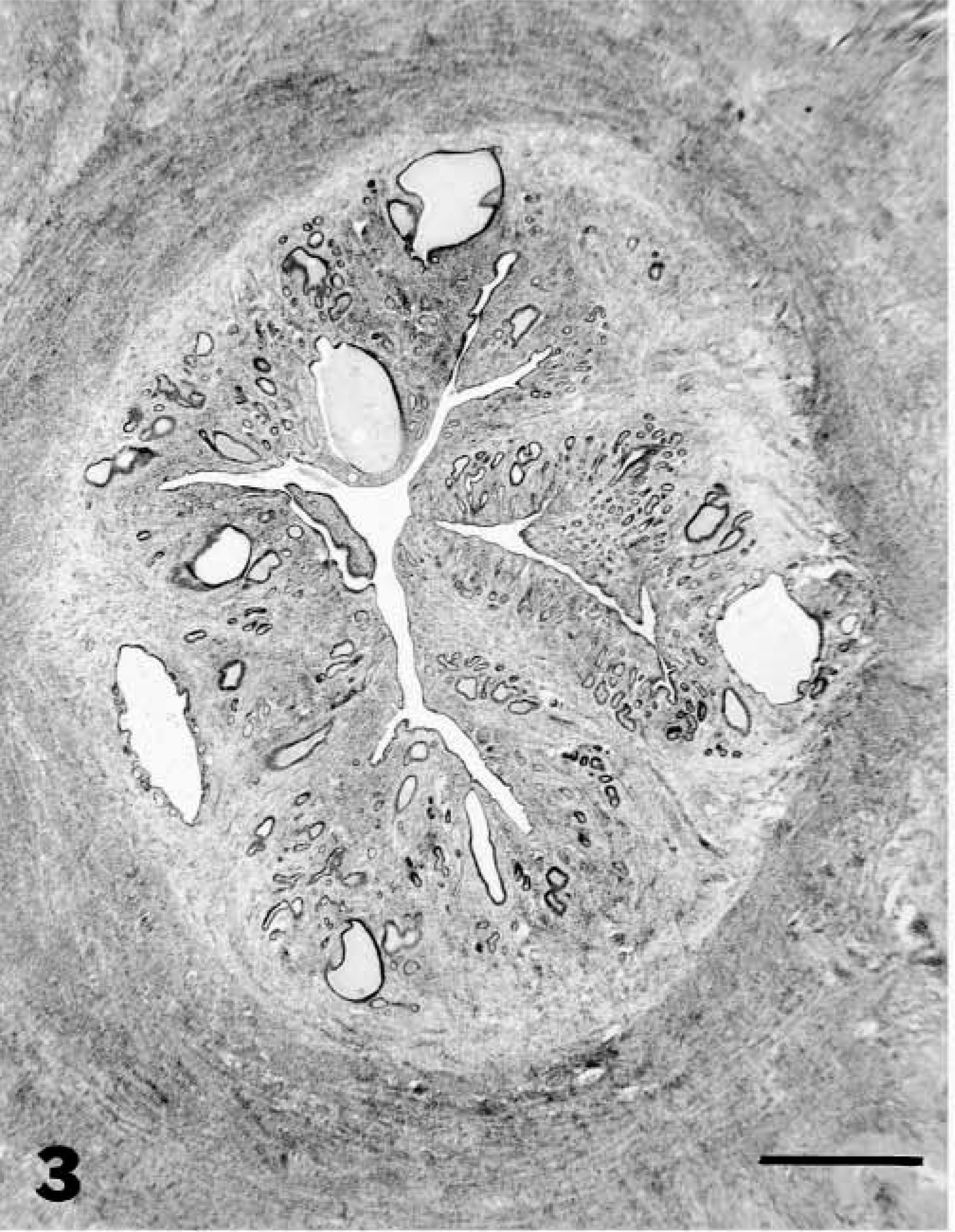
Uteri were examined histopathologically by one of the authors (L. Munson) without knowledge of species, age, or MGA exposure. The endometrium was evaluated for adenomatous and cystic hyperplasia of glandular epithelium (Fig. 2), papillary hyperplasia of luminal epithelium, polyps, adenomyosis, fibrosis, mineralization, and inflammation. Proliferative changes in the endometrium that were typical for stages of the estrous cycle were not considered hyperplasia.20 Cystic hyperplasia was distinguished from endometrial cysts by the crowding, pseudostratification, and hyperchromasia of glandular epithelium and the involvement of multiple glands. For the purpose of grading, cystic and adenomatous hyperplastic changes were combined to determine the overall cumulative increase in endometrial thickness because these changes were presumed to have a similar effect on fertility. The severity of endometrial hyperplasia (EH) was categorized as follows: grade 0 = no hyperplastic changes; grade 1 = minimal to mild proliferative and/or cystic changes in glands or surface epithelium without an increase in overall endometrial height (Fig. 3), grade 2 = moderate hyperplastic and/or cystic change with an increased endometrial thickness of ≤2 times normal (Fig. 4; grade 3 = severe hyperplastic and/or cystic changes with increased endometrial thickness of >2 times normal (Fig. 5). Normal endometrial thickness was determined from grade 0 uteri in species of similar body size and from approximately the same stage of estrus. Stage of estrous cycle was estimated by glandular length, tortuosity, and epithelial morphology.20 Uteri with variation in the severity of EH were graded based on the most severe lesion. Adenomyosis was defined as the presence of well-differentiated endometrial glands and stroma in the myometrium discontiguous with but morphologically similar to the endometrium. Endometrial polyps were defined as discrete, narrow-based, intraluminal projections of well-differentiated endometrial glands and stroma resembling the mural endometrium. Endometritis was defined by the presence of neutrophils, plasma cells, or lymphocytic aggregates. Small numbers of lymphocytes dispersed throughout the endometrium were considered normal. Fibrosis was distinguished from normal endometrial stroma by the circumferential arrangement of fibroblasts around glands, increased separation and nesting of glands, or any abnormal orientation or distribution of fibroblasts in the endometrium. Ovaries were examined for structures that could be a source of endogenous estrogen or progesterone, such as tertiary or cystic follicles, corpora lutea (CL), or granulosa cell tumors. Details of other ovarian lesions in this population have been previously reported.10
Endometrium; lion. Endometrial hyperplasia with cystic and adenomatous patterns. HE. Bar = 450 μm.
Endometrium; lion. Mild (grade 1) endometrial hyperplasia. HE. Bar = 300 μm.
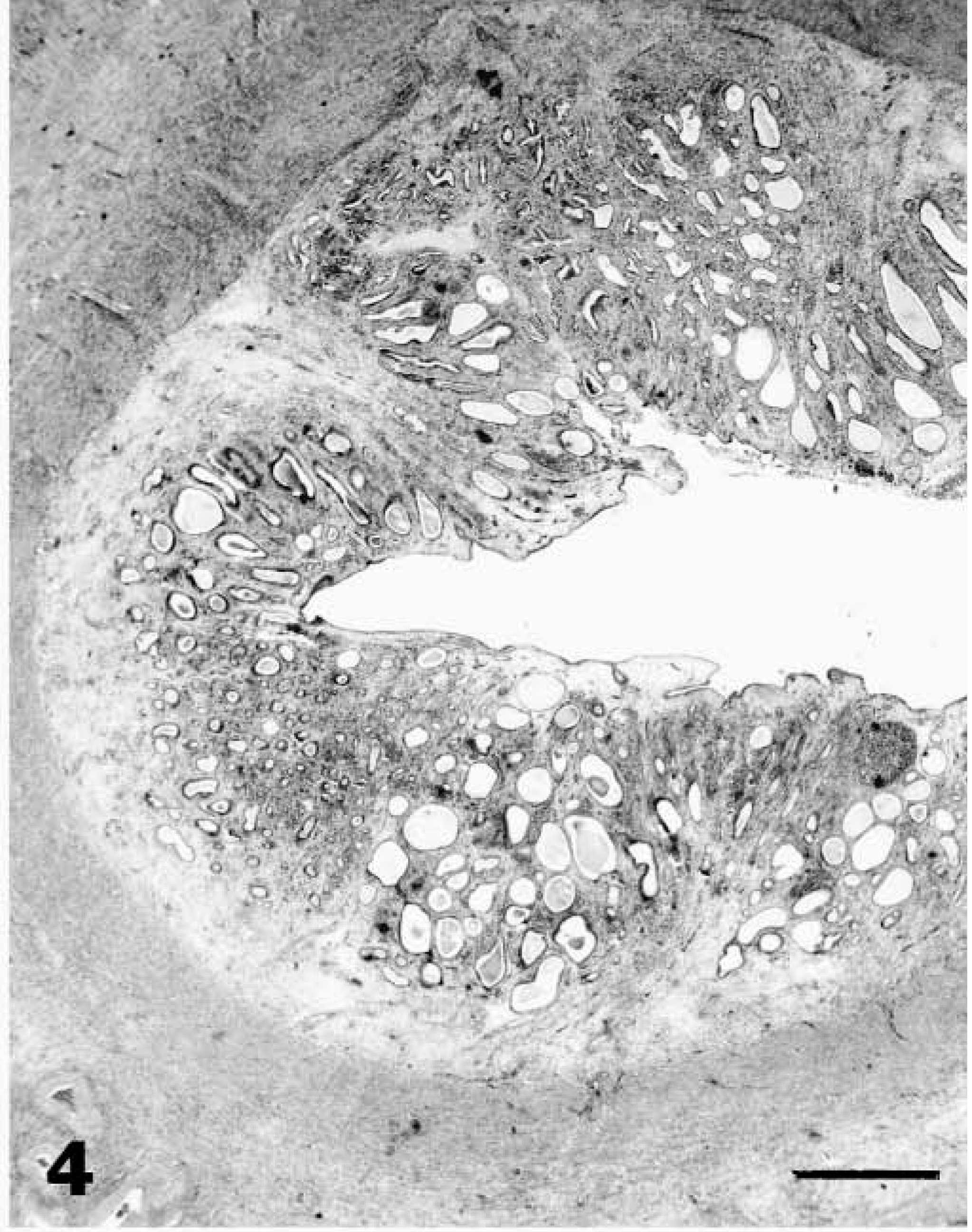
Endometrium; lion. Moderate (grade 2) endometrial hyperplasia. HE. Bar = 300 μm.
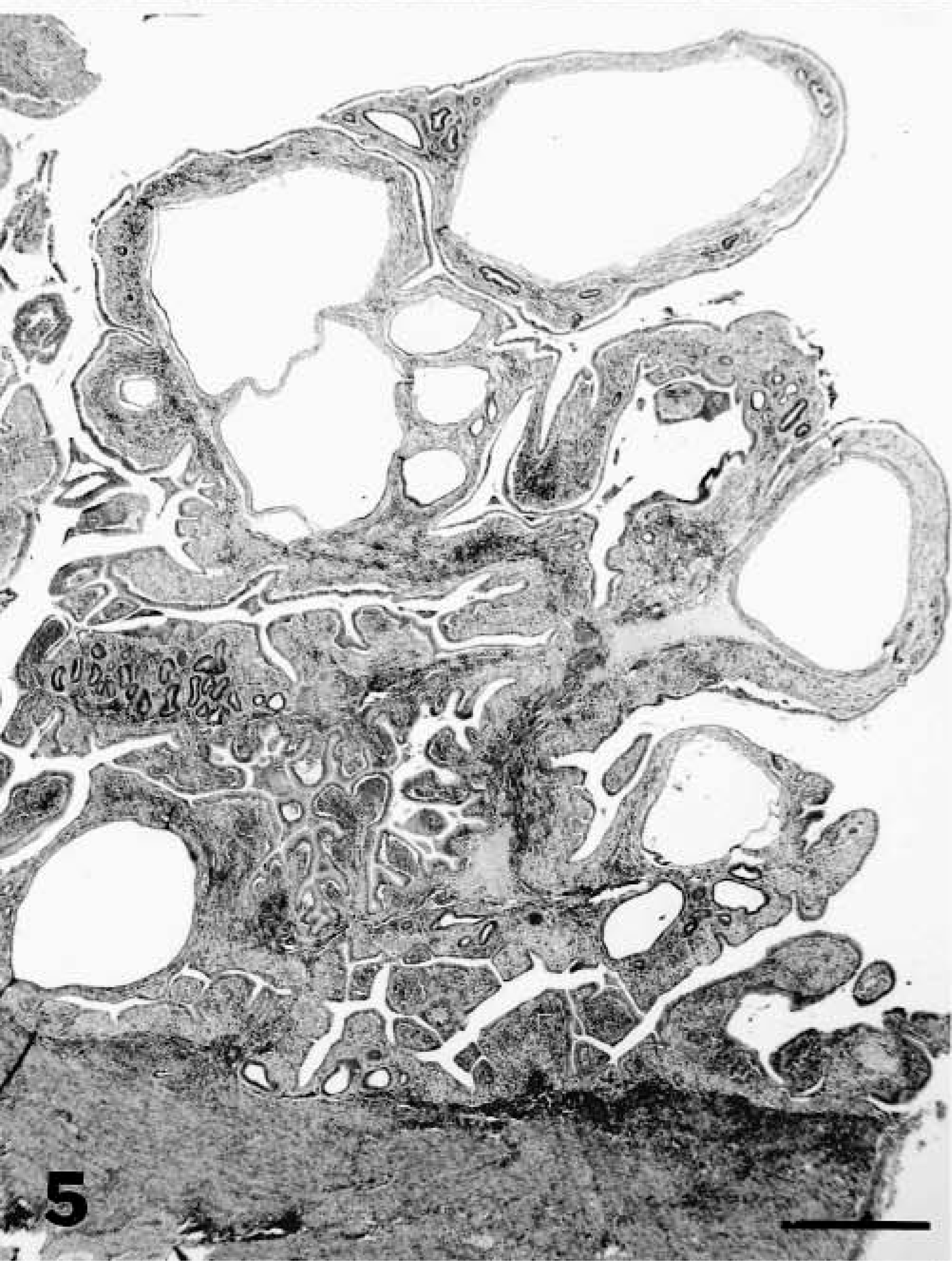
Endometrium; lion. Severe (grade 3) endometrial hyperplasia. HE. Bar = 300 μm.
Statistical analysis
Because both moderate and severe grades of EH were considered biologically important, we dichotomized this lesion as advanced (grades 2 and 3) or not advanced (grades 0 and 1) for all analyses. The association between MGA treatment and advanced EH (yes/no) was analyzed by logistic regression with age (months) and parity (nulliparous or parous) as covariates.8 For the subset of records with ovary data (n = 140), we also evaluated the effects of concurrent tertiary follicles or CL on advanced EH. Species was not included as a covariate because lesion character did not differ among the 19 species with lesions and adequate numbers of all species were not available for statistical tests. However, we grouped species into big cats (tigers, lions, jaguars, snow leopards, and leopards) and others (small cats, cheetahs, cougars, and clouded leopards) based on currently understood phylogenetic relationships16 and allowed this variable to enter models if significant. The association between MGA exposure and various proliferative, inflammatory, or degenerative lesions was analyzed by logistic regression with age, parity, cat grouping, and advanced EH as covariates. Logistic regression also was used to assess effects of MGA on the type of EH (adenomatous versus cystic), the effects of MGA dose and duration, including whether treatment was continuous or interrupted (>48 months between treatments), and the effects of current or previous MGA exposure on advanced EH. Logistic regression modeling followed recommended procedures. For final logistic models, adjusted odds ratios (OR) and 95% confidence intervals (CI) were calculated to measure the strength of association between exposure variables and outcomes. Overall fit of models was assessed using Hosmer–Lemeshow goodness-of-fit (HL) statistics.8 Pairwise associations between lesions were analyzed by McNemar's test for paired data. For all analyses, the significance level was set at P < 0.05.
Results
Proliferative endometrial lesions
The most prevalent endometrial lesion in the zoo cat population was EH, characterized by glandular proliferation in adenomatous clusters, dilated cystic glands, or hyperplasia of the surface endometrium resulting in irregular folds or polypoid projections into the lumen (Figs. 2–5, Table 2). EH (grades 1, 2, or 3) occurred in 153 (72.1%) of the 212 felids, and advanced EH (grades 2 or 3) occurred in 90 (58.8%) of these 153 animals. Most (70%) endometria with adenomatous changes had concurrent cystic glands, and both types of hyperplasia occurred in control and MGA-treated animals. Mild lesions were characterized by small irregular clusters of glands lined by a densely packed cuboidal to columnar epithelial cells that differed morphologically from those in unaffected glands and were asynchronous with the stage of the estous cycle (Fig. 3). Cystic glands were variable in size and were lined by densely packed epithelium that was usually compressed by retained secretory material (Fig. 2). Lesions of increasing severity had more abundant overall glandular and surface proliferation with more prominent stratification of the epithelial lining and increasing fibroplasia in the endometrial stroma (Fig. 5). Papillary hyperplasia of the surface epithelium occurred in 39% of the felids with severe hyperplasia, and notable cellular atypia (marked cytoplasmic pallor and vacuolation and multinucleation) was present in some areas (Fig. 6). Lesions of severe hyperplasia were often accompanied by extensive stromal mineralization (Figs. 6, 7). Statistical analysis indicated that advanced EH was positively associated (Table 3) with both adenomyosis and mineralization.
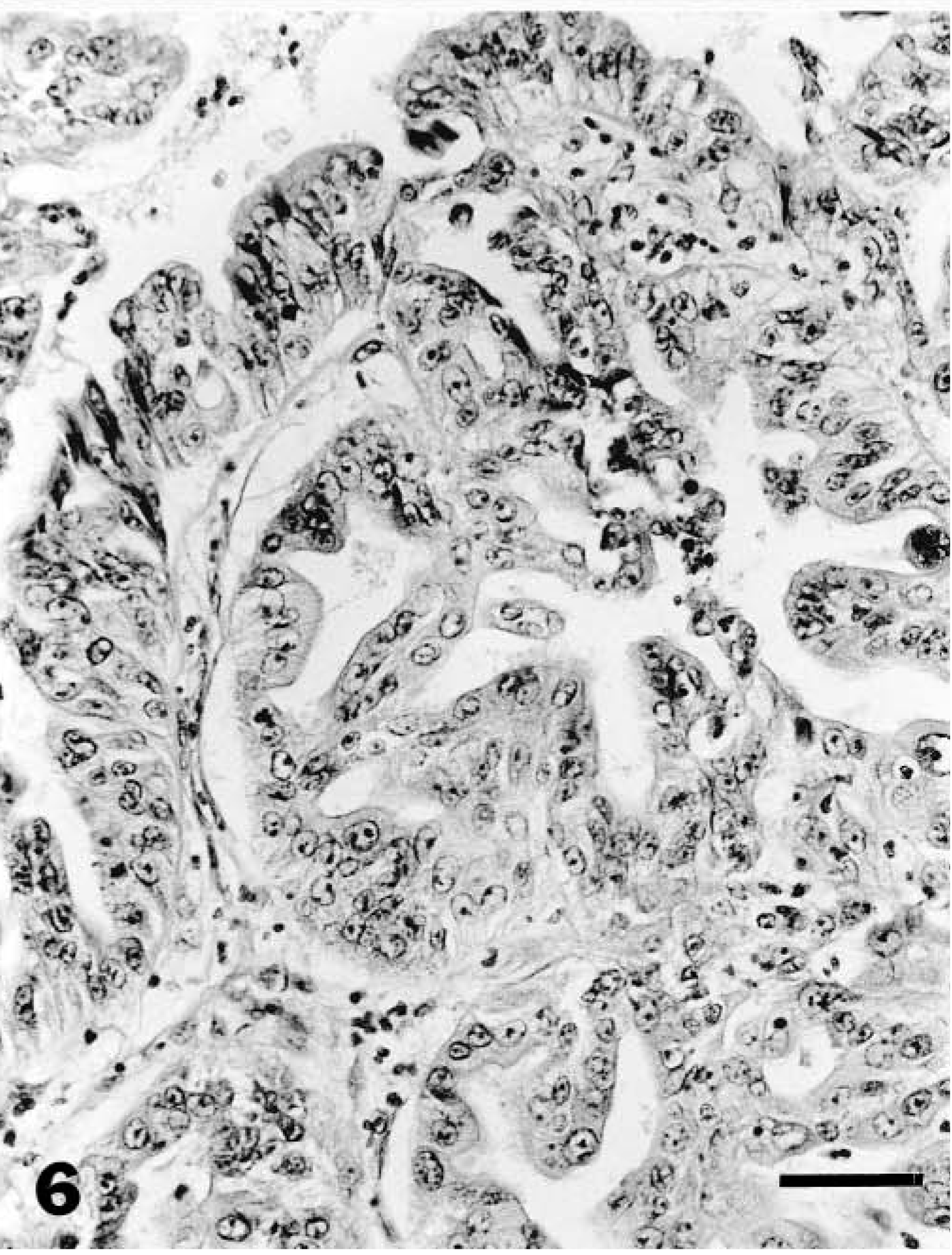
Endometrium; lion. Cellular atypia in the surface endometrium with severe hyperplasia. Note multinucleation, anisokaryosis, and disorientation of cells. HE. Bar = 30 μm.
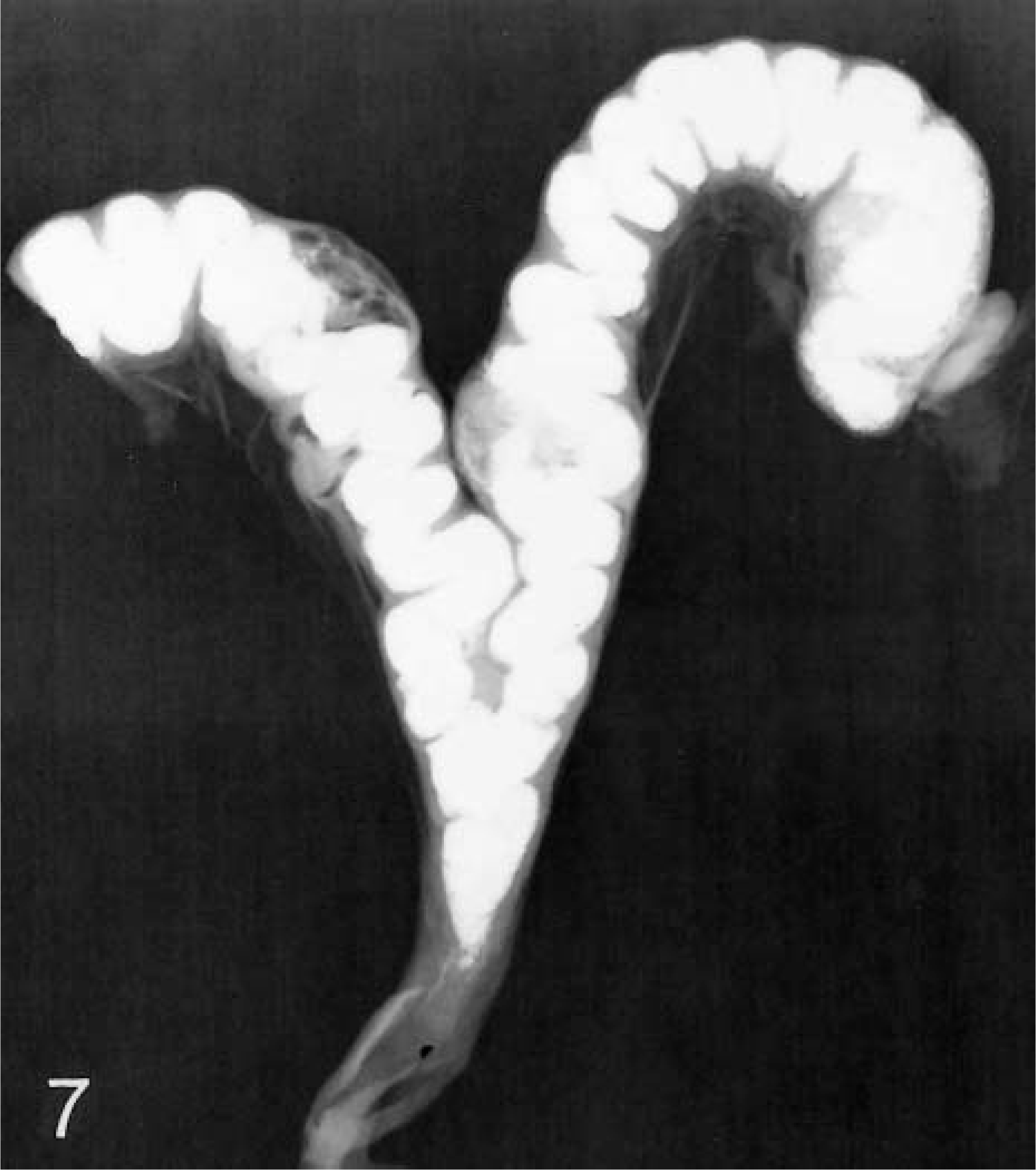
Uterus; tiger. Radiograph reveals severe endometrial mineralization.
Occurrence (%) of endometrial lesions in melengestrol acetate (MGA)-treated (n = 99) and control (n = 113) zoo felids.
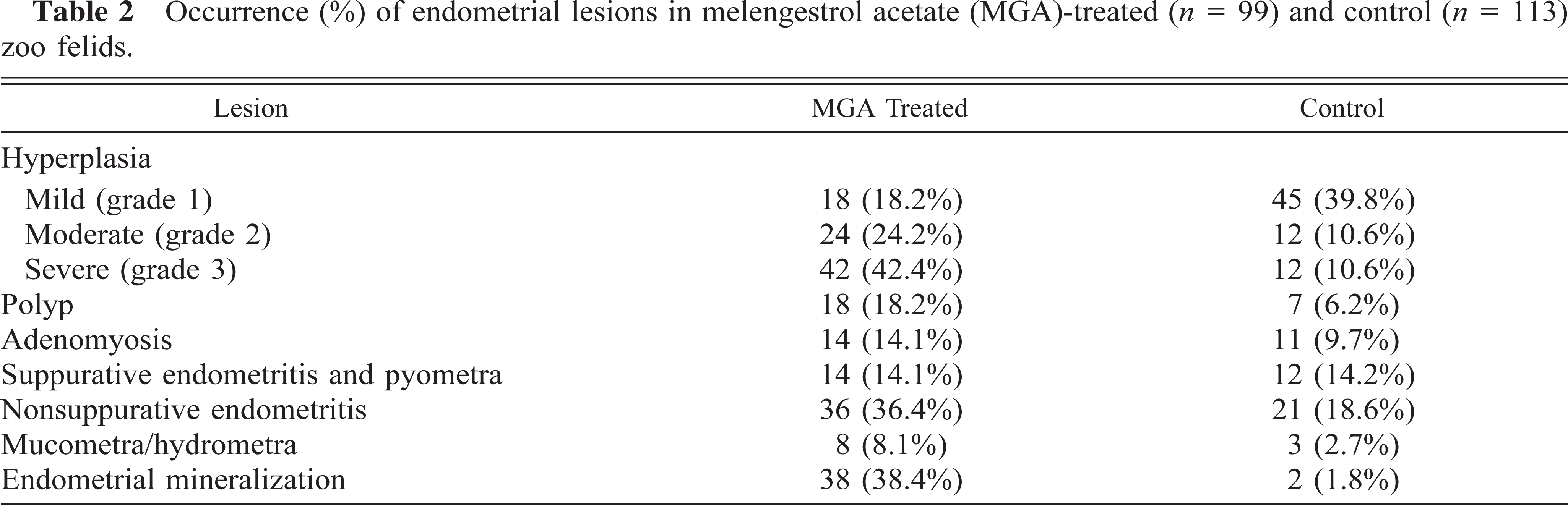
EH was present in all species except for the sand cat (Felis margarita), black footed cat (Felis nigripes), wild cat (Felis silvestris), and margay (Felis wiedii). However these four species of small cats were only represented by one or two animals, and those animals were not contracepted and were all <4 years old except for one margay (Table 1).
EH occurred in 85% of MGA-treated felids and 61% of controls (Table 2); 67% of MGA-treated and 21% of control felids had advanced hyperplasia. Before adjustment for confounding factors such as age and parity, MGA-treated felids had a more than threefold higher odds (OR = 3.6, 95% CI = 1.8–7.4) of EH and about a sevenfold higher odds (OR = 7.2, 95% CI = 3.8–14.4) of advanced EH than did control felids. The youngest cat with severe hyperplasia in MGA-treated group was 64 months of age in contrast to 96 months in the control group. The median grade of hyperplasia (grade 2) did not differ between those felids exposed to MGA at the time of sample collection and those previously but not currently exposed. All 21 felids with papillary hyperplasia of the surface endometrium were MGA treated.
Three variables were significantly associated with the risk of advanced EH: MGA treatment and age were associated with increased risk, and parity was associated with a decreased risk. The ORs were 8.2 (95% CI = 3.9–17.3) for MGA treatment, 1.04 (95% CI = 1.01–1.08) for each 1-month increase in age, and 0.31 (95% CI = 0.14–0.71) for parous females. There were no significant interactions between these variables, and final model fit was good (HL, P = 0.64). Felid species group (big or other) was not a risk factor for advanced EH (OR = 0.82, 95% CI = 0.38–1.74) after accounting for MGA treatment status, age, and parity. Adenomyosis was highly associated (OR = 5.1, Table 3) with advanced hyperplasia, although EH was not a requisite for this condition because four felids had adenomyosis without EH.
Odds ratios (OR) and 95% confidence intervals (CI) obtained from logistic regression models for significant associations between melengestrol acetate (MGA) treatment, advanced endometrial hyperplasia (EH, grades 2 or 3), and other endometrial lesions.
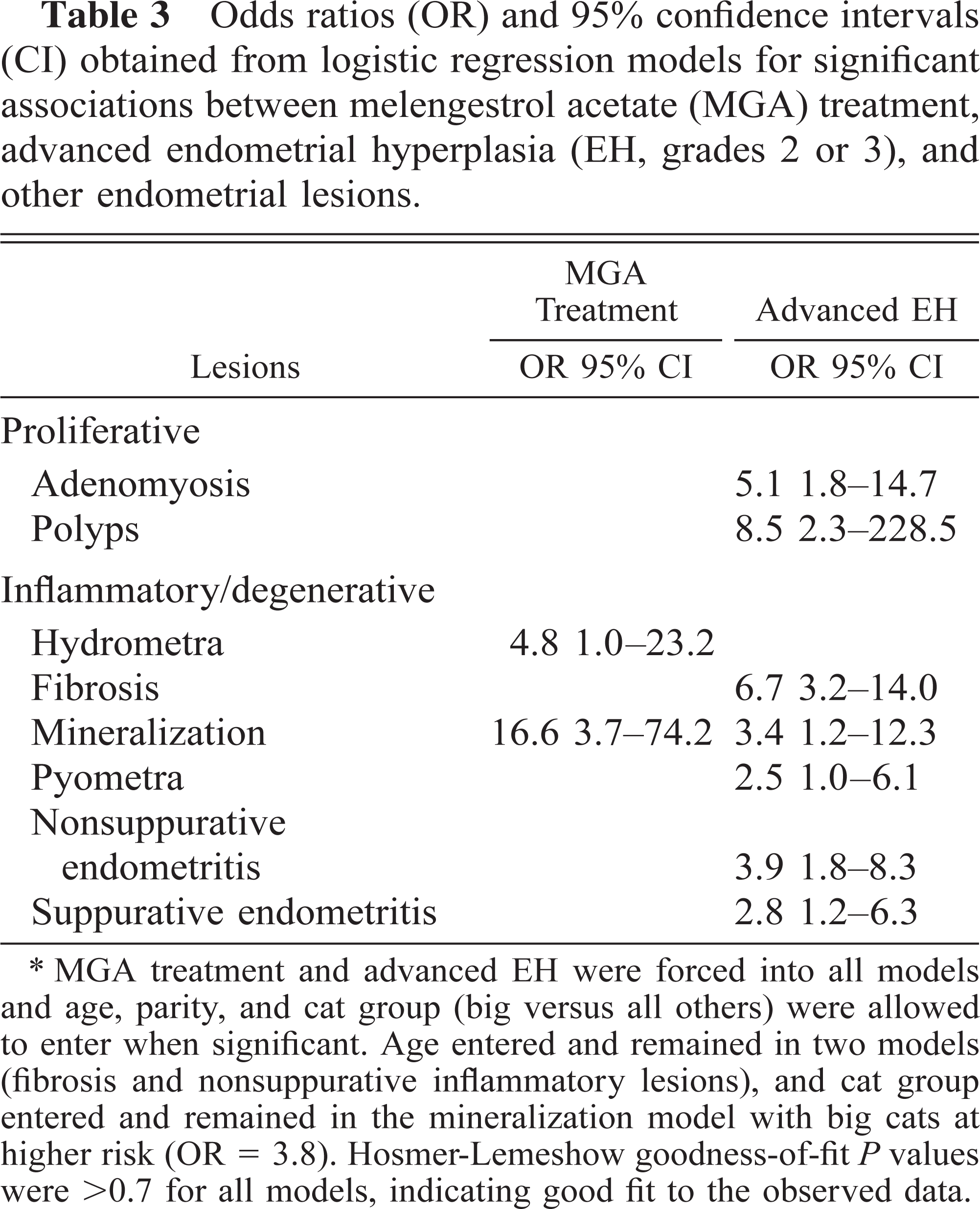
MGA treatment and advanced EH were forced into all models and age, parity, and cat group (big versus all others) were allowed to enter when significant. Age entered and remained in two models (fibrosis and nonsuppurative inflammatory lesions), and cat group entered and remained in the mineralization model with big cats at higher risk (OR = 3.8). Hosmer-Lemeshow goodness-of-fit P values were >0.7 for all models, indicating good fit to the observed data.
Because endogenous steroids could exacerbate endometrial lesions, associations between the occurrence of tertiary follicles, CL and advanced EH were evaluated in the subset of animals (n = 140) with ovarian data. Neither tertiary follicles nor CL were significant risk factors for advanced EH after MGA treatment, age, and parity were included in logistic models. Of these 140 felids, 125 had tertiary or preovulatory follicles, but the apparently increased risk (OR = 3.8) was not significant. CL occurred in both groups, but neither the severity of EH nor the prevalence of cystic or papillary hyperplasia were greater in felids with CL (OR = 1.3). One 11-year-old cheetah in the study had a granulosa cell tumor but had only mild EH. One 17-year-old leopard had cystic follicles and severe EH but also had been continuously exposed to MGA for 12 years.
The effect of MGA dose, duration of treatment, and whether treatment was continuous or intermittent on the development of advanced hyperplasia was assessed in 95 felids with complete data for these variables. Treatment duration was associated with advanced EH, but once duration was in the model, dose (either as categories or as a continuous variable), treatment constancy, parity, age, and cat group were not significant variables. Compared with the reference category of >6–24 months, OR estimates for treatment duration were 1.2, 2.7, and 13.8 for >24–48, >48–72, and >72 months, respectively. However, only animals in the >72-month category had a significantly elevated risk (95% CI = 2.1–89.5) of developing advanced EH after accounting for age effects compared with the shortest treatment duration. The duration model fit the data well (HL, P = 1.0).
Endometrial polyps (Table 2) occurred in felids 60–231 months of age. Most polyps consisted of a core of fibrous stroma separating widely spaced glands and covered by endometrial epithelium, although some polyps had a greater epithelial component consisting of compressed papillary folds similar in character to the surface epithelium. Many polyps caused marked compression atrophy of the adjacent endometrium, and five of the 11 felids with hydrometra or mucometra had endometrial polyps. Polyps were highly associated with advanced EH (OR = 8.5; Table 3), whereas hydrometra/mucometra were associated with MGA exposure (OR = 4.8; Table 3). Forty felids had endometrial glandular or stromal mineralization, and 38 of those 40 felids had been exposed to MGA compared with 2 of 113 controls (Table 2). Logistic analysis indicated a >16-fold increase in risk associated with MGA exposure (Table 3).
The distribution of mineral was regional or diffuse and was remarkably severe in some felids (Fig. 7, 8); Most affected animals (36/40) were big cats. Thirty-eight felids with endometrial mineralization had some degree of hyperplasia, and this association was stronger for felids with moderate to severe hyperplasia (OR = 3.4; Table 3); 48% of all felids with grade 3 hyperplasia had some degree of endometrial mineralization. Endometrial stromal fibrosis occurred in 101 felids, and in 85 felids this fibrosis was strongly associated with advanced EH (OR = 6.7; Table 3).
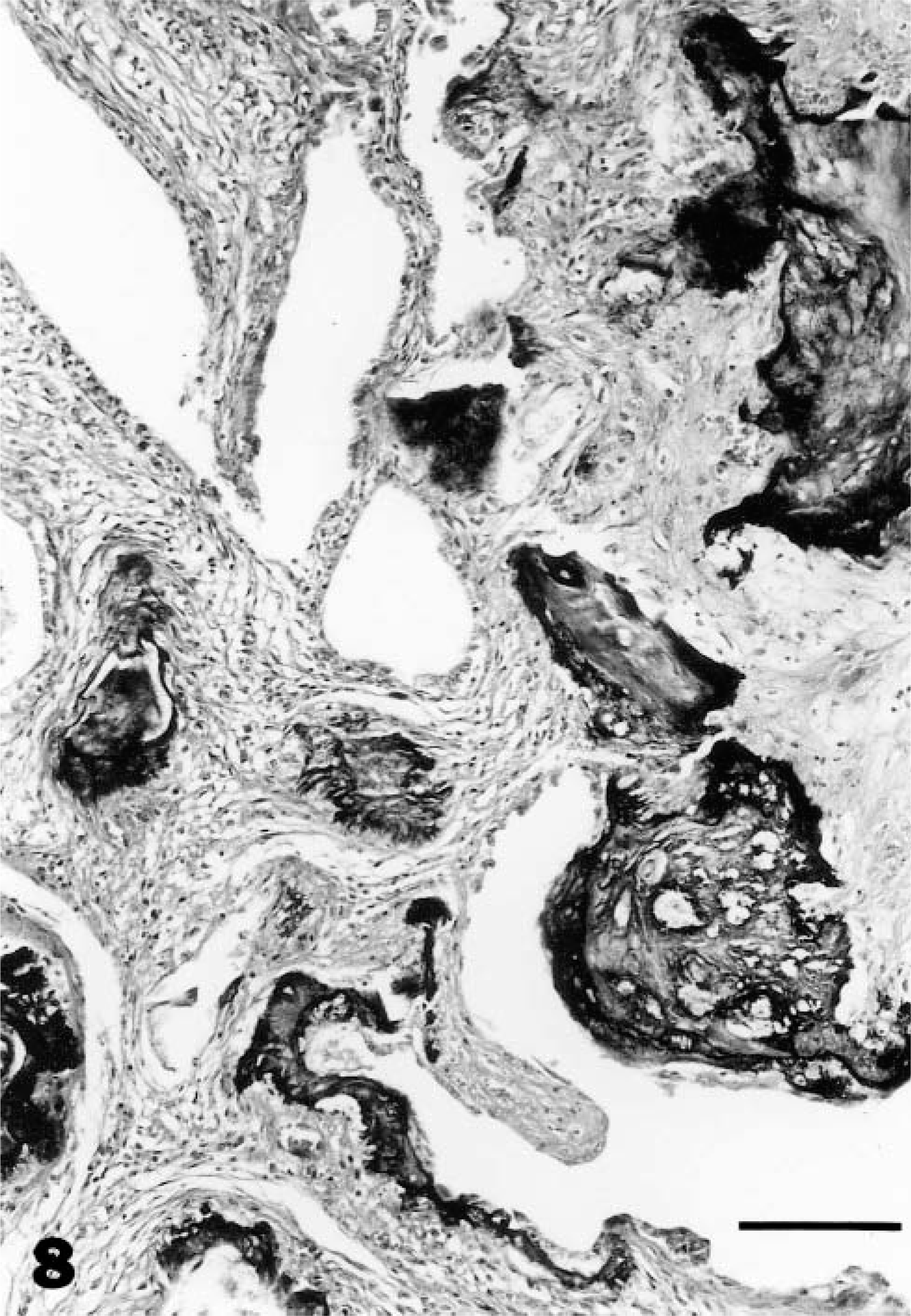
Endometrium; tiger in Fig. 7. Histopathologic appearance of endometrial stromal and epithelial mineralization. HE. Bar = 80 μm.
Inflammatory and degenerative endometrial lesions
Pyometra occurred in 26 felids, and 14 of those 26 were MGA treated. Six of 15 felids with pyometra from which ovaries were available had CL (three MGA treated and three controls). Occurrence of a corpus luteum was associated with nearly a threefold higher odds (OR = 2.8, 95% CI = 0.9–8.6) of pyometra, but sparse data precluded adjustment for other variables such as advanced hyperplasia. Sixteen of the 26 felids with pyometra had moderate to severe EH (OR = 2.5; Table 3), although pyometra also occurred in the absence of any hyperplastic changes. Mild neutrophilic infiltrates were noted in the endometrium of 17 other felids (13 MGA treated, four controls).
Of the 57 felids with nonsuppurative endometritis, 24 animals had prominent lymphocytic aggregates and 18 of these 24 were MGA treated. Nonsuppurative endometritis and advanced EH were significantly associated (OR = 3.9; Table 3), and neutrophilic endometritis and advanced EH were less so (OR = 2.8; Table 3). There was no effect of parity or MGA treatment on inflammation.
Discussion
This study clearly demonstrated an association between MGA contraceptives and the development of advanced EH and related lesions in zoo felids. The population of nulliparous and aged animals that are now found commonly in zoos as a result of improved husbandry, veterinary care, and breeding programs designed to prevent reproduction of surplus animals are at risk for developing these lesions spontaneously; however, treatment with MGA contraceptives increased this risk, leading to earlier development and more advanced lesions. The risk was significantly elevated when treatments extended beyond 72 months; thus, prolonged contraception with MGA implants is contraindicated in genetically valuable animals. Findings from the study were similar when the analyses were done with EH (grades 1, 2 and 3) versus no EH, indicating that our findings are robust.
The acceleration and exacerbation of spontaneous age-associated endometrial changes in zoo felids by MGA are not unexpected considering the comparable effects of progestins on other carnivores.1,15,23 Melengestrol is a potent progestin,11 and the method of administration in zoo cats provides constant exposure for prolonged periods of time. Similar EH has been noted in domestic cats5,12,18 (Munson, unpublished data), both spontaneously and as a sequela to progestin treatment. The cystic glandular changes in the endometrium of zoo cats, although less pronounced than those in cystic EH in domestic dogs,15,22 are a characteristic outcome of prolonged exposure to progestins, which induce secretory differentiation of endometrial epithelial cells. Because both cystic and adenomatous changes were present in 70% of the felids and the morphologic characteristics of the hyperplastic cells (hyperchromasia, pseudostratification, high nuclear/cytoplasmic ratio) were similar in both types of hyperplasia, these categories were considered different manifestations of the same disease process in which cystic changes occur when endometrial gland necks become occluded by the hyperplastic epithelium. Although induction of cystic EH in dogs by megestrol acetate was dose related,15 comparable dose-related changes were not noted in our study population. This lack of dose effect may indicate that even the lowest MGA doses used in zoo felids cause maximal deleterious changes.
Progestins can promote endometrial growth by activating estrogen receptors,9 which in turn upregulate epidermal growth factor receptors.14 MGA does not suppress folliculogenesis in the majority of animals;10 thus, endogenous estrogens could also have contributed to EH in zoo cats.10,14,19 Direct mitogenic effects of progestins have not been demonstrated.
The endometrial lesions in many MGA-treated cats appeared irreversible, and their persistence in felids previously but not currently exposed to MGA supports this assessment. The marked asynchrony between hyperplastic and normal endometrium in MGA-treated individuals suggests that the hyperplastic epithelium was unresponsive to hormonal signals that regulate normal growth and functional maturation.4 The marked stromal mineralization associated with MGA-induced EH was also probably permanent. These endometrial changes are important factors to consider if MGA contraceptives are to be used to regulate reproduction in endangered species.
Pyometra was a relatively rare finding and was not MGA associated, in contrast to progestin effects in dogs.3,7,15 The hyperplastic changes in zoo felids lacked the marked cystic dilation that is characteristic of progestin-induced hyperplasia in domestic dogs, which fosters bacterial growth and the development of pyometra. In zoo felids, endogenous estrogens from ongoing folliculogenesis (that was not suppressed by MGA treatment) may sustain adequate uterine tone and minimized endometrial secretions, thereby limiting the development of pyometra.
Endometrial polyps in the zoo population were similar to the polyps in domestic cats, which also are associated with EH, fibrosis, and advancing age. 6 However, hyperplasia was not a necessary precondition for developing polyps in our study population. Polyps also were more common in felids with nonsuppurative inflammation, suggesting that they may arise in response to chronic inflammation.
This large multiinstitutional, multispecies study disclosed a high prevalence of advanced EH in zoo felids and demonstrated that animals receiving MGA contraceptives have an increased risk of developing hyperplasia, mineralization, and hydrometra/mucometra. Although other lesions such as polyps and endometritis were not directly linked to MGA exposure, their association with advanced hyperplasia make their occurrence in MGA-treated animals likely. Together these lesions would likely lead to endometrial dysfunction and possibly sterility, outcomes that should be considered before using MGA in valuable zoo felids. Because endometrial lesions were more prevalent and severe in older and nulliparous animals, alternating MGA use with planned breedings may minimize the development of endometrial disease. Furthermore, we recommend that all animals intended for breeding programs should be bred early in their reproductive years
Footnotes
Acknowledgements
We thank Dr. L. Harrenstien and M. Mason for assistance with the project and J. Schneider for initial statistical analyses. This project was supported by the New York Zoological Society Nixon Griffis Fund for Zoological Research and the American Zoo and Aquarium Association Conservation Endowment Fund/Ralston Purina Big Cat Survival Funds. The following zoological societies and veterinarians contributed reproductive tracts to this study: Acadia Zoo (Dr. J. Rausch), Akron Zoo (Dr. G. Riggs), Zoo Atlanta (Dr. R. McManamon), Audubon Park Zoo (Dr. R. Aguilar), John Ball Zoo (Dr. R. Bennett), Baltimore Zoo (Dr. M. Cranfield), Binder Park Zoo (Dr. D. Rost), Birmingham Zoo (Dr. M. Shaw), Blank Park Zoo (Dr. D. Riordan), Brandywine Zoo (Dr. L. Klein), Brookfield Zoo (Dr. L. Phillips), Burnet Park Zoo (Dr. C. Wallace), Caldwell Zoo (Drs. D. Starnes, K. Reese), Calgary Zoo (Dr. R. Cooper), Central Florida Zoo (Dr. G. Kollias), Chaffee Zoological Gardens of Fresno (Drs. R. Gentzler and S. Lynch), Cheyenne Mountain Zoo (Drs. M. Burton and P. Calle), Cincinnati Zoo (Dr. M. Campbell), Columbus Zoo (Drs. L. Kramer and R. Wack), Dallas Zoo (Dr. B. Raphael), Denver Zoo (Dr. D. Kenny), Detroit Zoo (Dr. D. Agnew), Exotic Feline Breeding Center, Fort Wayne Children's Zoo (Dr. K. Casserly), Fossil Rim Wildlife Center (Dr. E. Blumer), Franklin Park Zoo (Dr. J. Curtin), Henry Doorly Zoo (Dr. D. Armstrong), Hogle Zoo (Dr. R. Anderson), Honolulu Zoo (Dr. B. Okimoto), Houston Zoo (Dr. J. Flanagan), Jackson Zoo (Drs. T. Lester and W. Maslin), Jacksonville Zoo (Dr. D. Page), Kansas City Zoo (Dr. W. K. Suedmeyer), King's Island (Dr. H. Reed), Knoxville Zoo (Drs. P. Morris and E. Ramsey), Lake Superior Zoo (Dr. S. Abelt), Lincoln Park Zoo (Drs. T. Mehan and R. Barbiers), Little Rock Zoo (Dr. M. N. Baeyens), Los Angeles Zoo (Dr. B. Gonzales), Louisville Zoo (Dr. R. Burns), Lowry Park Zoo (Dr. D. Murphy), Memphis Zoo (Dr. E. M. Douglass), Metro Washington Park Zoo (Dr. M. Finnegan), Miami Metrozoo (Dr. C. Miller), Micke Grove Zoo (Drs. N. Lamberski and L. Phillips), Miller Park Zoo (Dr. S. Murphy), Minnesota Zoo (Drs. P. Wolff and K. Petrini), Montgomery Zoo (Mr. B. Fiore), National Zoo and Conservation and Research Center (Dr. R. Montali), National Institutes of Health (Dr. L. Johnston), New York Zoological Society/Wildlife Conservation Society (Dr. J. Walberg), Oklahoma City Zoo (Dr. M. Barrie), Dr. J. Peddie, Philadelphia Zoo (Dr. G. Pierce), Phoenix Zoo (Dr. K. Orr), Pittsburgh Zoo (Dr. B. Wagner), Potawatomi Zoo (Dr. A. Duncan), Racine Zoo (Dr. F. Culbert), Reid Park Zoo (Dr. T. Miller), Rio Grande Zoo (Dr. B. Snyder), Riverbanks Zoo (Drs. T. Norton and N. Lamberski), Ross Park Zoo (Dr. D. Nielsen), Sacramento Zoo (Dr. L. Phillips), San Antonio Zoo (Dr. M. Richardson), San Diego Zoo and Wild Animal Park (Drs. M. Anderson and L. Lowenstine), San Francisco Zoo (Drs. A. Bennett and F. Dunker), Santa Ana Zoo (Dr. L. Boldrick), Santa Barbara Zoo (Dr. A. Smith), St. Louis Zoo (Drs. E. Miller and R. Junge), Seneca Park Zoo (Dr. D. Garell), Sunset Zoo (Dr. J. Veatch), Toledo Zoo (Dr. W. Shellebarger), Tulsa Zoo (Dr. W. C. Russell), Utica Zoo (Dr. C. Pertz), White Oak Conservation Center (Dr. S. Citino), Wildlife Safari (Dr. J. Mortenson), Wildlife Waystation (Dr. B. Gonzales), and Woodland Park Zoo (Drs. J. Ott-Joslin and D. Collins).
