Abstract
As husbandry practices have improved, safe and effective contraception for captive wildlife management has become a necessity. Melengestrol acetate (MGA), a synthetic progestin, is highly effective and has been used in many zoo species. Long-term use of MGA has been associated with uterine lesions in zoo felids, but effects in zoo canids have not been evaluated. This retrospective study documented spontaneously occurring lesions and investigated the impact of MGA on the reproductive health of zoo canids. Reproductive tracts from adult females were submitted by US zoos to the Association of Zoos & Aquariums' Wildlife Contraception Center Health Surveillance Program. Reproductive tracts were sampled and processed for histopathologic examination following standard protocols. Microscopic evaluations were performed without prior knowledge of MGA treatment status. Prevalence of uterine lesions was evaluated and compared between MGA-treated animals (n = 20) and control (untreated) animals (n = 61). Common lesions within the study population as a whole included endometrial hyperplasia (predominantly cystic) (53%), hydrometra (33%), and adenomyosis (25%). Treatment with MGA was a risk factor for endometrial hyperplasia, hydrometra, fibrosis, and adenomyosis. Uterine mineralization occurred exclusively in MGA-treated animals. Results indicate that MGA contraception can lead to lesions that may permanently impair the fertility of females. Therefore, if long-term contraception of zoo canids is necessary, the use of alternate methods of reproductive control such as gonadotropin-releasing hormone (GnRH) analogs or GnRH vaccines that reduce gonadal hormone exposure should be pursued.
Keywords
The zoo canid family is comprised of 36 species, at least 9 of which are considered threatened.44 Captive breeding programs manage the reproduction of threatened species intensively to maximize genetic diversity within a relatively small genetic pool. This requires that selected animals are allowed to reproduce, whereas reproduction in other individuals is curtailed. In addition, the reproduction of nonendangered canids also needs to be controlled due to the limited space in zoos. Contraception is a humane method to limit the offspring of specific animals and to provide reproductive rest for breeding animals without disrupting social groups.
Progestins have been the most commonly used form of contraception in zoos and comprise 81% of the reversible contraceptives used between 1975 and 2006 in zoo canids (Association of Zoos and Aquariums-Wildlife Contraception Center [AZA-WCC] Database). Synthetic progestins have been favored for their efficacy, ease of administration, cost effectiveness, and apparent reversibility. Because comprehensive experimental testing of new drugs before extensive use is not feasible in zoo species, it is essential to systematically monitor and assess the potential side effects of the contraceptive drugs used, as is done during human postmarketing pharmacovigilance. Progestins used as contraceptives in zoo canids include melengestrol acetate (MGA), megestrol acetate (MA), and medroxyprogesterone acetate (MPA). Eighty-five percent of the prescribed progestin contraceptives in zoo canids reported between 1975 and 2006 was MGA (AZA-WCC Database). MGA is administered subcutaneously or intramuscularly in a silastic implant, and during the course of at least 2 years, it delivers sufficient MGA to effectively prevent pregnancy in most treated animals.3
Exposure to endogenous progesterone or synthetic progestins (MGA, MA, and MPA) in domestic dogs has been associated with endometrial hyperplasia (EH), mucometra/hydrometra, pyometra, and diabetes.2, 7, 9, 10, 21, 24, 43, 52 In zoo felids, MGA is a risk factor for the development of EH, mineralization,34 and cancer (M. Smith and L. Munson, unpublished). Although a direct association between these lesions and fertility has not been established, an analysis of reproductive success in MGA-treated tigers (Panthera tigris) disclosed that only 50% of MGA-treated tigers reproduced after the MGA implant was removed,11 compared with 79% of control animals.11 EH may be a factor contributing to this reduced fertility because pathology surveillance revealed that 17 of 30 (59%) MGA-treated tigers had moderate-to-severe EH, whereas moderate-to-severe EH occurred in only 2 of 20 (10%) of controls (L. Munson, unpublished).26 Cystic endometrial hyperplasia (CEH) is considered an important cause of infertility in the domestic dog38 and cat.12, 33 In addition to the impact on fertility, EH also predisposes animals to pyometra, which has a mortality rate of 4% in dogs and 8% in cats.20, 30, 45 Synthetic progestins have the potential for more profound effects than endogenous progesterone, as their clearance rates are lower,15, 41 they have a higher affinity for the progesterone receptor (PR),5, 40 and are more potent.19, 47
Domestic and zoo canids have similar reproductive physiology and have been treated with MGA. The adverse effects of progestins including MGA on the uterus of domestic dogs have been documented;2, 7, 9, 10, 24, 52 however, the same has not been done for zoo canids. Because of the uterine lesions associated with progestin administration in domestic dogs, the increased potency of synthetic progestins and the potential for reproductive tract lesions to contribute to decreased fertility, there is a need to assess the risk of developing proliferative and inflammatory uterine lesions in zoo canids contracepted with MGA. With increasing age, intact domestic dogs will naturally develop proliferative and inflammatory uterine lesions.20, 36 Therefore, the risk of developing uterine lesions in zoo canids treated with MGA needs to be evaluated against the background of spontaneously occurring lesions.
The objectives of this study were to describe the reproductive tract lesions of female zoo canids and to assess the effect of MGA treatment on the prevalence and severity of these lesions. Because endogenous estrogens may exacerbate the effects of progestins by up-regulating the PR,23, 51 this study also evaluated whether folliculogenesis was suppressed in MGA-treated canids.
Materials and Methods
Study population
Reproductive tracts from female canids that were ovariohysterectomized or died at participating zoological institutions were submitted between 1992 and 2007 to the Wildlife Contraception Center-Health Surveillance Program (WCC-HSP) as part of ongoing surveillance for the AZA-WCC. From these submissions, the study population (n = 81) was selected based on the following inclusion criteria: known history of MGA exposure (MGA-treated) or no contraception (control), at least 6 months of MGA exposure for MGA-treated animals, known number of full-term pregnancies (parity), 24 months of age or older, at least 1 ovary and at least 2 sections of uterus. Twenty-four months of age was selected because it is the approximate age at which Canis spp. ovulate for the first time (WCC-HSP database; L. Munson, unpublished data; C. Asa, personal communication).4 Animals for which only 2 sections of uterus were available were included, because lesions caused by progestins are usually diffuse. Animals that were pregnant or recently pregnant were excluded because of the extensive remodeling that takes place during and shortly after pregnancy. Canids treated with a combination of progestins were also excluded to avoid the confounding effects of other progestins.
Evaluation of the ovaries was considered necessary to interpret uterine lesions in the context of endogenous ovarian steroid hormones. Because in some cases only 1 ovary was submitted, it was necessary to test whether 1 ovary would provide enough information on the endogenous hormonal milieu of the animal. This was expected to be true given that domestic dogs usually ovulate several ova synchronously from each ovary during an estrous cycle.1, 8, 39 In a subset of 40 animals for which both ovaries were available, Kappa coefficients of agreement beyond chance were calculated between follicles on the right or left ovary and follicles on both ovaries. Kappa coefficients were similarly calculated for corpora lutea (CL). There was good-to-perfect agreement between the presence of follicles on 1 ovary and those on both ovaries (Kappaleft ovary = 0.778 and Kapparight ovary = 0.844) and between the CL on right or left ovary and those on both ovaries (Kappaleft ovary= 0.942 and Kapparight ovary = 1.000). Based on the high coefficient of agreement, cases with only 1 ovary were included.
Histopathologic examination
Tissues were fixed in 10% neutral-buffered formalin by the contributing institution before shipment. Two sections from each uterine horn and 1 from each ovary were sampled, as well as any gross lesions. Tissues were embedded in paraffin, and 5-μm sections were routinely stained with HE. Histopathologic examination was performed by 2 investigators (LM, AM) without knowledge of treatment. Ovaries were examined for structures that could be a source of endogenous estrogen or progesterone, such as tertiary or cystic follicles, CL, or granulosa cell tumors (GCT). In addition, ovaries were examined for subepithelial structure cysts, rete cysts, follicular cysts, Wolffian duct remnants, neoplasia, and mineralization.
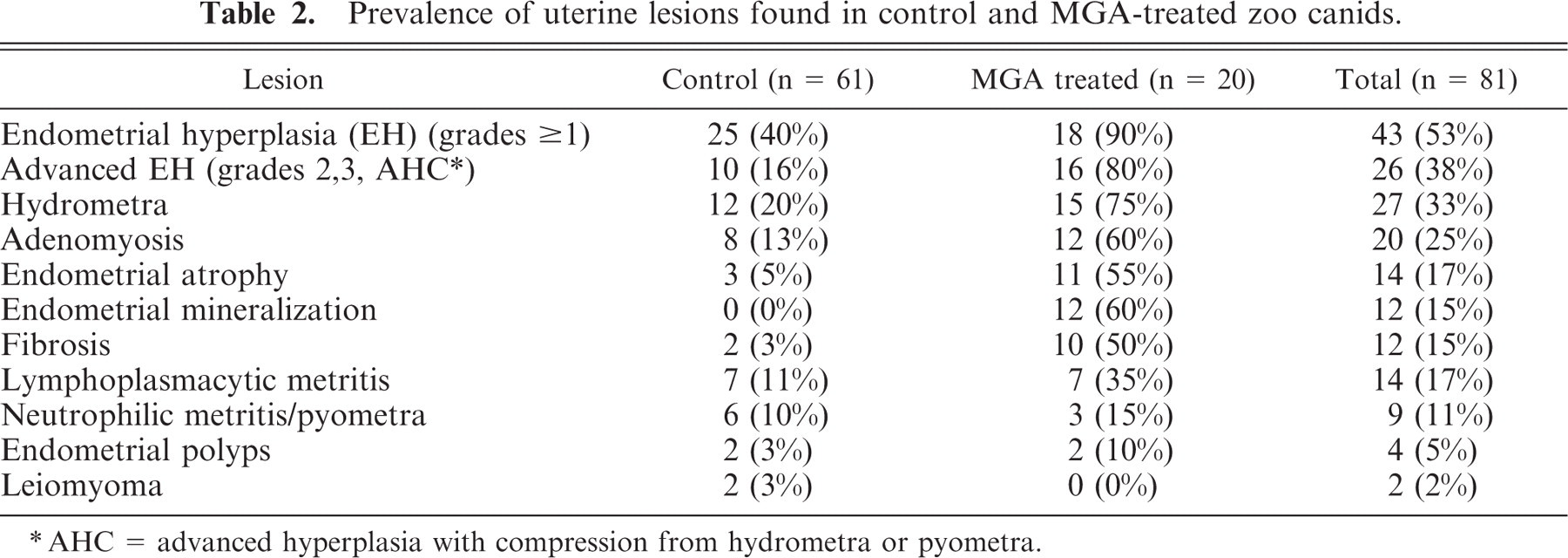
Uterine sections were evaluated for the presence of EH, hydrometra, pyometra, suppurative endometritis, lymphoplasmacytic endometritis, endometrial polyps, neoplasia, fibrosis, adenomyosis, and mineralization. EH was categorized as adenomatous, cystic, or papillary; some cases had more than one category of EH. Severity of EH was graded as in felids,34 based on the most severe lesion: grade 0 = no hyperplastic changes; grade 1 = minimal-to-mild proliferative or cystic change in glands or surface epithelium without an increase in overall endometrial height; grade 2 = moderate hyperplastic or cystic change with an increased endometrial thickness of <2 times normal; grade 3 = severe hyperplastic or cystic change with increased endometrial thickness of ≥2 times normal (Figs.1–3). Uteri that had marked hyperplasia in combination with compression due to hydrometra/mucometra or pyometra could not be scored with this grading system and were classified as advanced hyperplasia with compression (AHC) (Fig. 4). Normal endometrial thickness was determined from grade 0 uteri in species of similar body size and from approximately the same stage of the estrous cycle. Proliferative changes in the endometrium that were typical for stages of the estrous cycle were not considered hyperplasia. Hydrometra was defined as any amount of proteinaceous or mucinous fluid within the lumen of the uterus. Adenomyosis was defined as well-differentiated endometrial glands and stroma present in the myometrium, discontiguous with, but morphologically similar to the endometrium.34 Endometrial polyps were defined as discrete, narrow-based, intraluminal projections of well-differentiated endometrial glands, and stroma resembling the mural endometrium.34
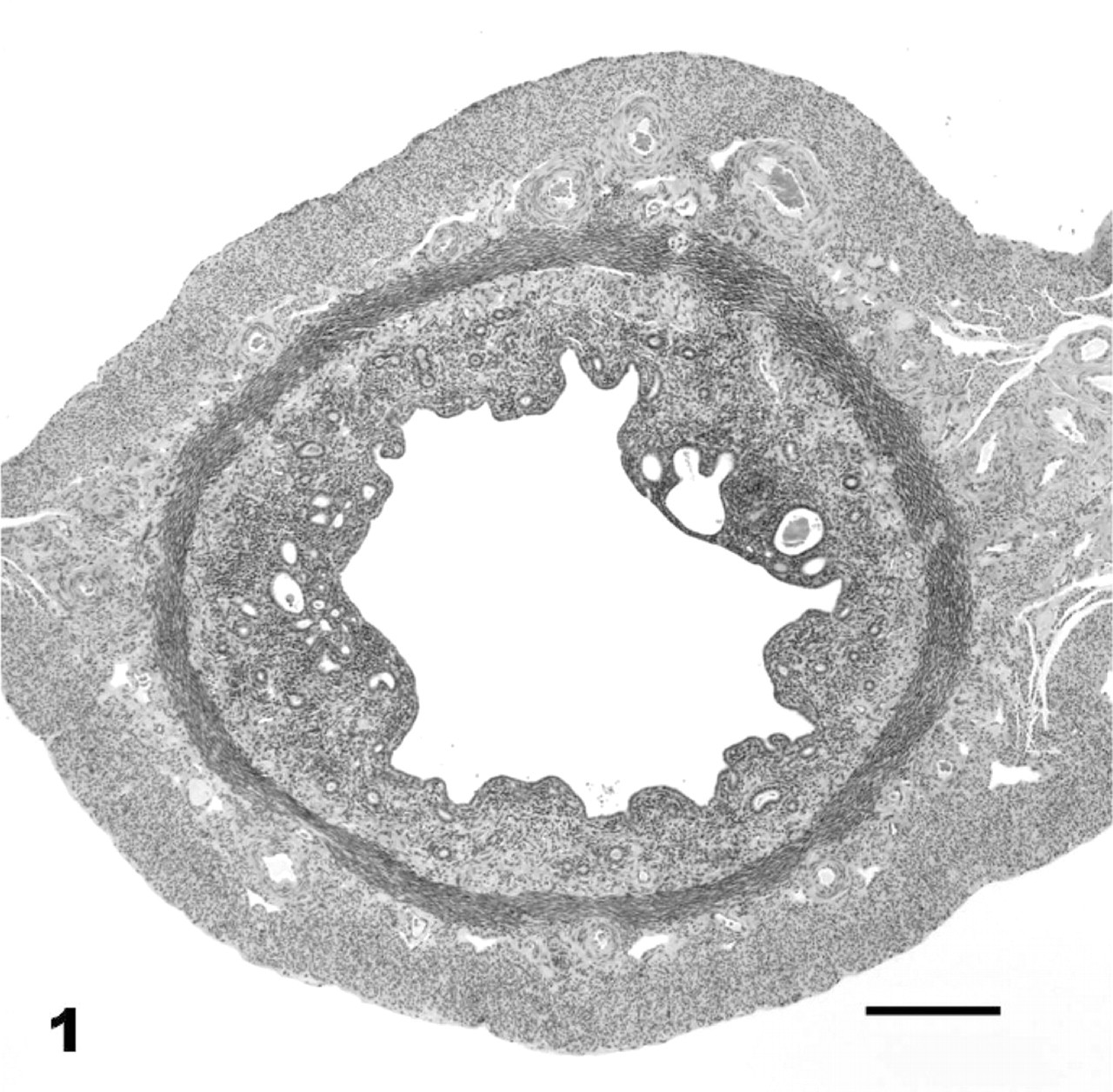
Uterus; bat-eared fox No. 25, control animal. Mild endometrial hyperplasia, grade 1. HE. Bar = 200 μm.
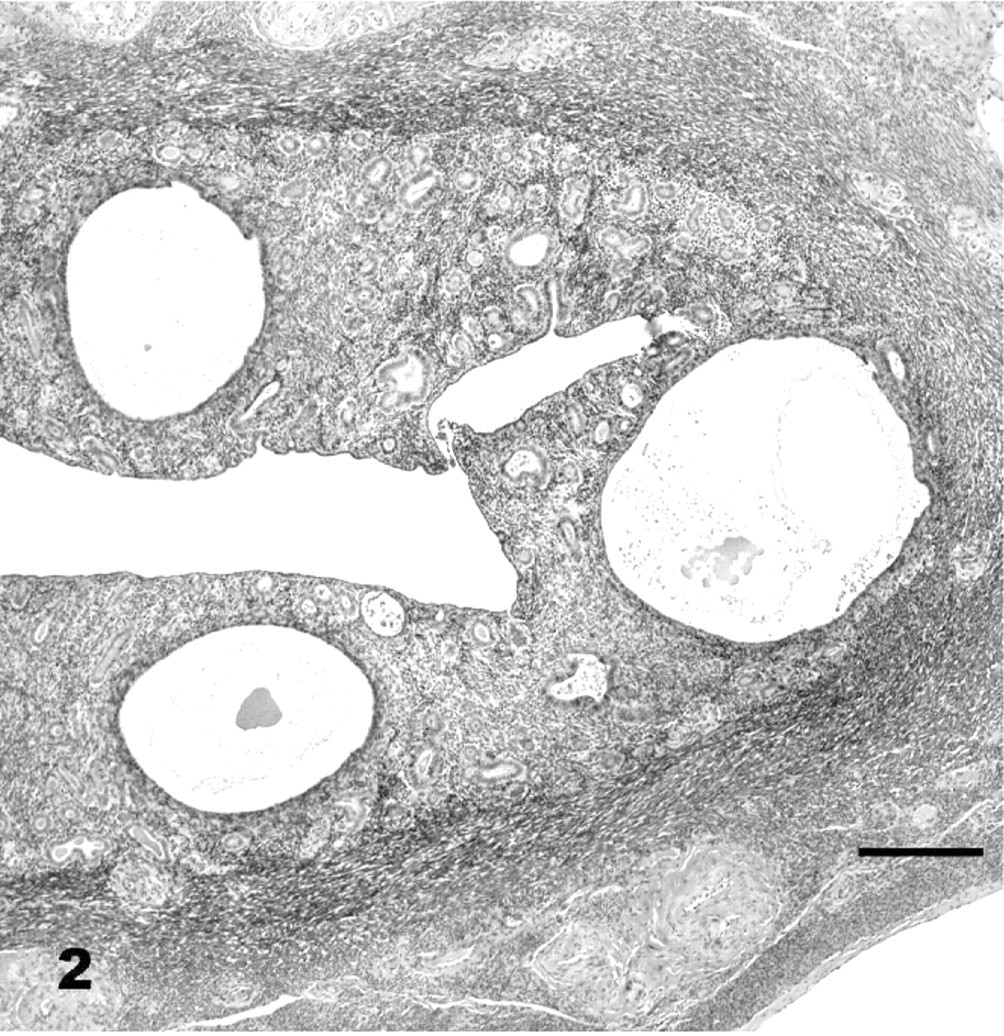
Uterus; maned wolf No. 18, control animal. Moderate endometrial hyperplasia, grade 2. HE. Bar = 200 μm.
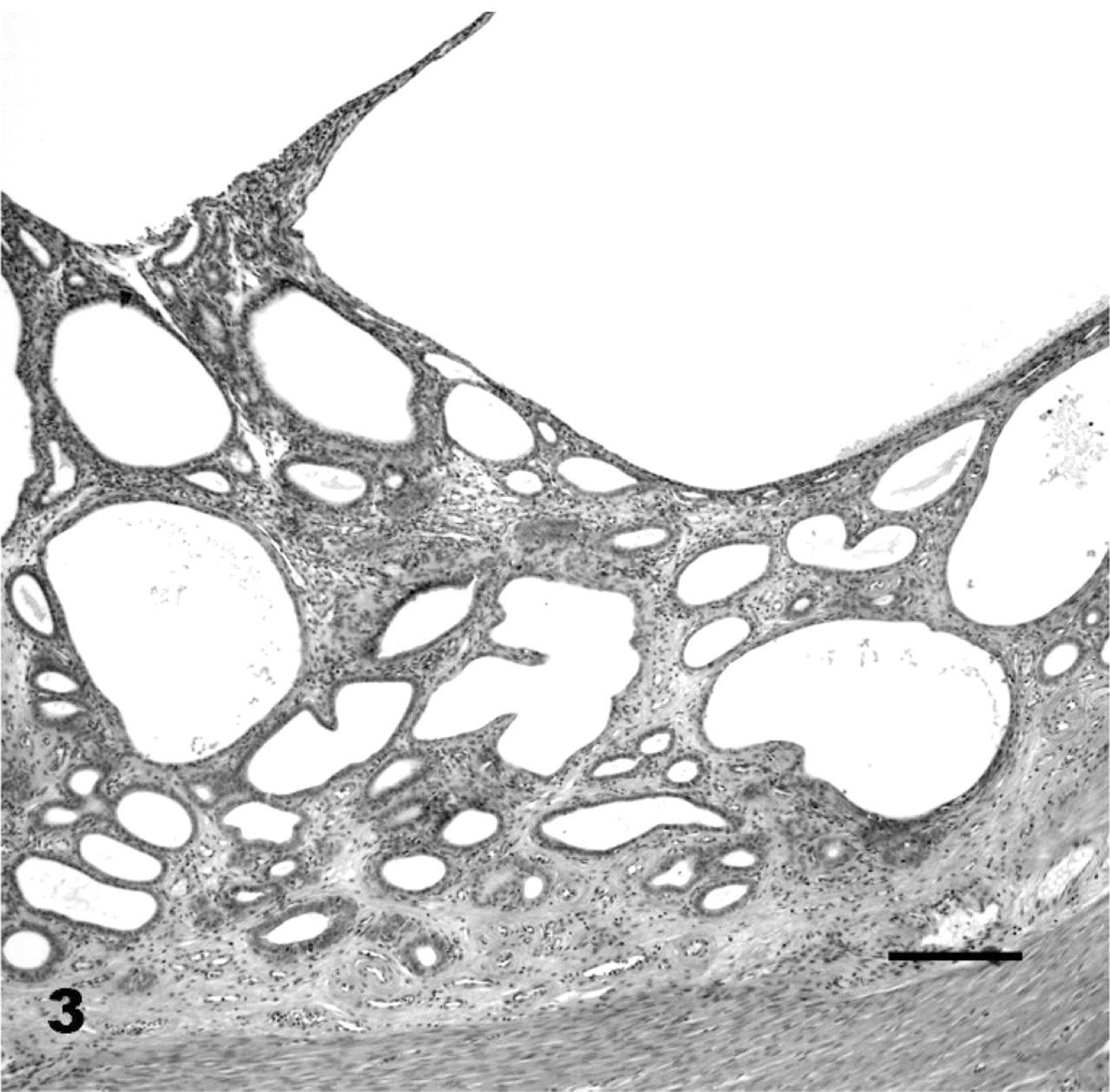
Uterus; bush dog No. 48, control animal. Severe endometrial hyperplasia, grade 3. HE. Bar = 200 μm.
Statistical methods
Age, duration of MGA exposure, and average dose were normally distributed and are reported as means ± SD; comparisons between treated and untreated animals were made using t-tests. For measures that were not normally distributed results are reported as medians with ranges; medians were compared with a Mann-Whitney test. The ability of MGA implants to suppress ovarian activity was evaluated in a subset of 6 animals that had an MGA implant in place when the reproductive tract was collected.
The presence of each lesion was tested for association with MGA exposure by Fisher's exact tests. Lesions with significant Fisher's exact test results were further analyzed by logistic regression to determine significant risk factors. EH was evaluated in 2 ways. First, EH was categorized as either present (any EH) or absent. Second, EH was dichotomized into mild EH (presumed clinically not relevant; grades 0 and 1) or advanced EH (presumed clinically relevant; grades 2, 3, and AHC). The remaining lesions were classified as either present or absent. Risk factors tested in the models were: MGA exposure, age in years, parous or nulliparous, presence or absence of suppurative endometritis, and presence or absence of CL. Because EH may develop spontaneously with age, all tested models of EH included age as a covariate. Suppurative endometritis was included in models evaluating EH because it has been associated with EH development.42 When comparing the severity of EH between the treated and the control groups, the AHC category was grouped with the grade 3 cases.
Results were considered statistically significant if P < .05; odds ratios (OR) and 95% confidence intervals (CI) were calculated for final models. Overall fit of models was tested using Hosmer-Lemeshow (HL) goodness-of-fit statistics.28
Results
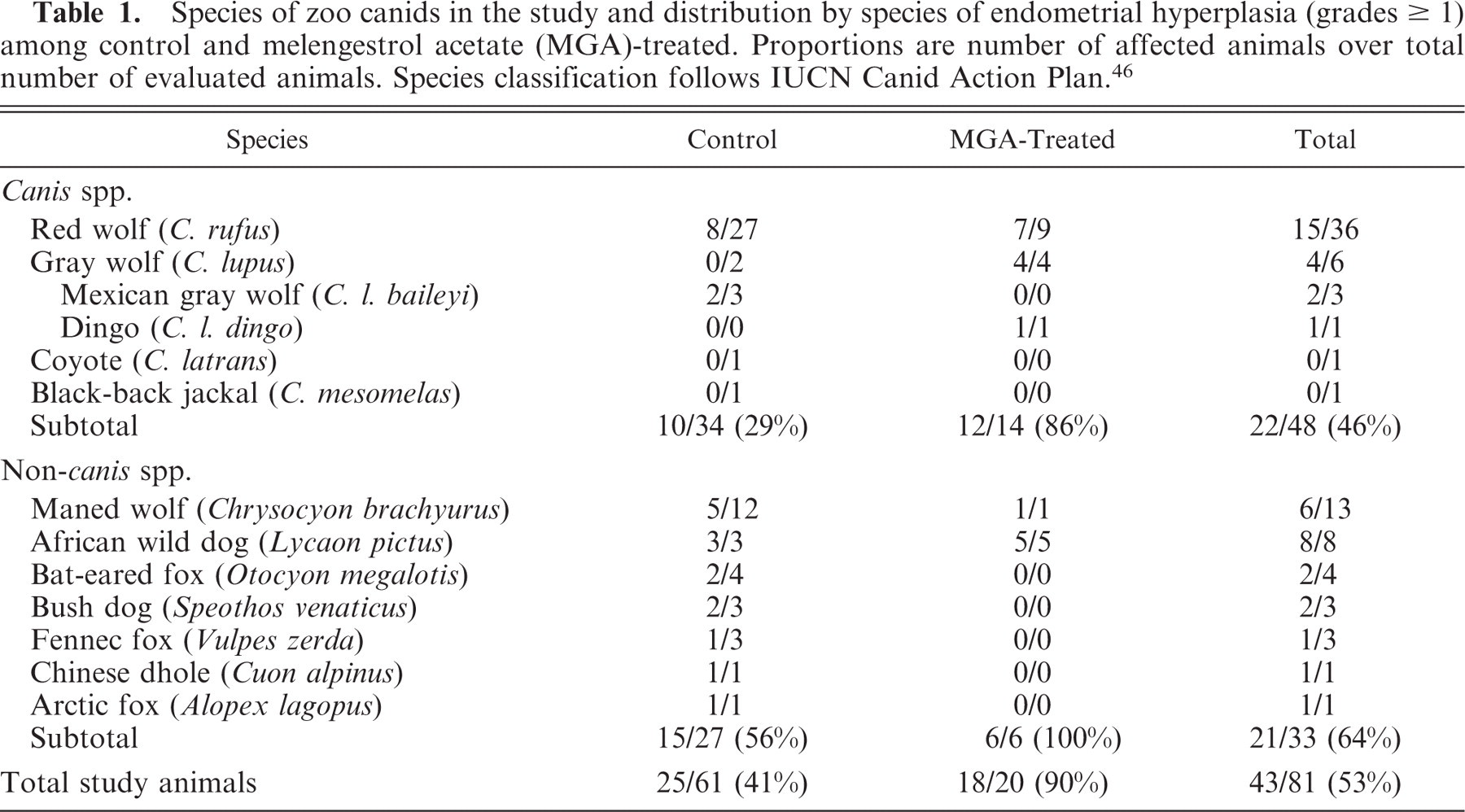
The study population included 20 MGA-treated and 61 control animals representing 11 species and 2 subspecies (Table 1).53 Mean (± SD) age did not differ (P = .416) between MGA-treated (114.1 months ± 47.8) and control animals (103.5 months ± 51.0). Median parity was lower (P = .047) for the MGA-treated group (median 0; range 0–3) than for the control group (median 1; range 0–7). The MGA-treated animals were exposed to an average dose per animal of 22.8 ± 6.3 mg/kg for 27.4 ± 11.7 months.
Species of zoo canids in the study and distribution by species of endometrial hyperplasia (grades ≥ 1) among control and melengestrol acetate (MGA)-treated. Proportions are number of affected animals over total number of evaluated animals. Species classification follows IUCN Canid Action Plan.46
Uterine lesions
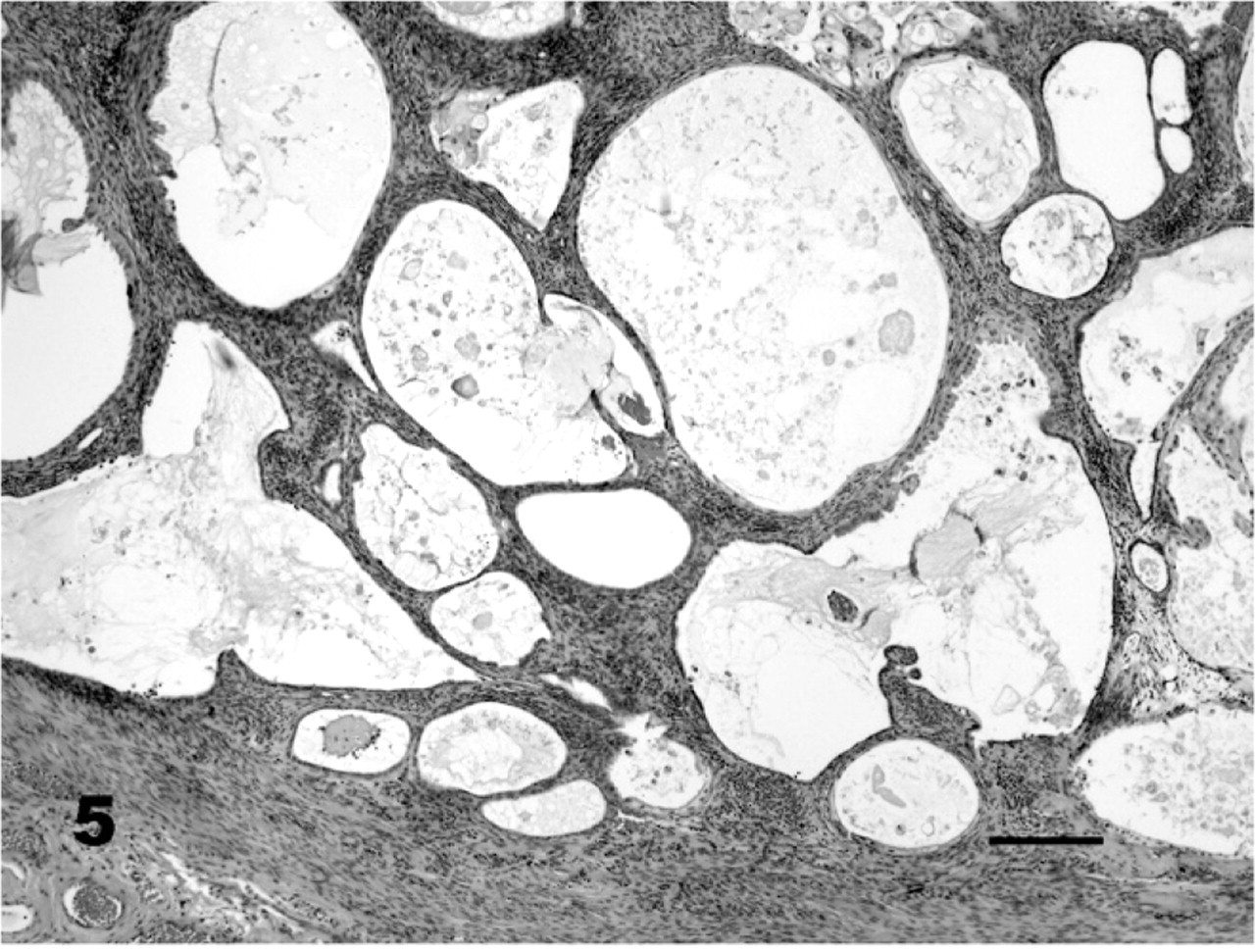
EH was the most prevalent lesion in the zoo canid population (53%) and prevalence was significantly higher in the MGA-treated than in the control group (P = .002) (Table 2). EH was characterized by glandular proliferation ranging from a few to multiple cystic glands, distributed diffusely throughout the endometrium or rarely in clusters (Figs. 1–3). Cystic glands varied in size and were distinguished from ectatic glands by the compression of the adjacent stroma. Cystic glands were lined by densely packed but variably compressed cuboidal epithelium and were filled with proteinaceous secretions. EH in zoo canids was predominantly cystic (of the cases with EH 90% had a cystic component, 44% had an adenomatous component, and 18.6% had a superficial component).
Prevalence of uterine lesions found in control and MGA-treated zoo canids.
AHC = advanced hyperplasia with compression from hydrometra or pyometra.
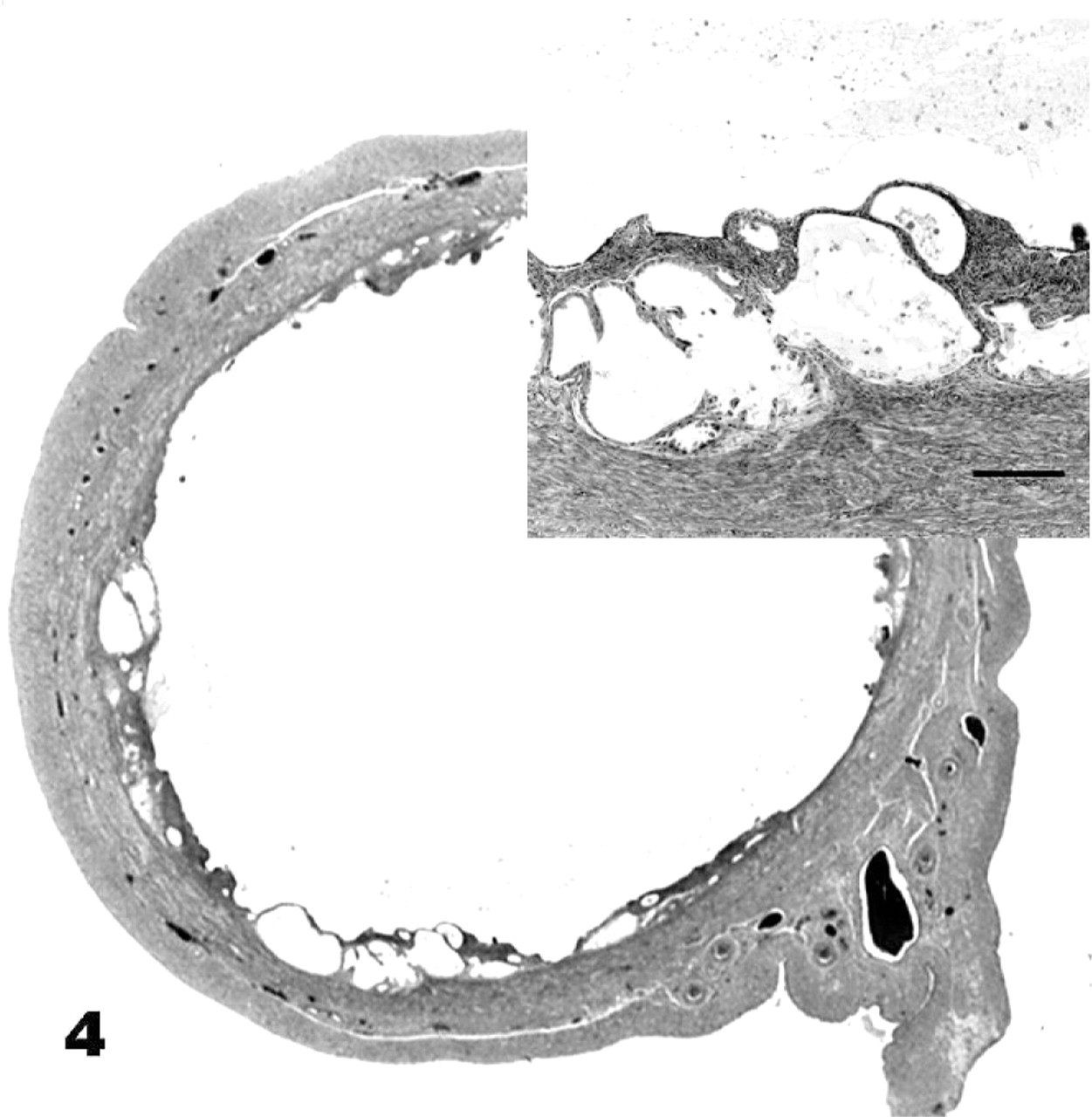
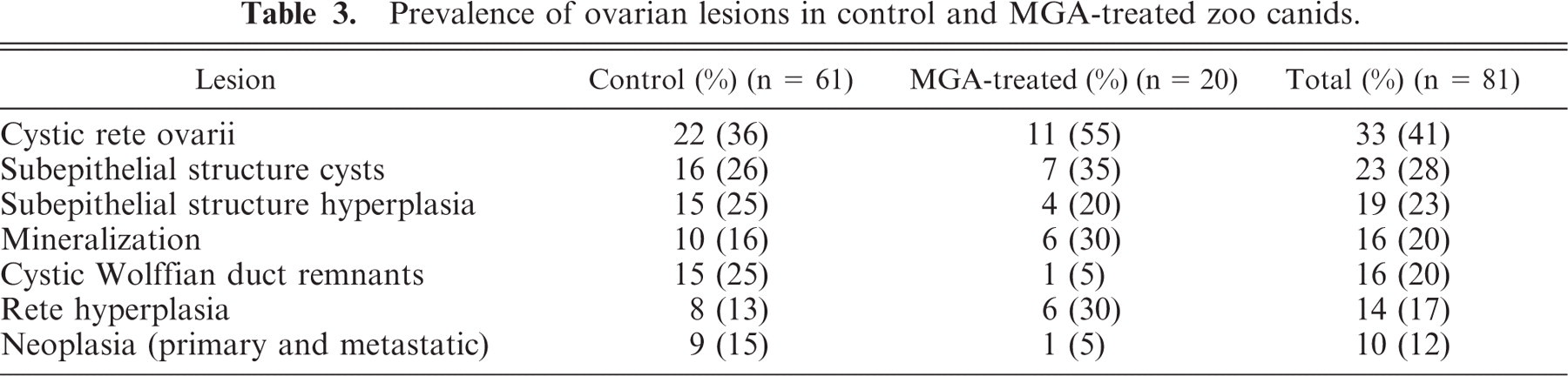
Although advanced EH was present in control and in MGA-treated animals, the morphology differed. Distinguishing features in MGA-treated animals included more disorganized, irregular cysts, more disorganized stroma, and more papillary surface hyperplasia than in control animals (Figs. 3, 5). The epithelium lining the cysts and luminal surface in MGA-treated animals had segmental regions of papillary hyperplasia with cells that had markedly vacuolated cytoplasm (Fig. 6) and karyomegaly. The segmental changes seen in MGA-treated animals are in contrast to the diffuse surface changes in normal diestrus. A notable feature found exclusively in MGA-treated animals was the presence of mineralized glands and stroma (Fig. 7). Periglandular and interglandular fibrosis were also more prominent in the MGA-treated animals.
Uterus; dingo No. 80, melengestrol acetate (MGA)-treated animal. Advanced endometrial hyperplasia with compression, AHC. HE. Inset: Bar = 200 μm.
Uterus; red wolf No. 44, MGA-treated animal. Grade 3 endometrial hyperplasia. Stroma and glands are markedly disorganized and endometrial cysts are irregular in MGA-treated animals. HE. Bar = 200 μm.
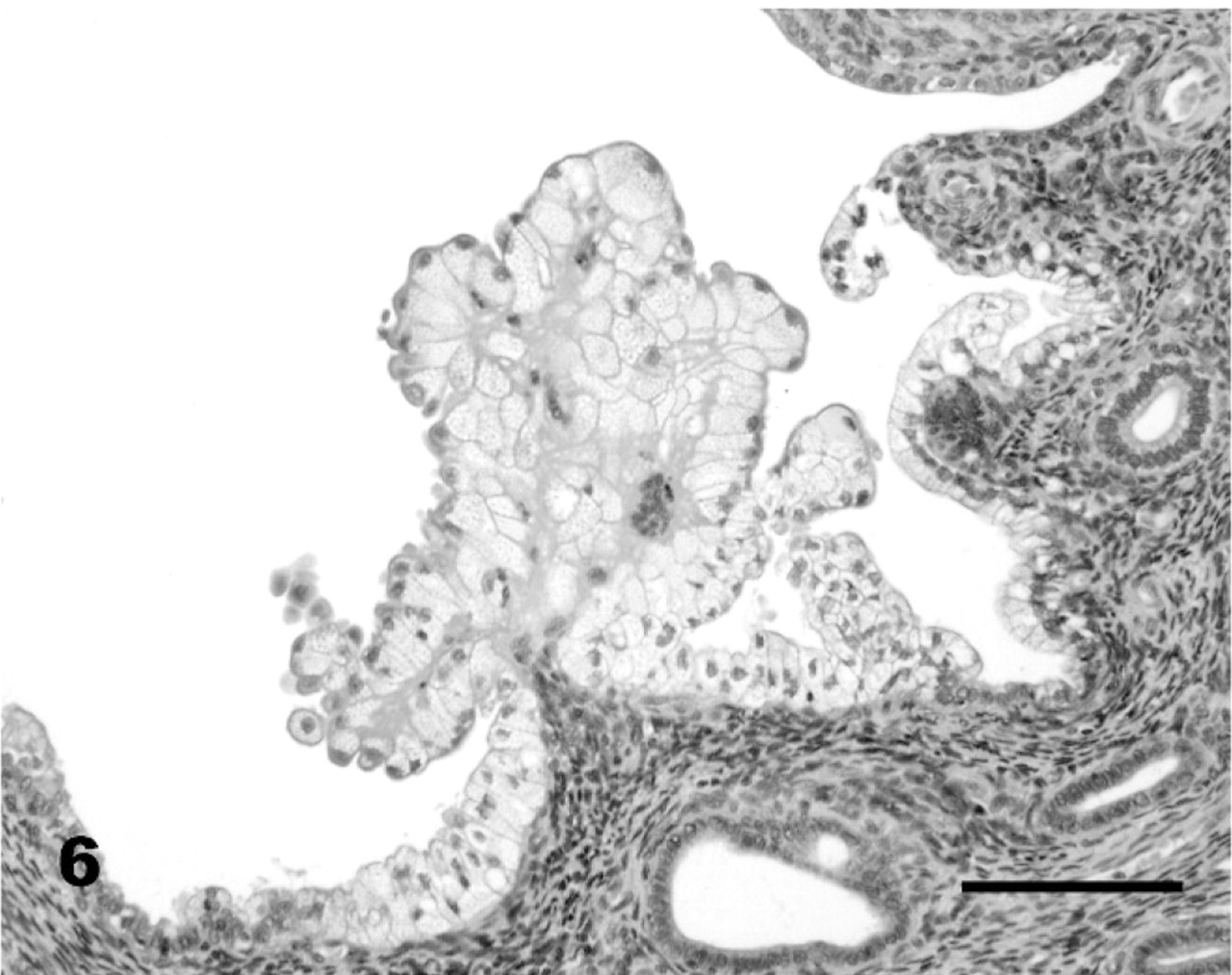
Uterus; red wolf No. 63, MGA-treated animal. Vacuolization and papillary hyperplasia of the surface epithelium are multifocal rather than diffuse. HE. Bar = 100 μm.
Hydrometra in control and MGA-treated animals was usually diffuse and grossly expanded the lumina of both uterine horns in a uniform diameter, but in some cases hydrometra was segmental. The degree of hydrometra ranged from mild distension of the lumen to severe compression of the endometrium (Fig. 4). The inflammation in control and MGA-treated animals consisted of neutrophilic and lymphoplasmacytic components. Although there were cases of severe inflammation in both groups, none of the control animals with endometritis had fibrosis, whereas 5 of 7 MGA-treated animals with endometritis had fibrosis.
Ovarian lesions
There were 37/81 animals (46%) that had tertiary follicles and CL present, 30/81 (37%) that had tertiary follicles only, and 6/81 (7%) that had CL only, the remaining 8 (10%) did not have either follicles or CL present. All 6 animals that were exposed to MGA at the time the reproductive tract was removed had tertiary follicles and no CL. Duration of MGA exposure in these 6 animals was 26–43 months, and average doses per animal ranged from 16 to 34 mg/kg.
The prevalence of ovarian lesions in control and MGA-treated animals is reported in Table 3. Mineralization occurred principally in the ova. Primary ovarian neoplasia was present in 10 animals (12%) and 1 of them also had metastatic neoplasia. Primary tumors found in the ovary were GCT (4 cases), leiomyomas (2 cases), papillary cystadenocarcinomas (2 cases), and 1 case each of papilloma, papillary cystadenoma, and dysgerminoma. Of these animals, 1 had a GCT as well as a leiomyoma; another had a primary papillary cystadenocarcinomas and a metastatic adenocarcinoma. The adenocarcinoma in the latter animal was presumed to be metastatic because it was multifocal in distribution and differed in appearance from primary ovarian carcinomas. Its origin was unknown. One of 4 animals with GCT had a grade 3 EH, 2 animals had grade 1 EH, and 1 animal, grade 0 EH; none of the GCT cases were MGA-treated animals. Neoplasms (1 leiomyoma and 1 leiomyosarcoma) were found in the ovarian stalk of 2 additional canids. Subsurface epithelial cysts were common (23/81, 28%) and were considered an incidental finding.
Prevalence of ovarian lesions in control and MGA-treated zoo canids.
Risk factors
The presence of EH (grade ≥1) was significantly associated with increasing age (P = .003), with MGA treatment (P = .006), and with the presence of endometritis (P = .007); however, the fit for this model was poor (HL P = .256). When the model only included MGA exposure and endometritis (and age was excluded), the fit improved substantially (HL P = .953). This effect is likely due to a small sample size in one of the age categories, introducing a disproportionate amount of variation when age was included in the model.
The presence of advanced EH (grades 2, 3, and AHC) was not associated with age (P = .335) but was significantly associated with MGA treatment (P < .001) and with suppurative and nonsuppurative endometritis (P = .004); the fit was good (HL P = .740). When the model excluded nonsuppurative endometritis, the fit improved (HL P = .909).
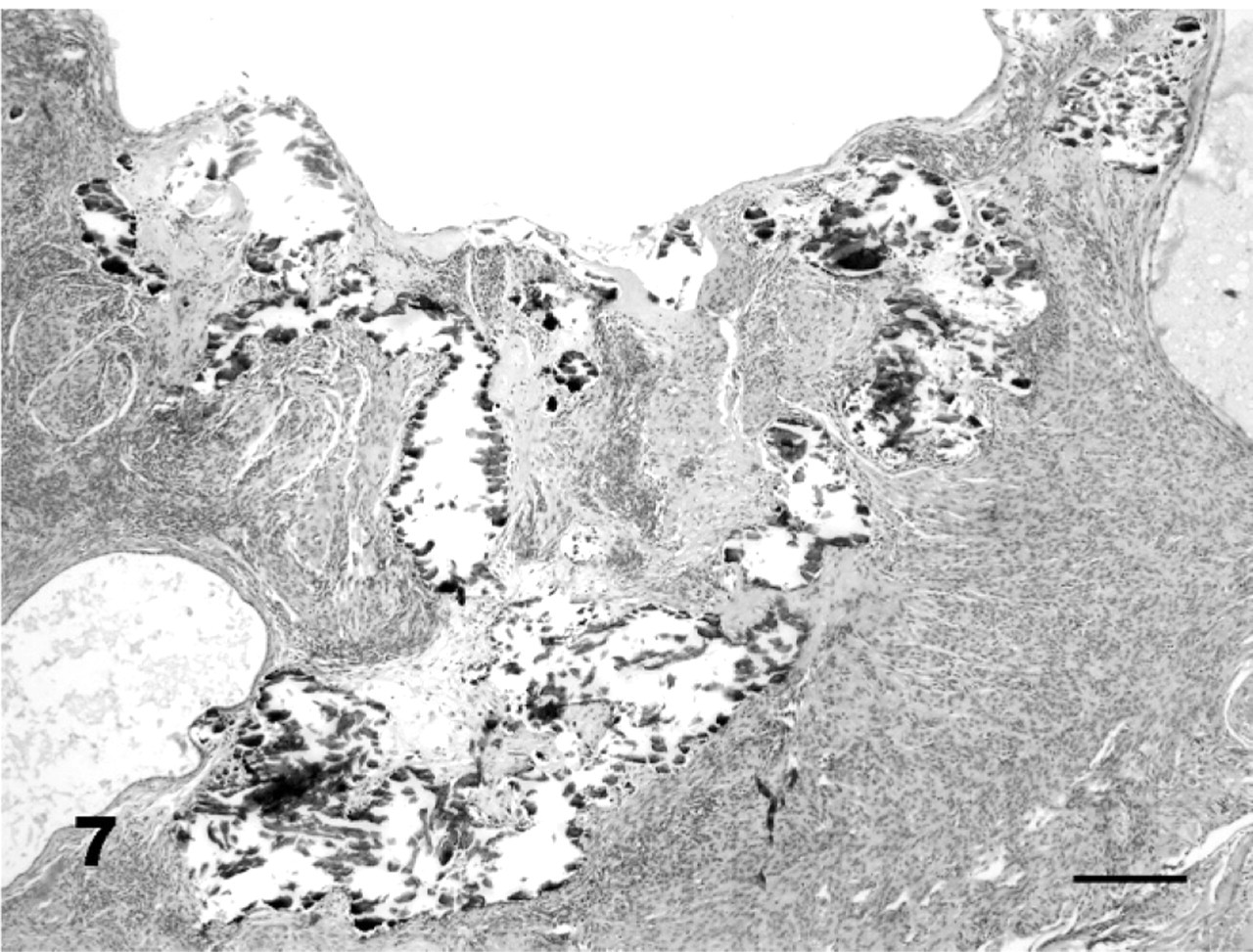
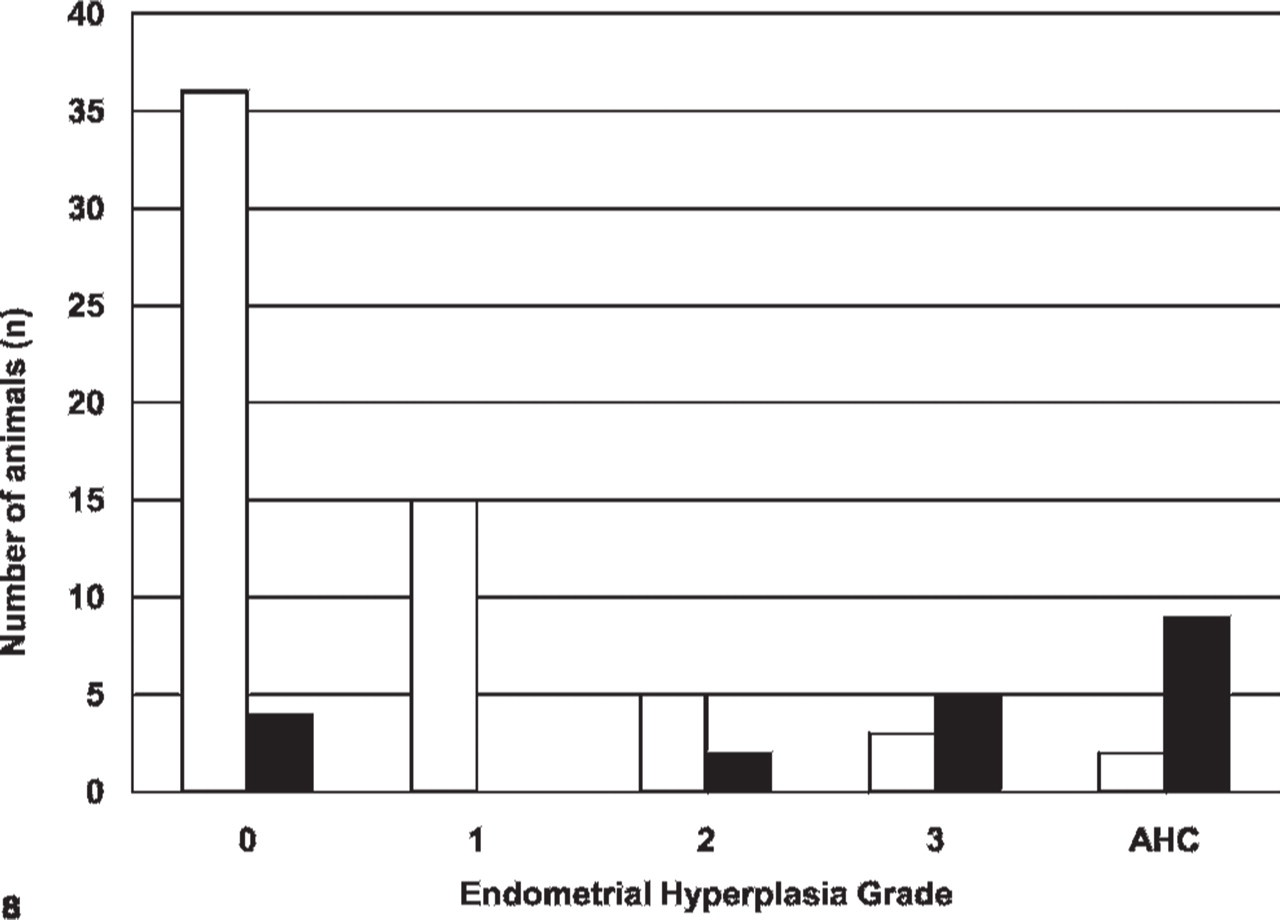
EH was more severe in MGA-treated than control animals (P < .001). In MGA-treated animals, the median grade was 3 (range 0–3/AHC) in contrast with the control group, in which the median grade was 0 (range 0–3/AHC) (Fig. 8). Advanced EH (grades 2, 3, and AHC) was more prevalent in the MGA-treated (16/20; 80%) than in the control group (10/61; 16%) (P < .0001). Advanced EH was associated with MGA exposure (P < .001; OR 22.25; 95% CI 5.74–86.04) and suppurative endometritis (P = .017; OR 7.81; 95% CI 1.44–42.45). Advanced EH was not associated with age or parity (P = .154 and .065, respectively). Advanced EH first developed at a younger age in the MGA-treated than in the control group (Fig. 9). The character of the EH (adenomatous, cystic, or superficial) was not associated with MGA exposure.
Uterus; African wild dog No. 52, MGA-treated animal. Mineralization occurred only in MGA-treated animals and was mainly localized in the endometrial glands. HE. Bar = 200 μm.
Distribution (by grade) of endometrial hyperplasia in zoo canids. Normal endometrium (grade 0); mild hyperplasia (grade 1); moderate hyperplasia (grade 2); severe hyperplasia (grade 3); advanced hyperplasia with compression (AHC). White bars represent control animals, black bars represent melengestrol acetate-treated animals.
Distribution of advanced endometrial hyperplasia (EH) by age, in control (white) and MGA-treated (black) zoo canids.
African wild dogs (AWD) seemed particularly susceptible to developing EH. All 8 AWD in the study had EH (5 MGA-treated and 3 controls); 6 of 8 had advanced EH and 5 of these 6 were treated with MGA. The 1 control animal with advanced EH had concurrent pyometra. Adenomyosis was also more common in AWD than in other species in this study (P = .024). In fact, AWD were 8.32 times (95% CI 1.27–54.60) more likely to have adenomyosis than the other species (P = .027). Adenomyosis was present in 6 of 8 AWD (2 of 3 controls and 4 of 5 MGA-treated) compared with only 14 of 73 other canids. The percentage of AWD with pyometra was higher (2/8, 25%) than in other species (5/73, 7%); however, this difference was not significant (P = .14).
Hydrometra was significantly associated with MGA treatment (P < .001), but not with advanced EH (P = .981) (Table 4). Adenomyosis in general was associated with MGA treatment (P = .001) (Table 4) and with the presence of endometritis (P = .001), but was not associated with the presence of advanced EH (P = .733). Endometrial mineralization was found only in treated animals, but was not independently associated with advanced EH in this group. Eleven of 12 animals with endometrial mineralization had grade 3 EH (n = 4) or AHC (n = 7). In the model, fibrosis was associated with exposure to MGA (P = .018) and advanced EH (P = .026) (Table 4), but not with endometritis (P = .714). Of the animals without endometritis, a higher proportion of the MGA-treated animals than controls had endometrial fibrosis (P = .003). Suppurative endometritis was positively associated with the presence of EH (P = .004) (Table 4), as well as the presence of CL (P = .027), was negatively associated with increasing age (P = .019), and not associated with MGA exposure (P = .718). The low number of animals with endometrial polyps or neoplasia did not allow risk factor analysis for these lesions.
Odds ratio (OR) and 95% confidence intervals (CI) for various endometrial lesions in zoo canids. Models include MGA treatment and advanced EH (grades 2, 3, or advanced hyperplasia with compression [AHC]).
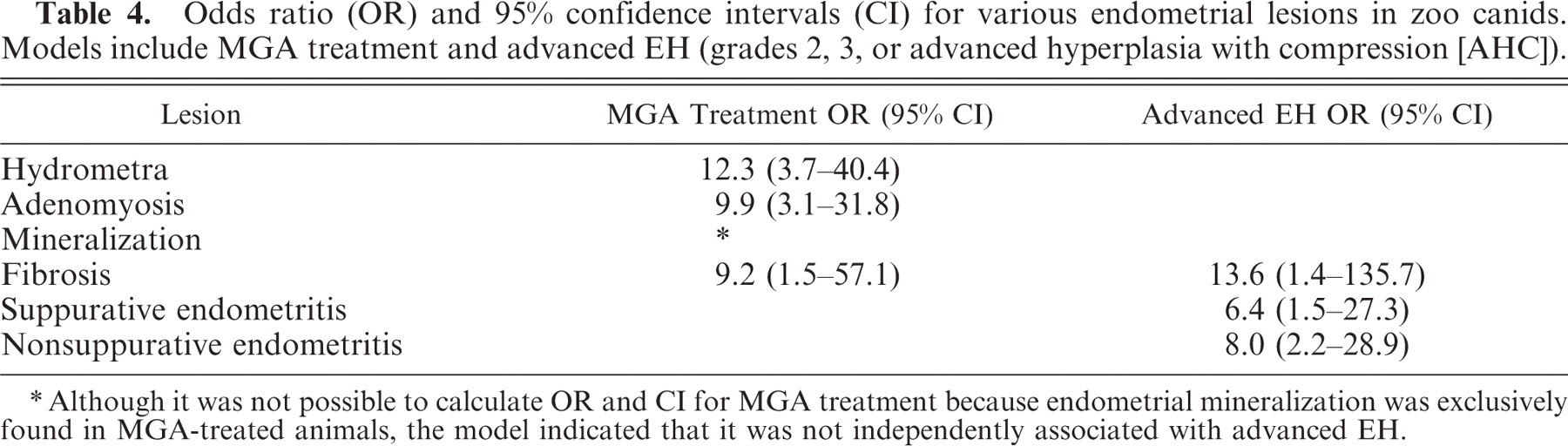
Although it was not possible to calculate OR and CI for MGA treatment because endometrial mineralization was exclusively found in MGA-treated animals, the model indicated that it was not independently associated with advanced EH.
None of the lesions in the ovaries or ovarian ligament was significantly associated with exposure to MGA (P = .102–1.000). Cystic subsurface epithelial structures were common in zoo canids and were associated with increasing age (P = .013).
Nine of 10 cases of primary ovarian neoplasia and the 2 cases with metastatic neoplasia were control animals. The presence of ovarian tumors was associated with age (P = .004), but not with MGA exposure (P = .141). There were not enough cases of GCT to statistically analyze their association with EH. Hosmer-Lemeshow goodness-of-fit P value for models was > .6.
Discussion
This study evaluated the prevalence of lesions in the uterus and ovaries of zoo canids to characterize spontaneously occurring lesions and determine whether MGA administration is a risk factor for these lesions. EH was the most common uterine lesion in the population as a whole. EH was significantly associated with exposure to MGA, developed at an earlier age, was more advanced in MGA-treated animals, and had unique morphologic features. Hydrometra, adenomyosis, fibrosis, and mineralization were also associated with MGA exposure. In this study population the occurrence of uterine inflammatory disease and ovarian lesions was not independently associated with MGA exposure.
Under the influence of endogenous progesterone, the canid endometrium increases proliferation and secretion.22 Therefore, the association of EH, hydrometra, and adenomyosis with MGA exposure in this study is in accordance with known actions of progestins. The association of MGA exposure with an increased risk of EH in zoo canids is in agreement with what has been reported in MGA-treated beagles and zoo felids,34, 46 as well as in domestic dogs treated with other synthetic progestins.7 Because not all treated animals developed EH, exogenous progestins do not necessarily lead to EH. The spontaneous occurrence of EH in untreated animals implicates endogenous hormones as an integral part of the pathogenesis.39 Although EH was significantly associated with increasing age, animals of advanced age do not necessarily develop EH. The oldest animal in the study, a 16-year-old fennec fox, had a normal (grade 0) endometrium despite not having been pregnant for 5 years. In the domestic dog, other factors in addition to steroid hormones may be involved in the development of EH, such as insulin-like growth factor (IGF-I)17 or inflammation due to bacterial infection or mechanical irritation.10, 37 An association between inflammation and EH was also found in zoo canids.
The predominantly cystic character of the EH in canids differs from the more adenomatous character of EH in felids.34 This difference may relate to the species differences in the estrous cycle hormonal pattern. Cats are inducible ovulators and although they are exposed to estrogen each estrous cycle, they are not necessarily exposed to progesterone. In contrast, dogs ovulate spontaneously, follicular luteinization starts before ovulation, luteal phase lasts approximately 2 months regardless of conception,14 and maximum serum progesterone levels are similar during nonpregnant luteal phases and during pregnancy.13 Canids are therefore exposed to long periods of elevated progesterone levels every estrous cycle, leading to a very secretory endometrium and facilitating the development of cystic gland lesions and hydrometra. Although control animals were repeatedly exposed to endogenous progesterone, the character of their EH differed from the EH in MGA-treated animals. Distinctive features in MGA-treated animals were endometrial mineralization, marked epithelial asynchrony, and disorganization of the endometrium. Endometrial mineralization only occurred in MGA-treated animals, similar to the predominance of this lesion in MGA-treated zoo felids.34
An association has been found between the development of EH in dogs and repeated estrous cycles without pregnancy.18, 49 Only at parturition are the normal cyclic hyperplastic changes in the canid endometrium extensively remodeled. Therefore, it would be expected that parity protects against developing EH. However, a protective effect of parity was not found in this zoo canid population. This finding is not likely attributable to small sample size as 46% of the population was parous. Possibly a better measure to assess the protective effect of parturition would be to evaluate the time since last parturition, but these data were not available for most animals in the present study.
Hydrometra was a common finding in both MGA-treated and control zoo canids. Identification of this lesion in endangered species is particularly concerning because it can permanently damage the endometrium by pressure atrophy and can serve as a nidus for bacterial infection, leading to pyometra and infertility. Treatment with MGA was a risk factor in zoo canids for the development of hydrometra and has been reported in domestic dogs treated with MPA.7 Hydrometra was also a relatively common lesion in control animals. The high prevalence of hydrometra in zoo canids (75% of treated and 20% of control canids) is in contrast to the lower prevalence found in zoo felids (8.1% of treated and 2.7% of control felids).34 This difference in prevalence might be explained by the more prolonged exposure of canids to the secretory effects of endogenous progestins on the endometrial epithelium.
The cause of adenomyosis remains unknown, but is likely multifactorial. Parity, progestin exposure, EH, inflammation, surgical trauma, and age have been suggested as potential risk factors.6, 25, 32, 42, 50 In the present study, adenomyosis was associated with MGA exposure, as well as with endometritis, but not with parity or EH. A direct role for progestins is further supported by findings in BALB-C mice where adenomyosis increased with prolonged progesterone exposure.32
The link between progestin exposure and the development of pyometra is still controversial. An association between progestins and endometritis/pyometra has been attributed to the secretory environment, altered immune resistance, and decreased myometrial contractility induced by progesterone,48 making the uterus fertile ground for bacterial infections. Suppurative endometritis/pyometra was not associated with MGA treatment in zoo canids in this study. This finding is in agreement with studies in domestic dogs and zoo felids where pyometra and progestins were not associated.16, 34, 36, 46 In contrast, other studies report an association between synthetic progestins and endometritis/pyometra in the domestic dog.2, 7, 31, 49 It is possible that the lack of association in the present study is due to the low number of suppurative endometritis and pyometra cases. Evaluation of a larger sample size would be valuable.
Findings of this study indicated that MGA does not suppress folliculogenesis in zoo canids. This finding is in agreement with the effect of MGA in zoo felids,29 as well as other progestins in domestic dogs.49, 52 MGA-treated animals were therefore exposed to endogenous estrogens while being exposed to exogenous progestins. Simultaneous exposure of animals to endogenous estradiol and MGA could potentiate the deleterious effects of MGA through the up-regulation of PR. In vivo and in vitro—canine endometrial epithelial cells up-regulate the number of PR when exposed to estradiol,23, 51 and priming with estradiol reduces the dose of progesterone needed for the development of EH.49 None of the animals implanted with MGA at the time the reproductive tract was obtained had CL present, therefore it is possible that MGA inhibits ovulation. However, canids ovulate only once or twice per year, minimizing the likelihood of detection of CL at the time of sample collection, therefore a larger number of animals would be needed to verify the inhibitory effects of MGA on ovulation.
Similar to felids,29 cystic rete ovarii were the most common ovarian lesion in this canid population, and were more common in MGA-treated animals, although this relationship was not significant. The secretory effect of progestins on the epithelial lining of the rete ovarii may contribute to cyst formation. The types of neoplasia found in the ovary of zoo felids were similar to those found in zoo canids in the present study.29 GCT have the potential to affect endometrial proliferation if they produce estrogens, although no association of GCT tumors with EH was seen in this study; however, the number of tumors was relatively small. As in domestic dogs,42 cystic subsurface epithelial structures were considered incidental findings that do not affect the general health or the fertility of females.
Although the number of AWD in the study was small, there seemed to be a propensity for AWD to develop endometrial disease. All AWD in this study had EH and in 75% of the cases EH were advanced. Seventy-five percent of the AWD in the current study had adenomyosis in addition to 1 case report,35 suggesting it is a relatively common lesion in this species. Although not statistically significant, pyometra was more prevalent in AWD than in other species in this study and additional cases have been noted (H. J. Bertschinger, personal communication).27
This study found an association between MGA treatment and advanced endometrial disease in zoo canids. In female domestic dogs infertility is considered the most important, often the only, clinical sign of CEH.38 In tigers MGA treatment was associated with lesions similar to those described in this study in zoo canids34 and with decreased reproductive success.11 Although the effects of the uterine lesions on the fertility of zoo canids could not be directly evaluated due to the retrospective nature of this study, it seems likely that the MGA-associated lesions described in these canids could potentially decrease fertility in zoo canids. Therefore, the risk of developing endometrial disease in MGA-treated canids may outweigh the benefits of long-term contraception, particularly for genetically valuable breeding animals. The occurrence of similar, but less severe endometrial lesions in zoo canids not exposed to synthetic progestins, suggests that repeated, prolonged exposure to endogenous progesterone may also be detrimental. Therefore, if long-term reproductive control is needed for zoo canids, alternative methods of long-term fertility control that suppress endogenous hormone exposure, such as gonadotropin-releasing hormone (GnRH) analogs or GnRH vaccines, should be considered.
Footnotes
Acknowledgements
We thank the following institutions for contributing tissues and information: Alligator River National Wildlife Refuge, Arizona-Sonora Desert Museum, Binder Park Zoo, Birmingham Zoo, Chaffee Zoological Gardens of Fresno, Chattanooga Nature Center, Brookfield Zoo, Detroit Zoological Park, Folsom Children's Zoo, Fort Wayne Children's Zoo, Fort Worth Zoological Park, Fossil Rim Wildlife Center, Gladys Porter Zoo, Great Plains Zoo & Museum, Henry Doorly Zoo, Houston Zoo, Kansas City Zoological Gardens, Miami Metrozoo, Mill Mountain Zoo, Milwaukee Country Zoological Gardens, Minnesota Zoological Garden, Montgomery Zoo, North Carolina Zoological Garden, National Wildlife Health Center-US Geological Services, Oregon Zoo, Oklahoma City Zoo, Phoenix Zoo, Point Defiance Zoo & Aquarium, San Francisco Zoo, Seneca Park Zoo, The Wilds, Pueblo Zoo, Riverbanks Zoological Park, Binghamton Zoo at Ross Park, Sacramento Zoo, San Antonio Zoological Gardens, Trevor Zoo, Tulsa Zoo & Living Museum, Wild Canid Survival & Research Center, White Oak Conservation Center, Wildlife Science Center, and Woodland Park Zoological Gardens. We thank C. Asa, A. Beyer, S. Boutelle, I. Porton, M. Rodden, P. Siminski, W. Waddell, D. Whitton, and J. Zambrano for providing information details for this study. We thank R. Higgins for assistance with photomicroscopy. This project was supported in part by the AZA Conservation Endowment Fund (grant #010007 or CEF file #483-03), the Bernice Barbour Foundation (#00309-2000), the Geraldine R. Dodge Foundation, and the Saint Louis Zoo. A. Moresco was supported by the NIEHS (Environmental Pathology training grant #ES07055).
