Abstract
Endometrial hyperplasia (EH) is a pathologic condition of the uterus with increased endometrial gland to stroma ratio compared to normal cyclic uterine proliferation. In domestic animals, EH often involves cystic distension of proliferating endometrial glands and may be concurrent with pyometra. In large captive nondomestic felids, an association between EH and pyometra is common; however, detailed species differences between the histological uterine findings in lions (Panthera leo) and tigers (Panthera tigris) and clinical manifestations have yet to be described. Uterine sections from 14 lions and 24 tigers with EH and/or pyometra were scored for several histological parameters and clinical histories were recorded. The percentage of endometrium affected by hyperplasia, endometrial gland to stroma ratio, and adenomyosis were significantly (P = .0385, P = .0008, and P = .0463, respectively) more severe in lions compared to tigers as univariate analytes. Although tubular complexity was not statistically significant (P = .3254), when combined as a proposed EH grading scheme, these 4 features confirmed lions had significantly (P = .0068) more severe EH compared to tigers. Endometrial hyperplasia severity significantly correlated with inflammation/pyometra severity when controlling for species (P = .0203). A significant correlation exists between pyometra-associated clinical sign severity and the presence of pyometra in tigers, (P = .0026) but not in lions (P = .1144). There was no statistical difference in the severity of clinical signs associated with pyometra between these species (P = .1986). This proposed grading scheme may have clinical utility in providing a more consistent and objective evaluation of EH in large captive felids.
Endometrial hyperplasia (EH) is a pathologic condition of the uterus characterized by an increase in the endometrial gland to stroma ratio when compared to the normal proliferation of the hormonally cycling endometrium in both animals and women.20,37 Among domestic animals, EH is most thoroughly described in canines and is thought to be an abnormal uterine response to long-term estrogen or progesterone exposure. 35 These unopposed estrogens/progesterones may come from several sources, including the normal estrous cycle, progesterone contraceptive therapy, functional tumors (such as granulosa-cell tumors), adrenal gland disorders, or in herbivores, consumption of estrogen-containing plant legumes.29,36 In humans, EH is associated with infertility and progression to premalignant or malignant endometrial cancer.3,32 Similarly, EH is frequently associated with infertility in both domestic and nondomestic felids, 25 and although EH is not currently considered a precancerous lesion in domestic animals, reports exist of feline and canine uterine tumors that also describe concurrent EH.2,34,37
In domestic dogs and cats, cystic endometrial hyperplasia (CEH) is so commonly associated with pyometra that it is often regarded as a single entity, called the CEH/pyometra complex (CEH-Pyo).8,27 However, recent literature has suggested these 2 diagnoses be separated as individual processes based on pathophysiology.8,33 Cystic dilation of hyperplastic endometria (CEH) has been documented in a wide range of domestic and nondomestic species, from African and Asian elephants to chinchillas.1,11,24,37 Pyometra and CEH have been reported concurrently in individual cases but has yet to be well established as a combined entity in nondomestic felids.4,26
Pyometra has been documented in older captive wild felids including lions (Panthera leo) and tigers (Panthera tigris), with previous reports indicating that lions may have an increased prevalence of pyometra compared to other nondomestic felid species.16,22 In the clinical experience of 2 authors and main caregivers (ER and DC), lions with pyometra frequently present with moderate to severe clinical illness while tigers with pyometra more commonly present with less clinical evidence of illness. In addition, it has been noted by another author (MS) that in association with pyometra, lions tend to have more severe proliferative lesions in the endometrium compared to tigers.
While there have been numerous histological analyses of EH in humans, domestic felines, canines, and nondomestic species, there is not yet a single broadly used grading scheme to allow for consistent characterization of histopathologic severity for the purposes of comparing clinical and histopathologic disease among animals of different species.8,9,28,38,43,45 The purpose of this retrospective study was to (1) characterize the histological differences in CEH between captive lions and tigers, (2) assess the correlation between EH severity, severity of uterine inflammation/pyometra, and the severity of clinical signs associated with pyometra in these animals, and (3) identify, apply, and evaluate current and previously graded features of EH in both humans and veterinary species to identify those which could best be used to establish an objective histopathologic scoring system to aid in future characterization and understanding of species differences in captive lions and tigers, and perhaps, in the future, more broadly across all veterinary species.
Materials and Methods
Lion and tiger necropsy and biopsy reports from the University of Tennessee Veterinary Medical Center Anatomic Pathology Service from 2003 to 2020 were reviewed. Cases were included if they met the following criteria: (1) female lions or tigers, (2) originate from Zoo Knoxville (Knoxville, TN), or Tiger Haven, a nondomestic felid sanctuary (Kingston, TN), (3) complete necropsy or biopsy reports were available, (4) a prebiopsy or prenecropsy reproductive history was available, (5) slides were available for histologic review, and (6) the animal had histologic evidence of EH and/or pyometra. Tissues from 9 lions and 9 tigers were used as normal controls if they met the first 5 criteria but lacked histologic evidence of uterine disease (Figs. 1 and 2). For each of these 9 cases, 3 measurements of the endometrium and myometrium, respectively, for up to 2 sections (as available), were averaged to establish normal endometrial and myometrial thickness. Uteri from each of the primary stages of the estrous cycle, excluding estrus, were included for each species.6,31 These were considered representative of the population of study animals as these animals had the same spectrum of ages, were from the same facilities (criterion 2), and were not in the estrus stage of the estrous cycle. Cases were limited to these facilities (criterion 2), as the reproductive health of privately owned large felids or those from neighboring zoological facilities were not clinically evaluated by one author (ER) and reproductive histories were unknown.
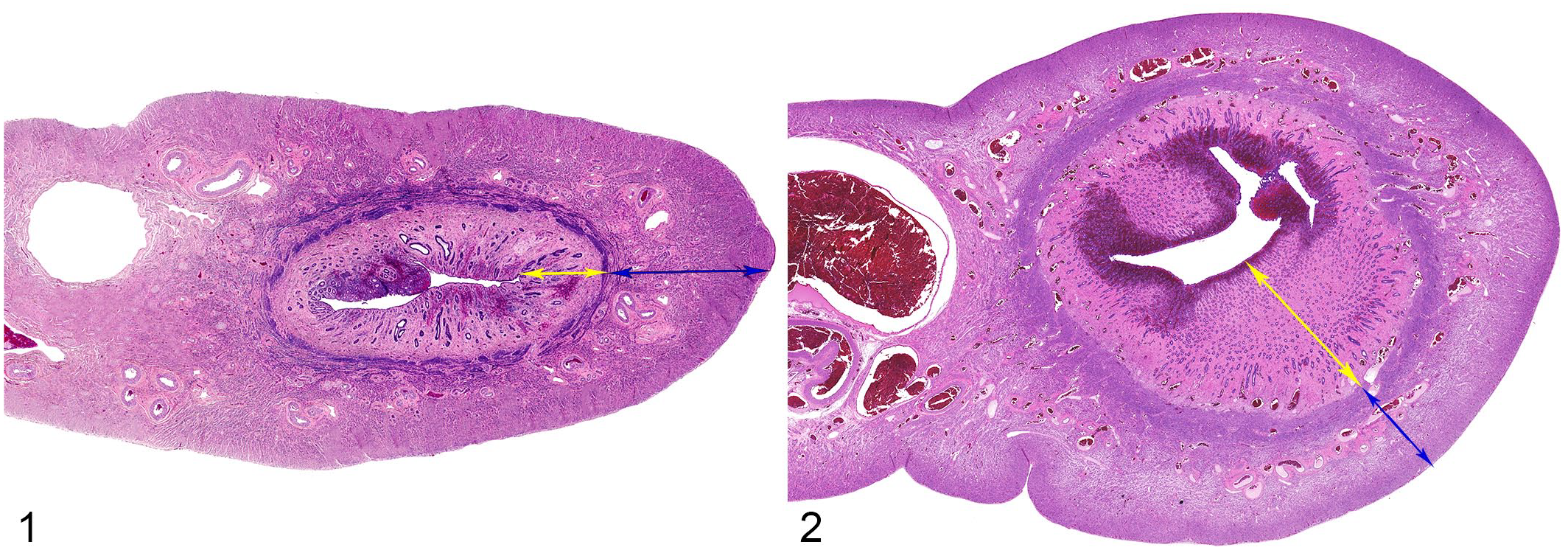
Normal uterus from a tiger (Panthera tigris). Hematoxylin and eosin (HE) (Fig. 1). Low numbers of lymphocytes within the endometrium (yellow arrow) are considered within normal limits for both tigers and lions. 23 The myometrium is denoted by the blue arrow. Normal uterus from a lion (Panthera leo). (Fig. 2, HE).
Hematoxylin and eosin (H&E)-stained slides were obtained from archives and reviewed together by the author (AC) and a board-certified anatomic pathologist (MS). The initial histopathologic findings and diagnoses were confirmed or revised based on current nomenclature. 36 Cases of EH were classified on the basis of the presence of irregular proliferation and arrangement of uterine glands compared to normal physiologic endometrial proliferation. When multiple samples were available from the same animal, the one exhibiting the most severe histologic changes was scored.
Histopathologic Scoring
Histopathologic parameters assessed on each animal were adapted from previous work in veterinary and human medicine and included: (1) percentage of endometrial tissue affected by dilated glands, 30 (2) ratio of endometrial glandular tissue to stroma (degree of glandular crowding), 42 (3) tubular complexity/tortuosity (length [perimeter] of basement membrane about the endometrial glands), 32 (4) presence and degree of adenomyosis,18,39 (5) mean and standard deviation of the shortest nuclear axis (nuclear atypia/pleomorphism, loss of polarity, and coarsening of chromatin) of hyperplastic endometrial cells,6,21,38,42 (6) changes in endometrium thickness, 26 (7) changes in myometrium thickness, (8) degree 10 and (9) depth of mucosal inflammation and (10) degree of luminal inflammation. Each histological parameter was categorized as follows: 0 = within normal variation, 1 = mild, 2 = moderate, 3 = severe (Table 1 and Figs. 3–14). The percentage of endometrium affected (1) was scored based on the most severely affected sample in each case. For the parameters of endometrium thickness and myometrium thickness (6 and 8), scores of 1, 2, or 3 represented values exceeding 1, 2, or 3 standard deviations from the relative mean for each species, respectively. The mean (±SD) values for normal endometrium and myometrium thickness were measured in control animals for both species. Each parameter was scored from 0 to 3 for every individual animal. In addition, edema, hemorrhage, fibrosis, and endometrial necrosis/ulceration were assessed and scored either “1” or “0” based on the presence or absence. A comprehensive score was performed to evaluate the significance of all parameters previously discussed in the literature in addition to changes identified in our study samples that had not been previously described (Supplemental information, Tables 1 and 2). After interspecies and intraspecies statistical analyses of all parameters, only 4 parameters (percentage of endometrium affected, endometrial gland to stroma ratio, glandular complexity/tortuosity, and adenomyosis) were statistically significant in univariate and/or multivariate analysis. Combined, these 4 parameters for each animal yielded a final EH assessment score ranging from 0 to 12.
Grading scheme for histologic parameters in lions (P. leo) and tigers (P. tigris) with endometrial hyperplasia.
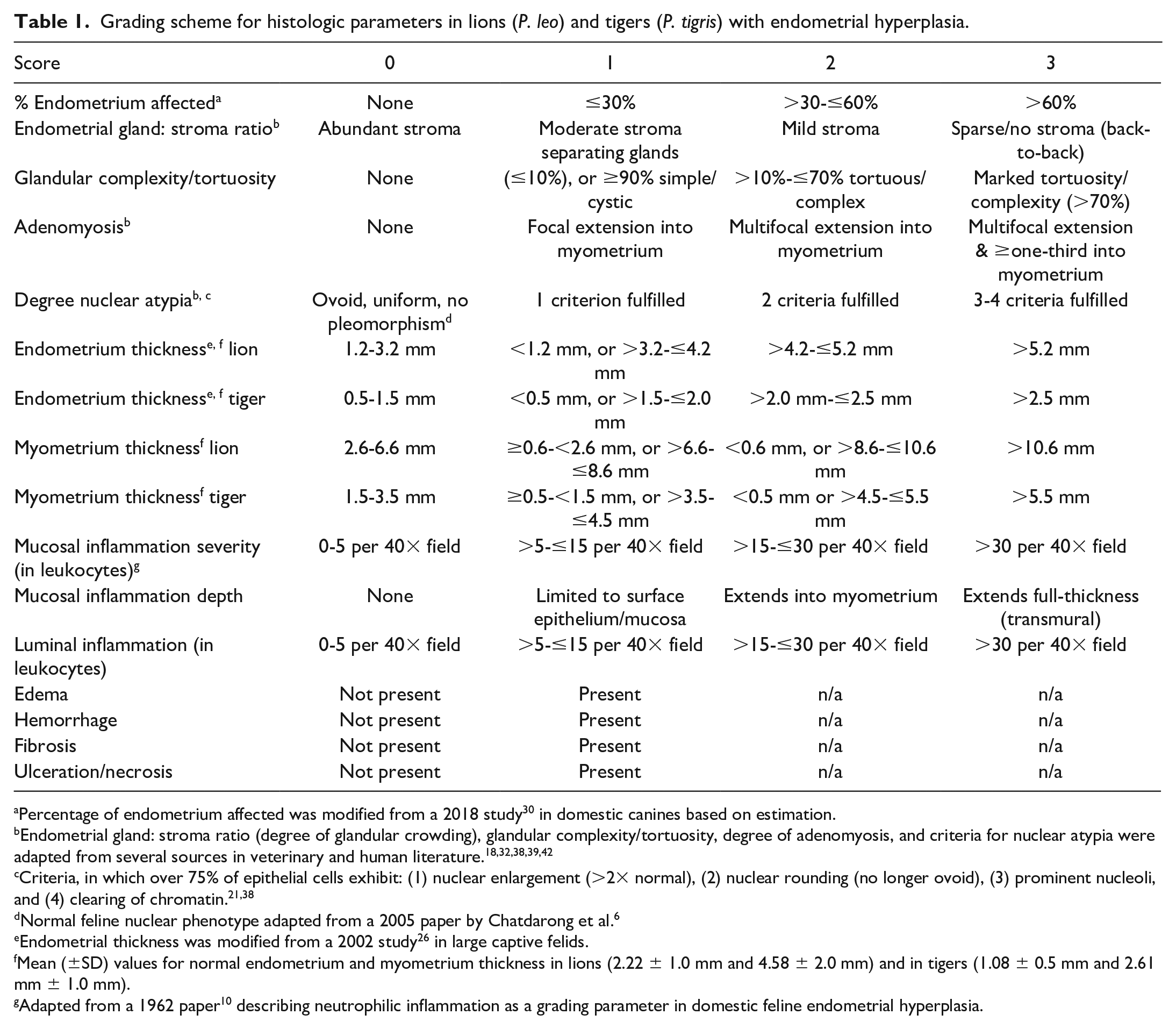
Percentage of endometrium affected was modified from a 2018 study 30 in domestic canines based on estimation.
Endometrial gland: stroma ratio (degree of glandular crowding), glandular complexity/tortuosity, degree of adenomyosis, and criteria for nuclear atypia were adapted from several sources in veterinary and human literature.18,32,38,39,42
Criteria, in which over 75% of epithelial cells exhibit: (1) nuclear enlargement (>2× normal), (2) nuclear rounding (no longer ovoid), (3) prominent nucleoli, and (4) clearing of chromatin.21,38
Normal feline nuclear phenotype adapted from a 2005 paper by Chatdarong et al. 6
Endometrial thickness was modified from a 2002 study 26 in large captive felids.
Mean (±SD) values for normal endometrium and myometrium thickness in lions (2.22 ± 1.0 mm and 4.58 ± 2.0 mm) and in tigers (1.08 ± 0.5 mm and 2.61 mm ± 1.0 mm).
Adapted from a 1962 paper 10 describing neutrophilic inflammation as a grading parameter in domestic feline endometrial hyperplasia.
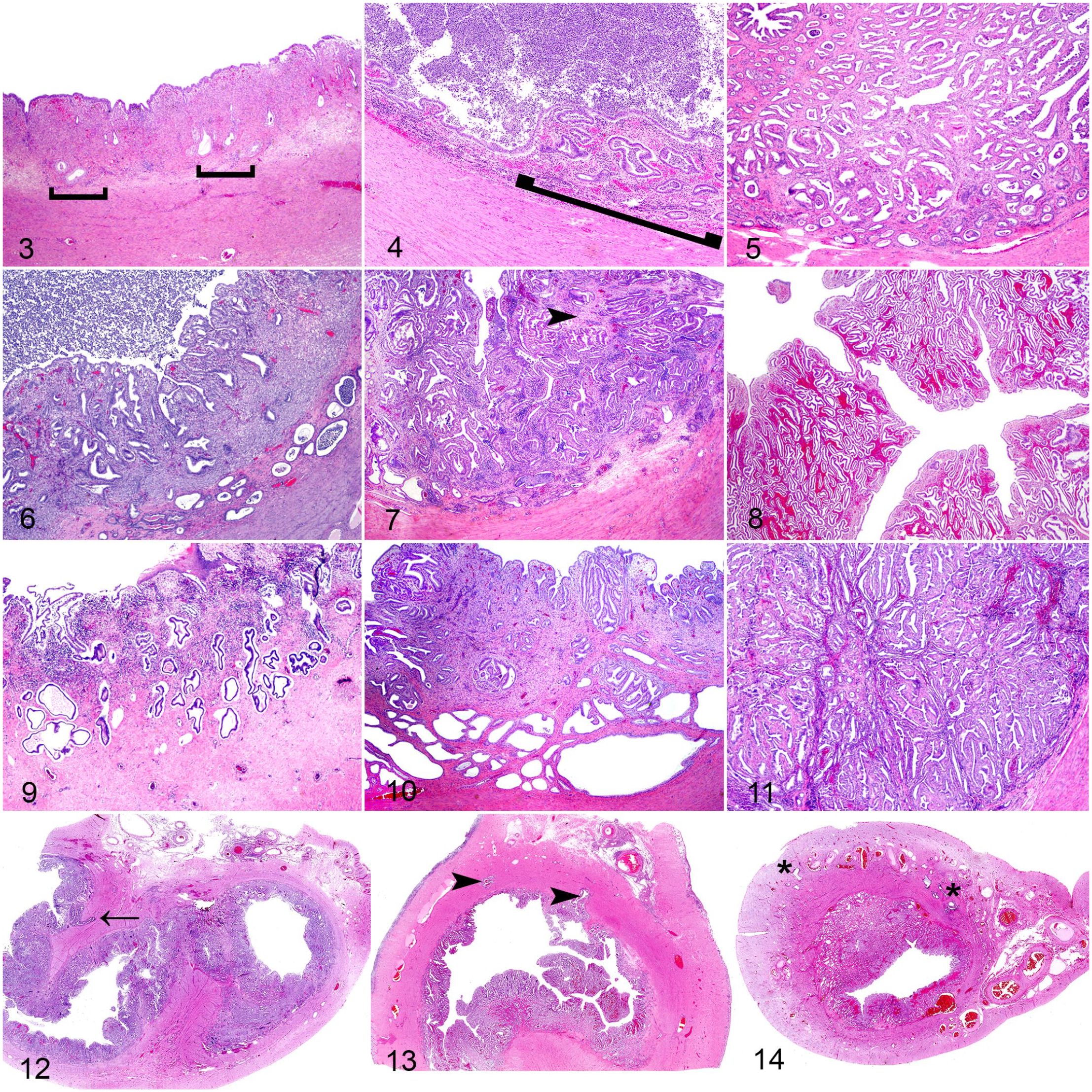
Histopathologic features of endometrial hyperplasia in uteri from lions (Panthera leo) and tigers (Panthera tigris). Hematoxylin and eosin (H&E).
Severity of inflammation/pyometra was quantified on a separate scale which focused solely on the related parameters (mucosal inflammation severity, (9) mucosal inflammation depth, and (10) luminal inflammation). Combined, these 3 parameters for each animal yielded a final inflammation/pyometra assessment score ranging from 0-9.
Clinical Signs Scoring
Reproductive clinical histories were separated into 5 groups on a 0 to 4 scale based on the severity of clinical signs. The absence of any clinical signs or reproductive issues were considered normal (Grade 0). Mild clinical signs included “not doing well” or “not acting like themselves” but continued to eat and drink with or without minimal vaginal discharge (Grade 1). Moderate clinical illness included lethargy, and loss of appetite with or without vaginal discharge (Grade 2). Vaginal discharge with the aforementioned signs plus fever and/or diarrhea and vomiting, were considered marked clinical signs (Grade 3). Vaginal discharge with additional critical illness including obtundation, lateral recumbency, or otherwise fulminant clinical illness were severe clinical signs (Grade 4).
Statistical Analysis
All histologic parameters were evaluated using univariate and multivariate analysis. The 4 most commonly physiologically relevant and diagnostically discriminating features of EH including percentage of endometrium affected, gland to stroma ratio, tubular complexity/tortuosity, and adenomyosis were combined in the grading scheme. Normality for the numeric outcomes was evaluated using Shapiro–Wilk test. The effects of species on nonnormal distributed histologic parameters such as EH scores and clinical signs severity were analyzed using nonparametric Wilcoxon test, while other normally distributed histologic parameters were analyzed using MANOVA. The linear relationships between (1) EH scores and clinical signs scores, (2) EH scores and inflammation/pyometra scores, (3) inflammation/pyometra scores and clinical signs scores, (4) age at pyometra diagnosis and EH scores, and (5) age at pyometra diagnosis and clinical signs scores, were estimated using regression analysis respectively for each species as well as for both species using general linear models controlling for species effect. The effect of species on the binary categorical outcome for occurrence of pyometra was analyzed using logistics regression. A P value <.05 was considered significant. Analyses were conducted in SAS9.4 TS1M7 for Windows 64 (SAS institute Inc., Cary, NC).
Results
During the study period, the authors’ institution histologically evaluated 27 lion and 85 tiger uterine samples. Fourteen lions and 24 tigers met the inclusion criteria. No animals in this study had been contracepted or bred while housed at their current locations. All uterine samples were attained either at the time of ovariohysterectomy (OHE) procedure or necropsy. There were no ovarian-sparing procedures in any animal. The ages of 2 lions and 3 tigers were missing from medical records. Known ages of affected lions and tigers were 6 to 15 years of age (average of 11.2 years), and 6 to 17 years of age (average of 12.6 years), respectively. The average ages for the control lions and tigers were 12.4 and 16.4 years, respectively.
Three lions (21%) and 4 tigers (17%) had EH without inflammatory disease. Eleven lions (79%) and 18 tigers (75%) had concurrent EH and pyometra. There were no lions and 2 tigers (8%) that had evidence of pyometra without EH; these animals were not included in the EH grading scheme statistics. None exhibited adenomyosis in the absence of EH.
The average endometrium thickness of healthy control lions is twice that of healthy control tigers (2.22 mm vs. 1.08 mm). The myometrium of healthy control lions is nearly 1.8 times as thick as in healthy control tigers (4.58 mm vs. 2.61 mm; Figs. 1 and 2). In response to long-term uterine disease, tigers have a more significant absolute increase (in microns) in endometrial thickness (P = .0097) and more significant myometrial hypertrophy (P = .0002) compared to the respective relative normal baseline measurements and established standard deviations for that species. Compared to 36% of lions, 91% of tigers had endometrial thickness and/or myometrial thickness scores of 3 (29 and 7% respectively in lions and 73 and 77%, respectively in tigers).
The average EH score for lions and tigers, out of a maximum of 12 possible points, was 8.14 and 5.82, respectively. As univariate analytes, percentage of endometrium affected (P = .0385), endometrial gland to stroma ratio (P = .0008), and adenomyosis (P = .0463) were statistically more severe in lions compared to tigers. All animals with adenomyosis (43% of lions and 18% of tigers) had concurrent EH; adenomyosis never occurred without the presence of EH. Glandular complexity was not statistically different between these species (P = .3254). Fourteen percent of lions and 36% of tigers had grade 1 glandular complexity/tortuosity, indicating the minority of the large captive felids in this study had simple, cystic glands, while most, 86% of lions and 64% of tigers, had noncystic, complex, serpiginous, or tortuous hyperplastic glands. In combination with the previous 3 features, glandular complexity strengthened the overall EH grading scheme in multivariate analysis. Combined, these 4 parameters confirmed lions had significantly more severe EH (P = .0068) compared to tigers with EH. Lions had more endometrial or myometrial fibrosis and more severe luminal inflammation compared to tigers (P = .0361 and P = .0385, respectively). Other histological features were inconsistently identified, and/or not significantly different between lions and tigers (Supplemental information, Tables 1 and 2).
Endometrial hyperplasia grading score was not significantly correlated to pyometra-related clinical signs score in either lions or tigers (P = .9424 and P = .2822, respectively). There was no significant difference between lions and tigers and the age at initial pyometra (P = .2030). There was also no correlation at the age at initial pyometra diagnosis, and the severity of clinical signs in either lions or tigers (P = .3705 and P = .9948, respectively). There was a correlation between the age at initial pyometra diagnosis and EH grading score when controlling for species and in lions (P = .0168 and P = .0370, respectively), but not in tigers (P = .177). There was not a significant correlation between the severity of clinical signs and the presence of pyometra in lions (P = .1144) but a positive correlation existed in tigers (P = .0026).
In animals with pyometra, vaginal discharge (grade 1 clinical signs score) was noted as the only pyometra-related clinical sign in 2 lions (18%) and 13 tigers (65%). Severe clinical signs (grades 3 or 4) were reported in 3 lions (27%) and 4 tigers (20%). The average clinical scores were 1.71 and 1.38 for lions and tigers, respectively. Comparing lions and tigers, there was no statistical difference in the severity of pyometra-related clinical signs (P = .1986) or in the severity of inflammation/pyometra (P = .2909). There was no significant correlation between inflammation/pyometra score and EH score in either lions or tigers (P = .1245 and P = .0955, respectively). However, when controlling for species, EH score significantly correlated with the severity of inflammation/pyometra (P = .0203). There was a positive correlation between inflammation/pyometra score and clinical signs severity score in lions and when controlling species (P = .0370 and P = .0168, respectively), but there was no correlation between these values in tigers (P = .1770). The depth of uterine inflammation did not correlate to EH score in lions (P = .0636), but was significant in tigers (P = .0243) and when controlling for species (P = .0029). Twenty-nine percent of lions and 27% of tigers had inflammatory infiltrate restricted to the surface epithelium while the remaining animals had inflammation that extended into the endometrial stroma or into the myometrium. Mucosal inflammation severity and luminal inflammation severity were not significantly different between the 2 species or while controlling species (data not shown).
Of the study population, 11 lions (79%) and 100% of tigers had ovaries available for histologic examination. Of these, 82% of lions and 73% of tigers had at least one corpus luteum.
Discussion
Similar to that previously described by Munson et al, 26 the authors prefer the term “endometrial hyperplasia” (EH) compared to “cystic endometrial hyperplasia” (CEH) for describing these uterine lesions in lions and tigers. Although “cystic” and “noncystic” EH are defined as 2 separate entities/diagnoses in domestic animals, as seen in this study, these terms more accurately describe a continuous spectrum of hyperplastic endometrial changes. This is similar to changes characterized in women.32,36 While a minority of animals in this study had simple cystic or minimal (grade 1) tubular complexity, most, 86% of lions and 64% of tigers, had segmental, if not entirely, noncystic or tortuous (grade 2 or 3) tubular complexity, or an overall more adenomatous proliferation, compared to what has been described in canids. 36 With the habitual and ubiquitous use of the clinical term “cystic endometrial hyperplasia,” pathologists and clinicians may run the risk of failing to identify diseased uteri that may lack grossly obvious cysts, particularly in cases where gross examination alone is used to identify suspect cases of EH. In this study, only 14% of lions and 36% of tigers had simple cystic (grade 1) tubular complexity. We support veterinary pathologists should adopt the term and diagnosis of “endometrial hyperplasia” for all species, and then further subclassify EH as either “simple cystic,” “noncystic,” or “complex.”
To achieve subclassification, we propose using the grading scheme herein with the 4 parameters [percentage of endometrium affected, endometrial gland to stroma ratio, glandular complexity/tortuosity, and adenomyosis] to grade these features on a 0 to 12 score. A score of 0 to 4 points would denote “simple cystic” EH, 5 to 8 points denotes “noncystic” EH, and 9 to 12 points denotes “complex” EH. Although endometria that are complex may occasionally have cystic glands, these tend to compose less than 10% of the endometrium and may not be readily apparent grossly. Given the limitations of this retrospective analysis, percentage of endometrium affected was estimated using the single most affected section of uterus in each case. Ideally, the authors recommend 2 representative sections from each uterine horn (4 sections total) be taken for histopathological review, encompassing both grossly normal and grossly abnormal portions of each horn, as applicable, and evaluate percentage of endometrium affected estimated over all 4 sections.
Differences in endometrium and myometrium thickness in both healthy and diseased lions and tigers further complicate gross interpretation of EH in these animals. Myometrial hypertrophy paired with EH and variably extensive inflammation in animals with or without accompanying pyometra is unsurprisingly quite difficult to accurately interpret grossly, thus routine histologic evaluation of the uterus is recommended in every case in which the uterus is removed to allow for more full characterization of the spectrum of disease in these animals.
Adenomyosis, or benign invasion of endometrial glands and/or stroma into the myometrium, is uncommon in domestic animals, typically reported in geriatric patients, and usually found as an incidental lesion with other uterine pathologies, such as EH, pyometra, endometritis, or, less commonly with uterine neoplasia.7,23,26,36,39,40 Adenomyosis is characterized as either focal to denote a single or nodular collection of endometrial glands extending into the myometrium, or diffuse to denote multifocal areas throughout the uterus. Adenomyosis was identified in 43% of lions in our study compared to 18% of tigers, with an average age of 10.5 and 12.3 years, respectively. All animals with adenomyosis had concurrent EH. As lions had significantly more severe EH compared to tigers (P = .0068), the increased prevalence of adenomyosis in lions compared to tigers suggests an association between adenomyosis and the presence and severity of EH rather than differences between the species, and supports the inclusion of this parameter within the grading scheme. In canines, it is proposed that invasion of endometrial glands into the myometrium may weaken the uterine wall thus predisposing to rupture. 35 The uterus was ruptured in 2 lions (14%) and no tigers in this study; however, neither lion had evidence of adenomyosis at the site of rupture (data not shown).
Concurrent EH and pyometra was identified in 79% of lions and in 75% of tigers. Pyometra occurring in concert with EH, identified as the CEH-Pyo complex, is commonly reported in domestic dogs and is less well established in domestic cats. It seems quite natural that hyperplastic endometrial glands with primed progesterone receptors producing a hypersecretory state may result in pyometra, mucometra, or hydrometra, thus the commonality of the complex. Conversely, uterine inflammation may also cause the development of EH. 44 While the frequency of concurrence of these 2 conditions in this study is intriguing to consider as evidence of “CEH-Pyo complex” in lions and tigers, recent literature reiterates that similar pathogenesis does not mean these conditions are always related, and suggest EH and pyometra/uterine inflammation should continue to be regarded as separate entities.8,33 In our study population, 3 (21%) lions and 4 (17%) tigers had EH without pyometra while no lions and only 2 (8%) tigers had histologically confirmed pyometra without EH (data not shown). These cases further support EH and pyometra as separate, but pathologically related entities in lions and tigers.
Pyometra is uncommon in domestic cats, with a prevalence of approximately 2.2% reported in some European countries where animals more commonly remain sexually intact into adulthood.2,13,15 Of the total 27 intact female lions and 85 intact female tigers that were evaluated histologically at the authors’ institution during the study period, the prevalence of pyometra in both lions and tigers was much greater than for domestic cats at 59.3% and 28.2%, respectively (data not shown). The prevalence of pyometra at the authors’ institution was 2.1× higher in lions compared to tigers; however, for those animals who met inclusion criteria and were evaluated as part of the study, the degree of mucosal inflammation, depth of inflammation, and degree of the luminal inflammation were not statistically more severe in lions. In domestic cats, pyometra is proposed to be caused by induced ovulation, disruption in the estrous cycle, and/or variance in seasonality, but other suggested causes run the gamut from endocrine disease to neoplasia. 14 There was no evidence of functional tumors or exogenous progesterone exposure in any lion or tiger included in this study.
Unique to nondomestic felids, the risk of developing EH has been correlated with long-term exposure to progesterone contraceptive therapy.4,10,26 However, despite the decreased use of progesterone-based contraceptives since the early 2000s, uterine and mammary diseases, including neoplasia, have continued at similarly high rates arguing that these progesterone implants could be coincidental to the development of these conditions in most cases, although additional studies are warranted for reproducibility. 19 For the animals in this study, it is not known if they were treated with contraceptives prior to their acquisition, but this is considered unlikely as progestin contraceptive implants were/are largely unavailable for privately owned animals (Edward Ramsay, personal communication, 2021).
Like the domestic dog, we highly suspect EH and pyometra in the lions and tigers of this study are directly related to hormonal effects of the estrous cycle. In some European countries, where 90% of domestic dogs are sexually intact well into adulthood, the prevalence of pyometra across all breeds approaches 25%, closely matching the prevalence of 28% of tigers evaluated at the authors’ institution. 15 For particularly at-risk canine breeds, the prevalence is closer to 50%, approaching the 59% of lions in this study. The cause of the markedly higher prevalence of pyometra in lions and tigers is speculative. Recent work has shown many of the tigers included in the present report, similar to domestic canids (and to a lesser degree, domestic felids), spontaneously ovulate, likely increasing the risk for pyometra in nonbreeding female tigers.5,12,41 Most of animals in this study (82% of lions and 73% of tigers) had at least one corpus luteum at the time of OHE or necropsy, suggesting a possible common source of endogenous progesterone driving EH in this population. Other sources of endogenous progestin cannot be entirely ruled out, namely induced ovulation from co-housed males of the same species; however, this is considered unlikely to have a role in these cases as none of the study animals were housed with or had direct contact with males (either castrated, vasectomized, or intact) since the time of transfer or residency at the zoo or rescue facility. In both species, documented housing history or access to males prior to acquisition is largely unknown. The main caretakers of these animals (DC and ER) note that 5 lions (36%) and no tigers had a known previous history of being housed with a male for an undetermined length of time prior to acquisition by the zoo or sanctuary. Although differences in ovulation rates between these species is unknown, rates would likely increase with access to males or endogenous progestin thus leading to predisposition of developing uterine changes including EH and/or pyometra.
The lack of a significant difference between the age at pyometra diagnosis between lions and tigers suggests that these species may be diagnosed with pyometra at approximately the same age. Lions and tigers are considered middle-aged adults at 5 to 15 years old while those >15 years of age are considered geriatric (Edward Ramsay, personal communication, 2021). When combining all animals in this study, the average age at the diagnosis of pyometra was 11.3 years, suggesting pyometra may be more common in middle-aged lions and tigers. The most common clinical signs of pyometra in domestic felids include weight loss, dehydration, polyuria/polydipsia, tachycardia, tachypnea, pallor, hyperemic mucous membranes, and unkempt appearance. 14 In the study population, there was a range of pyometra-associated clinical signs, from minor vaginal discharge to obtundation associated with peritonitis and sepsis, similar to those described in domestic cats. 14 In up to 40% of affected domestic cats, vaginal discharge may be absent due to self-grooming. 17 This may have occurred in our study population as well, as recognizing early or subtle clinical disease in solitary, large captive carnivores can be challenging. In concordance with the observations of the care staff, the mean score value in clinical sign severity (out of 4 points total) was greater for lions (1.71) than tigers (1.38); however, these values were not significantly different within our population. This may be because of the small number of animals for which we have carefully documented clinical signs, or may be due to the amnestic nature of the review, requiring some long-distance recall of changes that had happened years prior. Interestingly, in tigers, the severity of clinical disease statistically correlated with the presence of pyometra, indicating a clinically ill tiger is more likely to have pyometra. Moving forward, it would be ideal to more precisely document these clinical and clinicopathologic findings in animals with pyometra to assist practicing large cat veterinarians to more carefully delineate the differences in clinical presentation.
A Primary Limitation in Our Study Is Sample Size
Despite the relatively large sample size for a nondomestic animal pathologic study, a larger sample size would strengthen the data. The retrospective nature of this study created other limitations, as the extent of clinical histories and other patient information in some cases was limited. Furthermore, subtle clinical signs may not have been appreciated or remembered by caretakers or veterinary support staff when interviewed months to years after caring for said animal. In addition, the number of uterine tissue sections examined was variable; in some cases, only one sample of uterus was available for evaluation, and the possibility that the single sample did not represent the full spectrum of disease is of particular concern, given the segmental variation in disease severity and the challenges of grossly interpreting EH changes.
The EH grading scheme proposed here improves on former schemes by evaluating a comprehensive battery of EH features described in both veterinary species and humans and identifying 4 features that are consistently correlated to the clinical and histological differences between these 2 large cat species. Previous literature on EH in zoological felids has tended to group these large cats together 26 which has obscured the finer differences that may have a role in the clinical management of these animals. It is only through this more detailed analysis that we can better understand the differences and commonalities of this significant reproductive disease in these 2 similar, but certainly not identical felid species. It is our hope that this will be a stepping stone to entice a more critical evaluation and understanding of species differences among zoologically housed carnivores.
Finally, this study further underscores the importance of better assessing the degree or grade of EH, which may assist clinicians working with these patients, and has implications for long-term breeding management. Although uterine biopsies are not routinely performed in lions or tigers at the authors’ institution without an OHE procedure, this proposed EH grading scheme may be useful in species where uterine biopsies are more regularly performed for the use of prognosticating fertility in breeding programs. A large-scale analysis of lions, tigers, and other captive felids to better correlate histological findings of EH and the correlation to, and progressive development of pyometra would be of value. In nondomestic felids, we propose pathologists should grade all EH based on percentage of endometrium affected, endometrial gland to stroma ratio, glandular complexity/tortuosity, and adenomyosis generating the data that will allow for better characterization and correlation of the clinical and histological aspects of these disease processes.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221109094 – Supplemental material for Endometrial hyperplasia and pyometra in captive lions (Panthera leo) and tigers (Panthera tigris)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221109094 for Endometrial hyperplasia and pyometra in captive lions (Panthera leo) and tigers (Panthera tigris) by Ariel K. Carlson, Edward C. Ramsay, Xiaocun Sun, Deborah Chaffins and Mee-Ja M. Sula in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Ms. M. L. Haven and caregivers at Tiger Haven and Dr. Andrew Cushing, MRCVS, DACZM for their contributions to the clinical history for the animals. Thank you to Messrs. Nickolus Stahlman and Houston Dale for their necropsy assistance and the University of Tennessee histotechnician team. They would also like to thank the University of Tennessee College of Veterinary Medicine anatomic pathologists and anatomic pathology residents who collected samples and provided feedback in shaping this manuscript.
Supplemental Material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
