Abstract
In a survey of gynecologic lesions in female zoo felids conducted to determine if the widely used progestin contraceptive melengestrol acetate (MGA) had adverse effects, numerous leiomyomas and leiomyosarcomas were detected. This current study aimed to characterize the morphologic features of these tumors, determine their prevalence, and assess if MGA was a risk factor for their genesis. Genital tracts from 219 zoo felids representing 23 species were evaluated, and leiomyomas were detected in 24% of the felids. Leiomyomas were often multiple and occurred in the myometrium, ovary, or adjacent broad ligament. The risk of developing leiomyomas increased with age, but MGA treatment or parity had no effect. Five other felids had leiomyosarcomas. Leiomyosarcomas were distinguished from poorly demarcated leiomyomas by the presence of local invasion, metastasis, and cellular atypia, but necrosis and mitotic rate were not distinguishing criteria. Four of five felids with leiomyosarcomas had been treated with MGA. These results indicate that leiomyomas are common spontaneous lesions in the genital tracts of zoo felids and their genesis is not linked to MGA exposure. Whether progression to malignancy is promoted by MGA warrants further investigation.
Genital leiomyomas are common in domestic cats and comprise 80% of uterine tumors in dogs and up to 50% of female genital tumors in cattle, sheep, and swine.1,9,13,22,35,36 Leiomyomas have been reported in five lions and a black leopard,3,5,22 but no comprehensive survey of leiomyomas in zoo felids has been conducted. Uterine leiomyosarcomas are rare in all species, but seem to be more common in cats than in other species.13,15
The genesis of leiomyosarcomas is poorly understood, but the proposal has been made that they arise from either leiomyomas or de novo from transformed myometrial cells.32 Gonadal steroids are thought to promote the growth of leiomyomas, because ovariectomy often results in regression of these tumors, and leiomyomas only rarely occur in ovariectomized dogs.13,16 In women, leiomyoma growth increases during pregnancy, and tumor regression occurs after menopause.12 Treatment with the antiprogestin RU 486,10,26,28 GnRH agonists, or selective estrogen receptor modulators such as tamoxifen or raloxifene also arrests or reverses leiomyoma growth.12,28,33 In rats, progesterone treatment increases the number of proliferating cells in leiomyomas and increases overall tumor size.39
Many felids in zoos are treated with progestin contraceptives to prevent unplanned pregnancies. The most commonly used progestin, melengestrol acetate (MGA), is administered in a silastic implant that is placed subcutaneously or intramuscularly, thereby delivering MGA continuously for up to 2 years.31 During a survey to determine if MGA contraceptives adversely affected reproductive health in zoo felids, many felids were noted to have smooth muscle neoplasms. Because leiomyomas have been suspected to impede fertility in horses, dogs, and primates,4,13,17,20,30,37 and because maintaining fertility in zoo felids is essential for captive breeding programs, it was important to determine if MGA treatment was linked to the development of smooth muscle neoplasms. Morbidity and mortality from leiomyosarcomas also was of concern in these valuable species. Therefore, this study was conducted to characterize the morphologic features of these tumors, to determine the prevalence in the population, and to assess whether exposure to MGA was a risk factor for their genesis.
Materials and Methods
Genital tracts were requested from female felids that died or were ovariohysterectomized in United States zoological parks. Genital tracts received between 1988 and 1995 from felids whose age and contraceptive history were known (n = 219) were included in the study. Study animals were housed at 1 of 82 zoological parks. We grouped species into big cats (Panthera species, including tigers, lions, jaguars, snow leopards, and leopards; n = 122) and all other cats (Felis, Neofelis, and Acinonyx species, including all small cats, clouded leopards, cheetahs, and cougars; n = 97) based on current knowledge of phylogenetic relationships.24 The median age of all study animals was 143 months (range 2–24 years); for big cats the median age was 164 months (range 25–288 months), and for the other cat group the median age was 120 months (range 24–276 months). Ninety-eight felids were treated with MGA for at least 6 months and 121 had no history of exposure to progestins or other contraceptives (subsequently referred to as controls). Ovaries from 174 of these felids (79 MGA-treated and 95 controls) were also examined for structures that were potential sources of endogenous steroids, whereas ovaries from the other 45 reproductive tracts had been used for in vitro fertilization and embryo cryopreservation studies and were not available for histologic examination.
Reproductive tracts were fixed in 10% neutral buffered formalin at the contributing institution and then shipped to the study site. Two transverse sections of each uterine horn (taken at standardized areas) and sections of grossly visible lesions were taken for histopathologic analysis. A longitudinal section of ovaries also was sampled. Tumors from the vagina were excluded from the study, because this region of the genital tract was submitted from only a small portion of the population. Tissues were embedded in paraffin, sectioned at 7μm, and stained with hematoxylin and eosin. All sections were examined histopathologically by two authors (L. M. Chassy and L. Munson) without knowledge of contraceptive or reproductive history.
The number of leiomyomas and distribution within the myometrium were determined by microscopy. Information on the gross location of 29 tumors within the reproductive tract and gross measurements of 40 leiomyomas from 23 cases also were available for the study.
Because clinical outcome could differ by the site of tumors in the genital tract, the location of leiomyomas was recorded to determine if the tumors occurred more commonly in different regions of the myometrium. The myometrial regions were defined as subendometrial (arising or extending from the circumferential muscle layers adjacent to the endometrium), intramyometrial (within the circumferential or longitudinal layers), subserosal (arising in or extending from the longitudinal layers adjacent to the serosa), or in the broad ligament. The following microscopic parameters were evaluated: compression of surrounding tissues, clarity of tumor borders, arrangement and density of myocytes, mitotic index, and cellular atypia. Cell density was evaluated in each tumor at low magnification (100 × field) and recorded as high, medium, or low cellularity (Figs. 1–3). Tumors with variation in cell density were graded by the highest density. Cell necrosis, inflammatory cell infiltrates, hemorrhage, mineralization, and hyaline changes also were assessed.
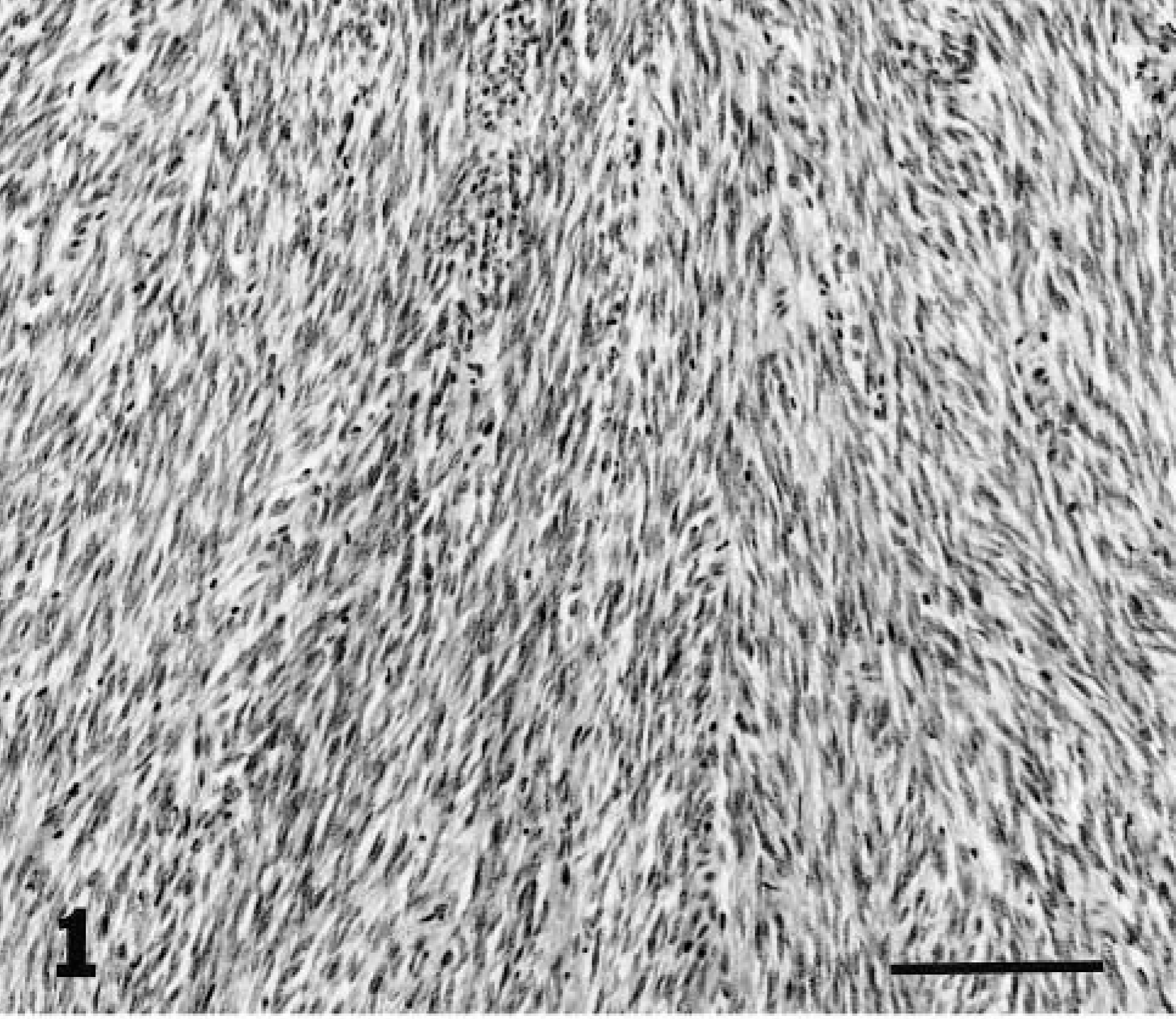
Myometrium; lion. Leiomyoma with high cell density. HE. Bar = 30 μm.
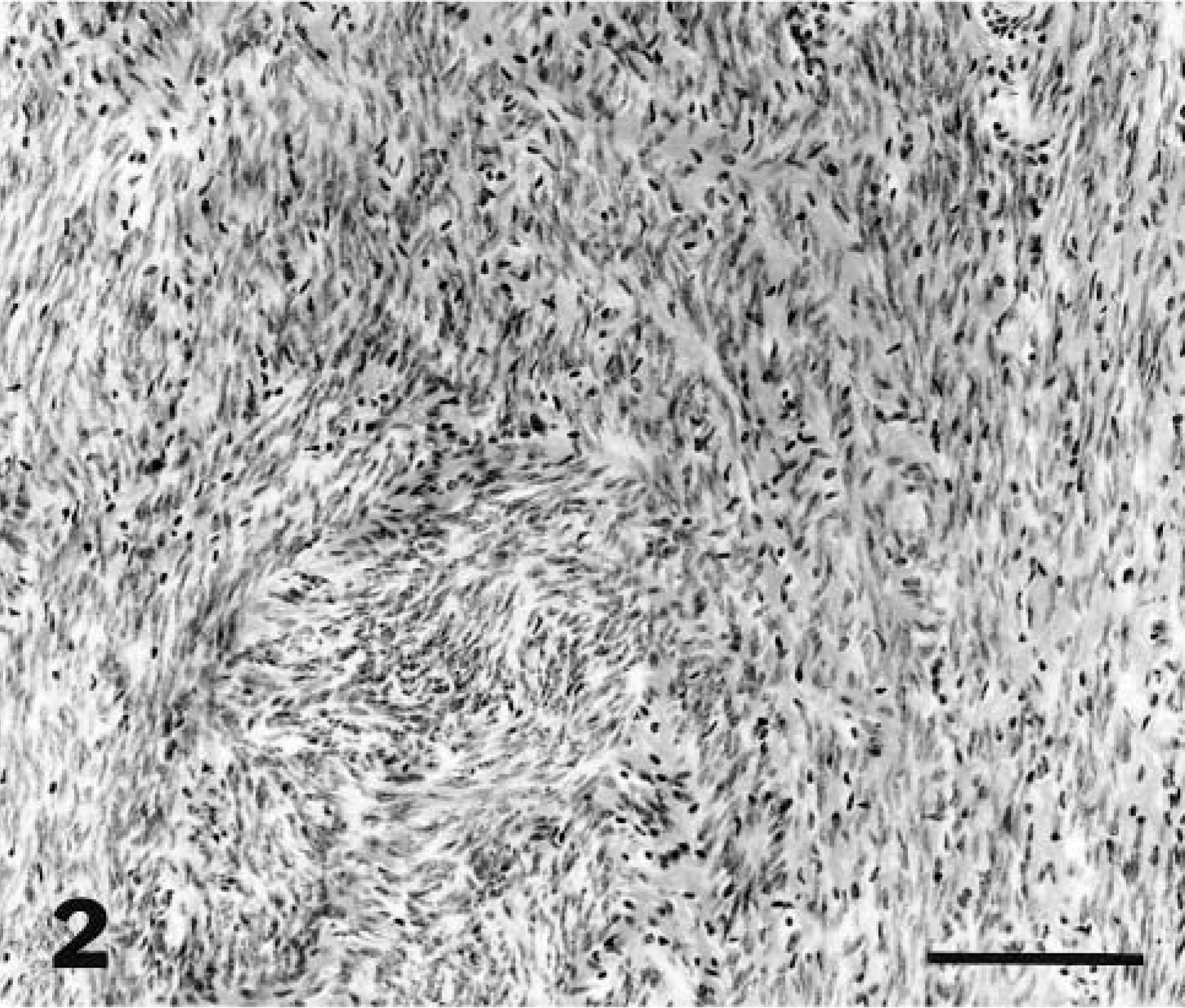
Myometrium; lion. Leiomyoma with medium cell density. HE. Bar = 30 μm.
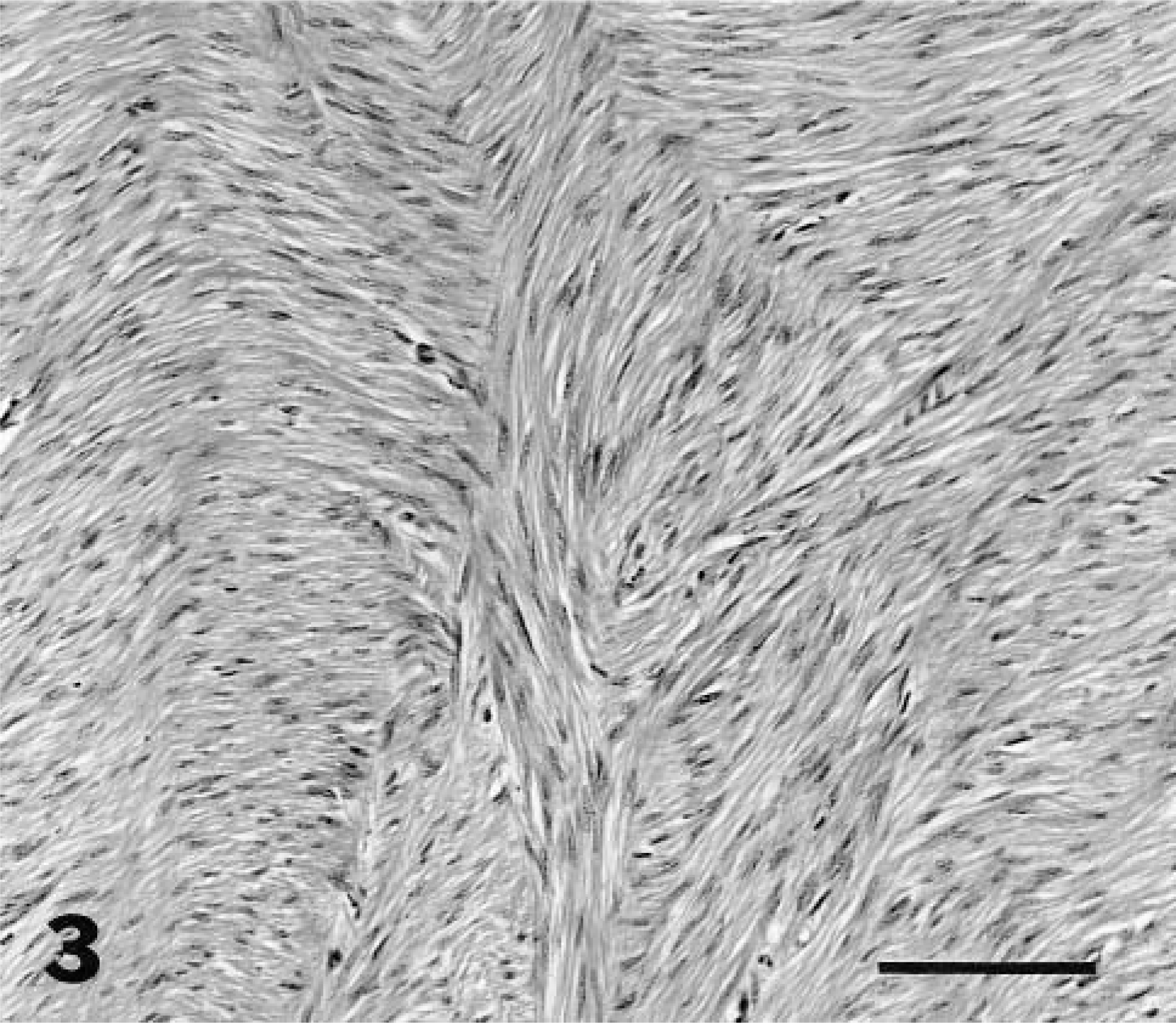
Myometrium; lion. Leiomyoma with low cell density. HE. Bar = 30 μm.
Leiomyosarcomas were distinguished from poorly demarcated leiomyomas by the presence of invasive growth, high mitotic index, or cellular atypia.14 Cells were classified as atypical if they had nuclear hyperchromasia, multinucleation, pleomorphism, anisokaryosis, increased nuclear size, variation in chromatin clumping pattern, and prominent or multiple nucleoli.25,27 Felids with leiomyosarcomas were excluded from statistics on leiomyomas because the extent of tumor invasion precluded indentifying benign masses in the same genital tract.
In all tumors, the number of mitotic figures in 40 high-magnification fields (HPF; 400 × area of field; diameter = 0.48 mm; area = 18 mm2) was recorded and then converted to the number of mitoses per 10 HPF to compare mitotic indices with those used to differentiate leiomyomas from leiomyosarcomas in women.25,27 A low mitotic index was less than one mitosis per 10 HPF; a medium mitotic index was one to four mitoses per 10 HPF; and a high mitotic index was five or more mitoses per 10 HPF.27
The association between MGA treatment and the occurrence of leiomyomas was analyzed by logistic regression11 with age and parity (nulliparous or parous) as covariates. Species was not included as a covariate because lesion character did not differ among the seven species with lesions and adequate numbers of all species were not available for statistical tests. Because the smooth muscle tumors occurred predominantly in big cats, we included cat group (big versus other) in the logistic model. For the subset of felids with ovary data (n = 140), we also determined the effects of concurrent tertiary follicles on leiomyoma development after accounting for MGA treatment, age, and parity.27
Because mitotic index is considered a measure of tumor aggression, Mann–Whitney tests were used to determine if the number of mitoses differed between tumors from control and MGA-treated felids and between tumors with or without atypia. An analysis of variance was used to compare mean mitotic indices among tumors with high, medium, and low cell densities. t-Tests were used to compare diameters of leiomyomas between MGA treated and control animals. Significance was set at P < 0.05 for all analyses.
Results
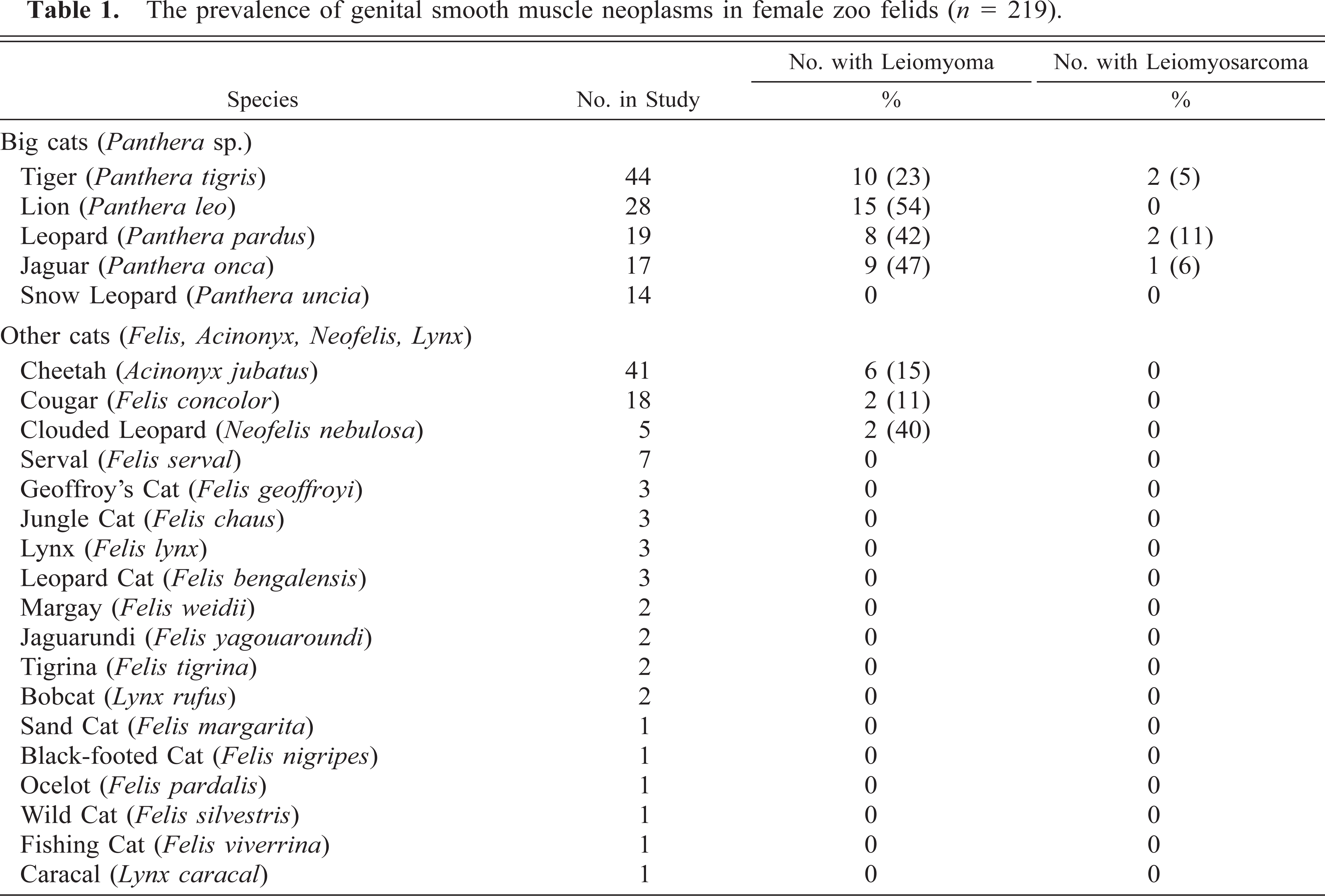
The prevalences of smooth muscle neoplasms in the species included in the study are listed in Table 1. Of the 219 animals in the study, 52 felids (24%) had one or more leiomyomas and 5 felids (2.2%) had leiomyosarcoma. Forty-two of those 52 animals were big cats, including all animals with leiomyosarcomas. Leiomyomas also occurred in cheetahs (n = 6), cougars (n = 2), and clouded leopards (n = 2). Except for cougars (Felis concolor), no felids from the genuses Felis or Lynx had smooth muscle tumors, although these genera represented 24% of the study population. The age distribution of felids with smooth muscle tumors is displayed in Fig. 4. The median age of felids in which leiomyomas were detected was 15 years (range 38–288 months), and the median age of felids with leiomyosarcomas was 16 years (range 116–248 months).
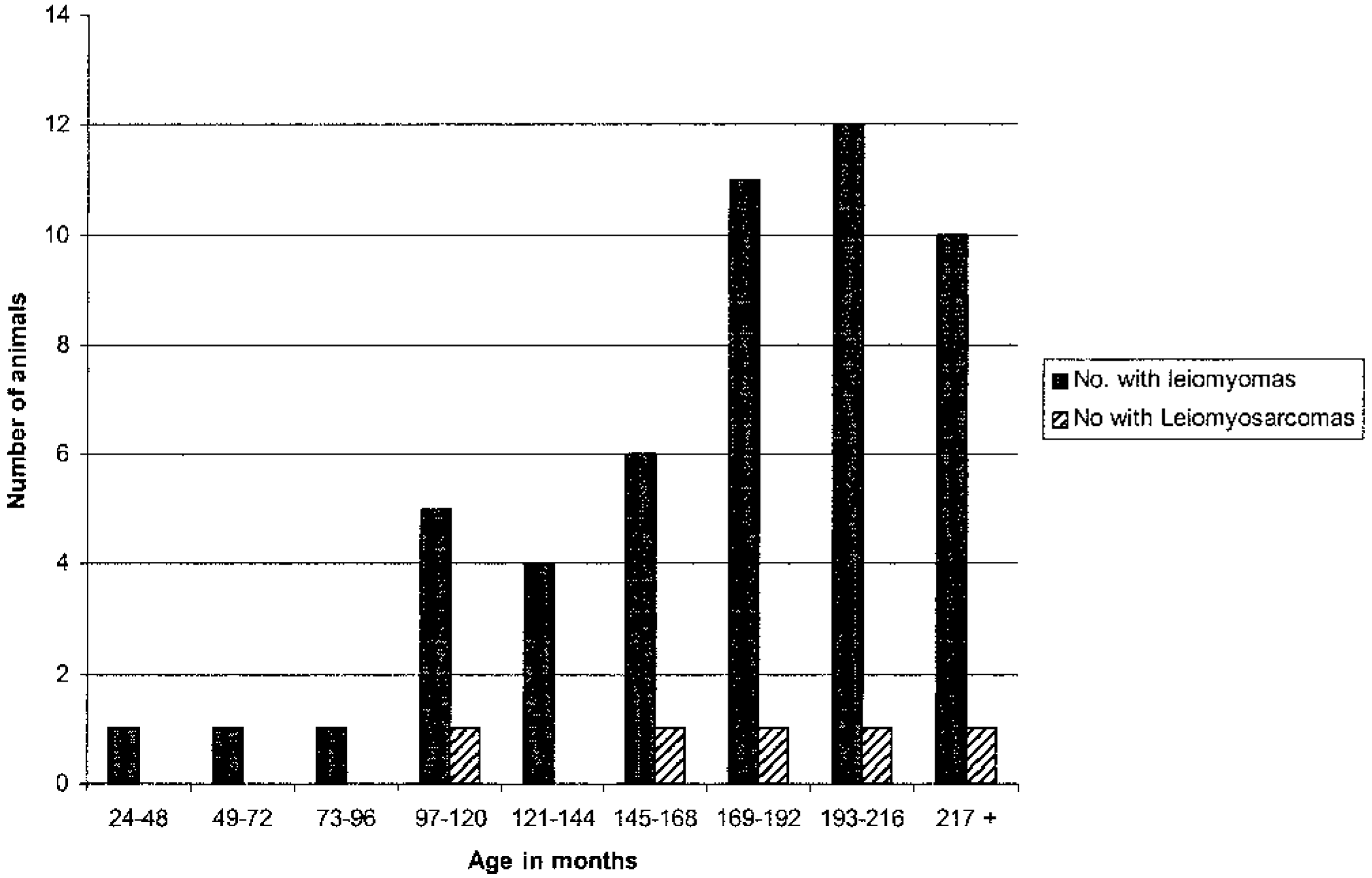
Age distribution of zoo felids with leiomyomas and leiomyosarcomas.
The prevalence of genital smooth muscle neoplasms in female zoo felids (n = 219).
Leiomyomas
A total of 111 leiomyomas in 47 felids was available to evaluate in detail. Seventeen of these 47 felids (36.2%) had solitary leiomyomas, whereas 30 felids (63.8%) had multiple leiomyomas. Of the 111 leiomyomas, 72 (64.8%) were present in the uterus, 36 (32.4%) were present in the ovarian medulla, and 3 (2.7%) were present in the broad ligament. Six leiomyomas were pedunculated, with three extending from the serosal surface of the uterus and two projecting into the uterine lumen. Forty of the 72 (55.6%) intrauterine leiomyomas were intramyometrial, 15 (20.8%) were subserosal, and 9 (11.1%) were subendometrial. The specific location of the remaining eight myometrial leiomyomas could not be determined from the histologic sections available.
The size of 40 leiomyomas for which gross measurements were available ranged from 0.1 to 16 cm in diameter. The mean diameter of leiomyomas in MGA-treated animals was 1.9 ± 0.53 cm (SEM) (range 0.1–16 cm) and in control animals was 2.9 ± 0.91 cm (range 0.1–13 cm; P = 0.15).
Sixty-seven leiomyomas (60.3%) had well-defined borders, 21 (18.9%) had borders that blended with the adjacent normal smooth muscle, and 19 (17.1%) had margins that varied from well defined to poorly defined in different regions. Tumor margins could not be determined in four large tumors (3.6%) because complete margins were not included in the sections sampled. Fifteen of 36 leiomyomas in the ovarian stalk (41.6%) were multilobulated.
All leiomyomas were composed of variably sized interlacing fascicles of well-differentiated smooth muscle cells. In seven leiomyomas, these fascicles were separated by dense fibrous connective tissue (fibroleiomyomas). Sixty of 111 leiomyomas (54.1%) had low cellular density, 39 leiomyomas (35.1%) had moderate cellularity, and 12 leiomyomas (10.8%) were highly cellular. Considerable variation in cellular density occurred within 15 leiomyomas (13.5%).
Mitotic figures were rare in all leiomyomas. Most tumors (98 [88.3%] of 111 leiomyomas) had less than one mitotic figure per 10 HPF, seven leiomyomas (6.3%) had from one to four mitoses, whereas only one leiomyoma had five or more mitoses per 10 HPF and this tumor was of low cellularity. Overall, the mitotic index was not correlated with the cellular density of the tumor (P = 0.7). The mean mitotic index in leiomyomas from MGA-treated felids was 0.3 ± 0.12 (SEM) (range 0–2) and from control felids was 0.3 ± 0.08 (SEM) (range 0–6.7; P = 0.48). Large, vesicular nuclei were present in 54 tumors (48.6%) from 27 animals, and 7 tumors from 3 of these animals also had irregular chromatin clumping or mild anisokariosis. Cellular morphology was otherwise unremarkable in the remaining 57 tumors.
Central necrosis was present in 18 leiomyomas (16.2%), and five leiomyomas (4.5%) had prominent individual cell necrosis. Three of the leiomyomas with necrosis had minimal neutrophilic infiltrates. Mineralization was present in four leiomyomas (3.6%) from two animals.
Age and cat species group were the only significant risk factors for leiomyomas in the logistic models. Odds ratios (OR) were 1.01 for each 1-month increase in age (95% CI = 1.0006–1.12), 3.69 for big cats compared with other species (95% CI =1.48–9.18), 0.64 for MGA treatment (95% CI = 0.29–1.42), and 0.78 for nulliparous females (95% CI = 0.6–2.77). For the subset of animals with ovarian data, age and big cat species remained as risk factors but the presence of a tertiary follicle did not affect leiomyoma occurrence (OR = 0.94, 95% CI = 0.27–3.10).
Leiomyosarcomas
Leiomyosarcomas were present in the genital tracts of five felids, all from the genus Panthera. Four of the five animals had been treated with MGA, but this association was not significant (P = 0.17).
Leiomyosarcomas appeared grossly as poorly demarcated, multilobulated firm tan to white masses that infiltrated and obliterated normal uterine architecture. In all cases, the neoplasm appeared to arise in the uterus (body or horns). In three felids, the neoplasm had metastasized. One felid had multiorgan metastases that included the heart, lung, brain, adrenal gland, thyroid, stomach, pancreas, kidney, eye, large and small intestine, vagina and ovary (Y. Schulman, personal communication), another felid had metastases to corpora lutea in the ovaries, and a third felid had metastases to other regions of the myometrium discontiguous with the primary tumor. In the latter two animals, other organs were not available to determine if further visceral involvement had occurred. In two felids with metastatic leiomyosarcoma, extensive serosal adhesions to pelvic viscera were present.
Histologically, all leiomyosarcomas were composed of interlacing smooth muscle cells that varied in degree of differentiation and density within the tumor. Three leiomyosarcomas had cellular atypia, high mitotic indices, and local invasion. Two leiomyosarcomas were locally invasive but had low mitotic indices and minimal atypia. Tumors with cellular pleomorphism had anisocytosis, anisokaryosis, multinucleation, hyperchromasia, and bizarre mitoses. The mitotic index ranged from 7 to 27 per 10 HPF among the five tumors. Larger neoplasms had foci of ossification and mineralization, as well as coalescing areas of necrosis that spared the tumor cells around blood vessels. Inflammatory infiltrates were common in areas of necrosis. Vascular invasion was evident in the three neoplasms that had metastasized.
Discussion
This study demonstrated that smooth muscle tumors are a common spontaneous neoplasm in the female genital tract of zoo felids. The proportion of tumors that were leiomyosarcomas (8.7%) is noteworthy when compared to domestic cats, dogs,13 and women, in whom only less than 1% of smooth muscle tumors are sarcomas.18,23 Many zoo felids had multiple leiomyomas as occurs commonly in domestic dogs and women, but in contrast to domestic cats, in which most leiomyomas are solitary.13,16 Although leiomyomas occurred most commonly in the myometrium, several leiomyomas occurred in the ovarian medulla and pedicle, which are not common sites for leiomyoma development in other species.38
As in other species, the prevalence of leiomyomas in zoo felids increased with age,2,13,17,23 and the high prevalence in this study may be biased by the older median age of this population (13.6 versus 10 years), although small felids up to 23 years old were included in the study. However, the tumor prevalence was notably higher in the genus Panthera, which may indicate a predisposition for tumor development in animals from this genus. Yet no tumors were evident in snow leopards (Panthera uncia). Unfortunately, insufficient numbers of many species of small cats in the study precluded determining if these trends were significant.
The detailed histologic analysis of uteri as part of the surveillance program could account for the higher prevalence of leiomyomas in zoo felids than in domestic animals. The effect of sampling method on prevalence statistics has been demonstrated in a study of uterine leiomyomas in women, in which the overall prevalence increased 48% when uteri were serially sectioned.7 Therefore, prevalence estimates of leiomyomas in any population should include careful scrutiny of both grossly detectable tumors and seemingly normal regions of myometrium.
Lack of association between smooth muscle tumors and MGA treatment contrasts with the relationship between progestins and smooth muscle tumors in women26,28 and rats.39 Progestin regulation of smooth muscle growth would be predicted because of the extensive physiologic myometrial hypertrophy and hyperplasia that occurs during pregnancy.19 However, tumor volumes also have been reduced in women receiving high doses of progesterone, indicating complex hormonal regulation of smooth muscle tumor growth.4 Because felids are induced ovulators, the tumors in control animals would have arisen in most cases in a progesterone-poor environment, indicating either growth promotion by estrogens or autonomous growth in these species.
In our study, nulliparous felids were no more likely than parous felids to develop leiomyomas, suggesting that parity was not protective as is true for women.12 Whether most leiomyomas affect reproductive function in zoo felids could not be assessed from available data, but those tumors that formed intraluminal polypoid growths would certainly impede fertility.
Distinguishing leiomyomas from well-differentiated leiomyosarcomas was difficult in zoo felids, as has been true for other species. Although discrete tumor margins are useful to distinguish benign from malignant neoplasms of other cell types, many leiomyomas in zoo felids had very ambiguous borders despite being sparsely cellular and composed of well-differentiated smooth muscle cells. However, these tumors at other margins compressed surrounding tissues and were not invasive, consistent with benign behavior.16 Although necrosis and hemorrhage are useful indicators of malignancy in smooth muscle tumors of women, these degenerative changes also have been reported in clinically benign tumors in women21,27,34 and were present in some large, seemingly benign tumors in the animals in this study, suggesting that these should not be the sole criteria for defining malignancy. In women, leiomyosarcomas also are distinguished from leiomyomas by cellular density, atypia, high mitotic indices, and invasive growth.23 In zoo felids, many densely cellular tumors had no atypia and low mitotic indices, and were compressive, not invasive. These tumors were interpreted as highly cellular leiomyomas and not leiomyosarcomas.27 In many otherwise benign-appearing tumors in our study, neoplastic cells had large vesicular nuclei with dispersed chromatin, suggesting that this nuclear morphology should not be considered atypical in feline leiomyomas. Mitotic index also was not a reliable criteria for distinguishing leiomyomas from leiomyosarcomas in zoo felids because some noninvasive leiomyomas with low cellularity had high indices. Also, all leiomyosarcomas had areas with low mitotic indices despite having the indisputably malignant behaviors of invasion and metastasis.
Malignant transformation of leiomyoma to leiomyosarcoma is believed to be rare in women, but has been suggested to occur in domestic animals.13,16,29 Three of five cats in this study with leiomyosarcoma had leiomyomas adjacent to their malignant tumors. However, these morphologic features also could arise from invasion of a concurrent leiomyoma by the leiomyosarcoma, so the relationship between leiomyosarcomas and leiomyomas in felids remains unknown. In the other two cats, the extent of tissue destruction by the leiomyosarcoma made determination of any origin within a leiomyoma unfeasible.
In summary, smooth muscle neoplasms are common spontaneous neoplasms in uteri and ovaries of zoo felids. The smooth muscle tumors in this study present a spectrum of morphologic characteristics that made distinguishing benign from malignant neoplasia difficult. The presence of local invasion (but not indistinct margins) was the most reliable predictor of aggressive behavior. Age, but not MGA contraceptive exposure, was a risk factor in the development of leiomyomas. In rare cases, the size or location of the neoplasms might have impeded fertility, but the majority of the leiomyomas in this study were incidental findings.
Footnotes
Acknowledgements
We thank Dr. L. Harrenstein, Dr. R. Mason, and M. Mason for assistance with the project. We also thank J. Schneider for preliminary statistical tests on SAS, K. Wiegel for assistance with photomicroscopy, and Dr. R. Donnell for reviewing the manuscript. Veterinarians at the following institutions generously contributed reproductive tracts to this study: Acadia Zoo, Akron Zoo, Zoo Atlanta, Audubon Park Zoo, John Ball Zoo, Baltimore Zoo, Binder Park Zoo, Birmingham Zoo, Blank Park Zoo, Brandywine Zoo, Brookfield Zoo, Burnet Park Zoo, Caldwell Zoo, Calgary Zoo, Central Florida Zoo, Chaffee Zoological Gardens of Fresno, Cheyenne Mountain Zoo, Cincinnatti Zoo, Columbus Zoo, Dallas Zoo, Denver Zoo, Detroit Zoo, Exotic Feline Breeding Center, Fort Wayne Children's Zoo, Fossil Rim Wildlife Center, Franklin Park Zoo, Henry Doorly Zoo, Hogle Zoo, Honolulu Zoo, Houston Zoo, Jackson Zoo, Jacksonville Zoo, Kansas City Zoo, King's Island, Knoxville Zoo, Lake Superior Zoo, Lincoln Park Zoo, Little Rock Zoo, Los Angeles Zoo, Louisville Zoo, Lowry Park Zoo, Memphis Zoo, Metro Washington Park Zoo, Miami Metrozoo, Micke Grove Zoo, Miller Park Zoo, Minnesota Zoo, Montgomery Zoo, Morehouse College, National Zoo and Conservation and Research Center, National Institutes of Health, NYZS/Wildlife Conservation Society, Oklahoma City Zoo, Philadelphia Zoo, Phoenix Zoo, Pittsburgh Zoo, Potawatomi Zoo, Racine Zoo, Reid Park Zoo, Rio Grande Zoo, Riverbanks Zoo, Ross Park Zoo, Sacramento Zoo, San Antonio Zoo, San Diego Zoo and Wild Animal Park, San Francisco Zoo, Santa Ana Zoo, Santa Barbara Zoo, St. Louis Zoo, Seneca Park Zoo, Sunset Zoo, Toledo Zoo, Tulsa Zoo, Utica Zoo, White Oak Conservation Center, Wildlife Safari, Wildlife Waystation, and Woodland Park Zoo. This project was supported by the New York Zoological Society Nixon Griffis Fund for Zoological Research, the American Zoo and Aquarium Association Conservation Endowment Fund/Ralston Purina Big Cat Survival Funds, and The Geraldine R. Dodge Foundation.
