Abstract
Cutaneous chlorellosis in a gazelle caused a chronic granulomatous inflammation in the subcutaneous tissue of the carpus. The diagnosis was based on the intense green color of the lesion and the presence of organisms identified as algae of the order Chlorellales. According to morphologic and ultrastructural features, the genus Bracteacoccus is most likely.
Compared with mycoses, algal infections are rare in mammals and human beings.6,10,12 The infection manifests as cutaneous lesions or systemic disease. In animals, infections have been described in cattle,14 sheep,2,8,16 a dromedary,9 and probably a beaver.15
The Chlorellales is an order of chlorophyll-containing coccal green algae. The various genera of the Chlorellales must be differentiated from the genus Prototheca. Prototheceae are unicellular, achlorophyllous organisms belonging to the same phylogenetic group as green algae of the genus Chlorella.13 Unlike Prototheca, members of the genus Chlorella contain abundant periodic acid–Schiff (PAS)-positive cytoplasmic granules, which represent starch and chloroplasts.1,4,7,11
The infected gazelle belonged to a flock of seven animals kept in an enclosure of gravel and scarce vegetation that was completely surrounded by a fence of wire netting and without a moat. The 10-year-old female gazelle suffered an injury, presumably caused by wire, in the left carpal region. The animal was immobilized, and a firm green lump approximately 4 cm in diameter, which was thought to be an abscess, was removed surgically. The wound was dressed and treated locally and systemically with antibiotics. Two days later, healing of the wound appeared to be progressing. Local treatment was repeated by antibiotic spray without immobilization. Seven months later, a mass 2 cm in diameter recurred in the same location and grew to 4 cm in diameter within 3 weeks. Because the gazelle was pregnant, it was not treated. Four months later, after the animal gave birth, the mass, now a suspected neoplasm, was removed surgically and submitted for histologic examination. The animal did well after surgery. However, 19 months after the first excision the lesion recurred again at the site of the operation, and the gazelle was euthanatized.
The specimen submitted for examination consisted of an ovoid mass approximately 4 cm in maximum diameter and covered by hairy skin. The cut surface was of a striking yellow-green color. It was well demarcated from the surrounding subcutaneous tissue by a capsule of fibrous tissue and had a lobular, almost cystic appearance due to fibrous strands within (Fig. 1).
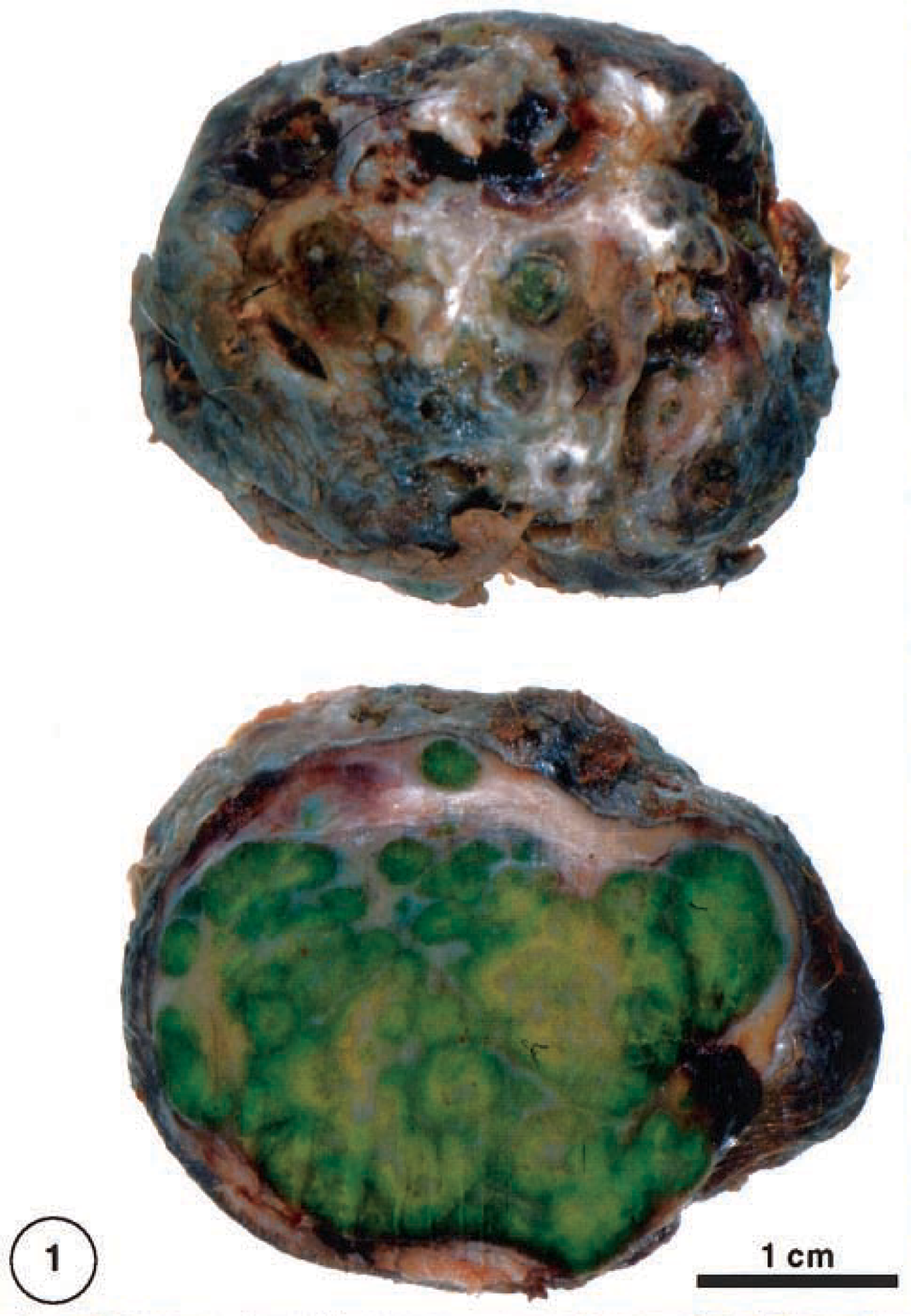
Forelimb; dorcas gazelle. Cutaneous mass has an ulcerated surface (above). The cut surface has a lobulated appearance (below) and a yellow-green color.
The tissue was immersion fixed in neutral 10% formalin, routinely processed, embedded in paraffin, and sectioned at 6 μm. For light microscopic examination, the sections were stained with hematoxylin and eosin (HE) and subjected to the PAS reaction. Methacrylate-embedded 1-μm sections were stained after Giemsa. For transmission electron microscopic examination, formalin-fixed material was embedded in Epon 812 after being washed several times in 0.1 M phosphate buffer and after subsequent postfixing in 1% osmium tetroxide and dehydration in graded acetones. Ultrathin sections (ca. 50 nm) were stained with uranyl acetate and lead citrate.
The mass consisted mainly of granulomatous inflammatory tissue. Between broad strands of fibrous tissue, there were large accumulations of macrophages, many of them ghost cells within areas of necrosis. A myriad of spheric to slightly ovoid organisms, identified as algae apparently were the cause of this tissue reaction (Fig. 2). Fibroplasia of variable degree was also present. Lymphoplasmacellular infiltration and multinucleated giant cells were scarce. The algal cells were 10–45 μm in diameter and obviously represented different developmental stages (Fig. 3). In HE-stained sections, two main types could be distinguished: small cells with thin cell walls and cytoplasmic basophilic granules that stain strongly by PAS (Fig. 4) and large cells with thick cell walls that were translucent with a slightly eosinophilic (cloudy) center.
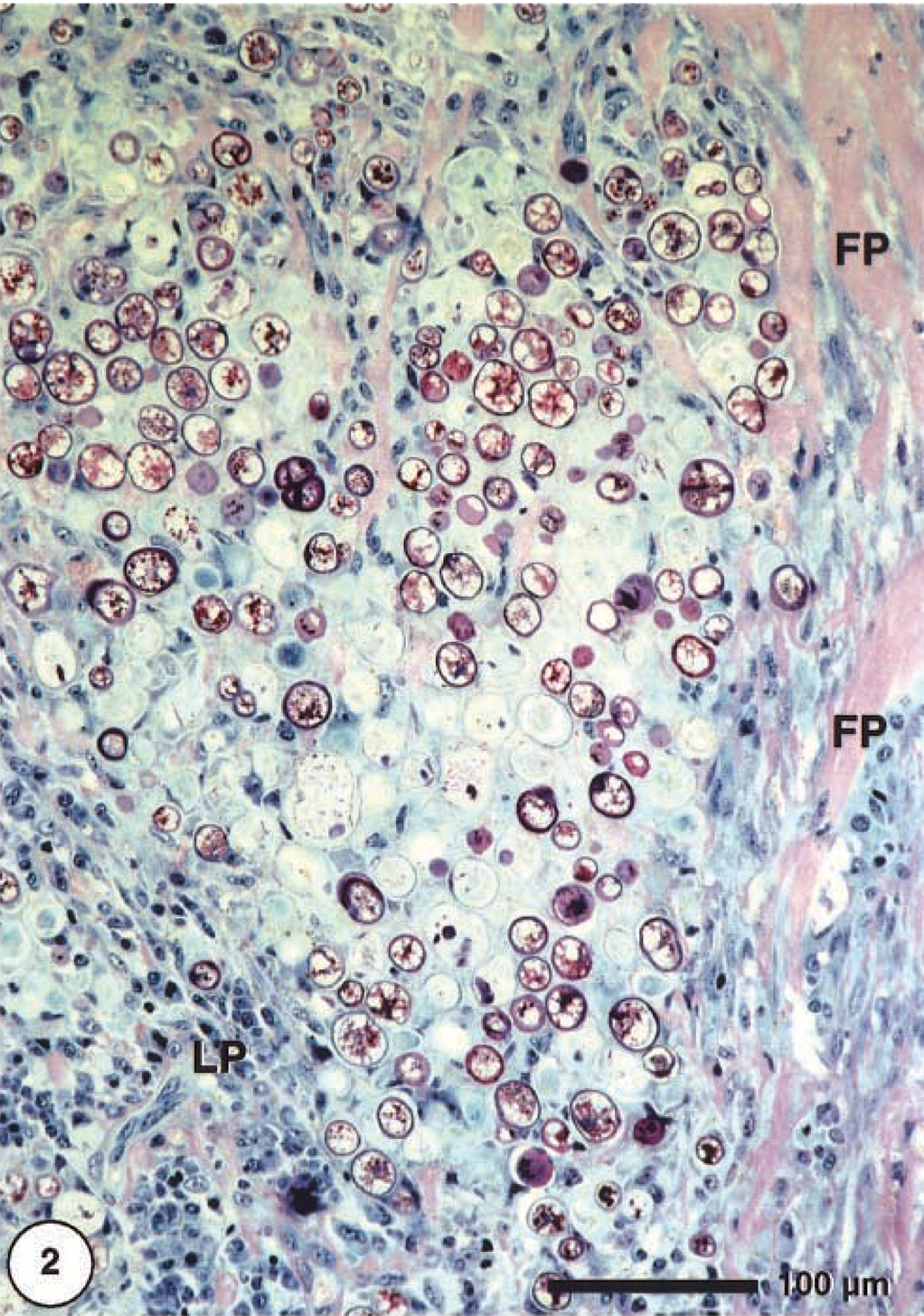
Cutaneous mass; dorcas gazelle. Granulomatous inflammation showing abundant algal cells surrounded by fibroplastic strand (FP) and lymphoplasmacytic infiltrates (LP). Methacrylate, Giemsa.
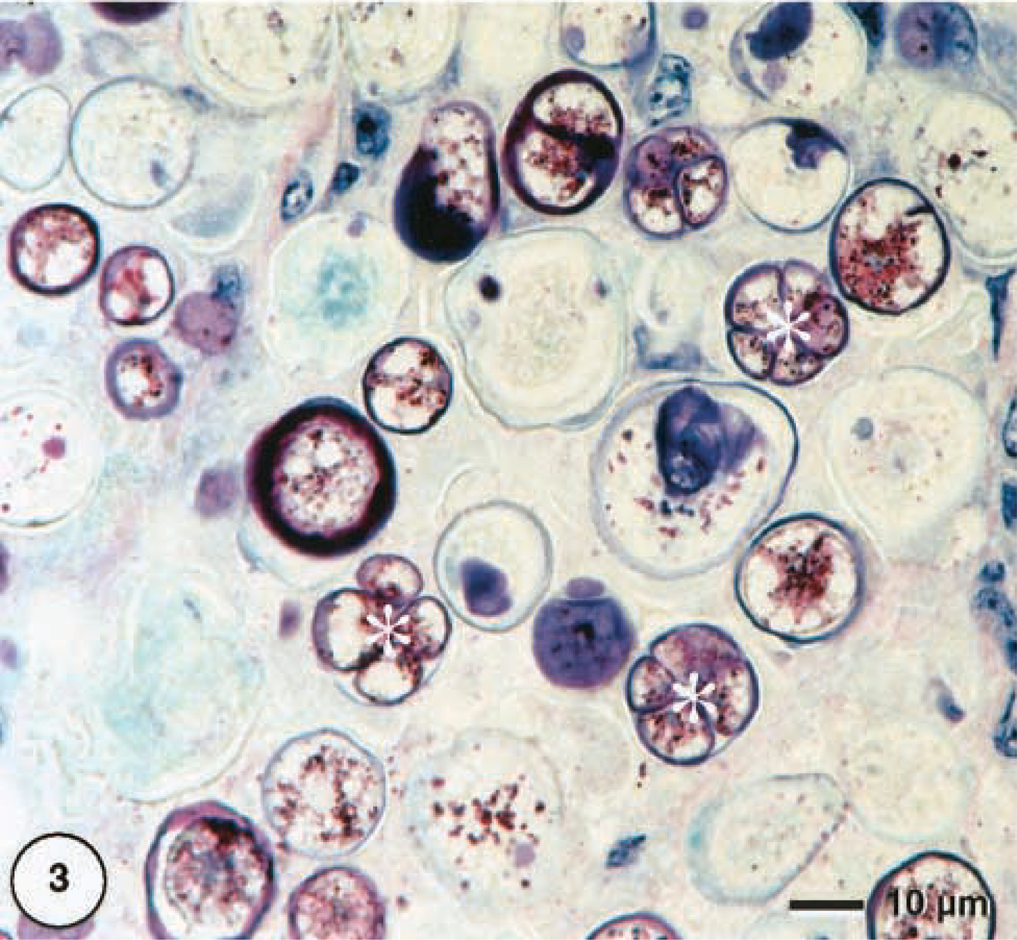
Cutaneous mass; dorcas gazelle. Single cells of coccal green alga with autospore formation (asterisks), which is a characteristic feature of the order Chlorellales. Methacrylate, Giemsa.
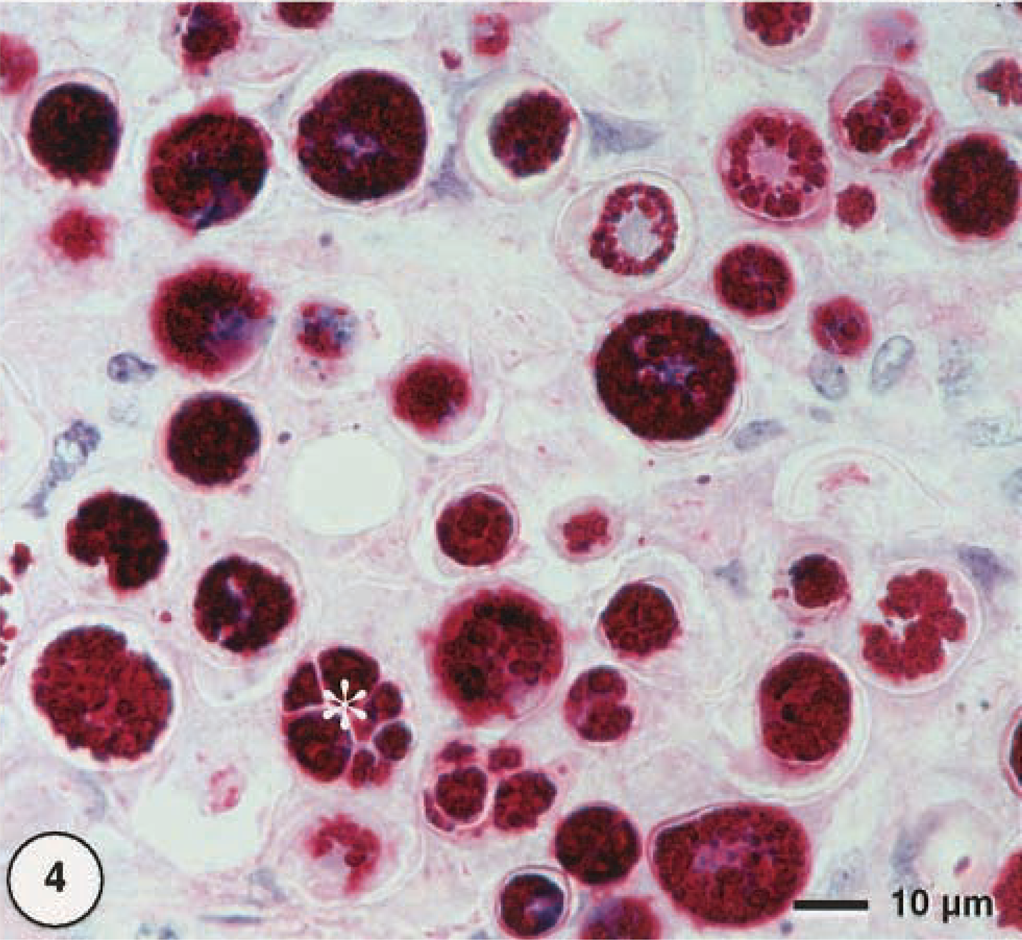
Cutaneous mass; dorcas gazelle. The algal cells contain abundant PAS-positive material (asterisk = autospore formation). Methacrylate, PAS.
Numerous organisms could not clearly be identified as belonging to the two main types described above. They were considered transitional stages. A few algal cells were in the process of cell division. In necrotic areas, dystrophic calcification was seen. Corresponding to the gross finding in the HE-stained paraffin-embedded sections (± 6 μm thick), a green color of some cells could be seen that was not clearly visible in methacrylate embedded Giemsa-stained sections (± 1 μm).
Only formalin-fixed material was used for transmission electron microscopic examination (Figs. 5–7). In cross section, single cells were more or less spherical with diameters of approximately 20 μm. Frequently, clusters of cells within a common cell wall were observed, representing typical autospores (Fig. 6), which are flagellum-free endospores that form within the cell wall of the mother cell. Because of the coccosiphonal organization (unicellular, multinucleated), cross sections of large algal cells exhibited up to seven randomly distributed nuclear profiles. The cell wall varied in thickness from 1 to 2 μm, with irregular local thickenings. Typically, a distinct coating (= sporopollenin layer) with a thickness of approximately 200 nm was visible as an electron-dense layer. Most of the cytoplasmic area was occupied by typical lipid bodies 1 μm in diameter with an electron-translucent homogeneous matrix and a half-unit membrane as a boundary. In addition, larger osmiophilic globules (diameter ca. 1.5 μm) were observed. These globules may represent a second population of lipid bodies with a different lipid composition. Chloroplasts were numerous; within one cross section about 25 rather small chloroplasts (1–2 μm) could be seen (Fig. 5). Thylakoid stacks, part of the internal membrane system of chloroplasts, were made up of three to five thylakoids. Plastoglobules (small plastidal lipid bodies) with diameters of 50–75 nm and starch grains were frequently observed (Fig. 7). Pyrenoides, definded as protein-rich globular structures involved in starch accumulation, were absent.
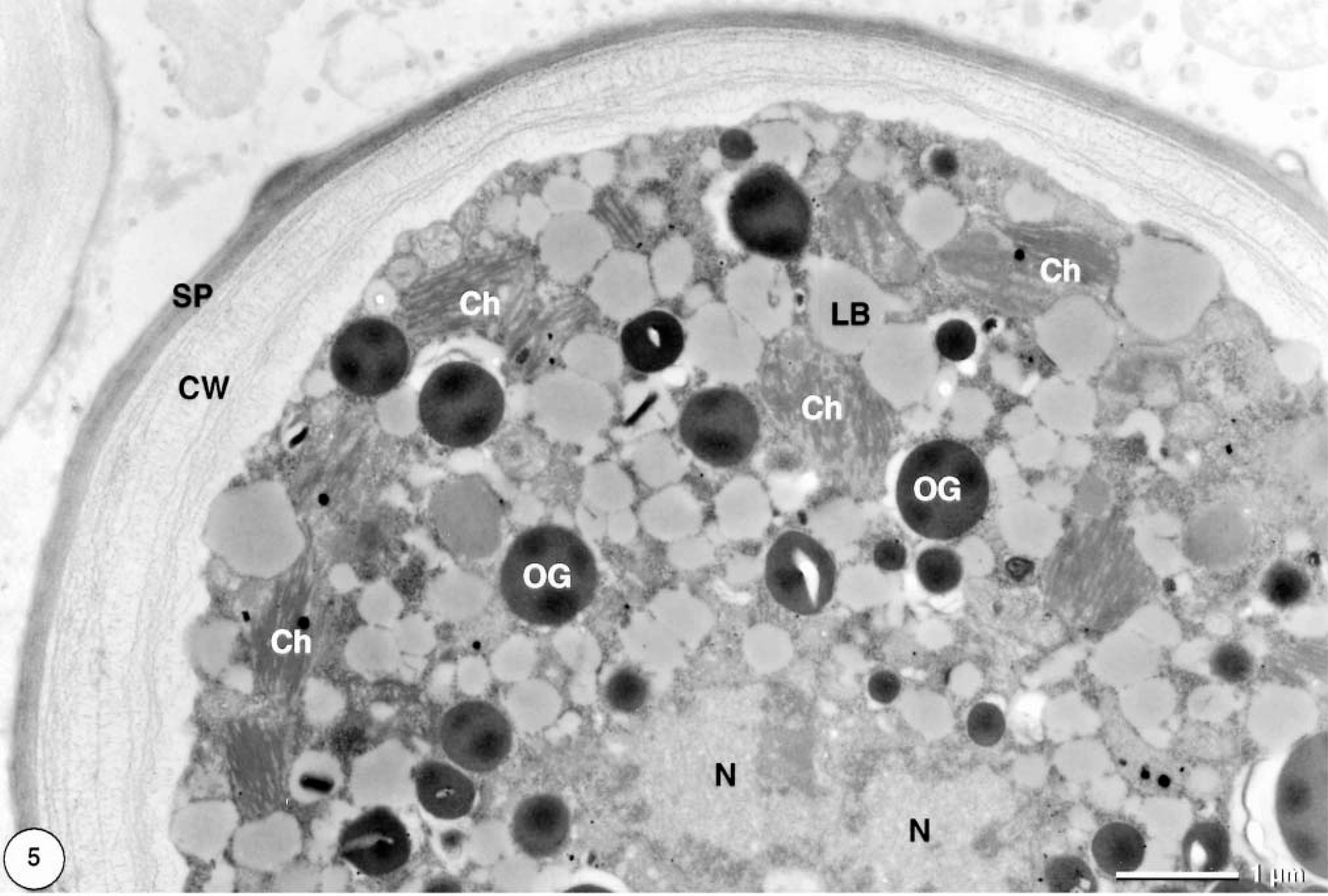
Transmission electron micrograph. Cutaneous mass; dorcas gazelle. Coccal green algal cell contains numerous chloroplasts (Ch) and lipid bodies (LB). A second type of lipid body–like osmiophilic globules (OG) is visible. The cell wall (CW) is rather thick and covered by a prominent sporopollenin layer (SP) (N = nuclei). Uranyl acetate, lead citrate.
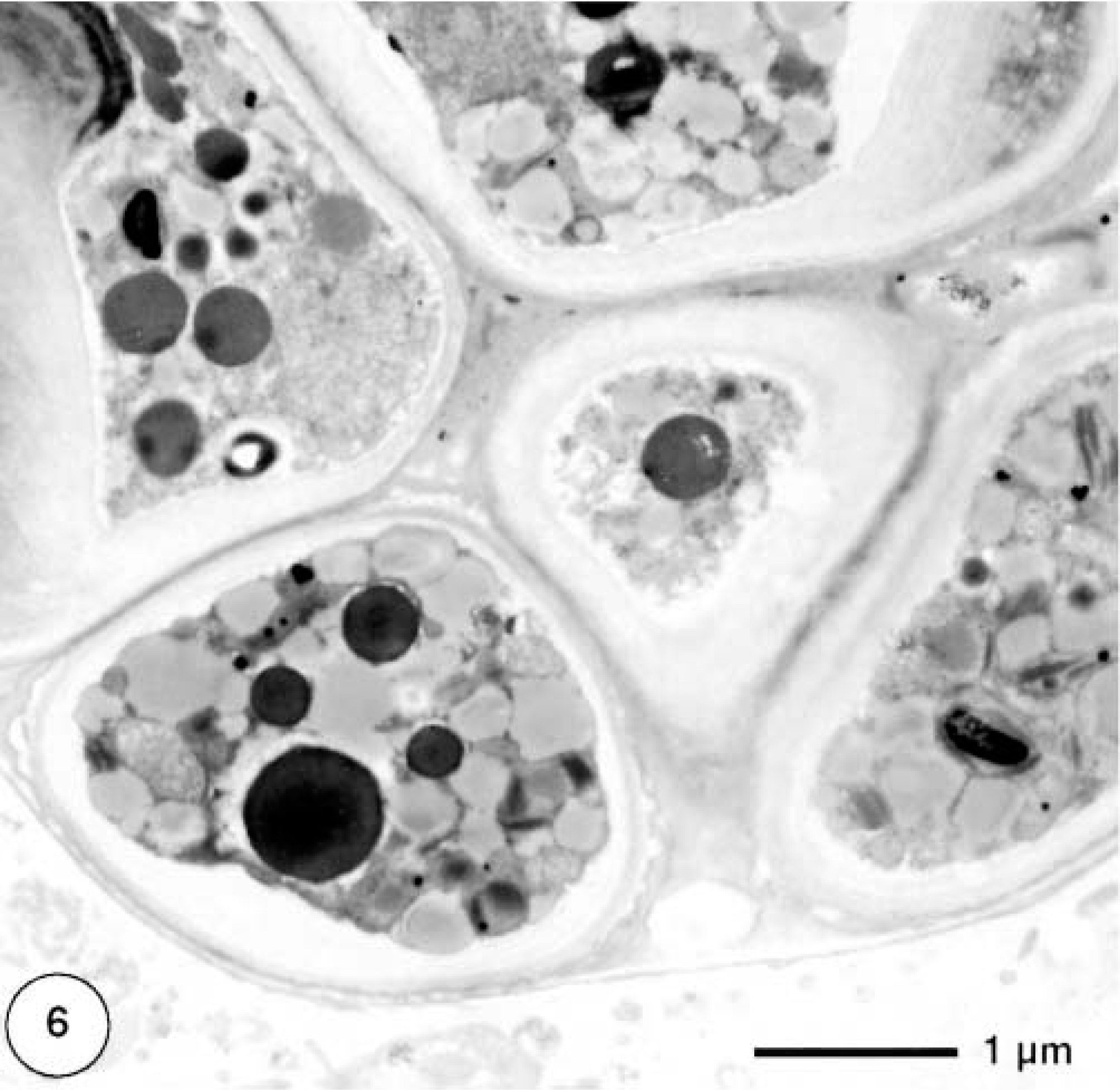
Transmission electron micrograph. Cutaneous mass; dorcas gazelle. Algal cells show autospore formation.
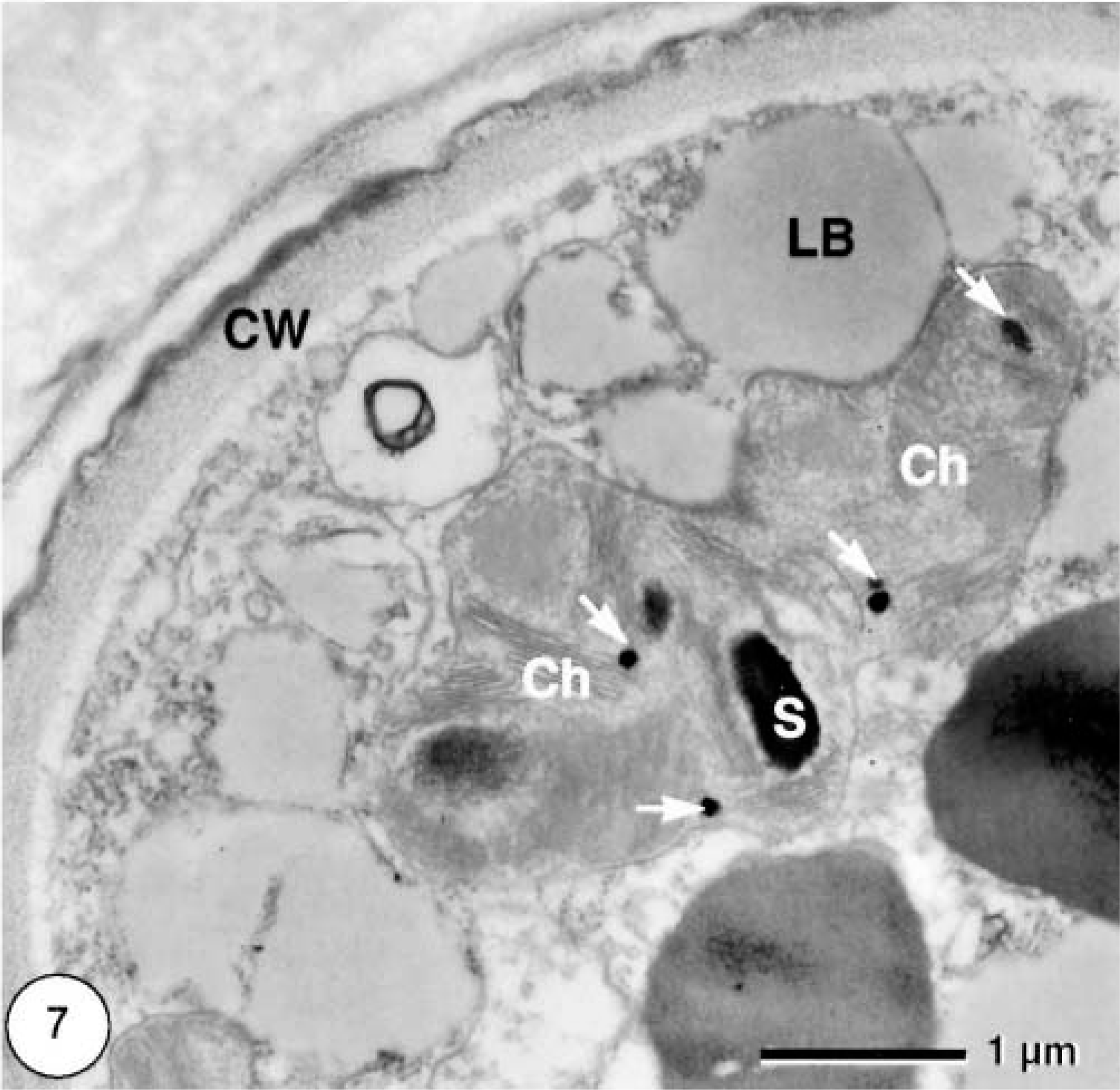
Transmission electron micrograph. Cutaneous mass; dorcas gazelle. Algal cells have chloroplasts (Ch) with small starch grains (S) and plastoglobules (arrows) (CW = cell wall; LB = lipid bodies).
The light microscopic morphologic features of the organisms and their green color are consistent with coccoid green algae, apparently producing chlorophyll. Although it is difficult in general to identify algae occurring in atypical environmental conditions (e.g., unusual humidity, light intensity, O2/CO2 concentration), there were some morphologic characteristics that allowed classification of these algae. According to van den Hoek et al.,5 we classify the algae as belonging to the section Chlorophyta. The four main criteria used for determination were 1) multinucleated cells, 2) numerous chloroplasts (up to 25 irregular profiles with a diameter of about 1–2 μm mainly located at the cell periphery and characterized by parallel thylakoid membranes, electron-dense starch granules, diameter 0.5–1 μm, and electron-dense spherical plastoglobules, diameter 50–75 nm), 3) absence of pyrenoids, and 4) characteristic cell wall architecture (1–2 μm thick with an electron-dense sporopollenin layer). According to the key of Ettl and Gärtner,3 the algae belong to the order Chlorellales (coccosiphonal organization, multinucleated, distinct cell wall with sporopollenin layer, numerous chloroplasts without pyrenoids) and the family Neochloridaceae (according to polyenergid cells and typical size and shape of the cells). Referring to size and shape and number of the chloroplasts, absence of pyrenoids, and cell wall characteristics, the genus Bracteacoccus is most likely.
Because most species of this genus are aerophytic or terrestrial, a secondary infection of the skin with a member of this algal genus is not unlikely. In addition, a member of this genus would be able to survive in tissues under these deficient conditions. Chlorellosis in animals might be caused by different species of the order Chlorellales. Ultrastructural investigation of granulomatous enteritis in a camel9 revealed much smaller green algal cells (5–11 μm in diameter) with a single nucleus, which obviously belong to another genus.
The mode of infection remains unclear. Probably an unobserved small wound facilitated the primary infection with bacteria and secondary infection with spores of the facultative pathogenic alga. A primary bacterial infection might have caused a purulent inflammation, providing conditions for the alga to multiply in the subcutaneous tissues and cause the granulomatous inflammation.
Footnotes
Acknowledgements
We thank Dr. Julia von Maltzan for providing the clinical data.
