Abstract
We report a case of cutaneous epithelioid hemangiosarcoma in a dog in which the majority of the neoplastic cells displayed histologic and ultrastructural features similar to those seen in granular cell tumors (GCTs). This intersection of hemangiosarcoma and granular cell change adds to the argument that GCTs are heterogeneous in histologic origin and underlines the fact that pathologists should not consider all GCTs as a single entity. The combination of histology in typical areas of the tumor with ultrastructural changes and the correct immunohistochemical markers can facilitate the accurate diagnosis of tumors with granular cell differentiation. Besides characteristic intracytoplasmic PAS-positive granules and ultrastructural proteinaceous accumulation within single membrane vesicles (presumably lysosomes and phagolysosomes), we suggest the following combination of markers for the diagnosis of granular cell angiosarcoma and/or hemangiosarcoma: vimentin positive, NSE and/or S100 negative, CD31 positive. We propose that the histologic granular appearance represents a metabolic defect of the neoplastic cells, which supports variability in cell origin for granular cell differentiation.
A 5-y-old neutered male mixed-breed dog was presented to Ross University Veterinary Clinic (Ross University School of Veterinary Medicine, Basseterre, St. Kitts, West Indies) because of a single bleeding cutaneous left parapreputial mass. The owner reported that the mass had developed over a year while living on the island of Nevis, West Indies. The mass had grown until it had ulcerated and was dripping blood. There were no other remarkable findings on physical examination. Complete blood count and serum biochemistry profiles revealed the following abnormalities: mild anemia, stress leukogram, decreased platelet count, acanthocytes, and increased alanine aminotransferase. Acanthocytosis is a frequent laboratory finding in patients with hemangiosarcoma, but it is not pathognomonic for this condition. 16 Differential diagnoses by the clinician were an ulcerated granuloma or skin neoplasia. The owner agreed to surgical excision, but declined preoperative workup for metastasis and treatment. The dog was anesthetized, and a 10-cm elliptical incision was made to remove the affected tissue. The excised mass (6.4 × 4.5 × 2.8 cm) was fixed in 10% neutral-buffered formalin and submitted for histologic analysis. The tissues were processed routinely and stained with hematoxylin and eosin, as well as periodic acid–Schiff (PAS), Luxol fast blue (LFB), Perl Prussian blue, and Fontana–Masson.
Immunohistochemical (IHC) markers (Table 1) included CD31 (endothelial cells), factor VIII–related antigen (FVIIIRAg; endothelial cells), S100 (many cell types), neuron-specific enolase (NSE; neural and neuroendocrine),1,7 vimentin (mesenchymal cells), pan-cytokeratin (epithelial cells), and lysosome (macrophages). IHC stains chosen were those proposed by the human literature for this rare subtype of hemangiosarcoma (Table 2) and also to rule out a collision tumor, particularly of histiocytic origin. Five-micrometer thick sections were deparaffinized, and then rehydrated in a graded alcohol series. The antigen was unmasked using pressure cooker heat-induced epitope retrieval with Diva Decloaker solution in a decloaking chamber (Biocare, Concord, CA) at 125°C for 30 s. Sections were incubated in 3% H2O2 for 15 min to block endogenous peroxidase activity. Sections were rinsed in Tris-buffered saline with 0.05% polysorbate 20 (TBST 0.05 M, pH 7.6). To block nonspecific binding sites, the sections were incubated with Background Punisher (Biocare) for 10 min. After excess blocking solution was drained, primary antibody was applied for 45 min at room temperature. Sections were rinsed with TBST and then incubated with the secondary antibody for 45 min at room temperature (EnVision horseradish peroxidase–labeled goat anti-rabbit immunoglobulin G, Dako, Carpinteria, CA). After rinsing in TBST, positive antigen–antibody reactions were visualized by incubating the slides with 3-amino-9-ethylcarbazole for 15 min; sections were counterstained with Mayer hematoxylin. Controls included substitution of the primary antiserum with diluted normal serum from non-immunized rabbits and inclusion of brain tissue with oligodendrocytes.
Antibody sources, manufacturers, dilutions, clones, types, incubation times, antigen retrieval method, and detection systems.

HIER = heat-induced antigen retrieval; Ms = mouse; NSE = neuron-specific enolase; Rb = rabbit. Dash (—) indicates no clone.
Summary of immunohistochemical and histochemical stains used to characterize granular cell angiosarcomas.

FVIIIRAg = Factor VIII–related antigen; NA = not assessed; PAS = periodic acid–Schiff.
Vascular lining cells were positive; granular cells were negative.
Vascular lining cells were positive; granular cells were occasionally positive; spindle cells were focally positive.
Weakly PAS-positive.
For ultrastructural examination, formalin-fixed neoplastic tissue was post-fixed in 0.166 M cacodylate-buffered, 3% glutaraldehyde (Electron Microscopy Sciences [EMS], Hatfield, PA), followed by a second post-fixation treatment in 1% osmium tetroxide (EMS). Using a graded series of ethyl alcohol, 1.0-mm3 tissue blocks were dehydrated and then embedded in Embed (EMS). Embedded samples were trimmed and sectioned (UC6 ultramicrotome, Leica Microsystems, Wetzlar, Germany). Thin sections (60–90 nm) were collected on 100-mesh copper grids (EMS), and then grids were stained with 5% uranyl acetate for 20 min and Sato lead citrate for 6 min. We examined samples (1200 EX II transmission electron microscope [TEM], JEOL, Tokyo, Japan) and obtained images (Veleta 2k × 2k camera with iTEM software, Olympus SIS, Munster, Germany).
Histologic examination revealed that, within, expanding, and replacing the dermis and extending into the subcutis, there was a relatively well-demarcated, non-encapsulated, infiltrative, moderately cellular neoplasm composed of blood-filled cavities separated by a sparse collagenous stroma (Fig. 1). The neoplasm consisted of 2 areas, one predominantly vascular area with plump spindle cells delineating variably sized blood-filled spaces, and a more densely cellular area with pleomorphic, polygonal neoplastic cells forming haphazard streams and bundles (Fig. 2). The neoplastic cells had indistinct cell borders and moderate-to-abundant amounts of fibrillar deeply eosinophilic cytoplasm, with variable numbers of intracytoplasmic hypereosinophilic granules 1–3 µm diameter. Nuclei were moderately pleomorphic (ranging from irregularly round to oval), centrally located, with finely stippled chromatin and 1–4 variably distinct magenta nucleoli. Anisocytosis and anisokaryosis were moderate-to-marked with scattered karyomegaly. The mitotic count was 68 per 10 consecutive high-power fields, with frequent bizarre mitoses. Multifocal areas of hemorrhage and necrosis were seen, as well as low numbers of lymphocytes, plasma cells, neutrophils, and hemosiderin-laden macrophages.
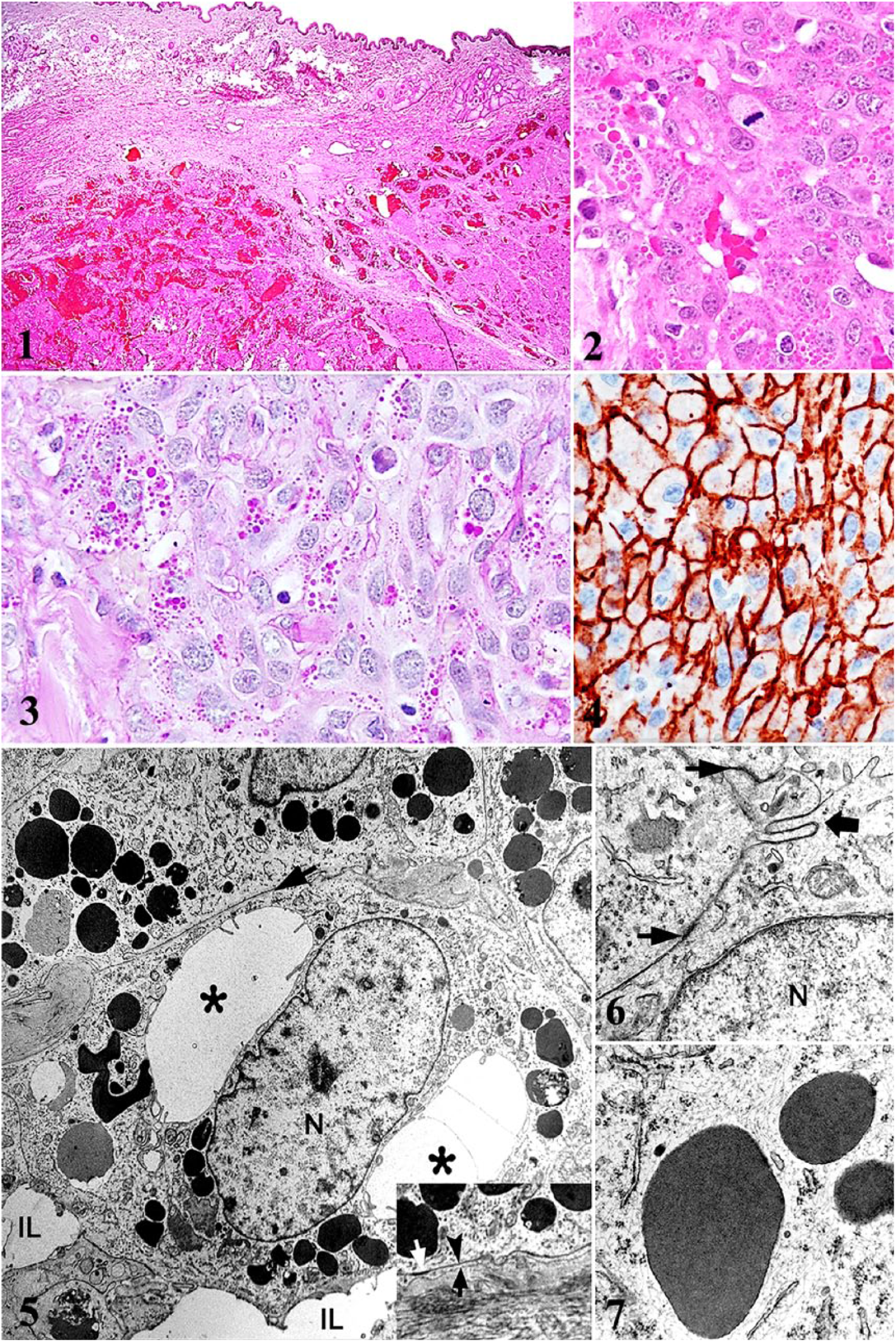
Granular cell hemangiosarcoma in a dog.
Histochemically, the intracytoplasmic granules were PAS-positive, indicative of the glycosidic nature of the granule’s contents (Fig. 3). However, LFB, Perl Prussian blue, and Fontana–Masson stains were negative for phospholipids, iron, and melanin, respectively. These findings were also confirmed ultrastructurally. The neoplastic cells had strong intracytoplasmic immunoreactivity for vimentin and CD31 throughout the neoplasm (Fig. 4). The neoplastic cells lacked immunoreactivity for lysozyme, FVIIIRAg, pan-cytokeratin, NSE, and S100. Positive and negative controls were appropriate. IHC and histochemical findings from the previous 3 cases reported in people are similar to our findings8,9,12 (Table 2).
On TEM, neoplastic cells had a plasma membrane with infolding and zonula adherens. Vascular lumina were formed by interaction between neoplastic cells (Fig. 5). Frequently, neoplastic cells formed internal lumens, and the basal membrane was discontinuous. The voluminous cytoplasm was composed of small rough endoplasmic reticulum cisterns, mitochondria, Golgi system, and numerous vesicles (Fig. 6). Sparse intermediate filaments were also present. Most of the cytoplasmic granules were accumulations of electron-dense amorphous material (probably proteins) bound by a single membrane (Fig. 7), consistent with storage vesicles, presumably secondary lysosomes. A proportion of granules were phagolysosomes containing heterogeneous tubular–vesicular–membranous electron-dense profiles within a single membrane. Weibel–Palade bodies, which are characteristic of endothelial cells, were not found in our case.
Based on histology, IHC findings, histochemistry, and ultrastructure, we concluded that the diagnosis was epithelioid hemangiosarcoma with granular cell differentiation. At follow-up examination, one year after the surgery, the dog was doing well, with no clinical signs of recurrence or metastasis.
In humans, granular cell angiosarcoma is an extremely rare neoplasm 3 with only 3 cases reported to date,8,9,12 and no prior reports, to our knowledge, in dogs. In human cases, the granules are reported to be PAS- and LFB-positive,12,13 whereas in our case, the granules were only PAS-positive. We confirmed the vascular origin of this neoplasm via CD31 immunohistochemistry. Although immunoreactivity to FVIIIRAg was not detected, this is not surprising given that differentiation of the neoplastic cells will impact expression of these 2 markers. IHC results for S100 and NSE were negative, excluding a possible proliferation of neural lineage (Schwann cell origin) as the main origin suggested for GCT in animals.1,7 The absence of Weibel–Palade bodies is equally unsurprising given that these bodies have been consistently seen in angiomas in the dog, but are often decreased or absent in angiosarcomas. 4
GCTs in veterinary species are histologically typified by large polygonal cells with abundant intracytoplasmic acidophilic granules that are PAS- and S100-positive. 11 In addition to GCTs, granular cell differentiation is widely seen in a variety of human neoplasms including those derived from epithelial, melanocytic, histiocytic, and muscle tissues. 3 The granules likely represent an increase in secondary lysosomes, 2 which was supported by the TEM findings of numerous secondary lysosomes and phagolysosomes in our case.9,10 We hypothesize that this feature may represent a degenerative phenotype common to many types of neoplastic cells as a result of some form of lysosomal enzyme dysfunction.
Epithelioid (histiocytoid) hemangiosarcoma in the dog is most commonly reported in the skin, but can also be a phenotypic variant of visceral hemangiosarcoma.6,15 It consists mainly of polygonal or plump spindle cells, with abundant, pale eosinophilic cytoplasm and large vesicular nuclei with prominent nucleoli and moderate-to-high mitotic activity.6,14 Patterns of growths vary from conventional vasoformative structures to acini, ducts, or solid areas. Immunohistochemically, even poorly differentiated endothelial cells are consistently CD31-positive, with variable expression of factor VIII depending on cellular differentiation.5,6,14
We suggest that granularity may represent heterophagy or autophagy consistent with neoplastic cell degeneration as a result of enzymatic deficiency. Therefore, we recommend that granular cell morphologic differentiation in a tumor should include vascular lineage as well, adding weight to the statement that GCTs are a heterogeneous group histogenetically.
Footnotes
Acknowledgements
This work is part of the Integrative Mammalian Research Center from Ross University School of Veterinary Medicine. We thank David Hilchie for the histopathology support and special stains, as well as Dr. Saundra Sample for reviewing the clinical pathology data of this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
