Abstract
Two greater kudu calves (Tragelaphus strepsiceros) born 7 years apart were found with fissures and thickened, scaly, cutaneous plates covering over 80% of their bodies. One was dead at presentation, and the other was euthanized shortly after birth. Both animals shared a common sire. On necropsy, chemosis, ectropion, eclabium, and bilateral valgus deformities of the tarsal joints were observed in one calf, presumed to be secondary to the plates restricting normal fetal development. The principal microscopic lesion was severe lamellar orthokeratosis, with focal mild parakeratosis. Ultrastructural epidermal lesions included the absence of normal lamellar granules, large dilated endoplasmic reticulum, and abnormal retention of organelles and vesicles. Gross, histopathologic, and electron microscopic findings in both kudu calves were consistent with those of harlequin ichthyosis, a rare dermatosis of humans believed to have an autosomal recessive inheritance pattern. The underlying genetic and molecular abnormality and heritability of this condition in this greater kudu herd were not determined.
Two deformed greater kudu calves with the same sire were found on exhibit at the North Carolina Zoological Park over a period of 7 years. Both animals were found within hours after birth and showed similar external abnormalities. The first calf, kudu No. 1, was found dead in October 1993, approximately 24 hours after its dam had displayed signs of premature labor. The calf was small, female, and had a hairless, pigmented integument with numerous fissures and cracks. The texture of the skin was described as that of “leather.” The lungs appeared dark pink and did not float in formalin. Tissues were reported to be moderately autolyzed, but other morphologic abnormalities were not noted on gross necropsy.
The second kudu calf, kudu No. 2, was found alive within 6 hours of birth with its 11-year-old mother in July 2000 after a normal gestation length. Over 80% of the calf's body was covered with a 0.3–0.5 mm thick dark gray/brown platelike material that was dry, fissured, and incorporated underlying hair (Figs. 1, 2). The limbs were tucked beneath the body in a fetal position and could not be extended. Additionally, both rear limbs had mild valgus rotation at the tarsal joints. Ocular abnormalities included chemosis, erythematous conjunctiva, and diffuse lens opacity bilaterally. Both aural pinnae were dry, thickened, and curled at the tips. The taut platelike tissue everted the lower lip and eyelids. Fifteen hours after birth the calf was unable to stand or nurse, apparently because of restriction of movement by the scaly plates, and was euthanized by intravenous injection of sodium pentobarbital.
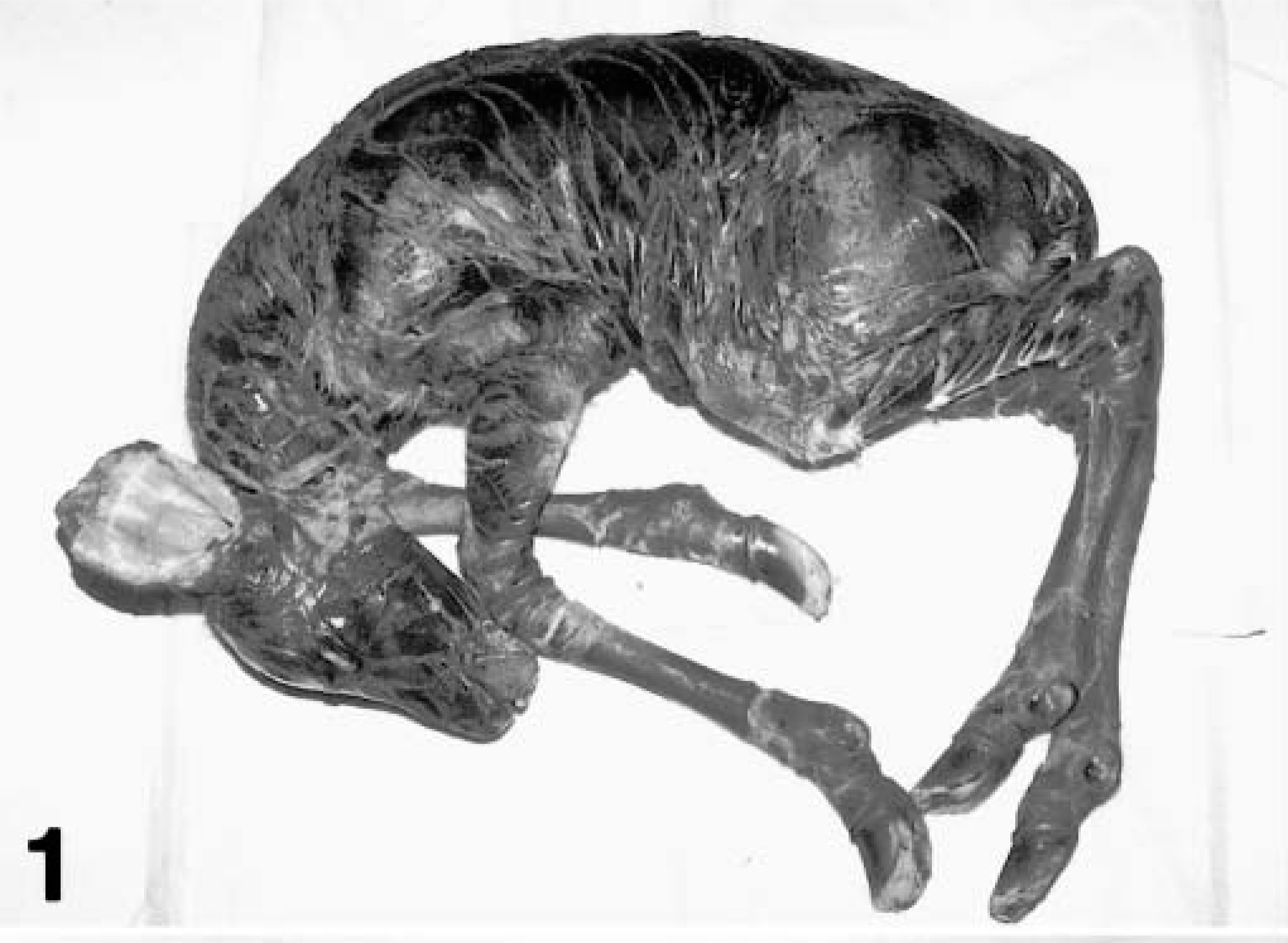
Whole body; greater kudu calf, kudu No. 2. Harlequin ichthyosis. Over eighty percent of the body is covered with scaly plates.

Ventral abdomen; greater kudu calf, kudu No. 2. Harlequin ichthyosis. Scaly plates are thickened and fissured.
Postmortem examination revealed a 9.4-kg male neonate that was fully haired underneath the gray/brown plate carapace. On internal examination a hemorrhagic patent urachus was filled with clear yellow urine. Testes were located inguinally. In the thorax a patent ductus arteriosis measured 6 mm in diameter. Excluding the skin lesions, all gross findings were consistent with the age of the animal. All other organs appeared within normal limits at necropsy.
Tissues from both calves were fixed in 10% neutral buffered formalin for histopathologic examination and included skin, adrenal glands, liver, kidneys, spleen, lung, heart, and esophagus. Pancreas, trachea, and placenta were also evaluated in kudu No. 1, and pituitary gland, urinary bladder, thyroid glands, testes, tongue, reticulum, omasum, abomasum, small intestine, large intestine, and brain were evaluated in kudu No. 2. Skin collection sites included the forehead, neck, and lip in kudu No. 2 but were not recorded in kudu No. 1. Tissues from a similarly aged, half sibling (same sire) kudu calf, that had died in 1999 from trauma and sepsis, were evaluated for comparison with the two affected individuals. All tissues were stained with hematoxylin and eosin.
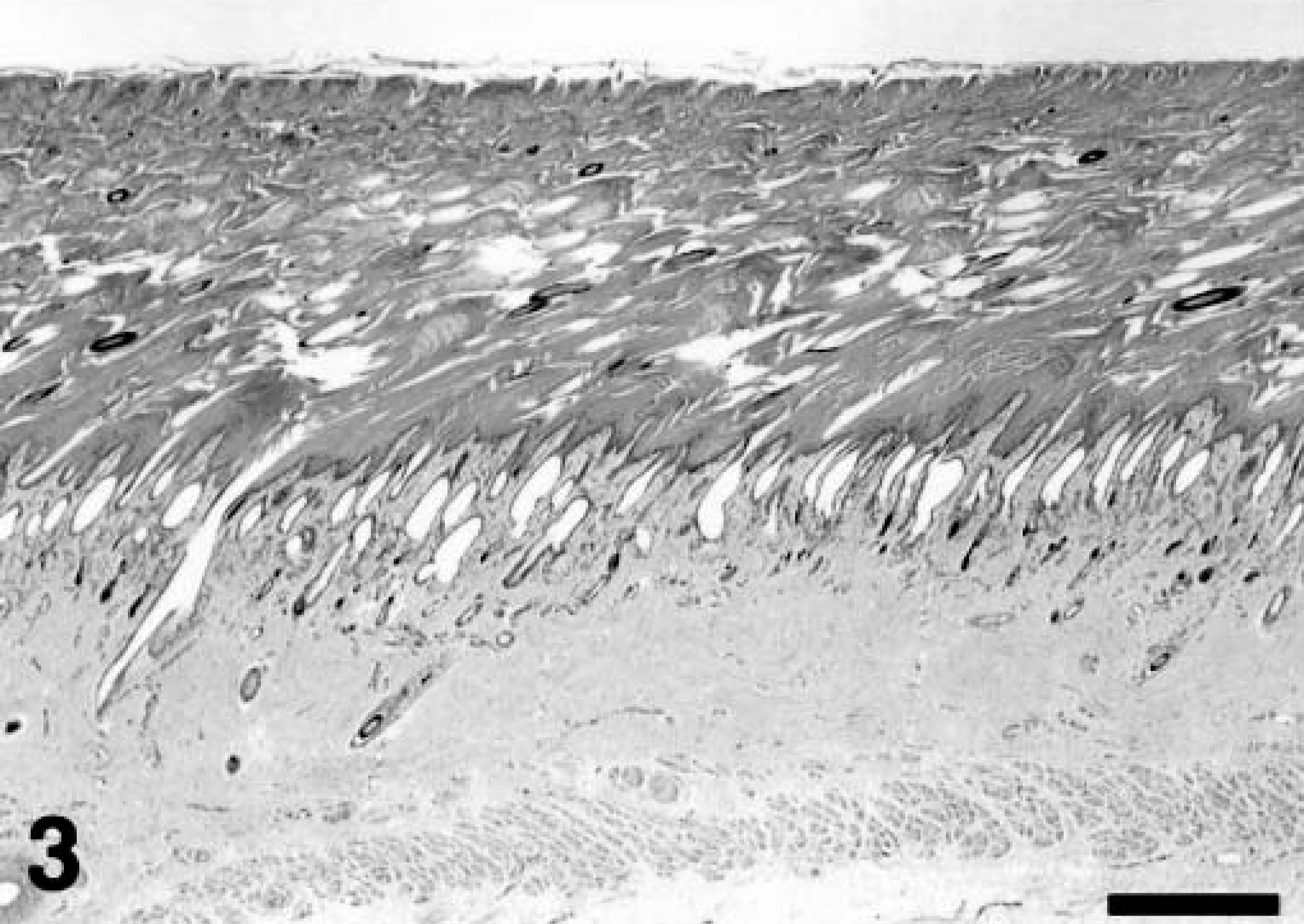
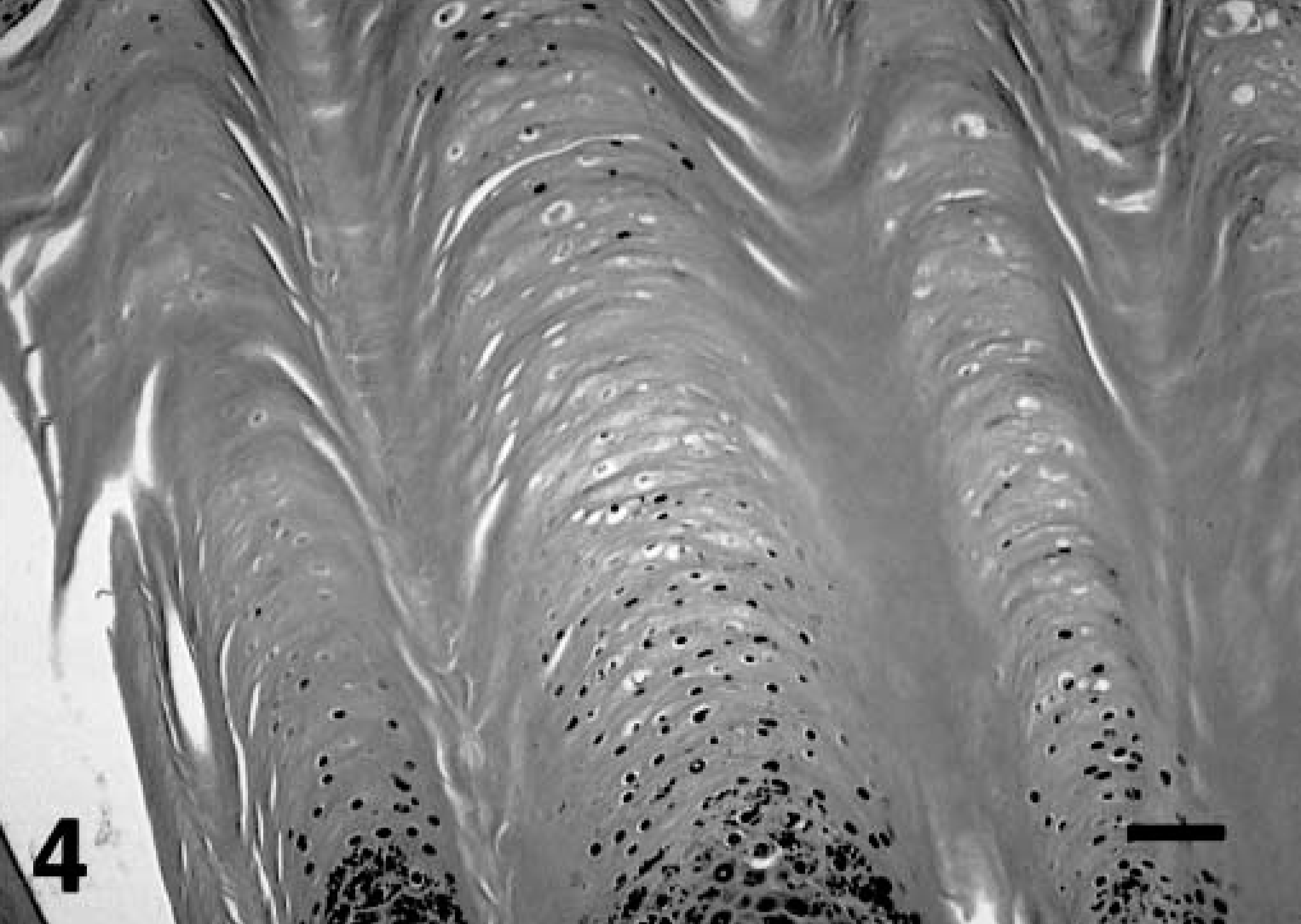
Histologic examination of the skin in both calves showed moderate to marked diffuse hyperkeratosis with no associated inflammation (Figs. 3, 4). In kudu No. 1, dense parakeratosis was predominant, whereas in kudu No. 2, lamellar orthokeratosis and occasional parakeratosis were seen. These thick layers of the stratum corneum covered a stratified squamous epithelium and dermis of normal thickness. In kudu No. 2, occasional keratinocytes exhibited clear, vacuolated cytoplasm. Numerous hair follicles were markedly dilated but otherwise appeared histologically normal. Occasional hair shafts were found embedded in the thick stratum corneum. No significant histologic lesions were noted in any other tissues evaluated.
Skin; greater kudu calf, kudu No. 2. Severe laminated orthokeratosis. Hematoxylin and eosin staining method. Bar = 175 μm.
Skin; greater kudu calf, kudu No. 2. Severe laminated orthokeratosis and occasional parakeratosis with vacuolated cytoplasm in keratinocytes. Hematoxylin and eosin staining method. Bar = 40 μm.
Formalin-fixed skin samples were further evaluated by electron microscopy. Tissues were excised, cut to 1 mm thickness in at least one dimension. After two rinses in 0.1 M sodium phosphate buffer (pH 7.2), samples were placed in 1% osmium tetroxide in the same buffer for 1 hour at room temperature. Samples were rinsed two times in distilled water and dehydrated in an ethanol solution series culminating in two changes of 100% acetone. Tissues were then placed in a mixture of Spurr resin26 and acetone (1:1) for 30 minutes, followed by 2 hours in 100% resin with two changes. Finally, samples were placed in fresh 100% resin in molds and polymerized at 70 C overnight. Semithin (0.25–0.5 μm) sections were cut with glass knives and stained with 1% o-toluidine blue in 1% sodium borate. Ultrathin (70–90 nm) sections were cut with a diamond knife, stained with methanolic uranyl acetate followed by lead citrate, and examined with a transmission electron microscope.
Electron microscopy of the epidermis in both cases showed abnormal structures in the stratum granulosum and stratum corneum. Despite formalin fixation that could result in some artifactual change, no distinct lamellar body structures were seen in the ultrastructural evaluation of either calf. In addition, large membrane-bound perinuclear vesicles filled predominantly with amorphous or granular material were found (Fig. 5). Because of their membrane-bound structure and position adjacent to the nucleus, they were interpreted as dilated endoplasmic reticulum. Similar structures were not evident in the nonaffected kudu calf (not shown). Within the stratum corneum of affected calves multiple layers of dead cells were seen, and many cornified keratinocytes contained retained organelles or vesicles (Fig. 7) that were heterogeneous in size and content and were not observed in normal tissue (Fig. 6). The unaffected calf exhibited a few thin layers of compact corneocytes (Fig. 6) and cells in transition from the granular layer.
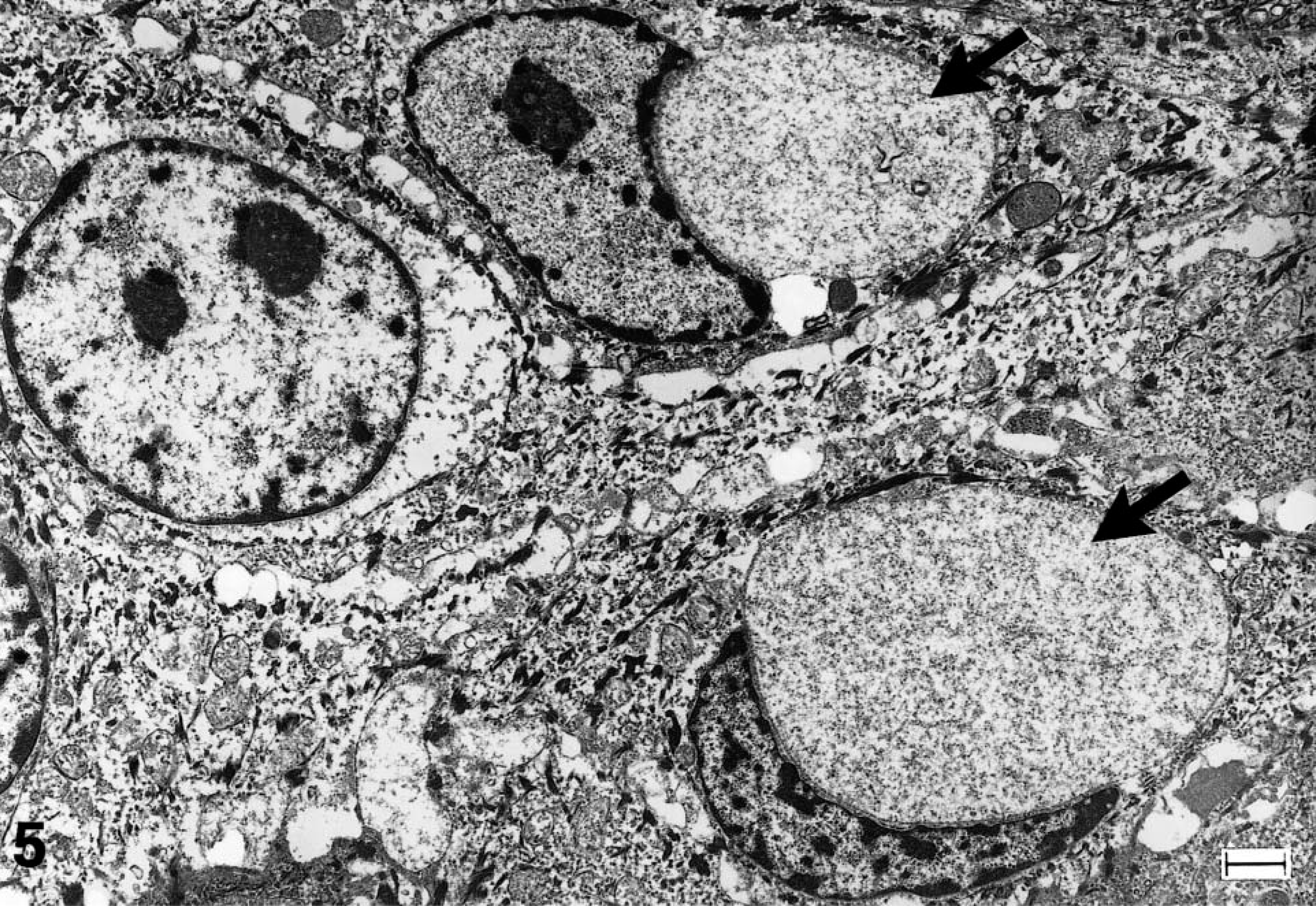
Epidermis, stratum granulosum; greater kudu calf, kudu No. 2. Several cells containing keratin filaments, mitochondria, desmosomes, and keratohyalin. Large vesicles (black arrows) interpreted as dilated endoplasmic reticulum lie adjacent to the nuclei. Methanolic uranyl acetate and lead citrate staining method. Bar = 1 μm.
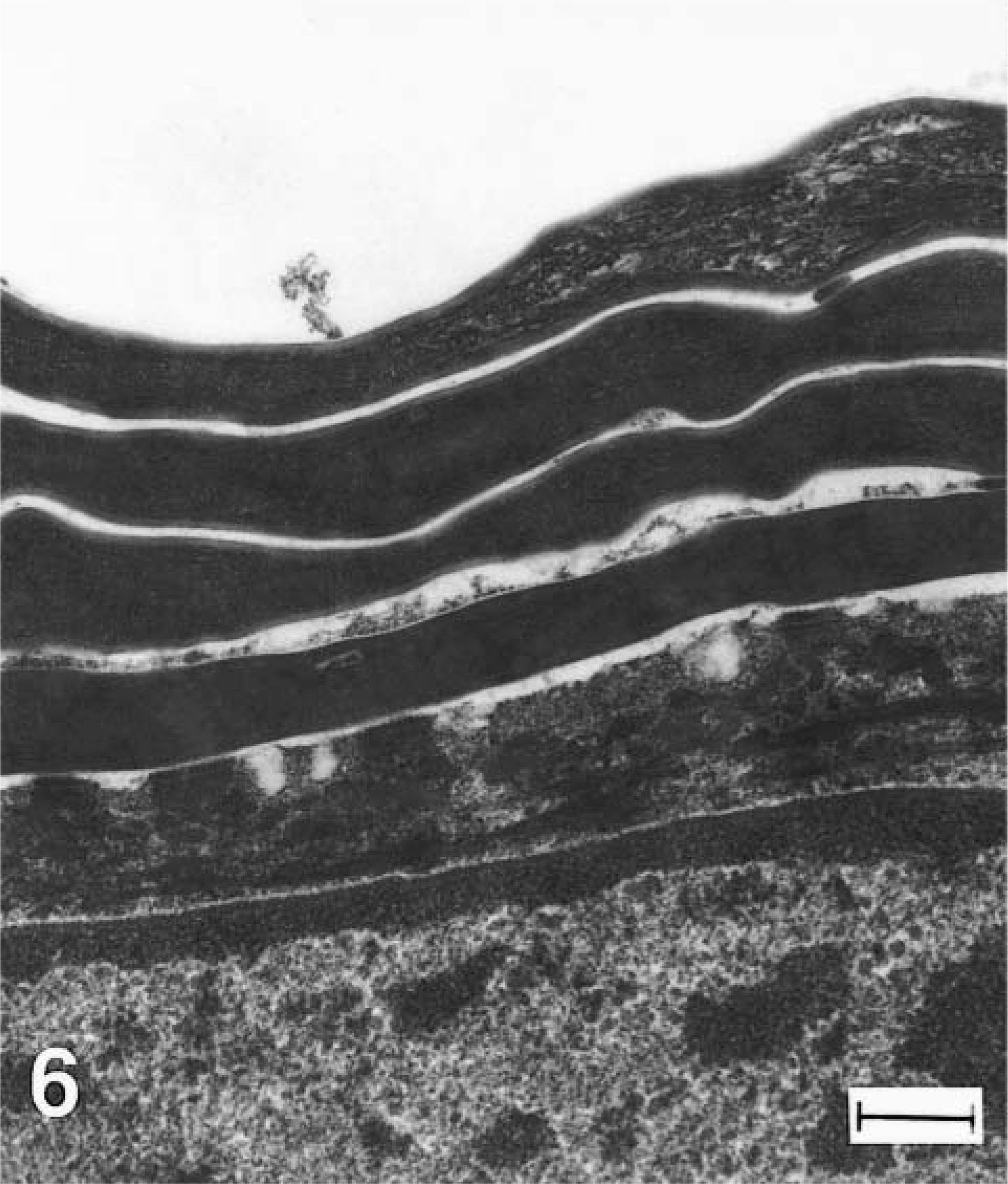
Epidermis, stratum corneum with cell in transition from stratum granulosum, greater kudu calf, unaffected. Note dense homogeneous structure of several layers of cornified cells superficial to transition cells that retain traces of keratohyalin (arrows). Methanolic uranyl acetate and lead citrate staining method. Bar = 300 nm.
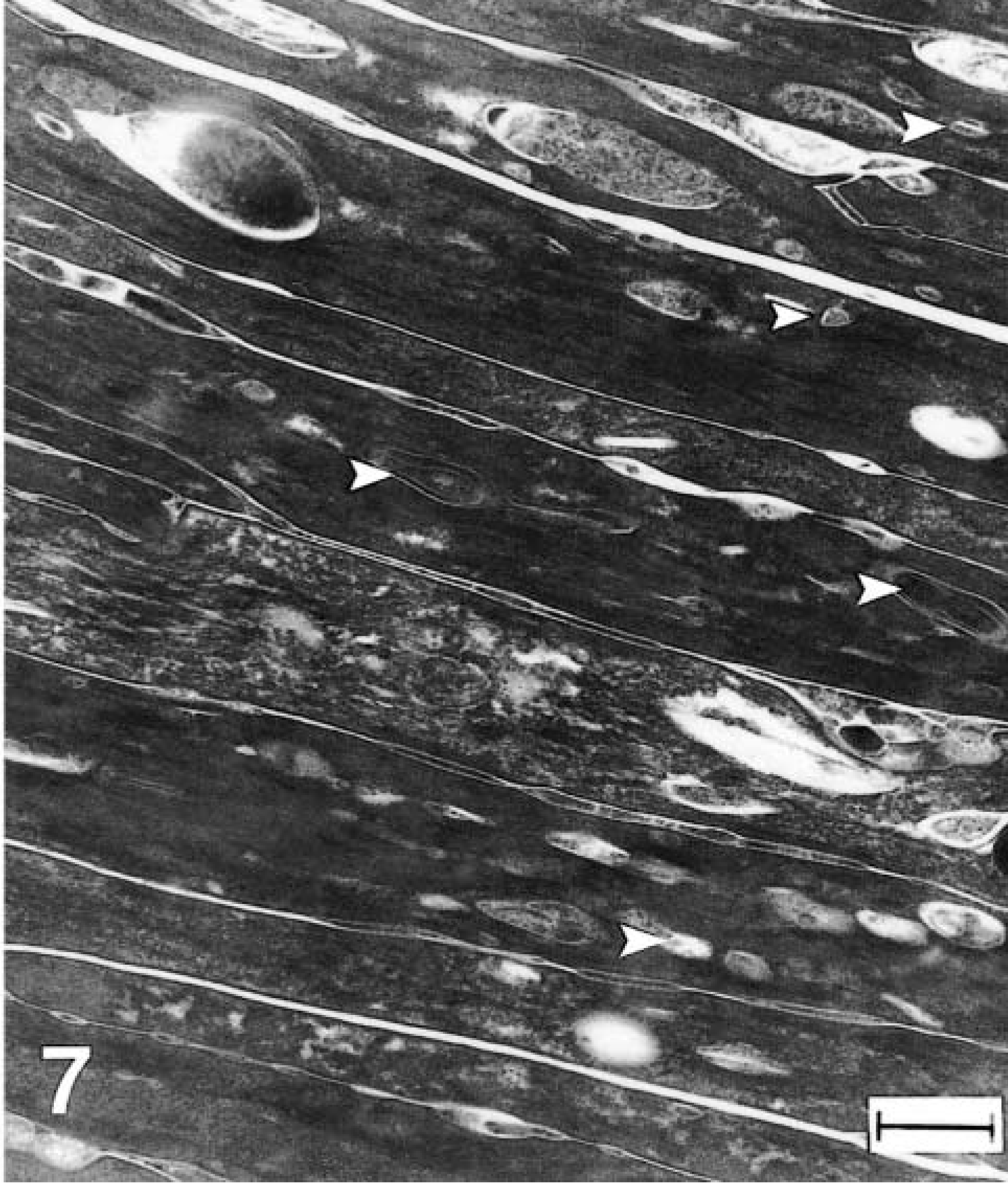
Epidermis, stratum corneum, greater kudu calf, kudu No. 1. Retention of dead cells and cellular debris. Cornified cells of the affected skin contain many retained organelles or vesicular structures of heterogeneous size (arrowheads) that are not seen in normal tissue. Methanolic uranyl acetate and lead citrate staining method. Bar = 300 nm.
The kudu calves in this report displayed gross, histopathologic, and electron microscopic signs consistent with an ichthyosis, a group of rare genodermatoses known to occur in several forms in cattle, dogs, pigs, mice, llamas, and humans.1,7,8,20,23,27,28 Human ichthyoses represent multiple diseases with different underlying genetic defects, although all result in scaly skin as the name suggests, derived from the Greek word for fish. Harlequin ichthyosis (HI), characterized by the accumulation of dense scale at birth, is considered to be the most severe of the nonbullous recessive congenital ichthyoses.1 Unlike lamellar ichthyosis, another recessive, though less severe, ichthyosis resulting from a defect in the epidermal transglutaminase critical for the formation of the cornified envelope, the genetic defect for HI is not known.1,15 A mouse model of HI has been described with many of the features seen in human HI.27
Variable clinical presentations and poorly defined pathogeneses complicate the classification of ichthyoses in animals.28 Currently, two congenital forms of ichthyosis are described in artiodactylids, ichthyosis congenita and ichthyosis fetalis.28 Bovine ichthyosis congenita is the milder disorder of the two, characterized by hyperkeratosis apparent at birth or developing over several weeks. This condition results in variable hair covering, from alopecia to hypertrichia, and large-scale formation predominating over the limbs, abdomen, and muzzle.7,28 Cataracts, microtia, and thyroid abnormalities have been reported in Pinzgauer calves affected with this disorder.28 Bovine ichthyosis congenita most closely resembles lamellar ichthyosis of humans.
Ichthyosis fetalis, the more severe form of bovine ichthyosis, results in abortion or neonatal mortality within a few days of birth.7,28 It appears similar to HI, as described in humans and mice, in that both disorders result in severe platelike scale formation diffusely over the body and early neonatal death.1,27 Distinguishing morphologic features of harlequin ichthyosis include the presence of abnormal lamellar bodies on electron microscopic examination of the epidermal granular layer and retention of vesicles in the stratum corneum,14,16,17,22 whereas parallel ultrastructural characteristics of bovine ichthyosis fetalis are yet to be determined. In this report, gross, histopathologic, and electron microscopic findings in two greater kudu calves showed several similarities with those reported with HI in humans.1,12
Clinically, the kudus' severe, diffuse, thick, carapacelike plates and gross periocular abnormalities were consistent with HI, characterized in newborn infants by severe diffuse scaling, thickened platelike scales, eclabium, and ectropion.1 Hyperkeratosis extended over 80% of the cutaneous surface in both calves and resulted in eversion of mucocutaneous junctions and curling of ear pinnae in kudu No. 2. Furthermore, these plates were presumed to restrict normal development and extension of the legs in kudu No. 2, likely resulting in the valgus deformities of the tarsal joints and the calf's inability to stand or nurse postpartum. Lack of musculoskeletal histopathology from the limbs of this animal, however, does not allow us to preclude the possibility of musculoskeletal disease contributing to the leg abnormalities.
Extensive hyperkeratosis with variable degrees of parakeratosis or orthokeratosis have been described in human HI and have been suggested to be the result of topographic variations in the skin.14 Varying amounts of parakeratosis and orthokeratosis were present in both cases of this study. Epidermal or dermal inflammations were not noted in either kudu, though chronic dermatitis has been reported occasionally in humans.10,22 Other histologic descriptions of human HI include follicular hyperkeratosis, changes in keratohyalin granules, and occasional papillomatosis and acanthosis, although these features are not universally diagnostic for human HI.6,11,12,16,22 None of these histopathologic features were noted in either affected kudu calf. Although no follicular hyperkeratosis sensu stricto was seen in these cases, the infundibuli of hair follicles often were distended, and hair fibers extended throughout the hyperkeratotic stratum corneum. Mice with HI show proliferation of anagen hair follicles in the dermis, but only a few weak or broken hair fibers emerge from affected skin.27 Hair follicles emerge postnatally in mice, but prenatally in kudu, possibly accounting for this difference. Profound abnormalities in hair follicle cycling stages were not noted in these kudu calves, but we cannot rule out that follicular cycling defects could be present.
The distinct ultrastructural findings observed herein distinguish these kudu calves from those affected with the other ichthyotic diseases described in the veterinary literature. The hallmark of HI in humans and mice is the presence of abnormal lamellar bodies, which are lacking internal structure and are variably sized in the granular layer of the epidermis.12,14,17,27 Additionally, there are large numbers of retained vesicular structures in the cytoplasm of stratum corneum of HI patients. Absence of normal lamellar bodies, retention of debris and multifocal vesicles in the stratum corneum in both affected kudu calves were consistent with findings in human and mouse HI cases.4,13,27 One distinguishing ultrastructural feature of this disease in both calves was the presence of large perinuclear structures in the granular layer, possibly representing abnormally dilated endoplasmic reticulum. Other ultrastructural features of HI in humans and mice can include focal absence of intercellular lipid lamellae,5,9,12,13 large mitochondria with vesicular changes,4,16,17,27 reduced size or distribution of keratohyalin granules,27 and fewer tonofilaments in the granular layer.9,11 None of these findings, however, were noted in the kudu cases in this report. Lack of these findings could indicate difficulties with formalin fixation and loss of cell membranes and detail, or could indicate the unique ultrastructural changes of this particular form of harlequin ichthyosis in greater kudu or artiodactylids.
The definitive inheritance pattern for HI is generally considered to be autosomal recessive, though some have suggested that dominant mutations may also result in a HI phenotype.1,2,13,23 The calves in this report shared the same sire, and the dam of kudu No. 2 was also the offspring of the same sire. Such inbreeding may have contributed to the manifestation of congenital disease and supports the hypothesis of an autosomal recessive inheritance pattern in these cases.
The molecular genetic pathogenesis for HI is currently under investigation. Lack of normal fusion and extrusion of contents from lamellar bodies could result in alteration of the lipid and proteolytic chemical content of epidermal intercellular spaces, increased cohesion of keratinocytes, and reduced desquamation.13,22 Abnormal lamellar body function could be mediated by enzymatic, lipid, or genetic defects.22 Defects in lipid biosynthesis as well as the conversion of profilaggrin to filaggrin in the epidermis of human HI patients have been identified.2,9,12,13,18 In vitro studies of keratinocytes from human HI patients show decreased epidermal protein phosphatase 2A (PP) activity, which could affect both lipid and profilaggrin pathways and ultrastructural findings of the disease.18 In mice models with HI, overexpression of keratin 6 and filaggrin in the epidermis has been linked to a mutant gene locus on mouse chromosome 19.27 Alternatively, impaired retinoic acid metabolism could explain poor differentiation of keratinocytes in HI. Systemic retinoid treatment of infants affected by HI or other severe inherited keratinization disorders results in exfoliation of excess keratin and improvement of skin condition.19,24,25 Recently, abnormalities in epidermal calcium-mediated signal transduction have also been considered as possible pathogeneses for HI. Reduced concentrations of calpain I, a calcium-activated neutral protease in the basal layer of the epidermis, could indicate such altered signaling events in fetal skin development of HI patients.21
Three subtypes of HI have been described in humans and a fourth HI-like syndrome has been reported in mice.12,27 The variable expression of structural proteins as well as the differing ultrastructural characteristics of the epidermis in HI patients demonstrate site-specific differences or a possible heterogeneity of this disease.3,12,27 More than one genetic defect could cause abnormal lamellar body function, keratinocyte development, or epidermal differentiation, or desquamation that could result in the HI phenotype. In this report, the cause of the ichthyosis in the kudu calves was not determined. The historical, gross, histopathologic, and electron microscopic findings, however, are consistent with the recessive congenital dermatosis, HI.
Footnotes
Acknowledgements
We thank Drs Mark Cesta, David Malarkey, P. Blackshear, F. Muntz, Michael Loomis, North Carolina State University's histopathology laboratory staff, the North Carolina Zoo veterinary technicians, and the African Plains exhibit keepers for their assistance with these cases. We thank Dr Michael Dykstra and the North Carolina State University's Laboratory for Advanced Electron and Light Optical Methods for their help with electron microscopy and photography, and Dr Norman Barlow and the Biomedical Communications Department for their assistance with photographic materials. We also thank Drs Norman Barlow and Michael Stoskopf for their help with manuscript review. Funding was provided by the North Carolina Zoological Society and the North Carolina State University College of Veterinary Medicine.
