Abstract
Testicular gonadoblastoma is an uncommon tumor in domestic animals. The current study describes a testicular gonadoblastoma in 2 pet domestic rabbits (Oryctolagus cuniculus domesticus) based on the histomorphological, immunohistochemical, and ultrastructural characteristics of the tumor. The tumor tissue consisted of discrete tubular structures separated by a fibrous stroma. The tubular structures were composed of large round cells similar in appearance to germ cells in the seminiferous tubules, and small spindle cells with oval or elongated nuclei resembling Sertoli cells. The spindle cells showed peculiar structural patterns arranged in a coronal or follicular pattern, often forming Call–Exner bodies like those in an ovarian granulosa cell tumor. One case was concomitant with seminoma. Immunohistochemistry revealed large round cells that were positive for c-kit and placental alkaline phosphatase, while spindle cells were positive for vimentin and Wilms tumor protein. Ultrastructurally, the spherical eosinophilic material (Call–Exner body) consisted of duplicated basal lamina, and sex cord components surrounded a single degenerative cell similar to a germ cell.
There are only a few reports about tumors arising in the testes of rabbits, one of the most common of which is interstitial (Leydig) cell tumor.4,5,12,24 Other types include seminoma,1,2 teratoma, 13 adenocarcinoma, 8 granular cell tumor, 9 and mixed cell-type tumors.15,22 A gonadoblastoma (an uncommon tumor in the gonads of animals) is histologically defined as a tumor composed of 2 principal cell types: germ cell components similar to those of seminoma, and sex cord components resembling Sertoli cells and granulosa cells. Most gonadoblastomas were reported to occur in dysgenetic gonads or undescended testes in human beings, 19 and can be rarely found even in apparently normal ovaries and testes.3,19 A gonadoblastoma assumes an “in situ” form as a malignant germ cell neoplasm, 16 and sometimes develops into high-grade invasive malignant tumors such as seminoma, embryonal carcinoma, yolk sac tumor, endodermal sinus tumor, choriocarcinoma, or teratoma.7,16,18,23 Among animals, gonadoblastoma has been reported in 2 dogs. The first had developed in the differentiated testis of a phenotypic male, 20 while the second was a bilateral gonadoblastoma with gonadal dysgenesis. 14 The current study describes a gonadoblastoma in 2 rabbits and the histological, immunohistochemical, and ultrastructural findings of this neoplasm.
The subjects of the present report were 2 male pet domestic rabbits (Oryctolagus cuniculus domesticus) of an unknown breed, and sexually intact based on the external appearance of penis and testes. The rabbits were 6 (case no. 1) and 10 (case no. 2) years of age. In both cases, enlargement of the left testicle was presented in the scrotum. Because of suspected testicular neoplasia, a bilateral orchiectomy was performed. All surgically resected samples were fixed in 10% neutral buffered formalin, and submitted to the authors’ laboratories for histopathologic examination. The testicular tissues were routinely processed, and embedded in paraffin. Sections were cut at 4 mm and were stained with hematoxylin and eosin and a periodic acid–Schiff (PAS) reaction. Immunohistochemically, c-kit a and placental alkaline phosphatase (PLAP) b served as the primary antibodies for germ cell markers. Vimentin a and Wilms tumor protein a (WT1) were used as Sertoli cell markers. A universal immunoenzyme polymer method c was used for immunoreaction. Normal rabbit testicle was used as a positive control. For a negative control, normal rabbit immunoglobulin G (IgG) or mouse IgG was used for each primary antibody. For electron microscopy, small formalin-fixed specimens of tumor from case no. 1 were fixed with 2.5% glutaraldehyde, postfixed in 2% osmium tetroxide, and embedded in resin. Ultrathin sections were cut and stained with uranyl acetate and lead citrate. Specimens were examined and photographed with a transmission electron microscope. d
Grossly, the enlarged left testicle of case no. 1 was approximately 1.7 cm × 1.7 cm × 1.7 cm, globular, and firm. The cut surface was equally gray-white in color and was flat and smooth in all its parts (Fig. 1). The right testicle was 1.5 cm × 0.5 cm × 0.5 cm and appeared to be atrophic. The enlarged left testicle of case no. 2 was approximately 2.5 cm × 2.0 cm × 2.0 cm and roughly globular in form. The cut surface was dark-brown and was almost solid with a partly cystic area. The right testicle was 1.7 cm × 0.6 cm × 0.6 cm and appeared to be atrophic.
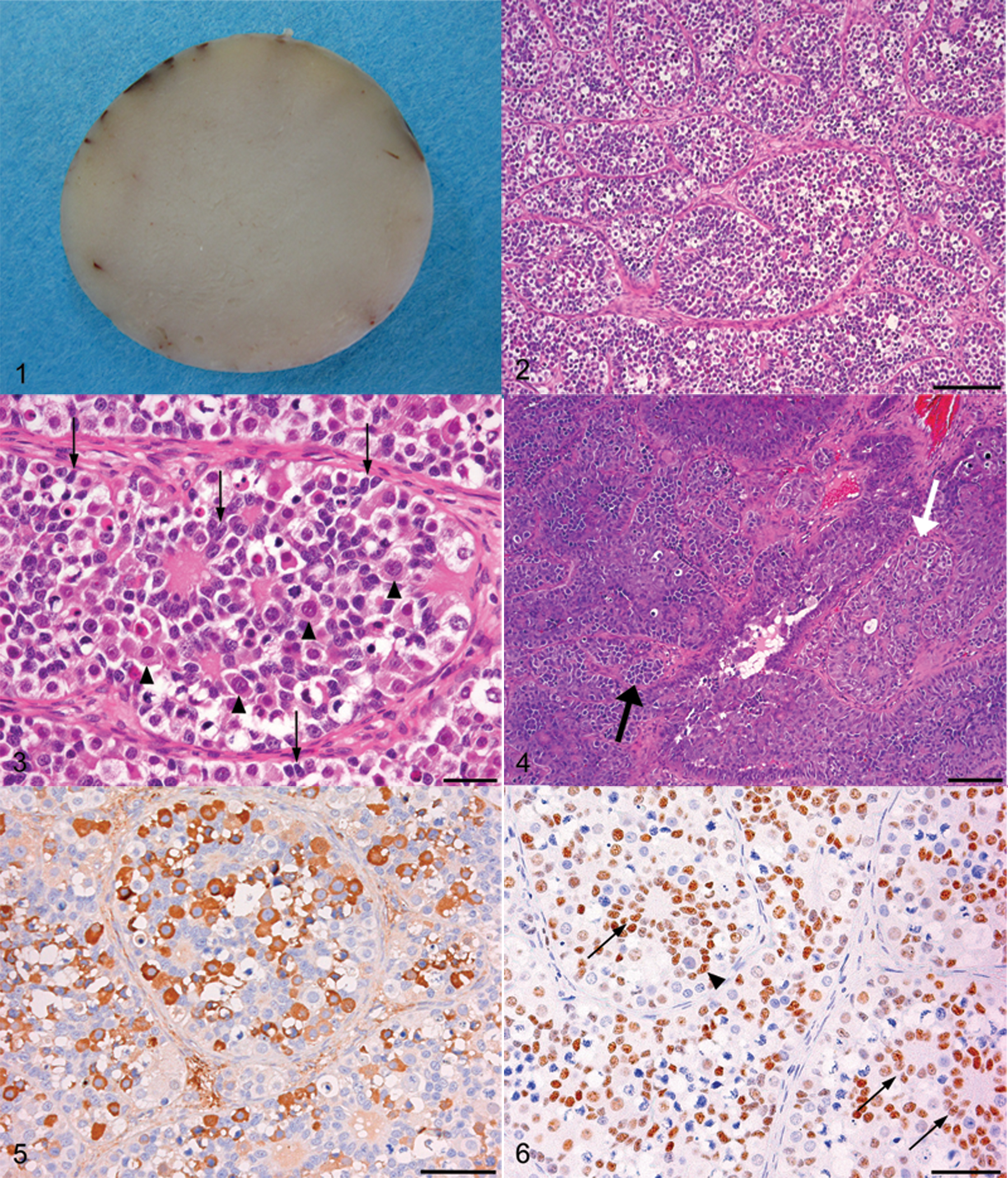
Rabbit, gonadoblastoma, case no. 1. The sectioned surface is gray-white in color, flat, and smooth.
Histologically, the enlarged testicles of both cases were almost entirely replaced by tumoral tissue consisting of discrete tubular structures surrounded by a thin fibrous stroma (Fig. 2). The tubular structures were composed of an intermixture of large round cells similar to germ cells and small spindle cells with oval or elongated nuclei resembling Sertoli or granulosa cells (Fig. 3). The spindle cells were mainly located on the periphery of the tubular structures forming a peripheral palisade surrounded by basement membrane (coronal pattern). Spindle-shaped tumor cells arranged in a follicular pattern often contained spherical eosinophilic material at their center (Call–Exner bodies). The spherical eosinophilic materials were PAS-positive. Tumor cells in the follicular pattern less frequently contained large round cells resembling germ cells instead of eosinophilic materials, assuming an appearance similar to the follicular epithelium of normal primary follicles (follicular pattern). Degenerative germ cells or cell debris were observed in the eosinophilic materials or in the center of the follicular structure. The large round cells (germ cell components) had abundant clear cytoplasm and round, vesicular nuclei with prominent nucleoli. The spindle cells (sex cord components) had scant but clear cytoplasm and small oval or elongated nuclei with indiscernible nucleoli. Mitotic figures were occasionally observed in the germ cell components, but were not detected in the sex cord components. In all cases, aggregates of Leydig cells were often present within the intertubular stroma. The Leydig cells were non-neoplastic and lacked crystals of Reinke. In case no. 2, germ cell components predominantly proliferated in some areas of gonadoblastoma. In the area with mild proliferation of germ cell components, tubular structure and Call–Exner bodies remained (Fig. 4), but in the severely proliferating area, they were entirely replaced by a diffuse sheet-like proliferation of cells resembling those of seminoma. From the morphologic features, the tumor was diagnosed as a seminoma arising in the gonadoblastoma. The contralateral testicle of case no. 1 was atrophic and consisted of small-sized seminiferous tubules lined only with Sertoli cells, with a thickened basement membrane and interstitial fibrosis. The testicle of case no. 2 was also atrophic with a small nodule of interstitial (Leydig) cell tumor. Immunohistochemically, the germ cell components were positive for c-kit (Fig. 5) and PLAP, whereas the sex cord components were positive for vimentin and WT1 (Fig. 6).
Electron microscopic examination revealed that tumor cells of the germ cell component had abundant cytoplasm with a few organelles and large round nuclei with dispersed chromatin and prominent nucleoli. Tumor cells of the sex cord component had a primarily scanty cytoplasm containing many mitochondria and irregular oval-shaped nuclei with few or small nucleoli. Spherical material surrounded by sex cord components consisted of electron-dense duplicated basal lamina and somewhat fewer electron-dense amorphous granular architectures (Fig. 7). Sex cord components also surrounded a single degenerative cell similar to a germ cell. On the basis of histological, immunohistochemical, and ultrastructural findings, testicular tumors in the present 2 cases were diagnosed as a gonadoblastoma (case no. 1) and a gonadoblastoma with seminoma (case no. 2).
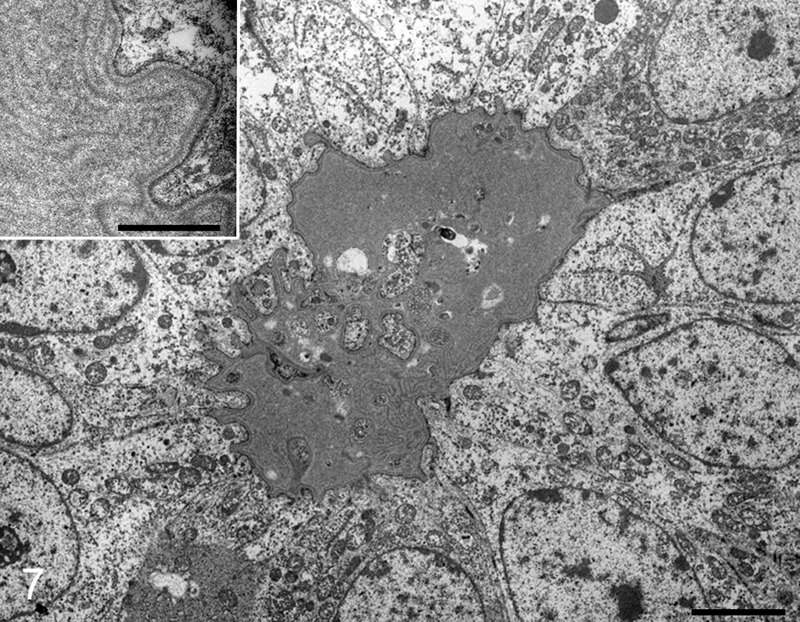
Rabbit, gonadoblastoma, case no. 1. Spherical material is surrounded by sex cord components. Bar = 2 µm. Inset: higher magnification of the spherical material, which consisted of electron-dense duplicated basal lamina and slightly less electron-dense amorphous granular architectures. Bar = 0.25 µm. Electron micrograph.
Histopathologically, differential diagnosis of gonadoblastoma included a mixed germ cell–sex cord–stromal tumor (MGSST), a seminoma, and sex cord tumor with annu- lar tubules (STAT). MGSST generally lacks discrete tubular structures like those of gonadoblastoma, and usually exhibits proliferative activity in the sex cord component unlike that in a gonadoblastoma, since germ cells are thought to be non-neoplastic and enveloped by a neoplastic sex cord–stromal tumor in MGSST other than a gonadoblastoma. The 3 typical patterns of sex cord elements within the discrete tubules (i.e., the coronal, follicular, and formation of Call–Exner bodies) are most characteristic and pathognomonic for the diagnosis of gonadoblastoma. In a gonadoblastoma, more than 1 growth pattern of neoplastic cells is usually found in an individual tubule. 19 The presence of these patterns supports the diagnosis of gonadoblastoma in the cases in the present study. Seminoma lacks a sex cord component and discrete tubular structures as well as some focal infiltrated lymphocytes. STAT has a growth pattern similar to gonadoblastoma and contains eosinophilic amorphous bodies and, frequently, calcified material, but lacks a germ cell component. Histopathologic features of the present cases were conclusively distinguished from the morphologic characteristics of these tumors.
The tumor from case no. 2 was judged a malignant tumor due to its coexistence with seminoma, which is the most common form of malignant germ cell tumor arising in gonadoblastomas and occurs in more than half the tumors in human beings. 19 Because the tumor from case no. 1 showed no evidence of an invasive germ cell tumor, this case was considered as a true gonadoblastoma with a benign outcome. 19 To the authors’ knowledge, there were no previous reports among rabbits of a pure gonadoblastoma or a gonadoblastoma with seminoma.
In normal ovarian follicles, under an electron microscope, many species exhibit a Call–Exner body distinguished by the presence of a basal lamina–like structure.6,21 It has been reported that some gonadal neoplasms form a Call–Exner body.14,19,20 Electron microscopical features of Call–Exner bodies are reported to be composed of a moderately electron-dense fibrillar material and a slightly less electron-dense granular material.6,10,20 In case no. 1, spherical eosinophilic material under light microscopy was composed of an electron-dense duplicated basal lamina and a somewhat less electron-dense amorphous granular architecture. These 2 structures were homologous to those found in close proximity to the follicular basal lamina of a rabbit. 6
A Call–Exner body surrounded by sex cord components reportedly indicates areas of the sequestration of products of necrosis and degeneration. 11 Under light and electron microscopy, sex cord cells showed a rosette arrangement surrounding a single degenerative cell resembling a germ cell (Fig. 6). This phenomenon resembles the physiological function by which apoptotic spermatogenic cells are phagocytosed by Sertoli cells in normal testicles, suggesting that sex cord components may have a tendency to assemble in the area of cell dissolution, not just in a Call–Exner body.
Gonadoblastoma typically occurs within dysgenic gonads in children or young adults. Approximately 80% of cases occur in phenotypic females, and most of the remaining 20% are in phenotypic male pseudohermaphrodites. 17 In more than 90% of patients with gonadoblastoma, a Y chromosome or fragment can be identified, 19 though it has also been found in the testes of normal men.3,19 The rabbits in the current study showed no clinical symptoms suggesting sex hormone abnormality such as alopecia, feminization, or cryptorchism, and were apparently sexually intact. The 2 cases also involved no dysgenic gonads. The atrophy of opposite testicle in these cases under a light microscope is regarded as a senile change, since an atrophic change ordinarily occurs in aging adults after the normal development of their genital organs. Therefore, the rabbits in the current study were judged to be normal adults. Consequently, the cases are considered very rare for 2 reasons. One is that the cases are the first report of a testicular gonadoblastoma in rabbits, and the other is that the cases have no gonadal dysgenesis.
Footnotes
Acknowledgements
The authors wish to thank Chihiro Azuma and Tadashi Noda for their technical help with the electron microscopic examinations.
a.
Dako Japan Inc., Tokyo, Japan.
b.
Zymed Laboratories Inc., South San Francisco, CA.
c.
N-Histofine Simple Stain Max PO (M) or (R), Nichirei Corp., Tokyo, Japan.
d.
JEM-1400, JEOL Ltd., Tokyo, Japan.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
