Abstract
Gastrointestinal stromal tumors (GISTs), leiomyomas, and leiomyosarcomas are common mesenchymal neoplasms in the gastrointestinal (GI) tract of dogs. As previously diagnosed smooth muscle tumors of the canine GI tract are increasingly reclassified as GISTs, it becomes important to identify additional criteria that may assist in the diagnosis of these neoplasms, provide prognostic information, and offer targets for therapy. Examination of cluster of differentiation (CD), molecule expression (such as KIT [CD117] and CD34) as well as gross, histologic, and immunohistochemical features (such as tumor size, tumor location, mitotic index, AgNOR, and Ki67 labeling) in human GISTs has revealed new and valuable prognostic, diagnostic, and therapeutic information. In this study, GISTs were examined for the gross, histologic, and immunohistochemical features listed above. Forty-nine cases of canine gastrointestinal mesenchymal neoplasms from the Animal Medical Center (New York, NY) were categorized as GISTs (KIT positive), leiomyosarcoma/leiomyoma (KIT negative, smooth muscle actin [SMA], and/or desmin positive), or other (KIT, SMA, and desmin negative). A proportion (55%) of canine cases previously diagnosed as smooth muscle tumors were reclassified as GISTs according to KIT immunoreactivity. Statistical correlations with survival data were not possible because of insufficient follow-up data. However, there was a significant difference between mitotic index, AgNOR, and Ki67 scores depending on the location of the tumor (small vs large intestine). This study represents the first time CD34 immunoreactivity has been demonstrated in canine GISTs.
Keywords
Primary gastrointestinal (GI) mesenchymal neoplasms in the dog include histologically similar entities such as leiomyomas, leiomyosarcomas, and gastrointestinal stromal tumors (GISTs). GISTs have been reported in humans, dogs, horses, and nonhuman primates. 4,23,24 Canine GISTs usually occur in older dogs, most commonly in the small intestine and colon. 4,23 In contrast, the majority of GISTs in humans occur in the stomach. 6,7,16,17,28 The clinical behavior of canine GISTs ranges from benign to malignant with metastases to the liver, spleen, mesentery, or mesenteric lymph nodes. 4
GISTs are hypothesized to derive from the interstitial cells of Cajal (ICCs). 7,16,21,28 These are pacemaker cells of the GI tract found within the wall between the neural plexi (Auerbach’s plexus, deep muscular plexus, and submuscular plexus) and smooth muscle, as well as between smooth muscle layers. ICCs are KIT positive with a subset that is also CD34 positive. 18,21,28 GISTs that are KIT positive and CD34 positive may derive from this double-positive subset of ICCs. 21 In contrast, leiomyosarcomas and leiomyomas typically lack both KIT and CD34 expression. 6,23 No previous studies on CD34 immunoreactivity of canine GISTs have been published because of lack of reactivity in canine tissues.
Histologically, GISTs are nonencapsulated, cellular masses composed of streams and short interlacing bundles of spindle to epithelioid cells primarily involving the smooth muscle of the tunica muscularis. These tumors can extend transmurally and form smooth to multinodular exophytic serosal masses. GISTs can exhibit a range of mitotic activity. 15 –17 Differentiation between GISTs and leiomyomas/leiomyosarcomas based on morphology alone is often difficult.
Recent developments in human classification of GI mesenchymal neoplasms have led to several reports of reclassification of similar GI neoplasms in dogs. 4,13,23 Previously diagnosed leiomyosarcomas and leiomyomas were found to exhibit immunoreactivity and genetic and ultrastructural properties more consistent with GISTs than smooth muscle neoplasms. 4,13,23 Previous studies have differentiated between mesenchymal tumors of smooth muscle origin and GISTs by use of immunohistochemical labeling, specifically by demonstrating KIT (CD117, c-kit) expression as the distinguishing feature of GISTs. 13,16,17,23 A majority (up to 60–80%) of human GISTs also exhibit CD34 immunoreactivity. 18,24,28 Double-positive reactivity to KIT and CD34 was also demonstrated in a case study of a GIST in a chimpanzee. 24 CD34 is a cell-surface glycoprotein that functions as a cell–cell adhesion factor and may also be involved in hematopoietic stem/progenitor cell development and inflammation. 10,19 In addition to a subset of ICCs, CD34 is expressed on hematopoietic stem cells, endothelial cells (particularly in small or proliferating vessels, and excluding those lining lymphatic vasculature), mast cells, Factor VIII–related antigen (von Willebrand factor), negative dendritic cells, and a few human sarcomas such as angiosarcoma and Kaposi sarcoma. 3,10,19 Demonstration of CD34 expression in canine GISTs would provide an additional marker to aid in the differentiation between GISTs and smooth muscle tumors, especially in the cases where KIT reactivity is not present and the tumor does not exhibit reactivity for smooth muscle markers. Tumors with this immunophenotype were classified as GIST-like by Maas et al 13 in 2007.
In humans, tumor site (intestines vs stomach), tumor size (≤2 cm), and mitotic index (<5 mitoses per 50 high-powered fields [HPF]) have been shown to be the most significant prognostic indicators. 7,9,15 –17,26,28 Cellular proliferation analysis, as determined by Ki67 and argyrophilic nuclear organizer regions (AgNOR) staining, also seem to provide predictive value. 22 No such correlations have been made for canine GISTs.
In this study, we examined the expression of CD34 in canine GISTs and evaluated possible prognostic indicators such as tumor size, location, and mitotic index as well as the proliferation markers Ki67 and AgNOR. Based on human case studies, we hypothesized that some canine GISTs would be immunoreactive for CD34 whereas canine GI leiomyomas/leiomyosarcomas would not be immunoreactive for CD34. In addition, we hypothesized that gross features, microscopic features, and proliferation activity of these tumors would correlate with survival. Specifically, gross features of canine GISTs including tumor size and tumor location (large intestine vs other GI location) would be positively associated with survival, whereas microscopic features of canine GISTs, specifically tumor necrosis and cellular proliferation indices (mitotic index, AgNOR, Ki67), would be negatively associated with survival. As such, there would be a positive correlation between gross features, microscopic features, and cellular proliferation indices of canine GISTs.
Material and Methods
Samples
Forty-nine client-owned animals diagnosed with GI mesenchymal neoplasms were identified from the AMC necropsy and surgical biopsy database. Tumors were from animals taken to surgery because of the intestinal mass based on historical information provided on the pathology submission forms. Multiple (6–8) 5-mm-thick, unstained sections of paraffin-embedded formalin-fixed tissue from respective cases were stained for histopathological, histochemical, and immunohistochemical evaluation. Tumor size and location were collected from submission forms. Tumor size was reported in 48 of the 49 animals, and tumor location was reported in all animals. Tumor location was categorized as stomach, small intestine, or large intestine. Tumor size was evaluated based on largest dimension as well as volume (volume = length × width × height × π/6), which was estimated from dimensions recorded on the pathology submissions forms. These measurements were taken at the time of submission and may have been from fresh as well as formalin-fixed tissues. The measurements for formalin-fixed tissue may slightly underestimate tumor size; hence, measurements are considered approximations only.
Histopathology and Histochemistry
Mitotic index was defined as the number of mitoses per fifty 400× fields. Necrosis scores, as summarized in Table 1 , were assigned to each GIST based on percentage necrosis in the sections examined. All available sections of the tumor were examined if more than 1 section was trimmed and the necrosis score averaged between all sections. AgNOR histochemical staining was performed using a standard protocol. 29 AgNOR scores were determined by calculating the average number of positive regions per nucleus after counting the total number of AgNOR in 500 nuclei per case. Resection was evaluated as complete or incomplete based on the presence of neoplastic cells at microscopic margins. Any section of tumor with neoplastic cells at the surgical margins was considered to be incompletely resected.
Scoring System for Necrosis in the Tumor Sections

Immunohistochemistry
A biotin–streptavidin–horseradish peroxidase detection system was used for each antibody (CD34, KIT [CD117], Ki67, SMA, desmin). The dilutions, clones, and sources for KIT, Ki67, SMA, and desmin antibodies are as follows: KIT (1:500, rabbit polyclonal, Lab Vision Corp, Fremont, CA), Ki67 (1:25, mouse monoclonal, clone BGX-297, Biogenex, San Ramon, CA), SMA (1:5000, mouse monoclonal, clone 1A4, Innogenex, San Ramon, CA), and desmin (1:100, clone 33, Biogenex, San Ramon, CA).
An optimized protocol for the CD34 immunohistochemistry (IHC) based on previous antibody titration and epitope retrieval experiments was used. Five-micrometer-thick tissue sections were cut from formalin-fixed, paraffin-embedded tissue blocks and mounted on positively charged OptiPlus fluid barrier slides (Biogenex, San Ramon, CA) and dried in a slide drier at 66°C for at least 1 hour. Adherent sections were manually deparaffinized and rehydrated by sequential treatment with Histoclear (National Diagnostics, Atlanta, GA), 100% ethanol, 95% ethanol, and water. Slides were then pretreated in heat-induced epitoperetrieval solution (pH 9.5). Antigen retrieval was performed by heating the mounted sections to 120°C for 3 minutes in a pressure cooker and then cooling to room temperature for 20 minutes and rinsing with water (Biocare Decloaking Chamber, Biocare Medical, Walnut Creek, CA). Immunohistochemical processing was performed using standard techniques on an automated stainer (Biogenex Automated Optimax) at room temperature. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide for 10 minutes. Slides were then incubated with biotin-bound mouse anti-canine CD34 (1:50) for 2 hours (CD34-biotin 2E9 monoclonal, BD Biosciences, San Jose, CA). The CD34 antibody was labeled with streptavidin-bound horseradish peroxidase for 20 minutes. The antigen–antibody reaction was visualized using 3,3′-diaminobenzidine (DAB) as chromogen for 10 minutes. The tissue was counterstained with hematoxylin for 1 minute. Between incubations the sections were washed 3 times in a phosphate-based wash buffer. Slides were manually dehydrated in 95% and 100% ethanol, cleared in Histoclear, and coverslipped with Cytoseal XYL (Richard-Allan Scientific, Kalamazoo, MI).
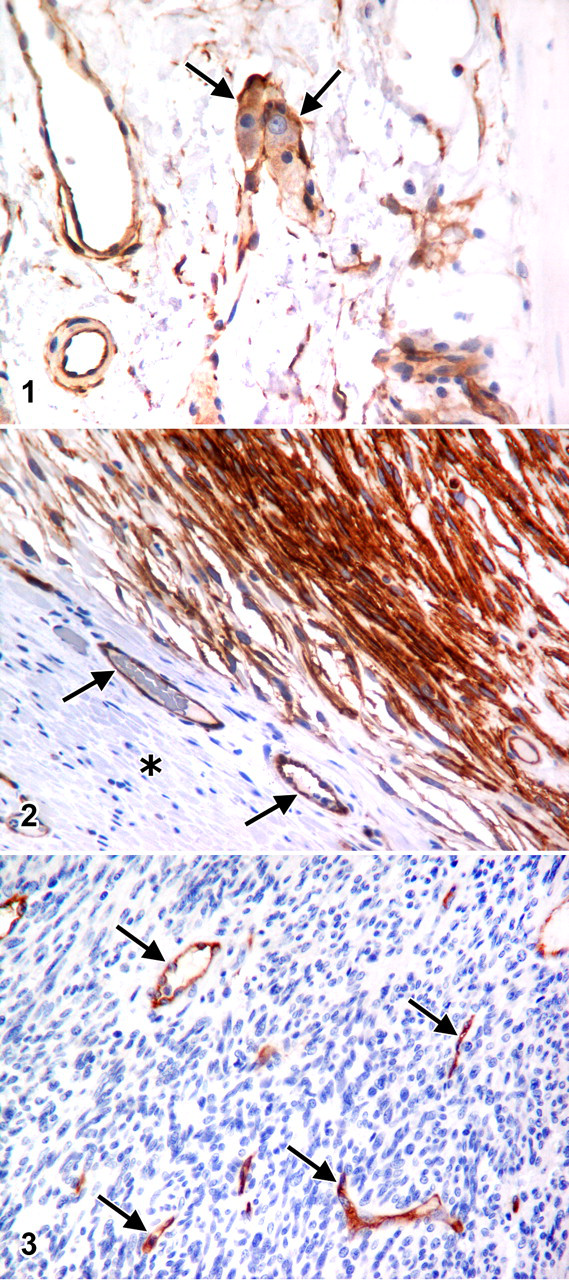
CD34 is normally expressed by some ICCs, vascular endothelium typically of newly formed or activated vessels (excluding lymphatics), and hematopoietic stem cells. Therefore, normal ICCs and vascular endothelium within evaluated specimens were used as internal positive controls (Fig. 1, 2, and 3), whereas intestinal muscularis and GI epithelium were used as negative internal controls (Fig. 2). Subsequent IHC revealed consistent positive staining of vascular endothelium and consistent negative staining of smooth muscle and GI epithelium confirming the appropriateness of these internal controls.
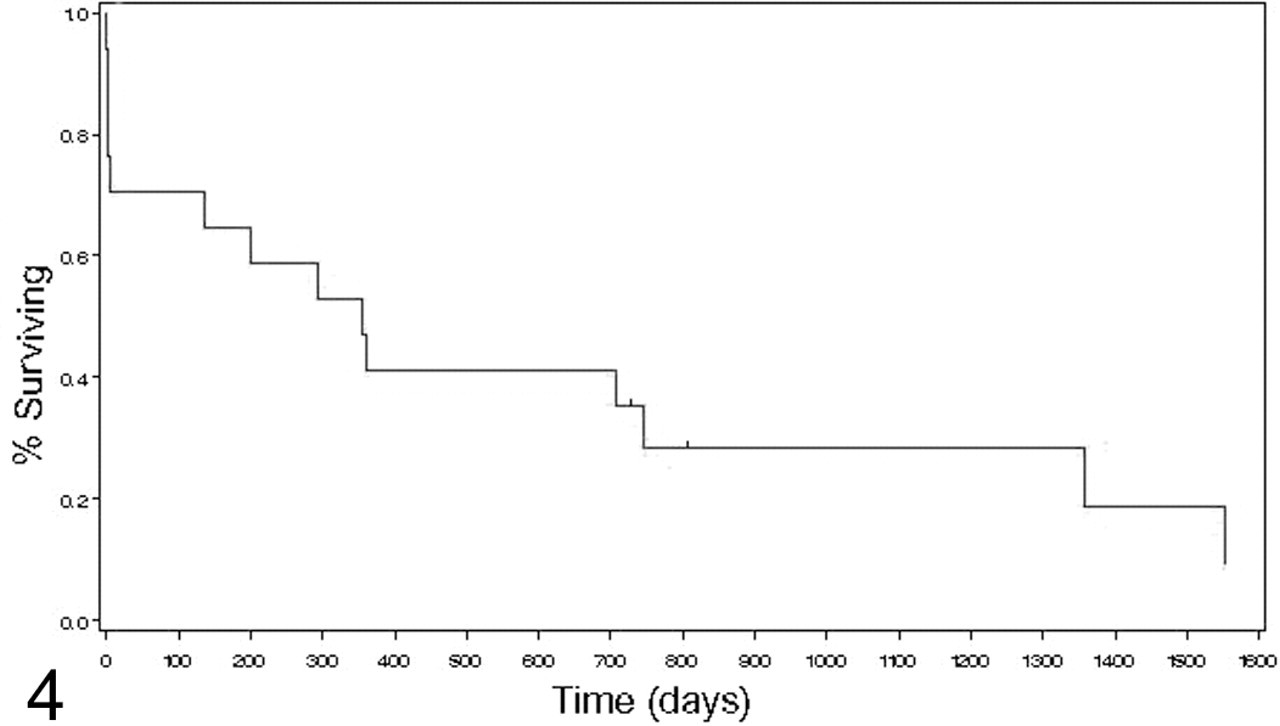
Survival curve with censors (Kaplan-Meier analysis).
The 49 neoplasms in this study were classified based on their immunohistochemical profile (summarized in Table 2 ): neoplasms that exhibited KIT immunoreactivity with or without CD34 immunoreactivity were classified as GISTs; neoplasms that did not exhibit KIT or CD34 immunoreactivity but were SMA and/or desmin positive were classified as smooth muscle tumors; and neoplasms that did not fit criteria for either GIST or smooth muscle tumors (negative for KIT, SMA, and desmin or CD34 and/or SMA and desmin positive, but KIT negative) were classified as uncategorized soft tissue sarcomas. Neoplasms classified as GISTs based on KIT positivity were stained for Ki67. Labeling indices for Ki67 were established by calculating the percentage of positive nuclei after counting 500 nuclei per case.
IHC Criteria for Classification of Canine Gastrointestinal Mesenchymal Tumors
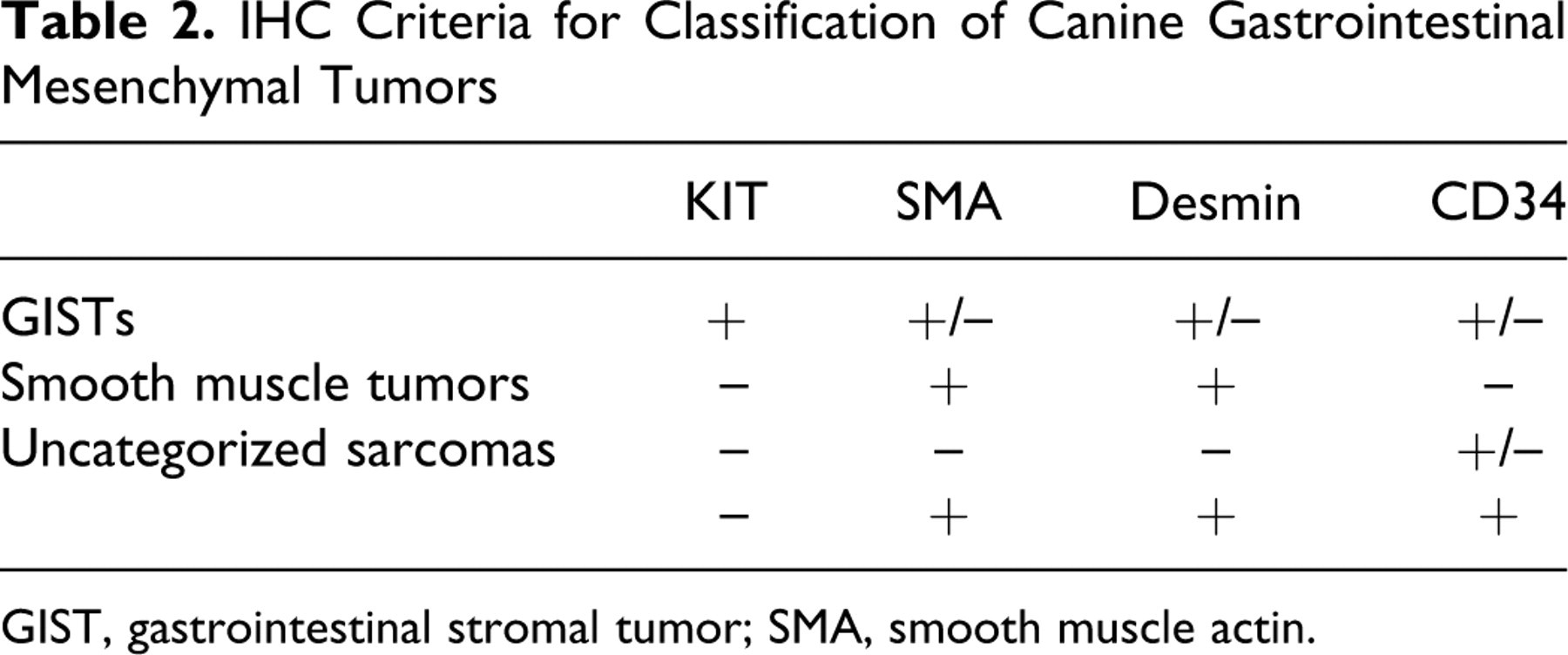
GIST, gastrointestinal stromal tumor; SMA, smooth muscle actin.
Clinical Follow-Up
Follow-up information was collected for each case classified as a GIST in which the medical record and owner contact information were available (16 of 27 GISTs). Information collected included signalment, tumor size and location (gastric vs small intestines vs large intestines), postsurgical disease-free interval (including local recurrence, presence of metastases, and time to development of metastases), survival (from time of diagnosis), and treatment.
Statistical Analyses
Correlations between continuous data (AgNOR, Ki67, tumor size, mitotic index) and between continuous and ordinal data (necrosis) were examined using Spearman rank correlation. Correlations between continuous and nominal data (tumor location) were examined using the Kruskal-Wallis test. A survival curve and median survival for all dogs with GIST was determined using the Kaplan-Meier method. For all statistical tests, P ≤ .05 was considered significant. Statistical analyses were performed using a standard statistical software package (SAS 9.0, SAS Institute Inc., Cary, NC).
Results
Table 3 lists signalment, locations, immunohistochemical findings (KIT, CD34, SMA, desmin), and diagnoses of the 49 cases of canine mesenchymal intestinal tumors examined. Twenty-six breeds, including mixed breeds, were represented. Twenty four of the dogs were male and 22 were female, with 3 canines of unspecified gender based on the pathology submission form. Age ranged from 1 to 15 years with an average of 11.2 years. Eighteen of the 49 tumors were located in the large intestines, 21 were in the small intestine, 8 were in the stomach, 1 was in stomach and proximal duodenum, and 1 was in an unspecified abdominal location. All 8 gastric tumors were consistent with smooth muscle tumors and all 18 of the large intestinal tumors were GISTs based on diagnostic criteria outlined in Table 2. Eight of the 21 small intestinal tumors were consistent with GIST, as was the one from the unspecified abdominal location
Signalment, Location, Immunohistochemistry (KIT, CD34, SMA, Desmin), and Diagnosis of 49 Cases of Canine Mesenchymal Intestinal Tumors Examined
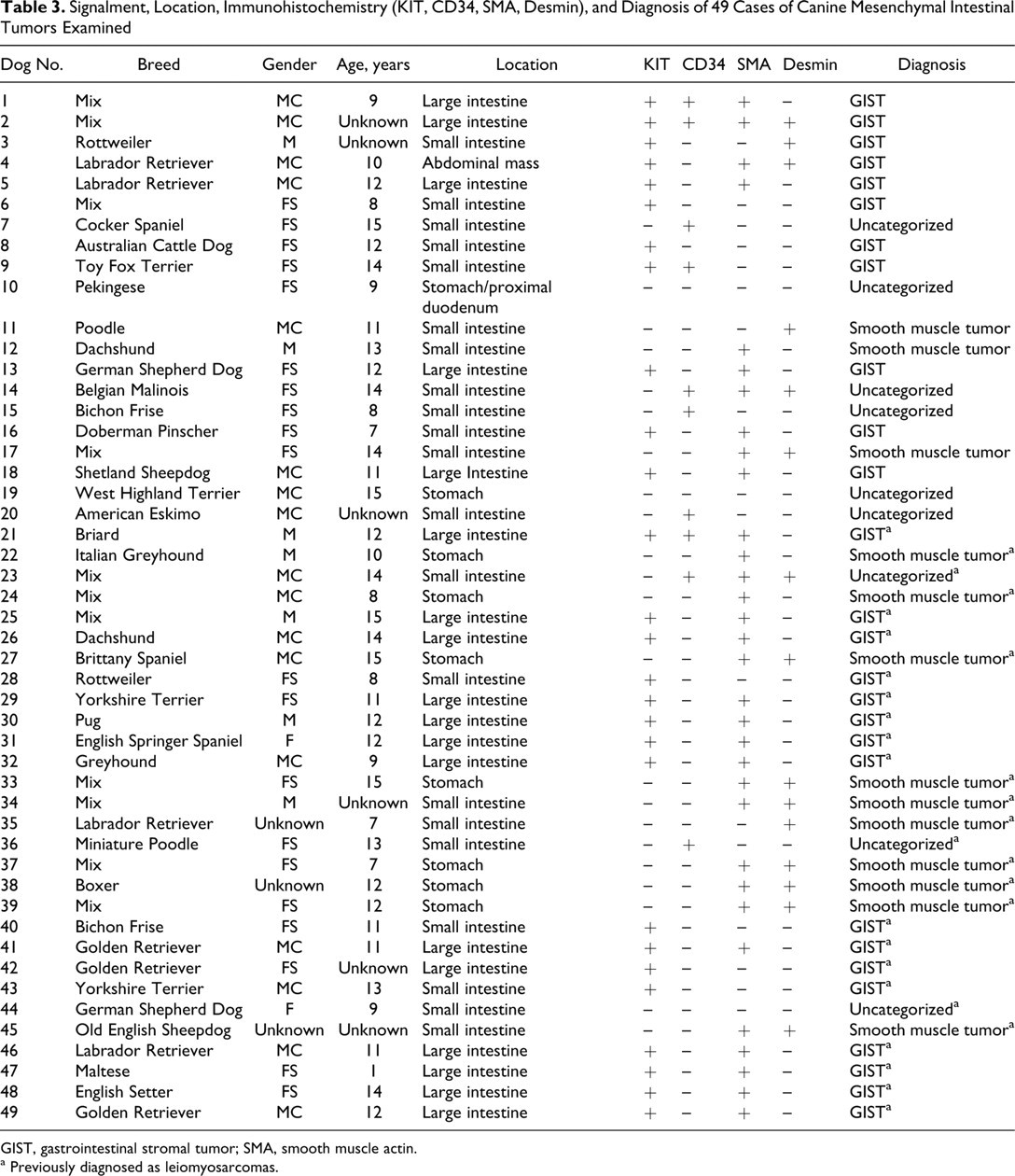
GIST, gastrointestinal stromal tumor; SMA, smooth muscle actin.
a Previously diagnosed as leiomyosarcomas.
Immunohistochemistry
Appropriate internal positive and negative controls were used, namely, vascular endothelial cells and GI smooth muscle, which were CD34 immunopositive and immunonegative, respectively (Fig. 1, 2, and 3). Tumor cells that were CD34 positive exhibited moderate to deep, diffuse, consistent staining of the cytoplasm (Fig. 2). A subset of spindle-shaped ICCs between neuronal bodies of GI neural plexi (Auerbach plexus, deep muscular plexus, and submuscular plexus) also displayed multifocal cytoplasmic labeling with CD34 (Fig. 1).
Based on the diagnostic criteria outlined in Table 2, 27 of the 49 neoplasms (55%) were classified as GISTs, 13 of the 49 neoplasms (27%) were classified as smooth muscle tumors (with no further classification as malignant or benign smooth muscle tumors), and 9 of the 49 neoplasms (18%) were uncategorized. Of the 27 GISTs, 4 were CD34 positive (14.8%). The immunophenotypes of the 9 uncategorized neoplasms are as follows: 4 of the 9 (44 %) were immunopositive for CD34 but immunonegative for KIT, SMA, and desmin; 2 (22%) were KIT immunonegative but immunopositive for CD34, SMA, and desmin; and 3 (33%) were immunonegative for all 4 antibodies.
Twenty-nine of the 49 neoplasms were originally diagnosed as leiomyosarcomas. Sixteen of these 29 (55%) were reclassified as GISTs after IHC. Twenty of the 49 neoplasms were originally diagnosed as sarcomas or mesenchymal tumors and given differential diagnoses of GISTs or smooth muscle tumors. Of these 20 neoplasms, 11 (55%) were classified as GISTs and only 3 (15%) were classified as smooth muscle tumors after IHC. No statistically significant associations were found between KIT and CD34 immunoreactivity.
Gross, Microscopic, Cellular Proliferation Parameters, and Follow-Up
Table 4 lists the survival time, mitotic index, tumor size, AgNOR and Ki67 scores, and necrosis score in the 27 canine GISTs. Mitotic indices ranged from 0 to 642 in 50 HPFs or an average of 0 to 12.8 per HPF. There was significant positive correlation between mitotic index and AgNOR (P = .0077) and between mitotic index and Ki67 (P = .0376). There was significant positive correlation between Ki67 and AgNOR scores (P = .0014). There was significant positive correlation between necrosis and both volume and largest dimension of tumor (P = .0380 and P = .0508, respectively). Tumor sizes ranged from 1 × 1 × 0.8 cm (0.4187 cm3) to 18 × 16 × 6 cm (904.32 cm3). There was significant difference between mitotic index, AgNOR, and Ki67 scores when stratified by tumor location (large intestine vs small intestine, summarized in Table 5 ). Mitotic index was significantly higher in the GISTs arising from the small intestines compared with those originating from the large intestines (median values of 26 and 9.5, respectively; P = .0027). AgNOR scores were significantly higher in small intestinal than large intestinal GISTs (median values of 1.308 and 0.746, respectively with P = .0205). Ki67 scores were significantly higher in small intestinal than large intestinal GISTs (median values of 2.9 and 1.0 respectively; P = .0014).
Survival Time, Mitotic Index, Tumor Size, AgNOR, and Ki67 Scores and Necrosis Score in 27 Canine GISTs
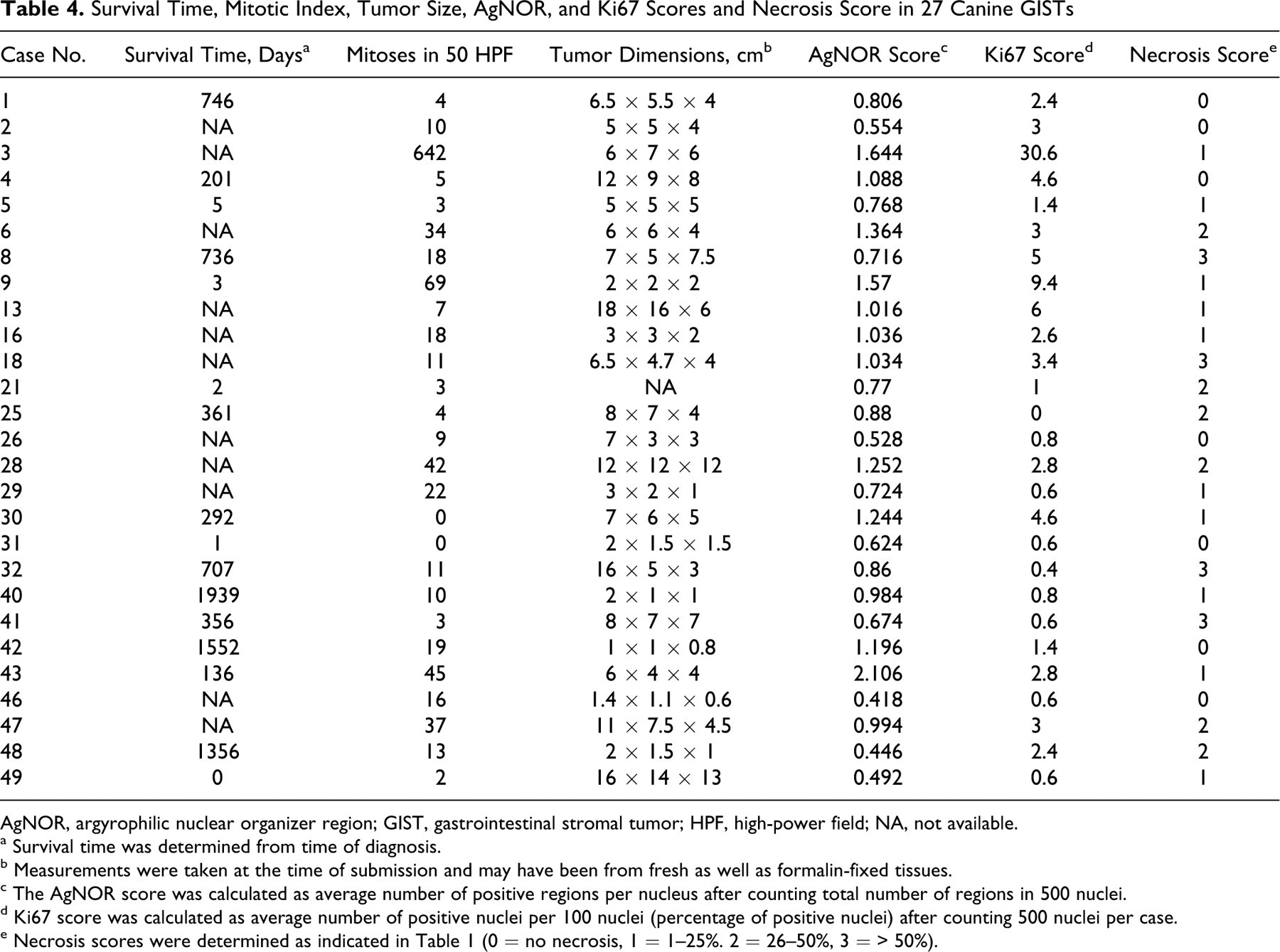
AgNOR, argyrophilic nuclear organizer region; GIST, gastrointestinal stromal tumor; HPF, high-power field; NA, not available.
a Survival time was determined from time of diagnosis.
b Measurements were taken at the time of submission and may have been from fresh as well as formalin-fixed tissues.
c The AgNOR score was calculated as average number of positive regions per nucleus after counting total number of regions in 500 nuclei.
d Ki67 score was calculated as average number of positive nuclei per 100 nuclei (percentage of positive nuclei) after counting 500 nuclei per case.
e Necrosis scores were determined as indicated in Table 1 (0 = no necrosis, 1 = 1–25%. 2 = 26–50%, 3 = > 50%).
Comparison of Median Mitotic Index, AgNOR, and Ki67 Scores of Small Versus Large Intestinal GISTs

AgNOR, argyrophilic nuclear organizer region; GIST, gastrointestinal stromal tumor; HPF, high-power field.
The dogs were not routinely examined at the Animal Medical Center for follow-up care or screened for metastases. Postoperative follow-up data as well as estimation of survival time were based on retrospective study of records and phone calls to the owner and referring veterinarian, which often did not include information about the presence of metastases. Two tumors (from dog Nos. 4 and 49) had gross evidence of metastases at the time of surgery as reported in the pathology submission forms. Dog No. 4 had metastases to the liver, peritoneum and spleen. Dog No. 49 had metastases to liver, omentum, pancreas, and duodenum. One of these (dog No. 49) was euthanatized at surgery.
Follow-up data were available for 16 of the 27 GISTs; however, the sampling was inadequate for statistical analyses. Overall median survival time was 356 days based on Kaplan-Meier analysis. Two dogs were still alive at the time of follow-up phone call. Of the deceased dogs, 8 (50%) were conservatively considered euthanatized for clinical disease related to the GIST based on available medical records and/or information relayed by the owners. Four (25%) of the deceased were euthanatized within 1 week of surgery (including No. 49 mentioned above), presumably from GIST-related disease; however, surgical reports were not available from these animals to evaluate whether euthanasia may have been due to surgical complications. Only 2 dogs were treated with chemotherapy in addition to surgical resection (No. 8 was still alive at 735 days, No. 4 survived 201 days). Both dogs were treated with doxorubicin (Adriamycin); dog No. 4 was given 1 dose 185 days prior to euthanasia and dog No. 8 was give a dose every 3 weeks for 5 doses with the last dose 603 days prior to last follow-up.
Discussion
To our knowledge, this is the first report to describe CD34 immunoreactivity in canine GISTs. CD34 immunoreactivity was lower in our canine cases (14.8%) than reported in humans (60–80%). This may be due to species differences in expression, specificity, or sensitivity or degradation of antigen from long-term storage or fixation. However, demonstration of CD34 immunoreactivity in our study suggests that canine GISTs exhibit CD34 immunoreactivity consistent with GISTs that may arise from KIT- and CD34-positive ICCs. In a previous study, CD34 expression using a monoclonal QBEND-10 antibody could not be evaluated in GISTs because of lack of reactivity in canine tissues. 4 Another specific monoclonal antibody, 2E9, with high affinity for CD34 in canine bone marrow was described previously 14 and was used in this study to elucidate CD34 expression in canine tissues.
The use of CD34 as a marker for canine hematopoietic progenitor cells has been explored given the importance of canine models of autologous and allogeneic stem cell transplantations in studies of graft versus host disease, recombinant hematopoietic growth factors, and gene therapy. 10,14 CD34 has also been used as a marker for putative stem cells in the canine hair follicle. 19 CD34 has demonstrated serine phosphorylation domains with potential sites for serine, threonine, and tyrosine phosphorylation, 10 which may provide targets for specific kinase inhibitors such as the tyrosine kinase inhibitor, imatinib (STI571; Gleevec).
Imatinib has been used in cases of certain canine neoplasms but is associated with hepatoxicity even at low doses. 23 It has also been shown to affect human in vitro CD34+ hematopoietic progenitors by inhibiting differentiation. 1 Demonstration of CD34 immunoreactivity in canine GISTs may provide further evidence of the utility of tyrosine kinase inhibitors as therapy for these tumors. Imatinib (and other tyrosine kinase inhibitors) are being examined in vitro and with mouse xenografts with regard to treatment of canine mast cell tumors. 5,8 Recently, Palladia (toceranib phosphate, SU11654, PHA-2913639E), a tyrosine kinase inhibitor, was approved by the US Food and Drug Administration for treatment of recurrent mast cell tumors in dogs. 2,20 In humans, imatinib is used in therapy for a variety of other KIT-immunopositive neoplasms, such as GISTs, mast cell tumors, and peripheral nerve sheath tumors. 5,8 Like other intrinsic tyrosine kinase receptors, binding of KIT to its ligand (stem cell factor or steel factor) results in phosphorylation cascades that activate signaling pathways such as MAP kinase and protein kinase B that lead to cell proliferation, survival, and differentiation. 11 KIT mutations in GISTs result in constitutive activation of receptor tyrosine kinases independent of ligand binding. 4,11,26 The most common mutations are in the juxtamembrane domain (exon 11), 4,7,16,17,26,28 a protein sequence that is highly conserved, with 100% homology between humans and dogs. 4 Similar mutations in KIT are demonstrated in dogs and humans, 4 suggesting that canine tumors with KIT mutations may behave similarly and respond in the same way to targeted therapies. There is some evidence in human studies that the type of mutation may affect the prognosis and response to treatment. 16,26,28
A significant proportion of neoplasms (55%) originally diagnosed as leiomyosarcomas were reclassified as GISTs after immunophenotyping. This is consistent with previous canine and human reports. Nine tumors could not be categorized based on IHC. Of the 9 uncategorized neoplasms, 4 were immunopositive for CD34 but immunonegative for KIT, SMA, and desmin, and 2 were KIT immunonegative but immunopositive for CD34, SMA, and desmin. The former 4 tumors may still be considered GISTs, because rare GISTs are believed to be KIT immunonegative, 28 possibly arising from KIT-immunonegative and CD34-immunopositive ICCs. If these 4 neoplasms are added to the GIST category, the percentage of CD34-immunopositive GISTs increases to 25.8%. The latter 2 neoplasms may be considered smooth muscle tumors as rare leiomyosarcomas are reported to be CD34 immunopositive. 28 If these 2 are added to the smooth muscle tumor category, the percentage of CD34-immunopositive smooth muscle tumors increases to 13%. Other potential reasons for these immunohistochemistry results include laboratory-based factors such as inadequate fixation, excess antigen extraction, and nonspecific staining, although internal controls minimized the impact of these factors. Other human GI tumors (i.e., glomus tumors, schwannomas, solitary fibrous tumors, and inflammatory fibroid polyps) have been reported to be KIT immunonegative and variably immunoreact with anti-CD34, anti-SMA, and/or anti-desmin antibodies. However, morphology on hematoxylin and eosin (HE) staining and immunophenotyping (such as S-100 and GFAP, which were performed on these sections and were immunonegative) should allow for differentiation of these other diagnoses. 6,16,18,21,28 Finally, 4 uncategorized neoplasms displayed no immunoreactivity to all 4 antibodies used in this study. The morphologies of the KIT-/CD34-/SMA-/desmin-immunonegative tumors are similar to differentiated GISTs, and although a diagnosis of undifferentiated soft tissue sarcoma was considered, these cases may still be appropriately diagnosed as GISTs or leiomyosarcomas, because immunophenotyping results should always be interpreted in the context of HE morphology. KIT immunoreactivity remains the standard for diagnosis of GISTs, and additional criteria and markers may be explored to aid in cases where immunohistochemical results are not definitive. Genotyping of the neoplasm may be helpful in these cases, as some neoplasms in human cases have KIT mutations without exhibiting KIT immunoreactivity. 7,28
No statistical conclusions could be made regarding associations between survival and various gross features, microscopic features, or proliferation markers due to insufficient follow-up data for statistical analysis. A recent study reported that tumor diameter was an important prognostic indicator and mitotic count was a less significant indicator in cases of canine GISTs compared with cases of human GISTs. 13 The difference may be due to the low mitotic index of these tumors, as the cutoff point for mitotic indices is reported per 50 HPF in human cases, which is considered the minimum number of HPFs necessary to accurately assess this proliferation parameter. 15
Positive correlations were noted between mitotic index, AgNOR and Ki67, which is expected since these are all indicators of proliferation and consistent with human studies. Ki67 antigen is expressed by proliferating cells in active phases of the cell cycle (G1, G2, M, S phases) and is not expressed in quiescent cells (G0). Detection and quantification of Ki67 antigen are thus very useful in evaluating proliferation or growth fractions of cell populations. 25 AgNOR are segments of DNA encoding clusters of ribosomal genes. These regions are more active in S-G2 phase of the cell cycle than G1, which is characteristic of rapidly dividing cells. The number of active regions and expression of AgNOR proteins is detected and quantified by silver stains. 27 The number of regions per nucleus is correlated with mitotic index in tumor cell lines and serves as a generation time marker. In human GISTs, analysis of AgNOR area and Ki67 resulted in statistically significant correlations with mitotic index, tumor size, and presence of necrosis. 9,22 AgNOR protein area (NORA) was statistically associated with a mitotic index ≥5 per 10 HPF and presence of necrosis, whereas a Ki67 score ≥9.69% was associated with mitotic index ≥5 per 10 HPF, size ≥5 cm, and presence of necrosis. 22 Positive correlations were noted between necrosis and tumor volume, most likely because larger, faster growing tumors tend to outgrow their blood supply, resulting in coagulative necrosis.
Interestingly, there was a significant difference between mitotic index, AgNOR, and Ki67 scores depending on location of the tumor, with small intestinal GISTs having greater values than large intestinal GISTs. This may suggest a difference in biological behavior. However, sufficient follow-up data were not available for statistical analysis. Location of the primary tumor has been shown to have prognostic significance in human GISTs, with gastric GISTs having better prognosis than small or large intestinal GISTs. 6,15,16,28 Different thresholds for determining malignancy were used depending on the location. For example, the size threshold is smaller in the small intestines than stomach for calling a tumor malignant. 6,28 In our study, none of the GISTs were located in the stomach, which is compatible with previous reports that most canine GISTs occur in the small and large intestinal tract. 4,12,13,23
Increased understanding of human GISTs has allowed for development of more accurate classifications and more reliable prognostic information and treatment protocols. GISTs, leiomyomas, and leiomyosarcomas are among the most common mesenchymal neoplasms in the GI tract of dogs. With increasing recognition of GISTs in the GI tract in the dog, it becomes important to examine additional diagnostic criteria that may assist in differentiation of GISTs from leiomyomas and leiomyosarcomas. This study shows that CD34 expression can be demonstrated in formalin-fixed canine tissues using a canine-specific antibody.
Prediction of potential biological behavior of human GISTs can be difficult and remains controversial, as some experts believe that these tumors range from truly benign to malignant and others believe that all GISTs have malignant potential. 6,7,16 Multiple prognostic indicators including tumor size, location, mitotic index, AgNOR staining, and Ki67 labeling have been correlated with survival in human GISTs to try to stratify these tumors into prognostically significant categories. The few cases with available follow-up data in this study suggest that canine GISTs have the same range of behaviors. In our study, AgNOR, Ki67, and mitotic index were all positively correlated, and significant differences were noted in the proliferation indices (AgNOR, Ki67, and mitotic index) between small and large intestinal tumors. Although we were unable to correlate these values with survival data, these results indicate that further examination of these factors in canine GISTs is warranted, as it may allow for more reliable prediction of behavior and a potential grading scheme, and therefore increase prognostic capability.
Footnotes
Acknowledgements
We thank Drs Serena Liu, Taryn Donovan, Amanda Beck, Linda Johnson, Sebastien Monette, and Julie White for editorial input and support.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
