Abstract
Exotic Newcastle disease virus (NDV) isolated from chickens during the 2002-2003 California outbreak (CA exotic Newcastle disease [END] virus) was inoculated into 4-week-old specificpathogen-free (SPF) White Leghorn chickens, 3-week-old SPF Beltsville White turkeys, 6-week-old commercial Broad Breasted White turkeys, and 10- to 20-week-old racing pigeons, and the clinicopathologic features of disease were compared. Birds were monitored clinically and euthanized sequentially with collection of tissues. Tissues were examined by histopathology, by immunohistochemistry to detect viral nucleoprotein, and by in situ hybridization to detect viral mRNA. Clinically, infected chickens and SPF turkeys showed severe depression, and all died or were euthanized because of severe clinical signs by day 5 postinoculation. In these birds, histologic lesions were widespread and virus was detected in multiple organs. All infected commercial turkeys showed mild depression, and incoordination was observed in some birds. Histologic lesions were mild, and viral distribution was limited. In pigeons, only 1 bird showed overt clinical disease, and histologic lesions and viral distribution were present in limited organs. Consequently, susceptibility to highly virulent NDV was shown to vary among chickens, SPF turkeys, commercial turkeys, and pigeons. Additionally, we have evidence of CA END virus subclinical infections that suggest pigeons could be subclinical carriers of other virulent NDV.
Keywords
Newcastle disease is one of the most important avian viral diseases because of its economic impact on the poultry industry. The causative agent, Newcastle disease virus (NDV), is synonymous with avian paramyxovirus type 1. 2 It has been classified in the order Mononegavirales, family Paramyxoviridae, subfamily Paramyxovirinae, and genus Avulavirus. 13, 14 NDV infects approximately 236 species of pet and free-living birds in addition to domestic avian species (chicken, turkey, goose, duck, and pigeon). 8 Among poultry, chickens are the most susceptible, whereas ducks and geese are the least susceptible. Virus shedding is usually of short duration, 1–2 weeks, in gallinaceous birds and some species of song birds, but can last several weeks to many months in psittacines. 4, 8 Long-lasting shedding of NDV might be observed in Columbiformes (pigeons and doves) and Passeriformes in cases where there is chronic infection of the kidney. 8
Exotic Newcastle disease (END) is a term used in the United States to define the illness caused in birds by infection with velogenic NDV. Velogenic NDV is endemic in many countries of Central and South America, the Middle East, and most of Africa and Asia. 4 The END virus has been introduced into the United States on several occasions, but each outbreak was subsequently eradicated. Exotic ND is so virulent that birds may die without showing clinical signs. 2, 4 A death rate of almost 100% can occur in unvaccinated poultry flocks. Exotic NDV can infect and cause death even in vaccinated poultry. 4
An outbreak of END was confirmed 1 October 2002, in Los Angeles County, California, in small flocks of backyard birds, including some used for illegal cockfighting. 15– 17 A separate introduction also occurred in Texas during the outbreak. 18 Signs of the disease in birds included sneezing, coughing, gasping for air, drooping wings, muscular tremors, paralysis, and sudden death. Laboratory testing confirmed infection with END virus. Phylogenetically, the END virus was found to be most closely related to recent velogenic NDV isolates from Mexico and Central America. 18 In this study, the END virus causing the outbreak was characterized by standard pathogenicity tests and by clinicopathologic features of disease in 4-week-old, specific-pathogen-free (SPF) chickens, 3-week-old SPF turkeys, 6-week-old commercial turkeys, and 10- to 20-week-old racing pigeons.
Materials and Methods
Virus
NDV (Chicken CA/S0212676) was obtained as a field isolate from chickens during the 2002–2003 California END outbreak. The isolate was received as the second embryo passage, and an additional passage was completed to provide the amnioallantoic fluid used as inoculum as specified in the experimental design. 6, 12
Eggs and birds
The source of embryonating chicken eggs and chickens was the Southeast Poultry Research Laboratory (SEPRL) SPF White Leghorn flock. Eggs were used for viral propagation, isolation, titration, and one of the standard pathogenicity tests, the mean death time (MDT). 1, 6 Chickens were used for the other 3 pathogenicity tests: the intracerebral pathogenicity index (ICPI) test in day-old chicks, the intravenous pathogenicity index (IVPI) test in 6-week-old chickens, and the intracloacal inoculation pathogenicity test in 6-week-old chickens. 1, 6 For the clinicopathologic assessment, 3-week-old SPF turkeys, 6-week-old commercial turkeys, and 10- to 20-week-old racing pigeons were used in addition to 4-week-old chickens. 10– 12 The source of the 3-week-old turkeys was the SEPRL SPF Beltsville White turkey flock. The SPF chickens and turkeys were NDV hemagglutination inhibition (HI) negative at the time of inoculation. Unvaccinated 6-week-old commercial Broad Breasted White turkeys (Ridgeway Turkeys, LaRue, Ohio) and 10- to 20-week-old racing pigeons that were provided by the American Racing Pigeon Union (www.pigeon.org) were used for this study after the acquired maternal antibodies to NDV had been depleted and the birds were HI test 1 negative for NDV antibody. All birds were housed in negative-pressure isolators under BSL-3 agriculture conditions at SEPRL and provided feed and water ad libitum. 6
Pathogenicity index tests
To characterize the virus, 4 standard pathogenicity tests were performed. 1 These included the MDT in 9- to 10-day-old embryonating SPF White Leghorn chicken eggs, the ICPI in 1-day-old SPF White Leghorns, the IVPI in 6-week-old SPF White Leghorn hatchmates, and the intracloacal inoculation test in 6-week-old White Leghorn chickens. 1
Clinicopathologic assessment
Birds (10 chickens, 10 SPF turkeys, 8 commercial turkeys, and 10 pigeons) were inoculated bilaterally intraconjunctivally (chickens) and unilaterally intraconjunctivally and intranasally (SPF turkeys, commercial turkeys, and pigeons) with 0.1 ml of amnioallantoic fluid harvested from infected embryonating eggs. Each bird received approximately 105.0 50% embryo infectious doses (EID50) (chickens, SPF turkeys, and commercial turkeys) or 106.0EID50 (pigeons) of viral inoculum. 6 Phosphate buffered saline was inoculated into chickens, SPF turkeys, commercial turkeys, and pigeons as a noninfected control.
The birds were clinically monitored every day. Tissues (eyelid, spleen, thymus, bursa, Harderian gland, proventriculus, small intestine, Meckel's diverticulum, cecal tonsils, large intestine, air sac, trachea, lung, heart, esophagus, tongue/pharynx, crop, brain, liver, kidney, comb-only from chickens, head of left femur including bone marrow, and turbinate) were collected from birds sequentially euthanized or after disease-associated death. 6 Confirmation that sampled birds were virus infected was based on virus recovery from oral or cloacal swabs collected before or on the day tissue samples were collected or by seroconversion based on the detection of NDV HI antibody in blood collected on or after 14 days postinoculation (dpi). Swabs were taken at 2, 3, 4, and 5 dpi in chickens; 2, 4, and 5 dpi in SPF turkeys; 2, 4, 6, 8, and 14 dpi in commercial turkeys 6, 12 ; or 2, 4, 5, 8, 14, and 18 dpi in pigeons. 10, 11 Because of very severe morbidity and mortality, the experiments with chickens and SPF turkeys were terminated at 5 dpi, except for the noninfected control birds, which were monitored with sampling until 14 dpi. The experiments with commercial turkeys and pigeons were terminated, respectively, at 14 and 18 dpi after blood sampling. Collected tissues were fixed by immersion in 10% neutral buffered formalin for approximately 52 hours. The sections of femur and turbinates were decalcified in 5% formic acid for 3–4 hours. All sampled tissues were routinely processed into paraffin, and 3-μm sections were cut for hematoxylin and eosin staining, immunohistochemistry (IHC), and in situ hybridization (ISH). 6
Immunohistochemistry
All sampled tissues were examined by IHC to detect NDV nucleoprotein. 6, 12 The protocol used was as follows: After deparaffinization, tissue sections were subjected to antigen retrieval by microwaving for 10 minutes at full power in Vector antigen unmasking solution (Vector Laboratories, Burlingame, CA) followed by blocking with universal blocking reagent (Biogenex, San Ramon, CA) as recommended by the manufacturer. The primary antibody, made in rabbit, was anti-peptide (nucleoprotein) used at 1°8,000 dilution. 10 The detection system was avidin-biotin-alkaline phosphatase (Vector Laboratories), and the substrate was Vector Red (Vector Laboratories). Sections were counterstained lightly with hematoxylin and coverslipped with Permount for a permanent record.
In situ hybridization
Selected tissue sections (specifically, all collected tissues from all chickens, except the pancreas, proventriculus, and intestine from a bird at 5 dpi, and the eyelid, spleen, thymus, bursa, and Harderian gland from all SPF turkeys, commercial turkeys, and pigeons) were probed with a negative sense digoxigenin-labeled 850-base riboprobe representing the 5′ end of the matrix gene of NDV Fontana (CA1083). Protocol was as described previously. 6, 10– 12 Briefly, tissues sections were deparaffinized, rehydrated, and digested with 30 μg/ml proteinase K for 15 minutes at 37°C. Hybridization was conducted overnight at 42°C with approximately 20 ng of probe in prehybridization solution. After stringent washes, anti-digoxigenin alkaline phosphate was added to the sections. The development was with the chromogen/substrate nitroblue tetrazolium/5-bromo,4-chloro,3-indolylphosphate. Tissues were counterstained lightly with hematoxylin and coverslipped with Permount.
Virus isolation from swabs
Oral and cloacal swabs were obtained from each bird as described in the clinicopathologic assessment design and placed in separate tubes containing 1.5 ml of brain-heart infusion broth with antibiotics (2,000 unit/ml penicillin G, 200 μg/ml gentamicin sulfate, and 4 μg/ml amphotericin B; Sigma Chemical Co., St. Louis, MO). 1, 9 Swab fluids were centrifuged at 1,000 × g for 20 minutes for inoculation of 9- to 10-day-old SPF embryonating chicken eggs. NDV-infected dead or surviving embryos were identified by hemagglutination (HA) activity in amnioallantoic fluid harvested from chilled eggs. NDV was confirmed in HA-positive samples by an HI test with NDV-specific antiserum. 1
Serology
The HA and HI tests were conducted by conventional microtiter methods 1, 9 with blood samples from all groups of birds at preinoculation and from infected and noninfected birds at 14 dpi (commercial turkeys) and at 18 dpi (pigeons). Four HA units of β-propiolactone inactivated LaSota virus was used as the test antigen in completing the HI tests.
Results
Pathogenicity index tests
The MDT for the California outbreak NDV isolate was 59 hours, ICPI was 1.76, and IVPI was 2.54, which were consistent with a velogenic virus. The intracloacal inoculation test showed 100% mortality with hemorrhagic lesions, which indicated that the California NDV isolate was a viscerotropic velogenic virus.
Clinical signs
The clinical observations among infected birds are summarized in Table 1. All infected chickens and SPF turkeys showed severe depression (Fig. 1) and died or were euthanized because of severe clinical signs by 5 dpi. In chickens, nervous signs, such as head twitches and tremor, were observed by 3 dpi, but nervous signs were not seen in SPF turkeys. Commercial turkeys had reddened conjunctiva and periocular edema by 6 dpi, which proceeded to nasal discharge and mild depression in all birds. In addition, 3 showed incoordination. One pigeon had a slight body tremor at 10 dpi. No abnormal clinical signs of disease were observed in any of the noninfected controls.

SPF turkeys. Front: an infected turkey with severe depression. Back: a noninfected turkey.
Clinical signs, morbidity, and mortality of chickens, SPF turkeys, commercial turkeys, and pigeons infected with California END virus.∗

SPF = specific-pathogen-free; END = Exotic Newcastle disease; dpi = days postinoculation.
Total = the number of birds remaining in each group when first clinical signs were observed. Sampling at 2 and 4 dpi reduced commercial turkeys from 8 to 6. Sampling at 2, 4, 5, and 8 dpi reduced pigeons from 10 to 6.
Gross findings
In chickens at 2 and 3 dpi, spleens were enlarged, friable, and mottled, and those at 4 and 5 dpi were small with multiple foci of necrosis. At 4 and 5 dpi, hemorrhage was present in the cranial tracheal mucosa posterior to the glottis opening (Fig. 2) and proventriculus, and the bone marrow was pale. Necrosis with hemorrhage was observed in the intestine, most prominently at the cecal tonsils at 5 dpi. In SPF turkeys at 2 dpi, enlarged and mottled spleens were observed. Bone marrow was pale at 4 dpi, and the spleens were small and mottled at 4 and 5 dpi. In commercial turkeys at 4 dpi, enlarged and mottled spleens were observed. At 8 dpi, the spleen and thymus were small, and there were multiple foci of hemorrhage in the pancreas. At 11 dpi, lymphoid organs (spleen, thymus, and bursa) were shrunken and air sacs were cloudy. At 14 dpi, the ileum had reddened mucosa, and the pancreas was slightly mottled. In pigeons at 5, 14, and 18 dpi, spleens were enlarged and dark.
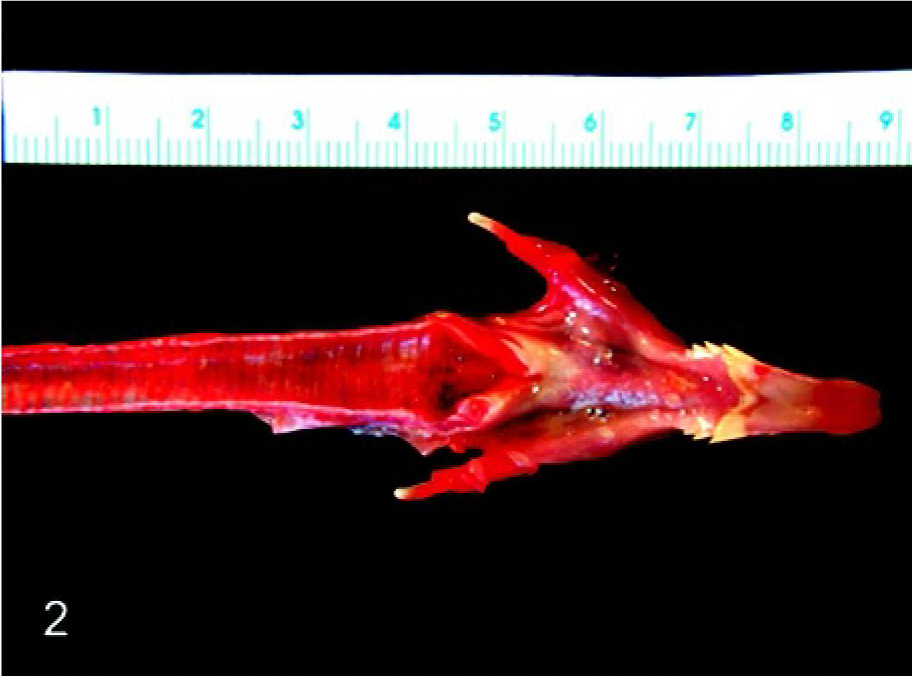
Cranial trachea with tongue; experimentally infected chicken. Necrohemorrhagic tracheal mucosa was present posterior to the glottis opening.
Histopathology
The microscopic findings are summarized in Table 2. In NDV-infected chickens and SPF turkeys, the most significant finding was severe lymphoid depletion along with lymphocellular necrosis and apoptosis in lymphoid organs such as spleen, bursa, and thymus. In chickens, lymphoid depletion was first observed in the bursa and spleen at 2 dpi. From 3 dpi, all lymphoid organs had moderate to severe lymphoid depletion with multifocal necrosis, progressing to diffuse areas of necrosis at 5 dpi (Fig. 3). In the cranial trachea, there were patches of necrotic epithelial cells and multifocal areas of lymphocytic infiltrates in the mucosa. At 5 dpi, most of the tissues examined, especially the gastrointestinal tract, liver, kidney, pancreas, and bone marrow, had extensive zones of necrosis. In SPF turkeys, lymphoid depletion with necrosis was observed in all lymphoid tissues at 4 and 5 dpi, but the lesion was less severe than that in chickens. In addition, necrosis was present in the gastrointestinal tract and pancreas. In commercial turkeys, the principal lesions were present in lymphoid organs and brain. The earliest brain lesions were observed at 8 dpi and were characterized by gliosis and perivascular cuffing in the cerebellum and brain stem. In addition, there was a drop-out of cerebellar Purkinje cells. At 11 dpi, moderate lymphoid depletion was observed in bursa and thymus, and mild lymphoid depletion was present in the spleen. Lymphocytic to histiocytic airsacculitis was observed in both infected and noninfected commercial turkeys. In NDV-infected pigeons, microscopic findings were lymphoplasmacytic airsacculitis and brain lesions, but mild airsacculitis was also present in the noninfected pigeons. Mild gliosis and perivascular cuffing were observed in the brainstem and cerebellum at 14 and 18 dpi. In addition, there was moderate intestinal lymphoid hyperplasia at 18 dpi.
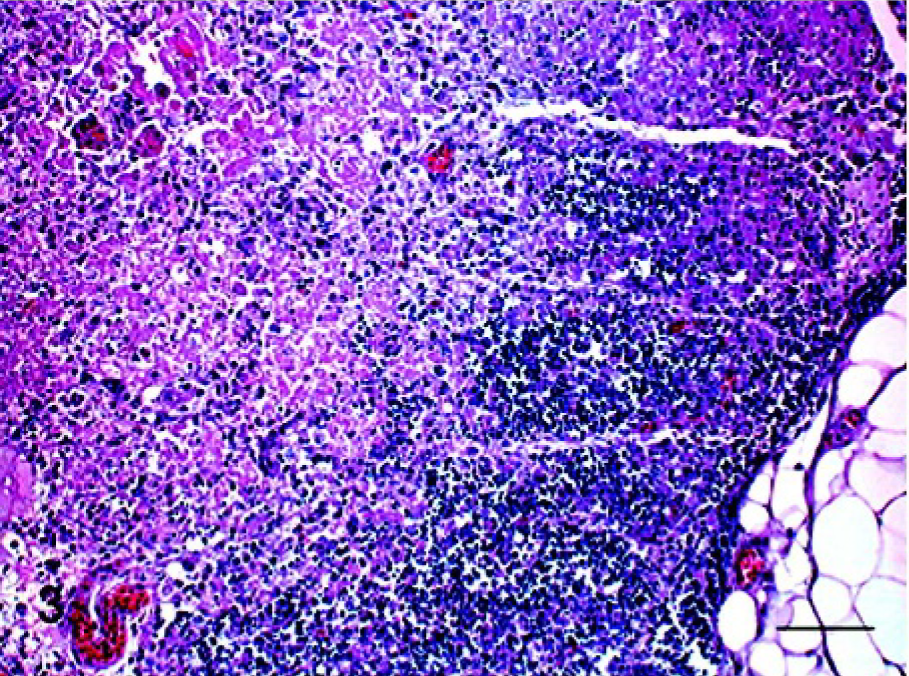
Thymus; chicken infected 5 days previously. Severe lymphoid depletion and extensive areas of necrosis were present. HE stain. Bar = 60 μm.
Main histologic lesions in organs from chickens, SPF turkeys, commercial turkeys, and pigeons infected with California END virus.∗

SPF = specific-pathogen-free; END = Exotic Newcastle disease; dpi = days postinoculation.
Immunohistochemistry
The results of IHC from NDV-infected birds are summarized in Table 3. The tissues of the noninfected controls were consistently negative by the IHC assay. In chickens at 2 dpi, there were positive mucosal epithelial cells in focal areas of the eyelid. Multiple macrophages and lymphocytes infiltrating in the superficial dermis of the comb were positive. After 3 dpi, positive cells were present in systemic organs, including lymphoid organs, respiratory and digestive tracts, heart, kidney, pancreas, brain, bone marrow, eyelid, and comb. At 3 dpi, lymphocytes and macrophages were the predominant types of cells with positive staining. These were diffusely and generously scattered in the spleen; however, in the thymus and bursa (Fig. 4), positive cells were observed only in limited areas, the medulla and interfollicular regions, respectively. In the respiratory and digestive tracts, positive cells were lymphocytes and macrophages infiltrating in mucosa, submucosa, or both. In the lungs, positive cells were present in bronchial-associated lymphoid tissue and multiple lymphoid aggregates. A few positive epithelial cells were observed in crop. Lymphoid aggregates admixed with monocytes in bone marrow were positive for viral nucleoprotein. In the kidney, interstitial lymphocytes and macrophages were also virus positive. In the heart, infiltrating macrophages along with multiple myocytes were positive for NDV. Lymphocytes in focal areas of leptomeninges were virus positive. By 4 dpi, positive lymphocytes and macrophages were present extensively in some areas of organs, and a large numbers of respiratory and intestinal epithelial cells were positive. In the thymus and bursa, positive cells were present diffusely. In the respiratory tract, epithelial cells of the larynx (Fig. 5) and trachea were strongly NDV positive. In the lung, positive cells, most likely macrophages, were scattered in alveolar septa. In the digestive tract, large numbers of epithelial cells were positive along with infiltrating lymphocytes and macrophages. The staining pattern was similar, and this increased by 5 dpi. Additionally, at this time point, multiple Kupffer cells in the liver and renal tubular epithelial cells became virus positive. In the brain, multiple cerebellar Purkinje cells and the cells involved in gliosis in the cerebellum and brainstem were positive.
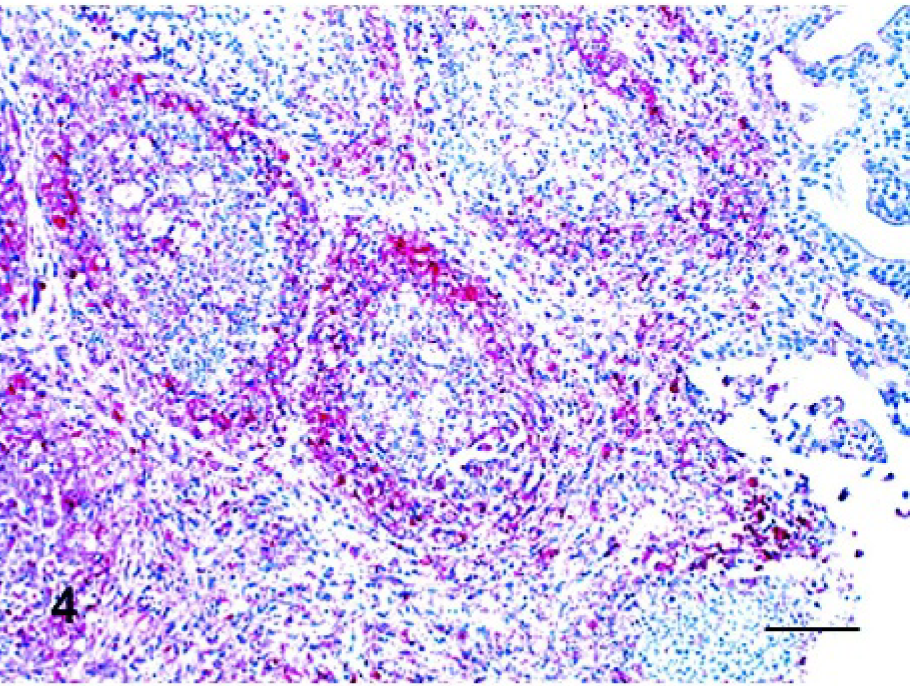
Bursa; chicken infected 5 days previously. Viral antigen (red stained) was detected in large numbers of lymphocytes and macrophages. Immunohistochemistry, Mayer's hematoxylin counterstain. Bar = 60 μm.
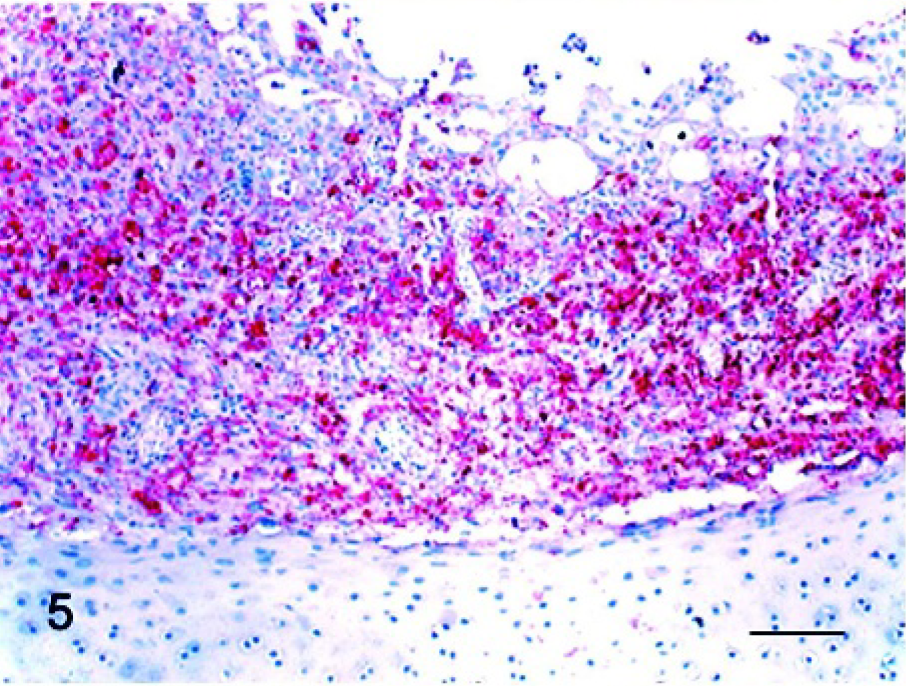
Larynx; chicken infected 4 days previously. Viral antigen (red stained) was detected in large numbers of respiratory mucosal epithelial cells and infiltrating lymphocytes and macrophages. Immunohistochemistry, Mayer's hematoxylin counterstain. Bar = 60 μm.
Extent of viral infection characterized by the presence of NDV nucleoprotein and mRNA detected by IHC and ISH, respectively, in tissues from birds infected with California END virus.∗
NDV = Newcastle disease virus; IHC = immunohistochemistry; ISH = in situ hybridization; END = Exotic Newcastle disease; SPF = specific-pathogen-free; dpi = days postinoculation; − = negative for NDV nucleoprotein by IHC; + = < 2 positive cells per high power field (HPF); ++ = 2–10 positive cells per HPF; +++ = >10 positive cells per HPF; −/∗ = negative by IHC but positive by ISH, indicating viral mRNA.
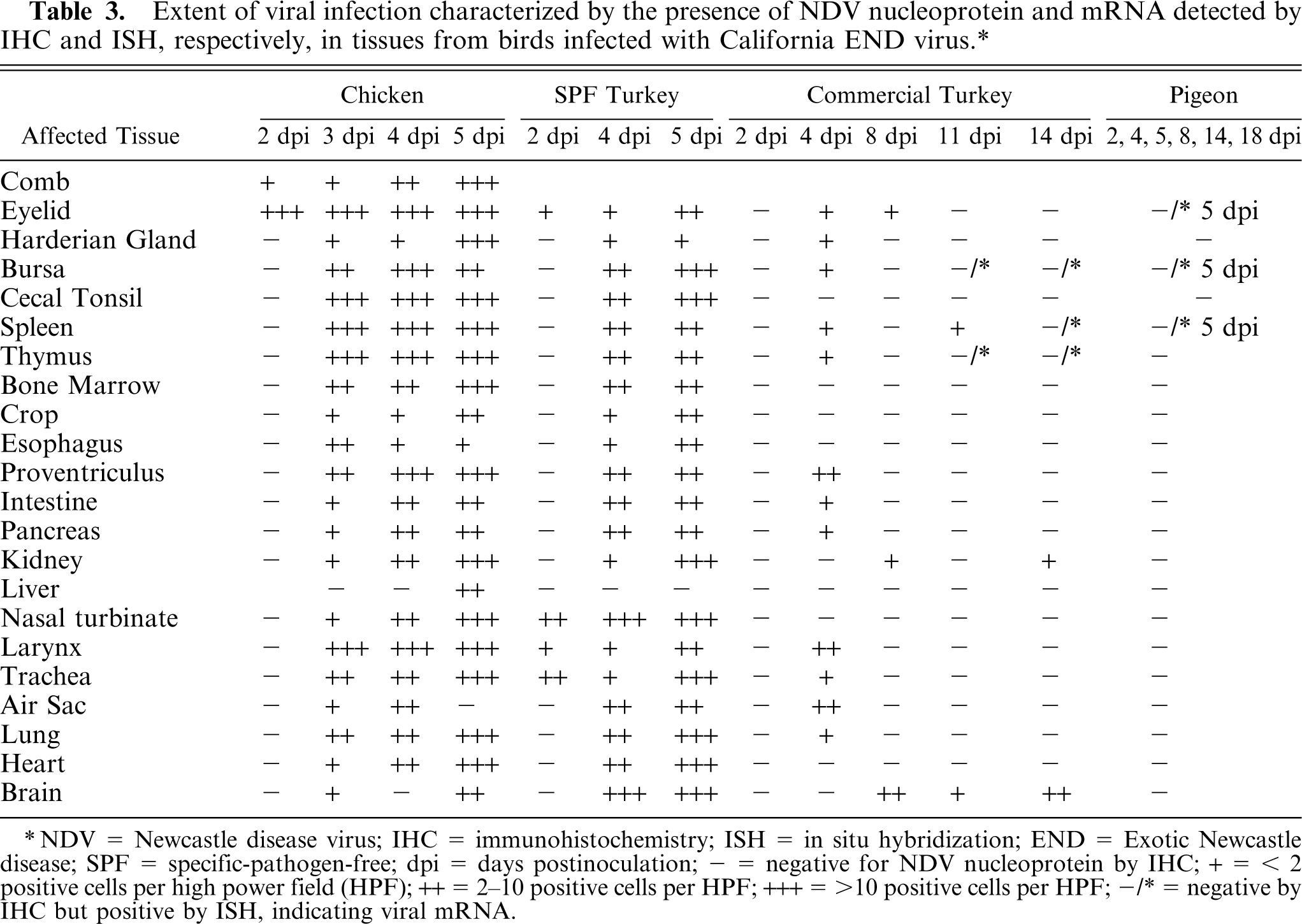
In SPF turkeys at 2 dpi, NDV-positive cells were present in the inoculated site and the upper respiratory tract. In conjunctiva, infiltrating macrophages and lymphocytes were stained positive. Virus-positive respiratory epithelial cells were present in the turbinate, larynx, and trachea. By 4 dpi, the positive cells extended to the systemic organs, including lymphoid organs, respiratory and digestive tracts, heart, kidney, pancreas, brain (Fig. 6), bone marrow, and eyelid. Large numbers of positive cells were present in the lymphoid organs; however, staining was less intense than in chickens. Compared to chickens, the SPF turkeys had greater amounts of viral nucleoprotein in the brain and lesser amounts in the larynx.
Cerebellum; SPF turkey infected 5 days previously. Viral antigen (red stain) was detected in large numbers of Purkinje cells and glial cells. Immunohistochemistry, Mayer's hematoxylin counterstain. Bar = 60 μm.
In commercial turkeys at 4 dpi, NDV-positive cells were scattered in small numbers but were present in multiple organs, including the eyelid, lymphoid organs, respiratory tract, and digestive tract. In lymphoid organs, lymphocytes and macrophages were virus positive. There were also positive respiratory epithelial cells in the larynx, trachea, and air sac. Small numbers of NDV-positive lymphocytes and macrophages were present in the lung. There were virus-positive mucosal epithelial cells in the proventriculus and small intestine, and multiple lymphocytes and macrophages infiltrating proventricular submucosa were positive. By 8 dpi, viral nucleoprotein was detected primarily in the brain among both neurons and glia in foci of gliosis in the cerebellum and brainstem. IHC in pigeons was consistently negative in all tissues throughout all time points following infection with the California outbreak virus.
In situ hybridization
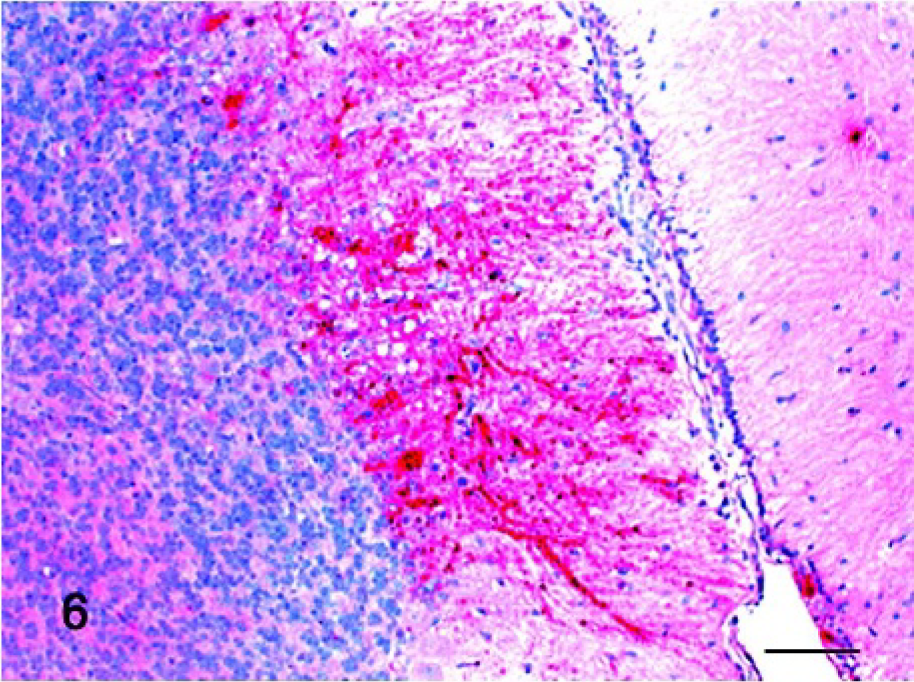
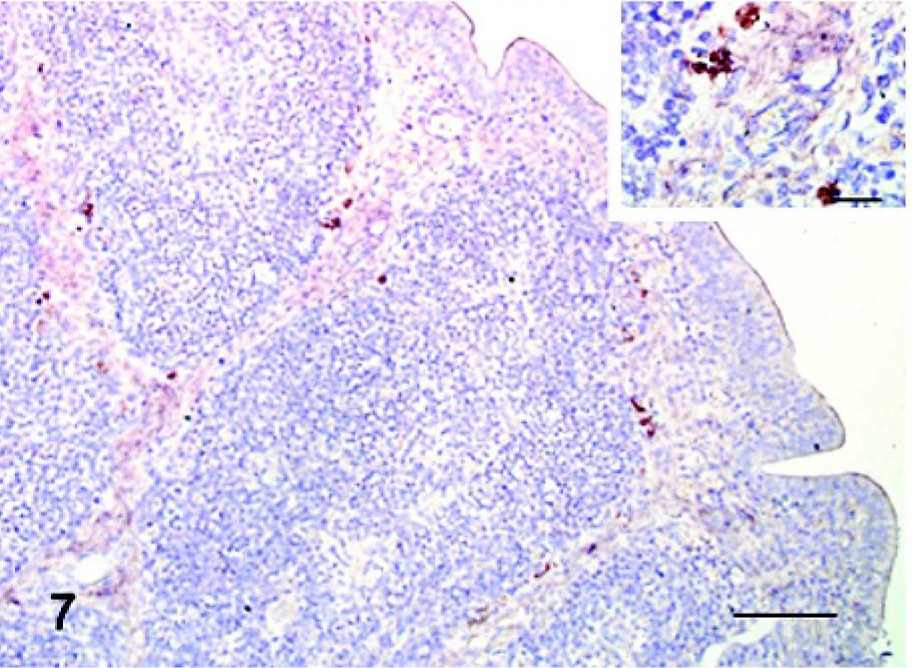
The selected tissue sections stained by ISH confirmed the distribution of the viral infection as detected by IHC. However, ISH detected several additional sites of viral replication in commercial turkeys and pigeons. In commercial turkeys at 11 and 14 dpi, small numbers of positive cells were observed in lymphoid tissues, including bursa (Fig. 7), spleen, and thymus. In pigeons at 5 dpi, small amounts of viral mRNA were detected in the eyelid, bursa, and spleen (Fig. 8). The tissues of noninfected controls were consistently negative by ISH.
Bursa; commercial turkey infected 11 days previously. Viral mRNA (brown stained) was detected in multiple perifollicular lymphocytes. In situ hybridization, Mayer's hematoxylin counterstain. Bar = 60 μm. Inset: higher magnification of IHC-positive lymphocytes. Bar = 20 μm.
Spleen; pigeon infected 5 days previously. Viral mRNA (brown stained, arrow head) was detected in a macrophage. In situ hybridization, Mayer's hematoxylin counterstain. Bar = 30 μm. Inset: higher magnification of an ISH-positive macrophage. Bar = 10 μm.
Virus isolation from swabs and serology
All oral and cloacal swab samples from inoculated chickens (collected at 2, 3, 4, and 5 dpi) and SPF turkeys (2, 4, and 5 dpi) were positive for virus isolation. Infection with virus in all inoculated commercial turkeys and pigeons was confirmed by swabs (either oral, cloacal, or both) (Table 4) or by seroconversion postinoculation (data not presented). At 2 dpi, which was the first time point collecting swabs after inoculation, the virus was isolated from all oral swabs and 40% of cloacal swabs from inoculated commercial turkeys. One of 2 commercial turkeys still shed the virus at 14 dpi, which was detected from both oral and cloacal swabs. In infected pigeons, virus was isolated from 75% of oral swabs and 62.5% of cloacal swabs at 2 dpi. Both oral and cloacal swabs from a pigeon at 8 dpi were positive for virus isolation, but no virus was isolated from swabs at 14 dpi. Virus was not isolated from any swab samples of noninfected control birds, and blood samples from all birds at preinoculation and from noninfected control birds at termination were negative by HI test.
Virus isolation from oral and cloacal swabs of commercial turkeys and pigeons infected with California END virus.∗
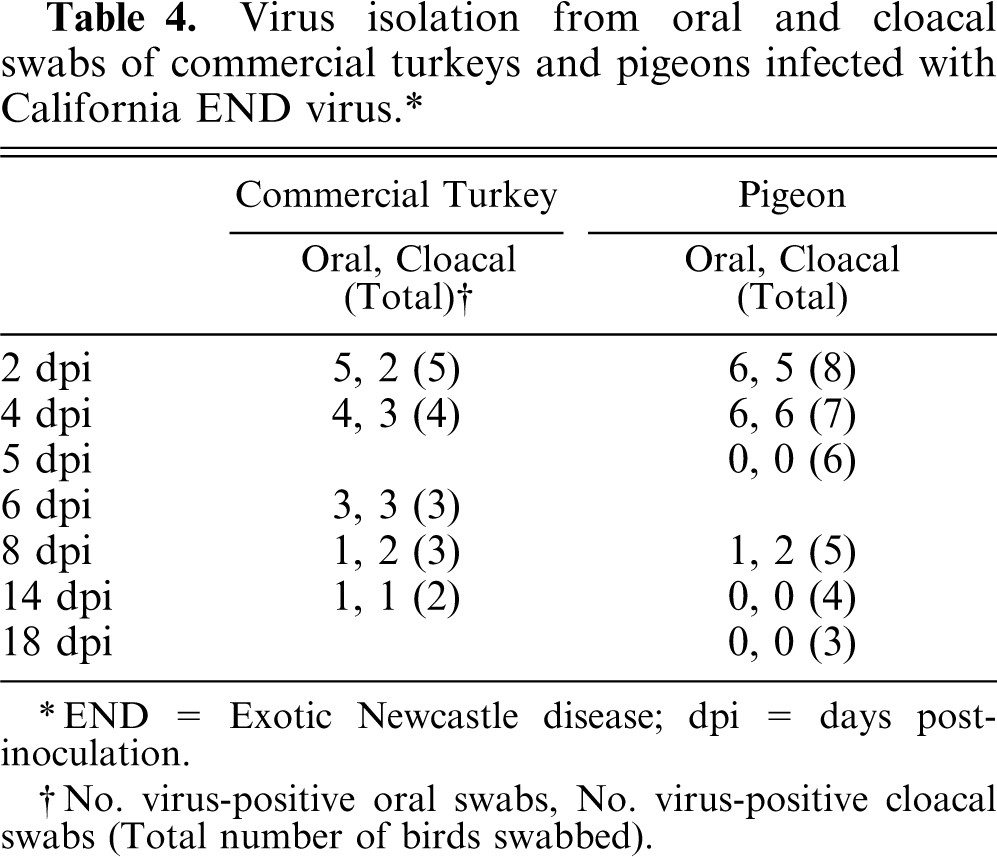
END = Exotic Newcastle disease; dpi = days postinoculation.
No. virus-positive oral swabs, No. virus-positive cloacal swabs (Total number of birds swabbed).
Discussion
Chickens, SPF turkeys, commercial turkeys, and pigeons exhibited different susceptibilities to the END virus isolated from a California outbreak during 2002–2003. Chickens and SPF turkeys were highly susceptible to the virus with 100% mortality by 5 dpi, and viral dissemination and replication occurred in multiple systemic sites. Severe lymphoid depletion and necrosis in lymphoid tissues were observed in both; however, chickens appeared to have more severe necrosis with foci of large amounts of positive cells indicating more extensive virus replication than did SPF turkeys. In chickens, an unusual finding was severe necrosis of cranial tracheal epithelial cells along with extensive lymphocytic infiltration. These tracheal lesions were also observed in SPF turkeys, but were less extensive relative to chickens. Conversely, SPF turkeys had greater damage and amounts of positive cells indicating more extensive virus replication in the brain than did chickens. At 2 dpi, viral distribution was limited to the inoculated sites in chickens; however, in SPF turkeys at this same time point, virus detection extended into the upper respiratory tract. This earlier viral dissemination in SPF turkeys may allow time for virus invasion into the brain, which may explain why greater amounts of positive cells indicating more extensive virus replication were present in the brains of SPF turkeys compared to chickens.
Commercial turkeys were less susceptible than chickens or SPF turkeys. At 4 dpi, virus distribution was present in multiple organs, but the amount of positive cells indicating virus replication was much less than in chickens or SPF turkeys. Virus distribution was limited to the lymphoid organs, the brain, or both after 8 dpi among commercial turkeys. Commercial turkeys were older than chickens or SPF turkeys; therefore, the lower susceptibility in commercial turkeys could potentially be attributable to a difference in age rather than to species or breed.
In the studies of Alexander et al., 3 turkeys appeared more resistant than age-matched chickens to viscerotropic velogenic NDV (GB 97/6 and GB/1). However, our study demonstrated that chickens and SPF turkeys appeared to have similar susceptibility to California END virus although there were some differences in viral distribution. In addition, commercial turkeys were less susceptible than chickens or SPF turkey.
Pigeons were the least susceptible to the California END virus. Clinical disease and histologic lesions were minimal. No viral protein was detected by IHC in tissues collected. However, at 5 dpi a small amount of viral mRNA was detected in the eyelid and lymphoid organs by ISH, which confirms infection and systemic replication. It has been reported that virus detection by ISH was slightly more sensitive than by IHC and that ISH may appear earlier than IHC because of the presence of viral mRNA before the manufacturing of viral protein. 5, 12 There were histologic changes in the air sacs and brain with no evidence of virus by either IHC or ISH. As previously described by Brown et al., 6 it could be a possible function of sampling of the air sacs, because only small sections of epithelium were present in paraffin-embedded sections. The exact cause of the brain lesions was unknown. Small amount of the virus that could not be determined by either IHC or ISH may cause these lesions. Also, pigeons seroconverted after inoculation with NDV. A previous study documented that juvenile and adult pigeons experimentally infected intraconjunctivally with viscerotropic velogenic NDV (Fontana 1083, 106.1 ELD50 per bird) developed clinical disease characterized by head tremors, wry neck, opisthotonous, wing droop, and leg paralysis, with some mortality. 7 In that report, the onset of the clinical disease was at 7 dpi in juveniles and at 18 dpi in adults, with higher mortality and morbidity in juveniles relative to adults. In our study, 1 pigeon had mild body tremors at 10 dpi; however, there was no mortality.
The results demonstrate that chickens and SPF turkeys were most susceptible to the California END virus. Commercial turkeys were less susceptible than chickens and SPF turkeys, whereas pigeons were least susceptible. In conclusion, the susceptibility to the NDV isolated from the outbreak in California during 2002–2003 varies among 4-week-old SPF chickens, 3-week-old SPF turkeys, 6-week-old commercial turkeys, and 10- to 20-week-old pigeons. Again, we have demonstrated that pigeons may carry highly virulent NDV without overt signs of clinical disease.
Footnotes
Acknowledgements
We acknowledge the excellent technical support of Phillip Curry and Tracy Smith-Faulkner. This work was supported by the US Poultry and Egg Association (project number 368) and the USDA Agricultural Research Service (CRIS number 6612-32000-038-OOD).
