Abstract
To shed some light on the mechanisms behind renal fibrogenesis, the present study immunohistochemically investigated the participation of different macrophage populations and myofibroblastic cells in rat renal interstitial fibrosis developed chronically after repeated injection of cisplatin (2 mg/kg body weight, once weekly for 7 weeks). During the 19-week recovery period after the final injection, fibrotic lesions progressively developed in the corticomedullary junction, with the greatest level at post-final injection (FPI) week 5, and then the lesions were gradually repaired by PFI week 19, indicative of a healing process. In conformity with the development of fibrotic lesions, the number of myofibroblastic cells reacting with an anti-α-smooth muscle actin antibody was increased, with a peak at PFI week 3, and collagens (types I, III, and IV), fibronection, and laminin were excessively accumulated in these areas. Interstitial cells forming the fibrotic lesions showed mitotic activity at the early stages, whereas they disappeared by apoptosis in the healing process. A large number of cells reacting with an antibody of ED1 (for exudate macrophages), ED2 (for resident macrophages), or OX6 (for major histocompatibility complex class II-presenting macrophages and interstitial dendritic cells) had already appeared at PF1 week 1, and then their numbers increased, with a peak at PFI weeks 7, 3, and 9 in ED1-, ED2-, and OX6-positive cells, respectively. Thereafter, the number of ED1- and ED2-positive cells decreased, whereas the number of OX6-positive cells persisted at a high level until PFI week 19. In the healing process, clusters of lymphocytes were present, the development of which might have been related to OX6-positive cells. The present study demonstrated that chronically developing rat renal interstitial fibrosis might be produced by the complicated mechanisms evoked by interactions between different macrophage populations and myofibroblastic cells, because macrophages show heterogeneous functions depending on microenvironmental factors.
Macrophages infiltrating injured tissues produce peptide growth factors such as transforming growth factor-β1 (TGF-β1) and platelet-derived growth factor.15,18,20,38,43 Such factors induce the development of myofibroblastic cells capable of producing extracellular matrix (ECM), such as collagens and fibronection, leading to the formation of fibrotic lesions as a result.2,30,39 Macrophages and myofibroblastic cells are key cells in fibrogenesis. Renal interstitial fibrosis, which is the common final pathway after renal glomerular and tubular injury,1,11,13–15,29,38 also has been characterized by the participation of such cell types.14,19,36,38,42,45,46 Thus, an expansion of infiltrating macrophages is well known to correlate with an increased number of myofibroblastic cells in the early stages of experimental rat renal fibrosis.1,11,14,20 Accordingly, the administration of essential fatty acid–deficient diet or protein-restricted diet, which inhibit macrophage infiltrate into lesions, ameliorated the degree of interstitial fibrosis in puromycin aminonucleoside–induced rat nephrosis.10,13 Sublethal x-irradiation to rats with ureteral obstruction also resulted in a decrease in numbers of macrophages and consequent development of myofibroblastic cells in the affected kidneys.11 Macrophages seem to act as an early trigger in the genesis of renal interstitial fibrosis.
Macrophages infiltrating in experimentally induced rat renal lesions have been identified by immunohistochemistry with ED1, a monoclonal antibody that recognizes monocytes and activated macrophages, particularly exudate macrophages.9,11,12,26,27,38,41 In addition to exudate macrophages, macrophage populations include resident macrophages (so-called histiocytes or fixed macrophages) and cells differentiating into dendritic cells (macrophage-related cells).31,37,44,47 These macrophage populations differ in ontogeny, morphology, immunophenotypes, and functions.16,37 Kupffer cells, which are the resident macrophages in the liver, have been reported to play a pivotal role in experimental hepatic fibrosis or granuloma by producing fibrogenic factors, such as blood monocyte–derived exudate macrophages.16,23,25 The participation of different macrophage populations in renal fibrosis has not yet been investigated.
Previously, we established a renal interstitial fibrosis model by administering cisplatin (
Materials and Methods
Animals and CDDP dosing
Thirty-nine male F344/DuCrj rats, weighing 100–131 g, were obtained from Charles River Japan (Hino, Shiga, Japan) at the age of 5 weeks, and used in the present study after 1 week of acclimatization. During this experiment, animals were housed in an animal room controlled at 22 ± 3 C and with a 12-hour light:dark cycle, and allowed free access to a standard commercial diet (MF, Oriental Yeast Co. Ltd., Tokyo, Japan) and tap water. The experiment was in compliance with our institutional guideline for animal care. Rats in the CDDP group were injected intraperitoneally with CDDP (Nippon Kayaku Co., Ltd., Tokyo, Japan) once weekly for 7 weeks at 2 mg/kg body weight.42 Three rats were examined each at weeks 1, 3, 5, 7, 9, 11, 13, 15, 17, and 19 after the final injection of CDDP. Rats in the control group received the equivalent volume of physiologic saline in the same manner, and three rats were examined each at weeks 1, 9, and 19 after the final injection. Serum samples from blood obtained under ether anesthesia at autopsy were tested for blood urea nitrogen (BUN) and creatinine with an Autoanalyser 705 (Hitachi, Tokyo, Japan). One hour before the autopsy, all animals received intraperitoneal injection of 5′-bromo-2′-deoxyuridine (BrdU, 50 mg/kg body weight) for detection of nuclear incorporation in S phase. Body weights were measured once a week during the 7-week injection and following 19-week recovery periods.
Histopathology and immunohistochemistry
All animals were necropsied. One kidney was fixed in 10% neutral buffered formalin and the other in Methacarn solution (methanol–chloroform–acetic acid, 6:3:1) for 1 hour at 4 C.45 Formalin- and methacarn-fixed renal tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (HE). Formalin-fixed tissue sections were also stained by the azan-Mallory method for evaluation of developed fibrotic areas.
The immunohistochemical analyses were performed by the avidin–biotin complex method (LSAB kit, Dako Corp., Carpinteria, CA) with primary antibodies listed in Table 1. The methods have been described elsewhere.26,44,45 Briefly, after pretreatments as described below, formalin- and methacarn-fixed, deparaffinized sections were incubated with 1.5% skimmed milk for 15 minutes at room temperature for protein blocking, and incubated with each primary antibody for 14 hours at 4 C. One-hour incubation with biotinylated goat anti-mouse antibody for monoclonal antibodies or goat anti-rabbit antibody for polyclonal antibodies followed. Final incubation was carried out for 1 hour with an avidin–horseradish peroxidase complex, and positive reactions were visualized with 3,3′-diaminobenzidine (DAB). Sections were lightly counterstained with hematoxylin. Nonimmunized mouse and rabbit sera, used instead of primary antibodies, served as negative controls. To quench endogenous peroxidase, sections for ED1, ED2, OX6, α-smooth muscle actin (α-SMA), laminin, and fibronection were pretreated with 0.1% trypsin for 15 minutes at 37 C and then 3% H2O2 for 10 minutes, whereas sections for collagen types I, III, and IV were incubated with 0.0004% pepsin for 15 minutes at 37 C and then 3% H2O2 for 15 minutes. For BrdU staining, sections were pretreated with 3% H2O2 for 10 minutes and then treated with 4 N CHl at 37 C for 20 minutes to denature DNA. The immunohistochemical method for TGF-β1 has been described.45 Sections for TGF-β1 were pretreated with 6 M urea at room temperature for 30 minutes and then with 0.5% periodic acid for 15 minutes to block endogenous peroxidase. To confirm the specificity, a primary TGF-β1 antibody that had been preincubated with TFG-β1 (1 µg/ml; UBI, Lake Placid, NY) was used as a negative control.
Primary antibodies.
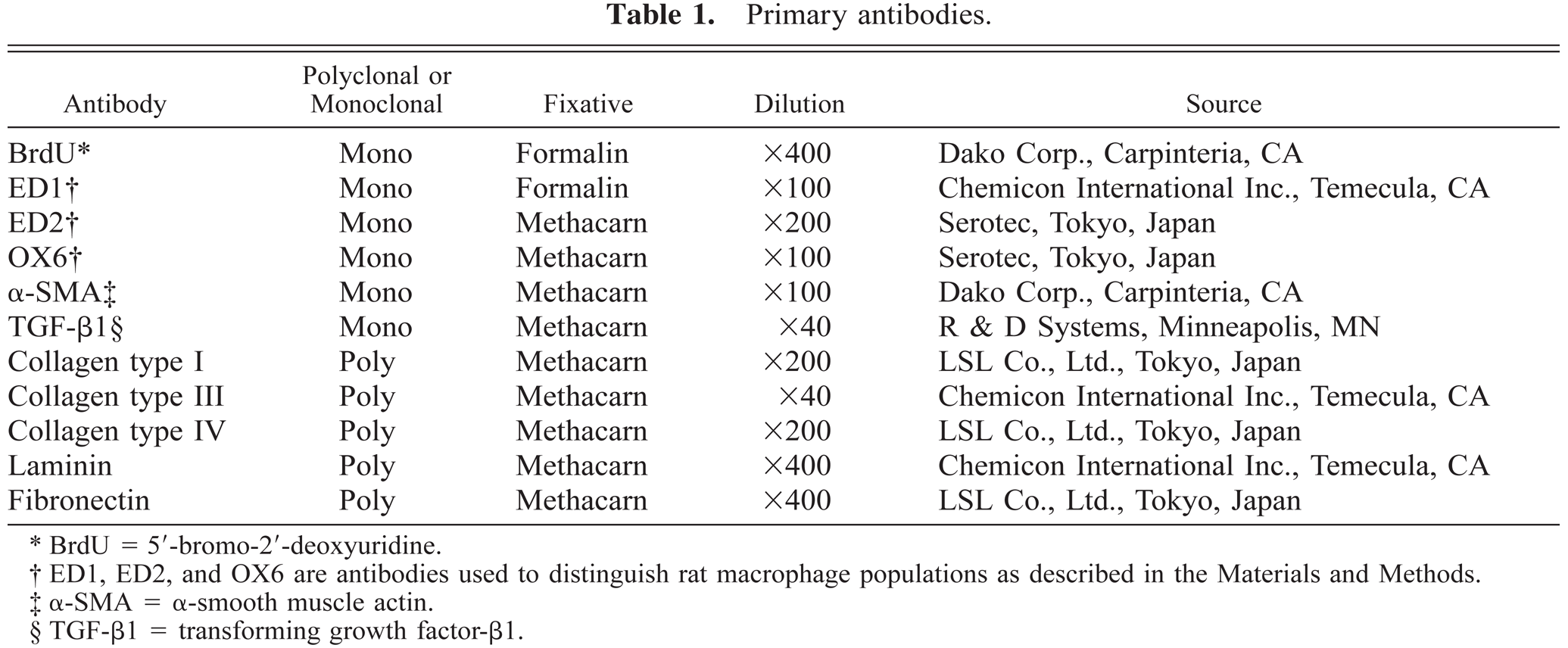
BrdU = 5′-bromo-2′-deoxyuridine.
ED1, ED2, and OX6 are antibodies used to distinguish rat macrophage populations as described in the Materials and Methods.
α-SMA = α-smooth muscle actin.
TGF-β1 = transforming growth factor-β1.
ED1, ED2, and OX6 antibodies were used to distinguish macrophage populations. ED1 labels blood monocytes and exudate macrophages in rats, and antigens recognized by ED1 are on the membrane of cytoplasmic granules, especially phagolysosomes.7,12,41 ED2 reacts with cell membrane antigens of rat resident macrophages, but the functions of ED2-recognizing antigens are still undetermined.7,12,35 OX6 is an antibody against rat major histocompatibility complex (MHC) class II antigen (Ia) that is mainly expressed on activated macrophages, dendritic cells, and B cells.35,49,50 α-SMA has been recognized to be induced in developing myofibroblastic cells in fibrotic lesions, and anti–α-SMA antibody has been used to detect the cells.2,11,15,26,27,33,43,45
Terminal deoxyribonucleotide transferase (TdT)–mediated deoxyuridine triphosphate nick end labeling (TUNEL) for apoptotic cells
A standard in situ TUNEL (Apop Tag Kit, Oncor Inc., Gaithersburg, MD) method was used for detection of DNA fragmentation in apoptotic cells.9,22 After treatment with proteinase K for 20 minutes at 37 C and then with 3% H2O2 for 20 minutes at room temperature to quench endogenous peroxidase activity, formalin-fixed, deparaffinized sections were incubated with TdT and digoxigenin DNA labeling mixture in the TdT reaction buffer for 60 minutes at 37 C. Subsequently, the sections were blocked with 10% normal goat serum and 10% fetal calf serum for 30 minutes at 37 C, and then treated with horseradish peroxidase–conjugated anti-digoxigenin for 60 minutes. Peroxidase was detected with DAB. The sections were counterstained lightly with hematoxylin. In negative controls, TdT enzyme was replaced with distilled water.
Evaluation and statistical analysis
Cells showing a distinct positive reaction for ED1, ED2, OX6, or α-SMA immunohistochemistry were counted in 10 randomly selected areas (0.0625 mm2/area) in the corticomedullary junction of cross sections at a magnification of 400× under a light microscope.42,45,46 Because OX6 also labels B cells,35,49,50 small round, lymphoid cells with scant cytoplasm and a hyperchromatic nucleus and cells constituting lymphoid follicles as mentioned in the Results were omitted from the count in OX6 immunohistochemistry. Because cells undergoing apoptosis were seen infrequently, the positive cells were counted in 30 randomly selected areas (0.0625 mm2/area) at a magnification of 400×. The fibrotic areas stained blue by the azan-Mallory method were measured by a color image analyzer (Mac SCOPE, Mitani Inc., Tokyo, Japan) in five different portions (0.5 mm2/area) of the corticomedullary junction; the percentage of fibrotic tissues per unit area was calculated.42,45,46 Because no differences in these data were observed among three control groups (weeks 1, 9, and 19), they were treated as one control group for statistical analysis. Paired samples were compared by Student's
Accumulation of extracellular matrix (ECM) components.∗

± = very faintly positive staining; + = faintly positive staining; 2+ = moderately positive staining; 3+ = severely positive staining. The grading evaluation was made in each ECM component by comparing with grade of control; see more detailed evaluation methods in the Materials and Methods.
Results
Cisplatin-induced renal failure
During the 7-week injection period and at post–final injection (PFI) weeks 1–13 and 16 and 17, the body weights of animals in the CDDP group were significantly decreased, but gradually returned to those of animals in the control group for the 19-week recovery period (Fig. 1). Both BUN and creatinine values showed the highest levels at PFI week 1, indicative of renal failure due to repeated injection of CDDP, but they recovered to levels of controls at PFI week 19 (Fig. 2).
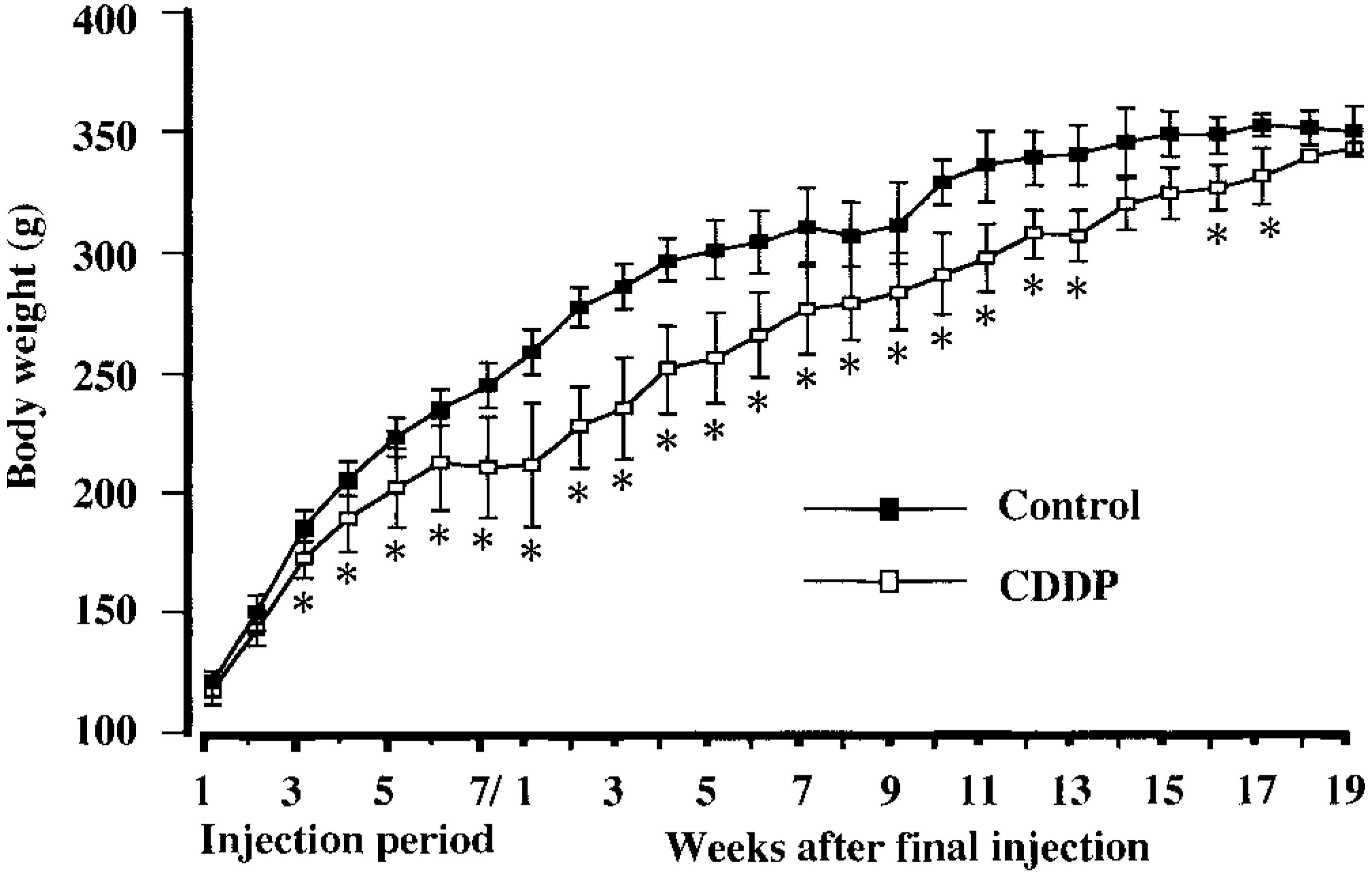
Body weight changes during the 7-week injection period of cisplatin (CDDP; once a week) and following 19-week recovery period. ∗ Significantly (
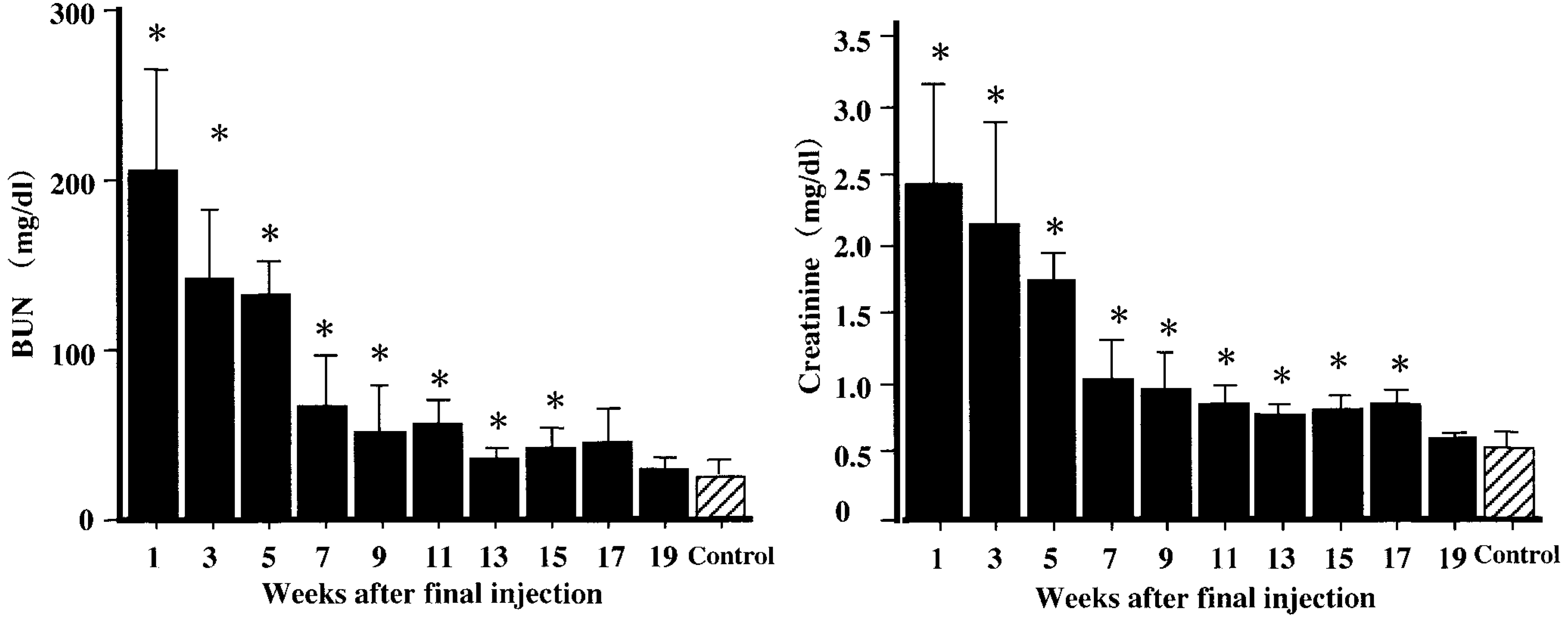
Blood urea nitrogen (BUN) and creatinine values. ∗ Significantly (
Initial lesions by CDDP seen at PFI weeks 1–5 were necrosis or degeneration or both of renal tubular epithelial cells in the corticomedullary junction. In addition to such lesions, fibrotic areas developed around the affected tubules, accompanied by infiltration of inflammatory cells such as macrophages and lymphocytes. Fibrotic tissues increased as early as at PFI week 1; the percentage of fibrotic tissue reached a maximum at PFI week 5, and gradually subsided to PFI week 19 (Fig. 3). The fibrotic tissues seen in the early stages at PFI weeks 1–7 were widely distributed in the damaged corticomedullary junction (Fig. 4). Thereafter, the damaged areas gradually were replaced by renal tubules rimmed by regenerated epithelial cells, and at PFI weeks 15–19, the fibrotic areas were seen only around the atrophied renal tubules with thickened basement membrane (Fig. 5). Lymphocyte clusters were seen in the affected areas (Fig. 5), and, interestingly, the clusters often formed follicles in the late stages at PFI weeks 11–19 (see Fig. 18).
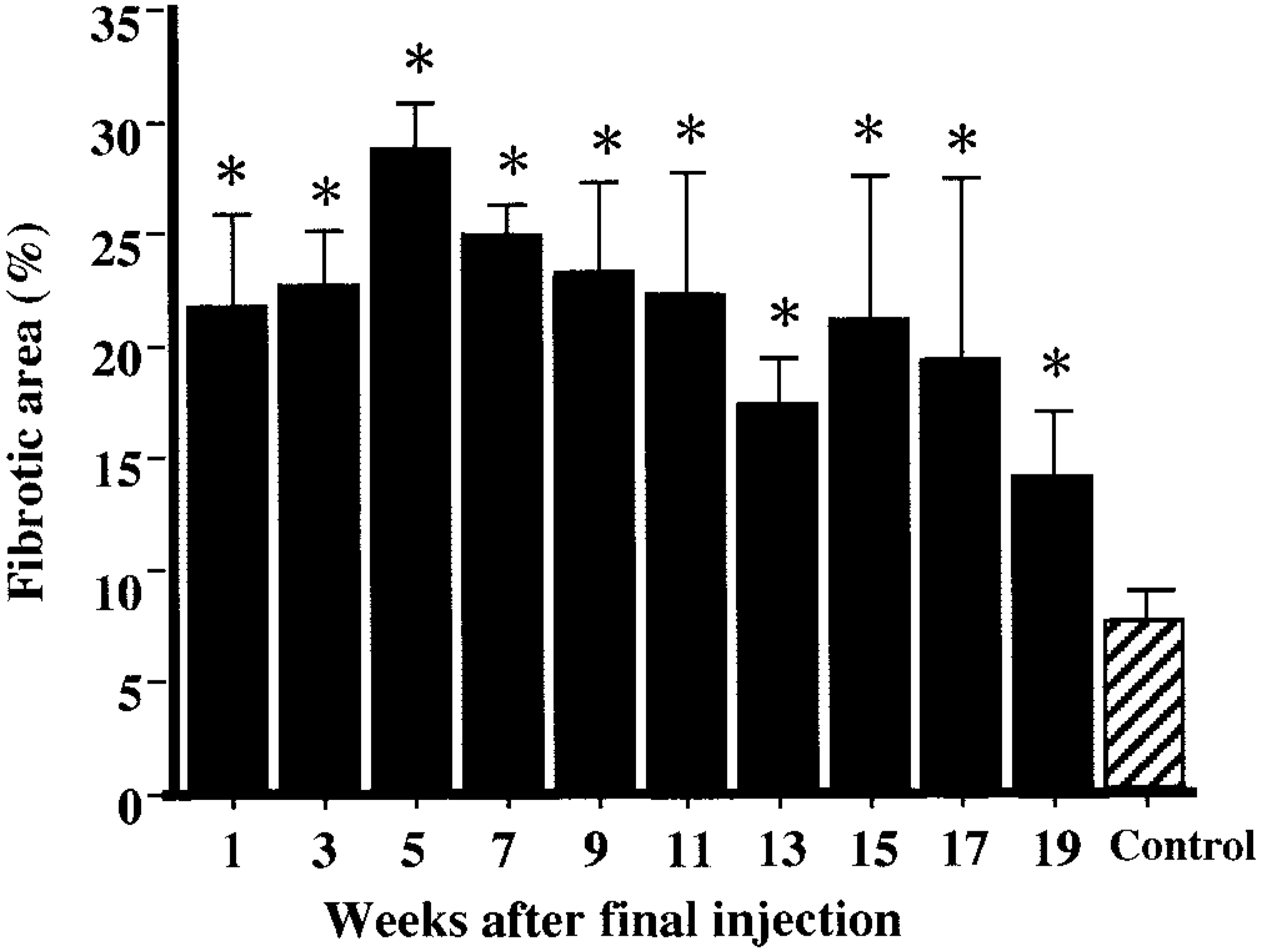
Morphologic analysis; fibrotic area per unit area (%). ∗ Significantly (
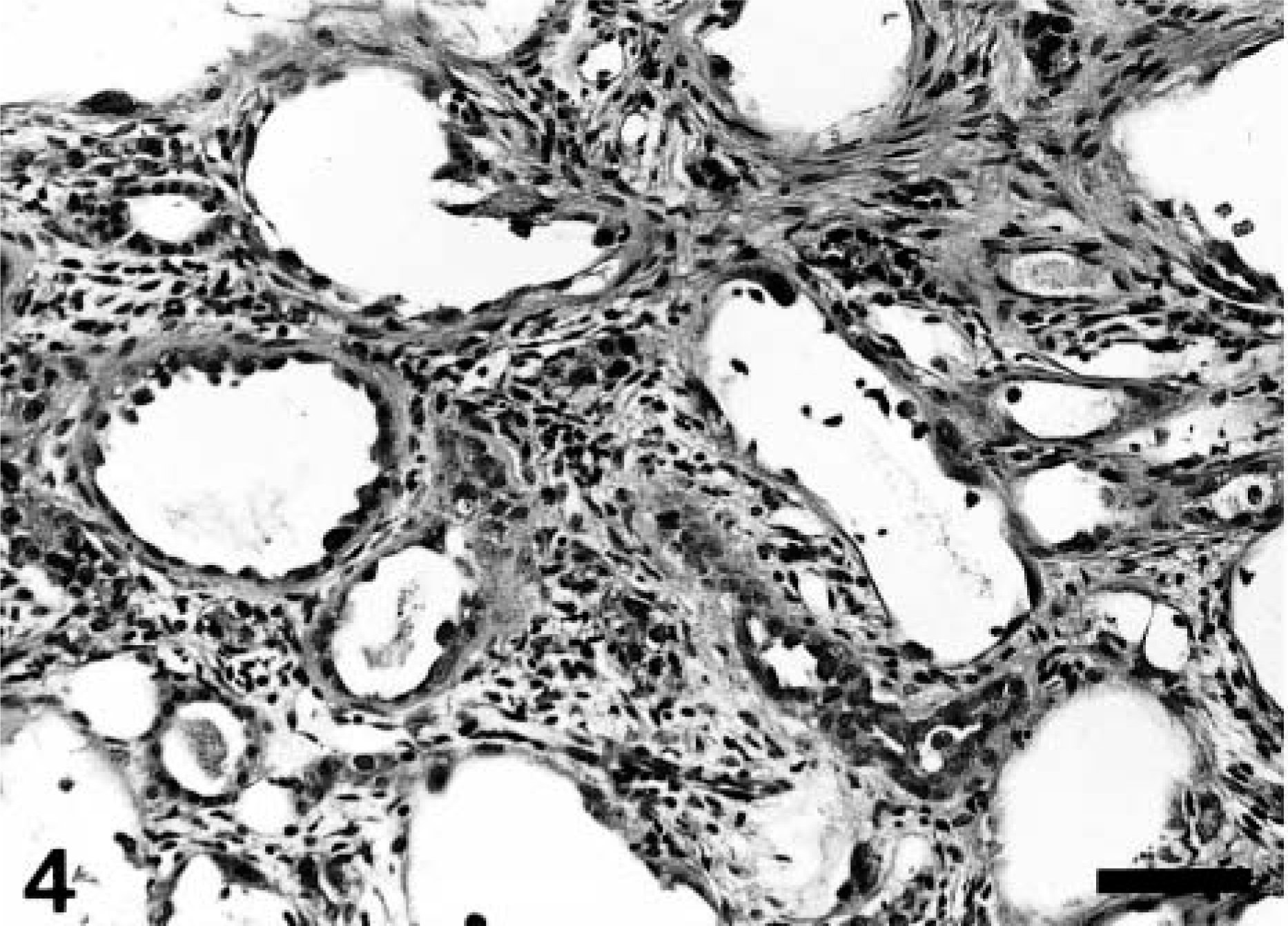
Kidney; CDDP-treated rat at post–final injection (PFI) week 5. Severely developed fibrotic lesions with variously dilated renal tubules and infiltration of inflammatory cells such as macrophages and lymphocytes in the corticomedullary junction. HE stain. Bar = 40 μm.
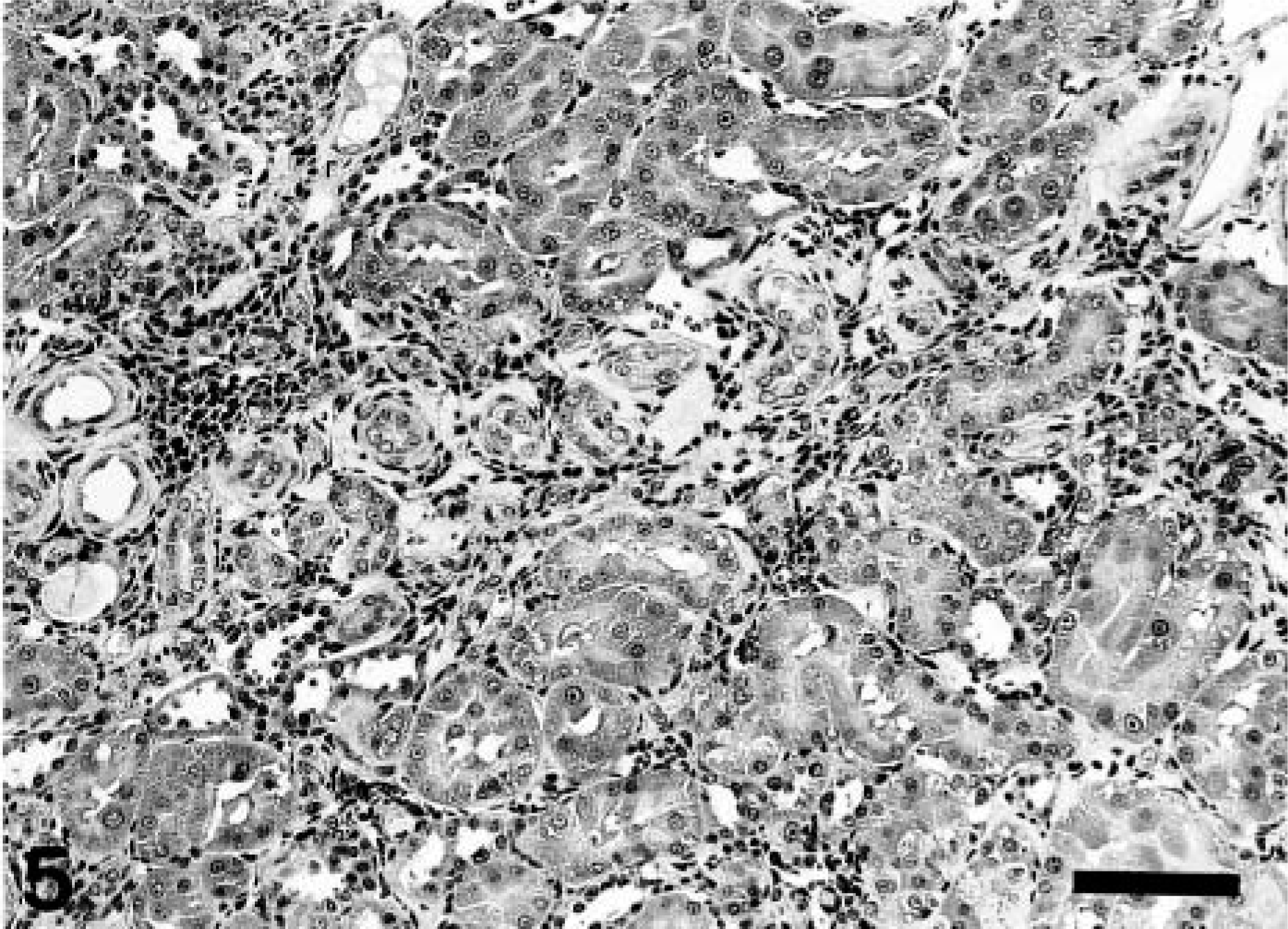
Kidney; CDDP-treated rat at PFI week 19. Fibrotic lesions consisting of atrophied renal tubules with thickened basement membranes and lymphocyte aggregate. HE stain. Bar = 50 μm.
Control rats examined at PFI weeks 1, 9, and 19 showed no renal lesions indicative of chronic progressive nephropathy, which has been reported in rats more than 1 year old.27
Myofibroblastic cells, BrdU-immunopositive cells, and ECM deposition
During the 19-week recovery period, the number of myofibroblastic cells reacting with an anti–α-SMA antibody was significantly increased, with a peak at PFI week 3, and decreased thereafter (Fig. 6). The myofibroblastic cells were observed mainly in the fibrotic areas and around cystically dilated renal tubules in the corticomedullary junction (Fig. 7).
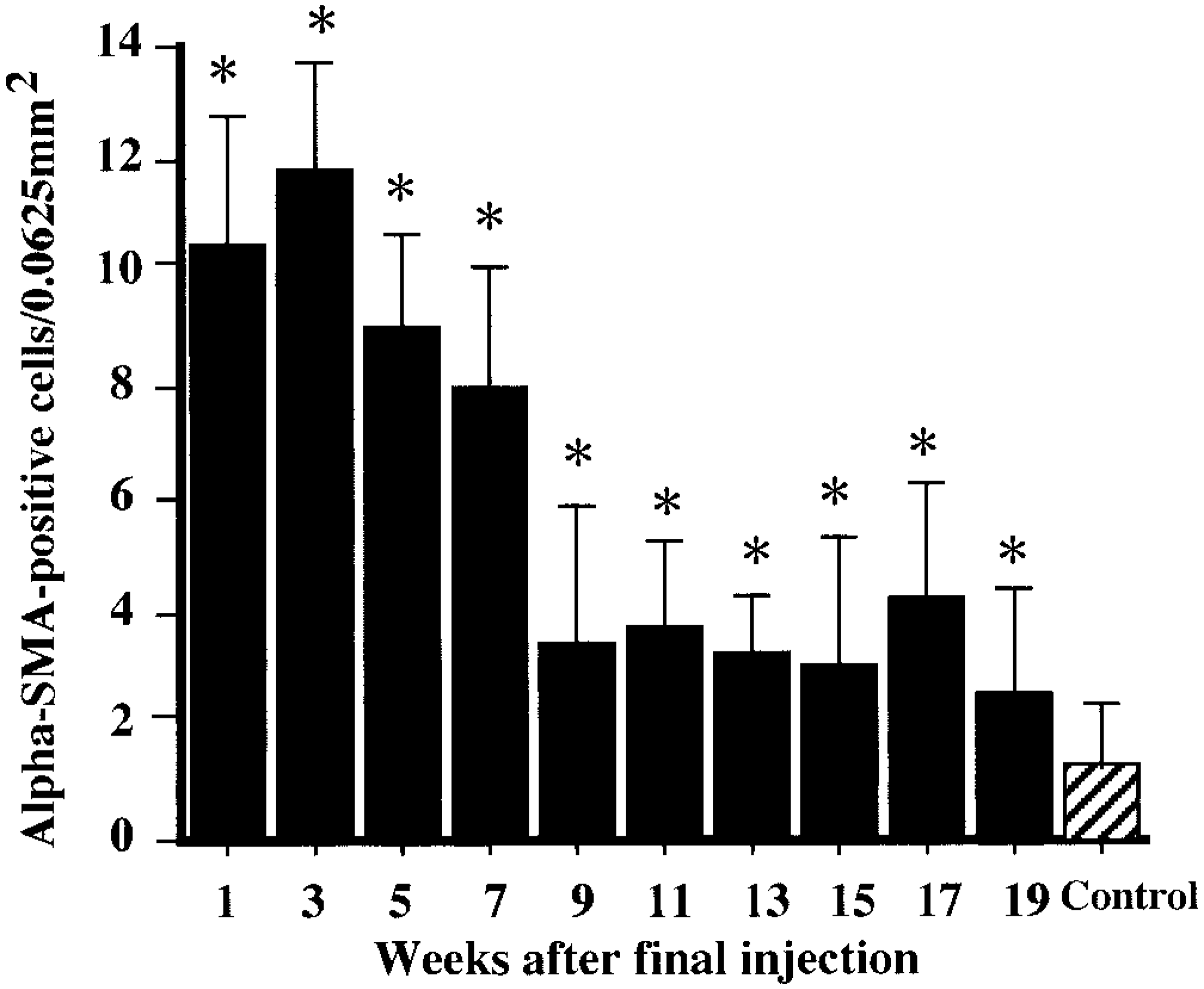
Kinetics of myofibroblastic cells reacting with an antibody against α-smooth muscle actin (α-SMA). ∗ Significantly (
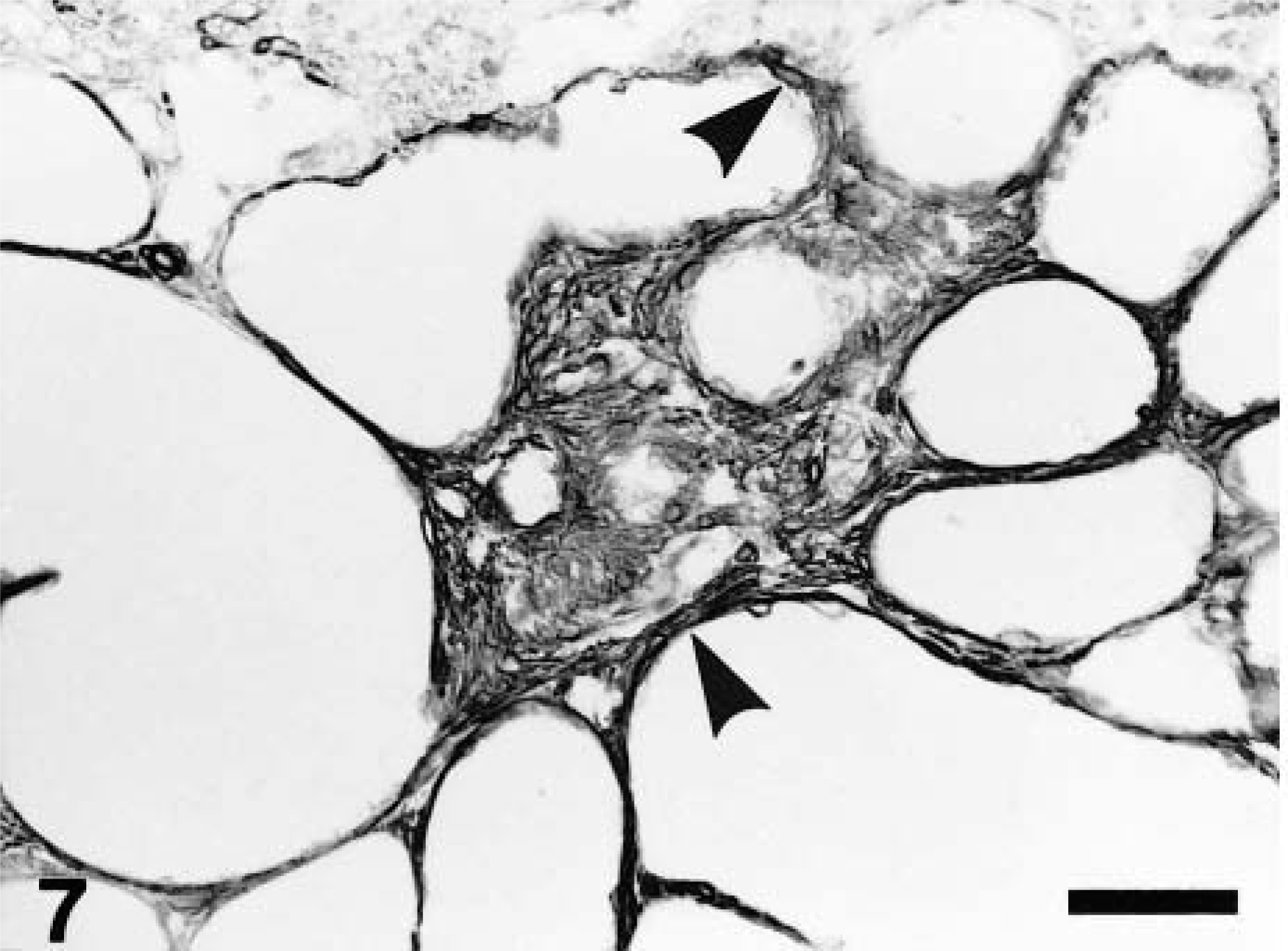
Kidney; CDDP-treated rat at PFI week 3. α-SMA-immunopositive myofibroblastic cells (arrowheads) are present in the fibrotic areas and around cystically dilated renal tubules. Immunohistochemistry, counterstained with hematoxylin. Bar = 40 μm.
BrdU-immunopositive interstitial cells in the fibrotic areas frequently were observed at PFI weeks 1–7 (Fig. 8), but from PFI week 9 onward, such interstitial cells reacting to BrdU were rarely present in the fibrotic areas (Fig. 9). As reported previously,42 BrdU-immunopositive renal epithelial cells also were observed especially at PFI weeks 1–7 (data not shown), indicating active regeneration of renal epithelial cells for repair after the injury.
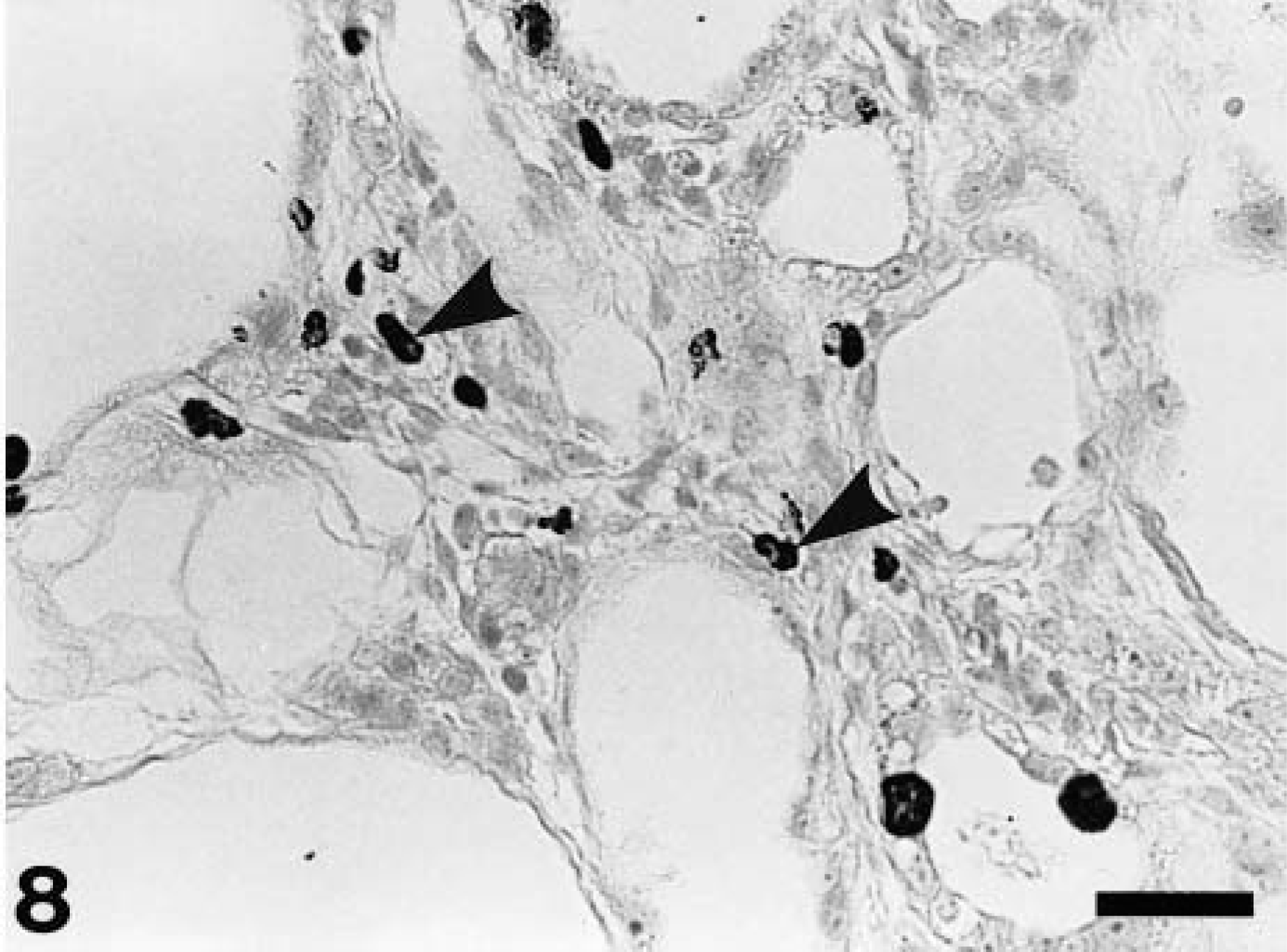
Kidney; CDDP-treated rat at PFI week 3. Bromodeoxyuridine (BrdU)-immunopositive interstitial cells (arrowheads), indicative of proliferative activity, are seen in the fibrotic areas. Immunohistochemistry, counterstained with hematoxylin. Bar = 40 μm.
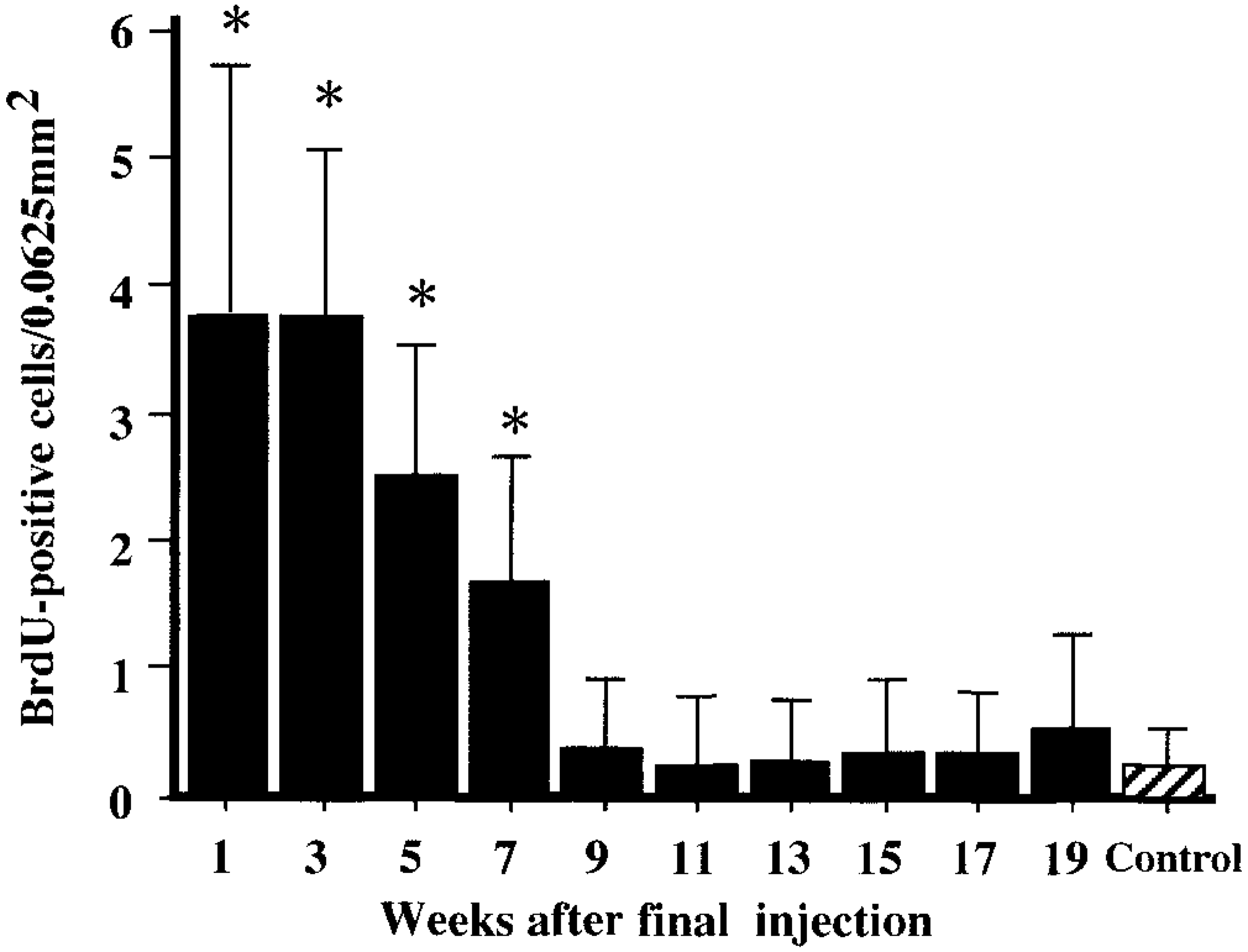
Kinetics of BrdU-immunopositive interstitial cells. ∗ Significantly (
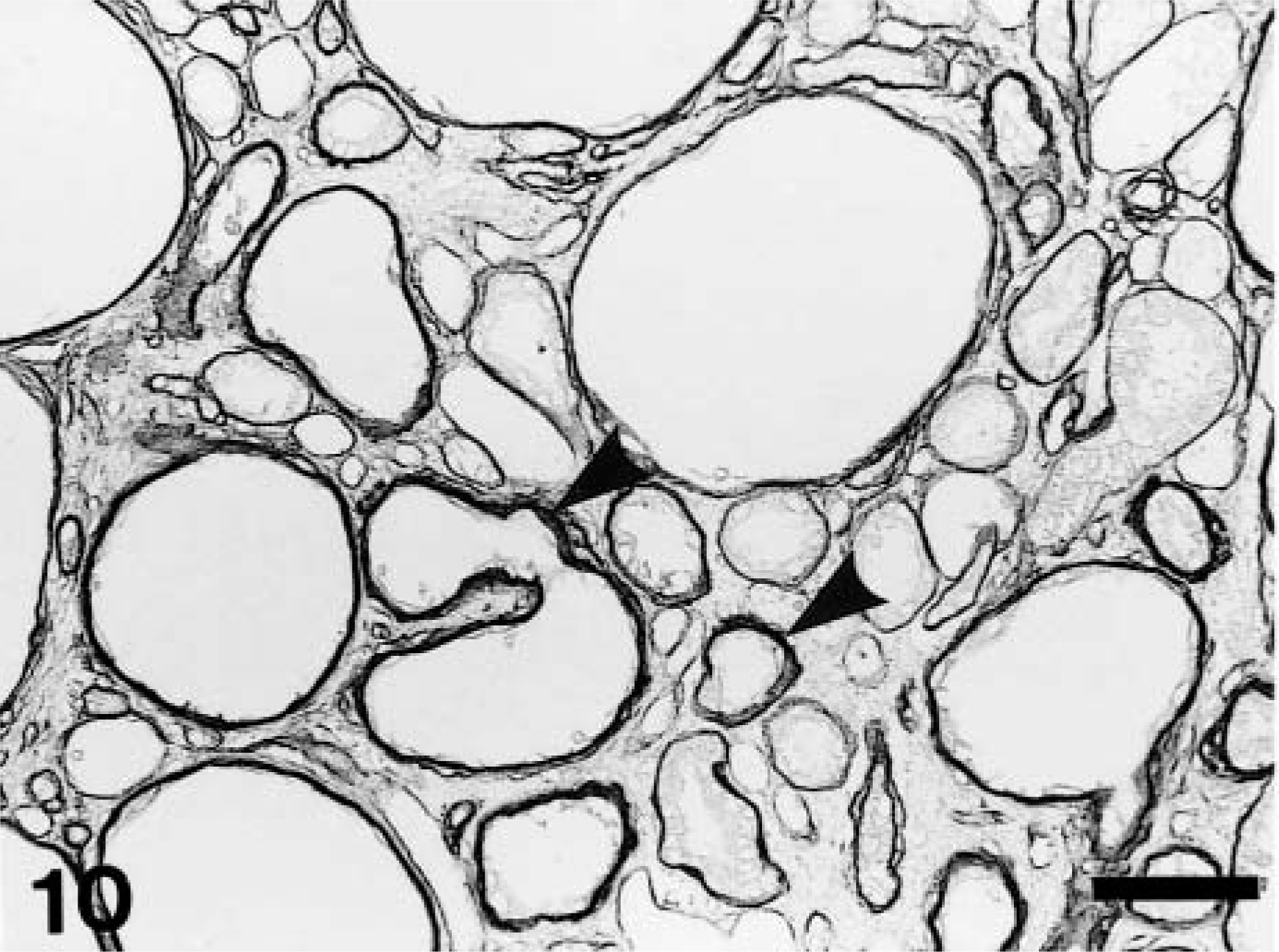
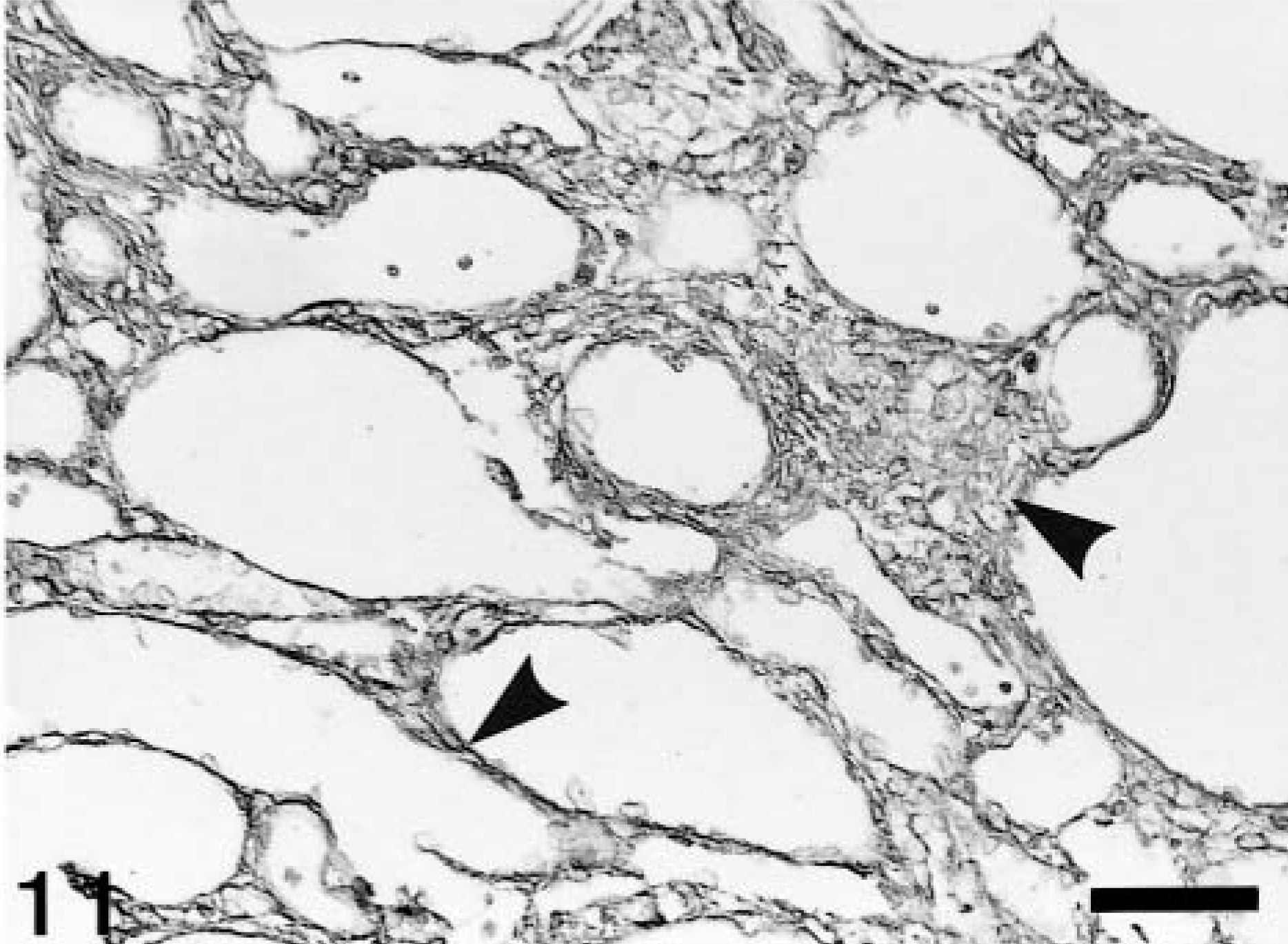
In conformity with an increment in fibrotic areas, excessive accumulation of ECM components such as collagens (types I, III, and IV), laminin, and fibronection was seen. All ECM components had already deposited severely at PFI week 1, and the severity (3+) was retained through PFI weeks 9–11 (Table 2). Thereafter, the accumulation of all ECM components gradually was decreased by PFI week 19. Although basement membrane–associated ECM components (laminin and collagen type IV) were markedly accumulated around the dilated or atrophied renal tubules (Fig. 10), interstitial ECM components (collagen types I and III, and fibronection) were present mainly in the fibrotic areas and around the affected renal tubules (Fig. 11).
Kidney; CDDP-treated rat at PFI week 5. Abnormal deposition of laminin around variously dilated renal tubules (arrowheads). Immunohistochemistry, counterstained with hematoxylin. Bar = 50 μm.
Kidney; CDDP-treated rat at PFI week 3. Abnormal deposition of collagen type III in the fibrotic areas and around dilated renal tubules (arrowheads). Immunohistochemistry, counterstained with hematoxylin. Bar = 50 μm.
Macrophage populations
In control rats, ED1-, ED2-, and OX6-immunopositive cells were rarely seen; some few cells were present in the interstitium and around arteriola in the corticomedullary junction, at not more than three cells per 0.0625 mm2. In the CDDP group, a large number of ED1-, ED2-, and OX6-positive cells already had appeared at PFI week 1, and their numbers subsequently increased (Figs. 12–14). ED1-positive cell number peaked at PFI week 7, and thereafter gradually was decreased by PFI week 19 (Fig. 12). The ED1-positive cells were round or oval in shape, and they were distributed mainly in the fibrotic areas and lumina of the dilated tubules (Fig. 15). The number of ED2-positive cells was highest at PFI week 3, and thereafter the number was rapidly declined by PFI week 9 and more slowly to PFI week 19 (Fig. 13). The majority of ED2-positive cells showed round morphology, but occasional cells were spindle-shaped; positive cells were present in the fibrotic areas but not in lumina of renal tubules (Fig. 16). The number of OX6-positive cells reached a peak at PFI week 9, and decreased at PFI week 11; however, the high level was retained with more than 25 cells per area at each PFI week until PFI week 19 (Fig. 14). The number of OX6-positive cells was consistently greater than that of ED1- and ED2-positive cells at each PFI week. OX6-positive cells revealed various configuration such as round, spindle-shaped, and dendritic; the cells were distributed in the fibrotic areas and around the dilated or atrophied renal tubules (Figs. 17, 18). Cells constituting lymphoid follicles also gave a positive reaction to OX6 (Fig. 18), indicating that they were B cells.
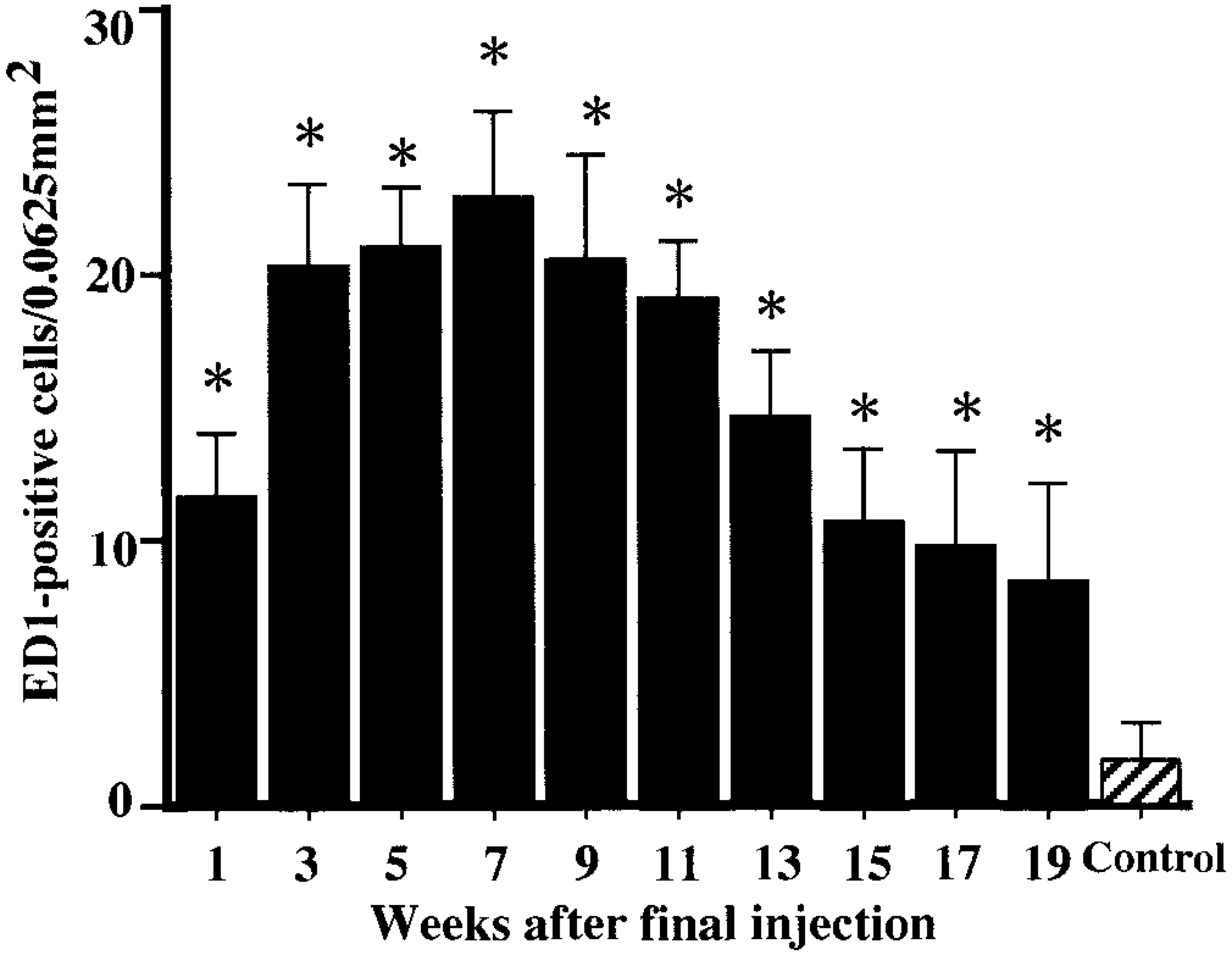
Kinetics of ED1-immunopositive macrophages. ∗ Significantly (
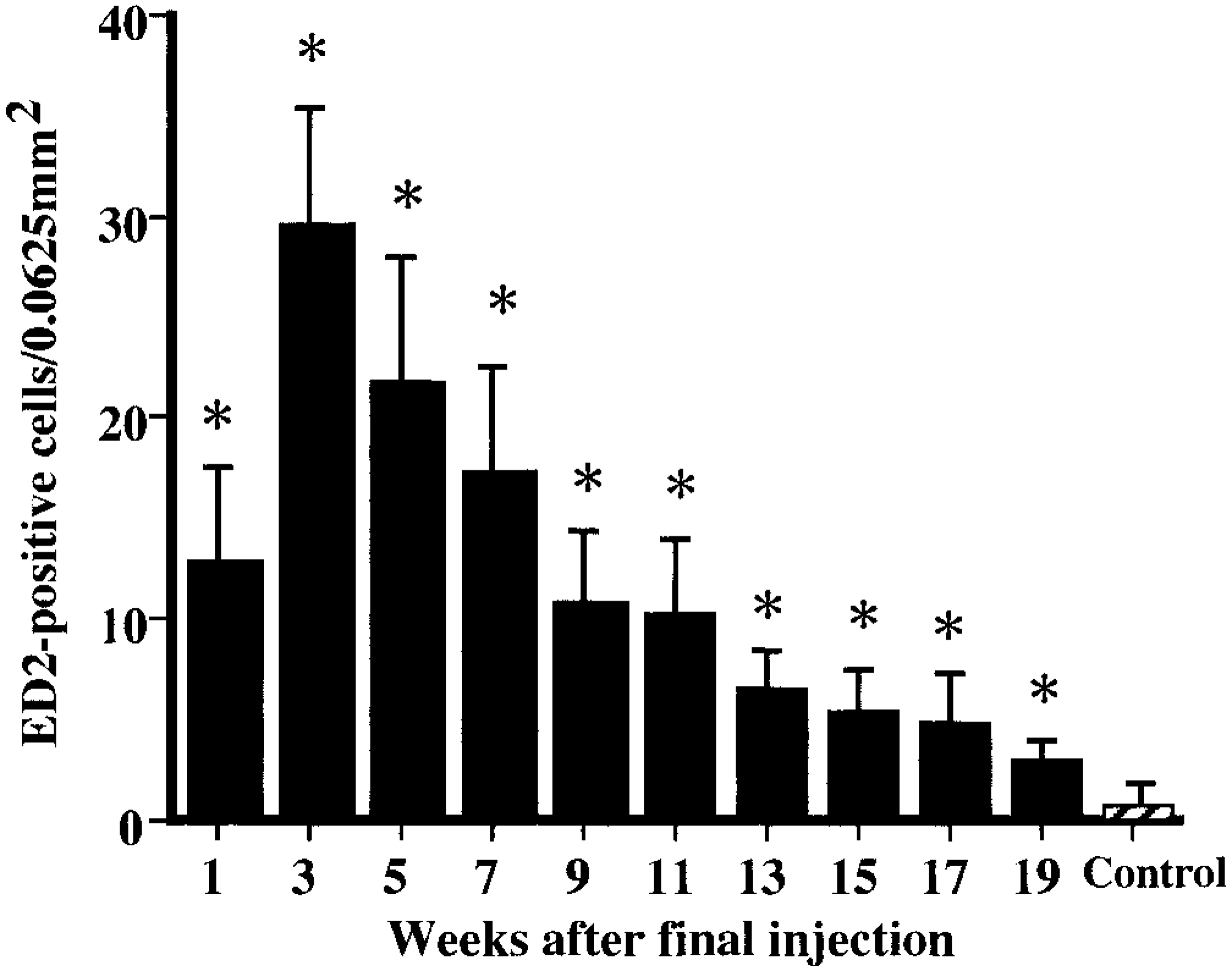
Kinetics of ED2-immunopositive macrophages. ∗ Significantly (
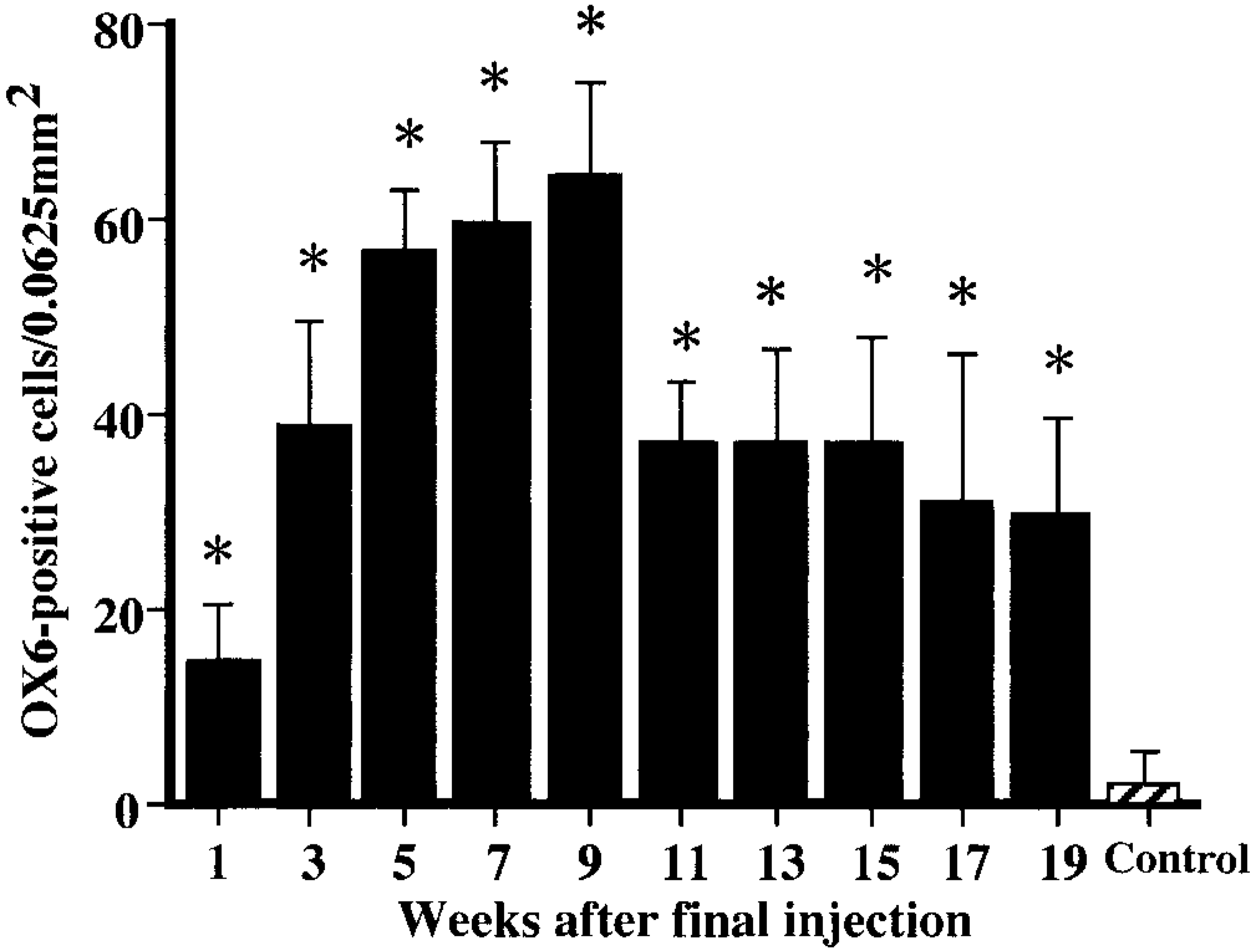
Kinetics of OX6-immunopositive macrophages. ∗ Significantly (
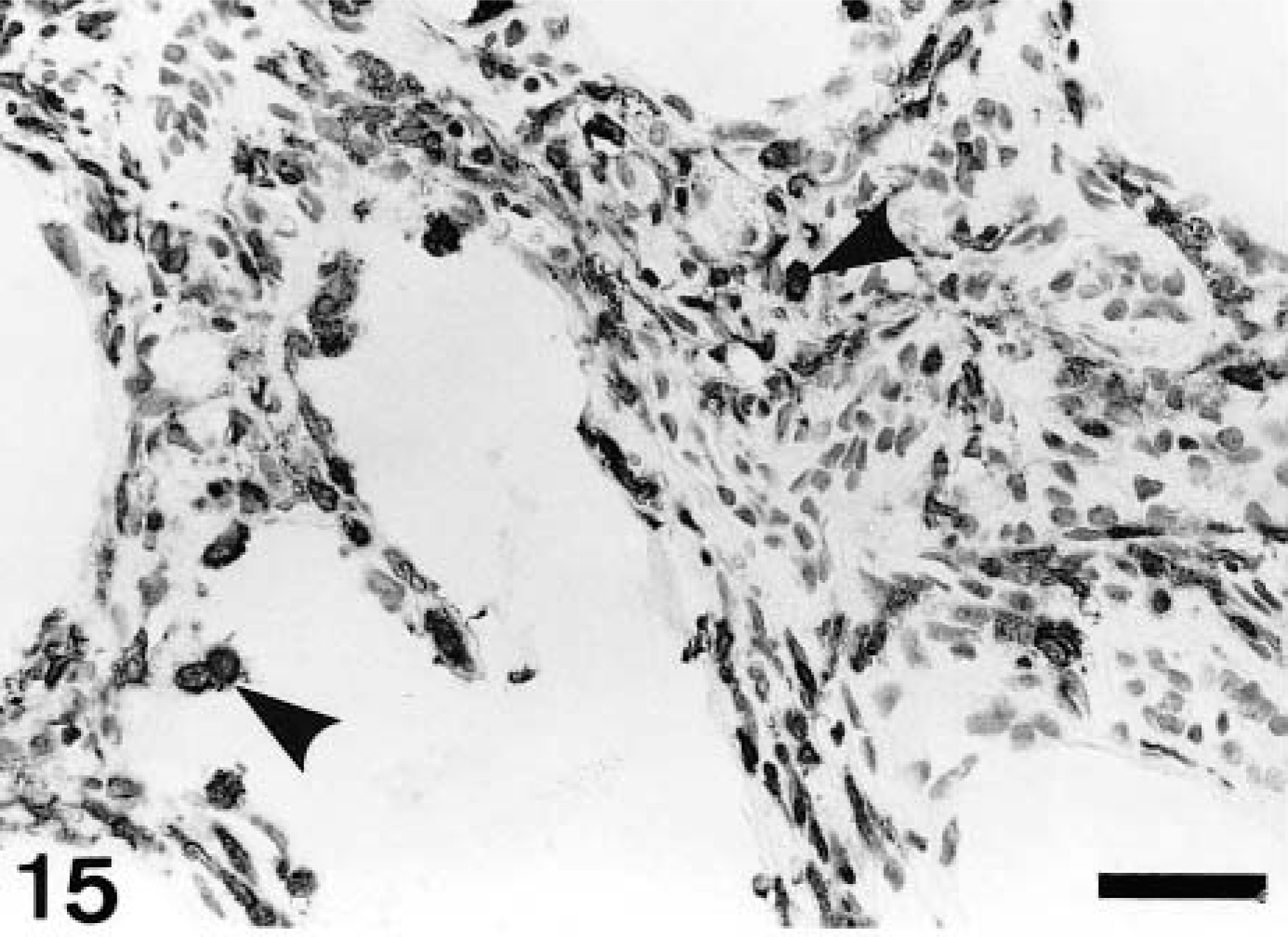
Kidney; CDDP-treated rat at PFI week 5. ED1-immunopositive exudate macrophages (arrowheads) are distributed in the fibrotic areas and lumina of dilated renal tubules. Immunohistochemistry, counterstained with hematoxylin. Bar = 40 μm.
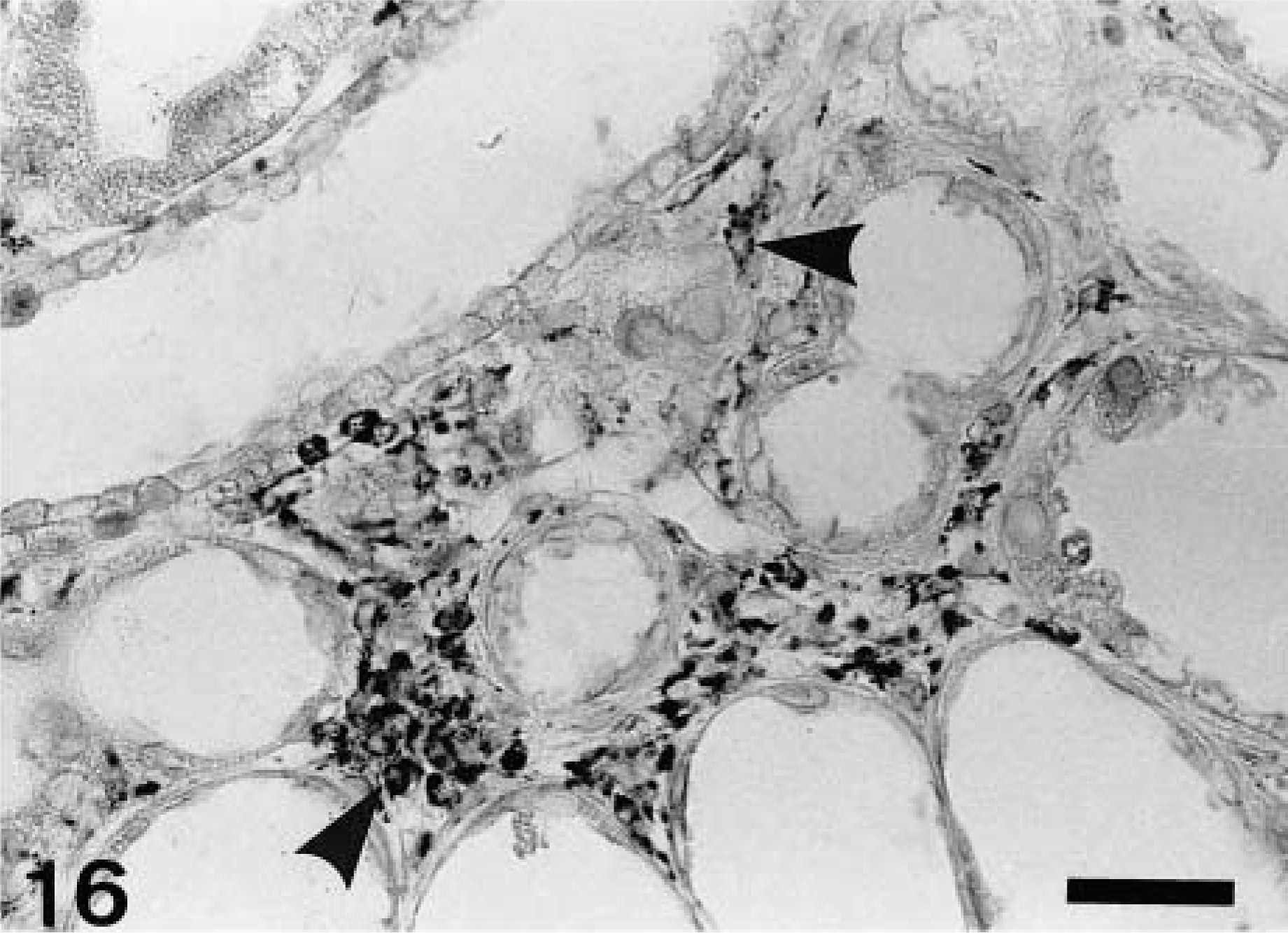
Kidney; CDDP-treated rat at PFI week 5. ED2-immunopositive resident macrophages (arrowheads) are seen in the fibrotic areas. Immunohistochemistry, counterstained with hematoxylin. Bar = 40 μm.
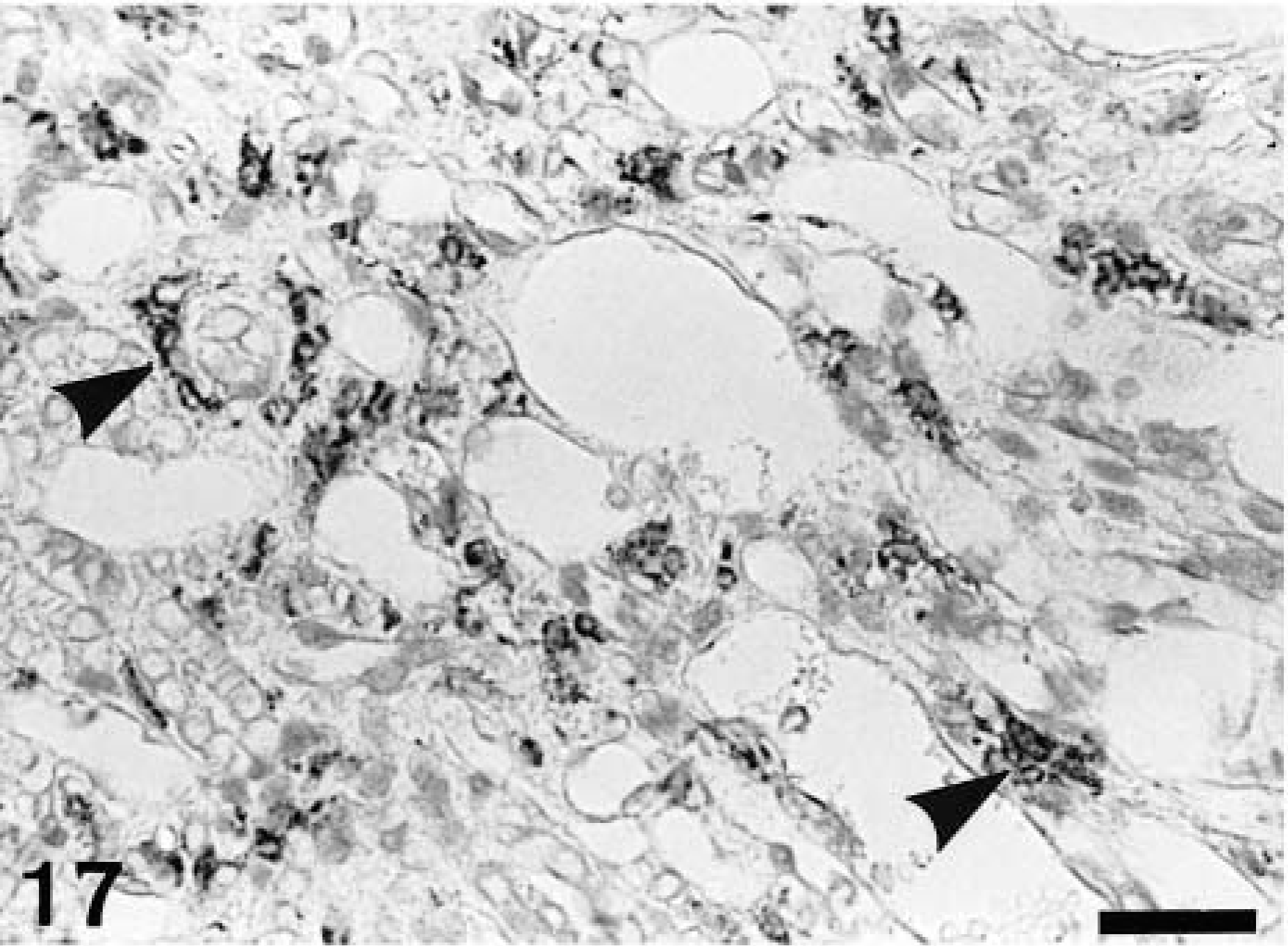
Kidney; CDDP-treated rat at PFI week 5. OX6-immunopositive cells (arrowheads) are distributed in the interstitium. The positive cells show various configuration such as round, spindle-shaped, and dendritic. Immunohistochemistry, counterstained with hematoxylin. Bar = 40 μm.
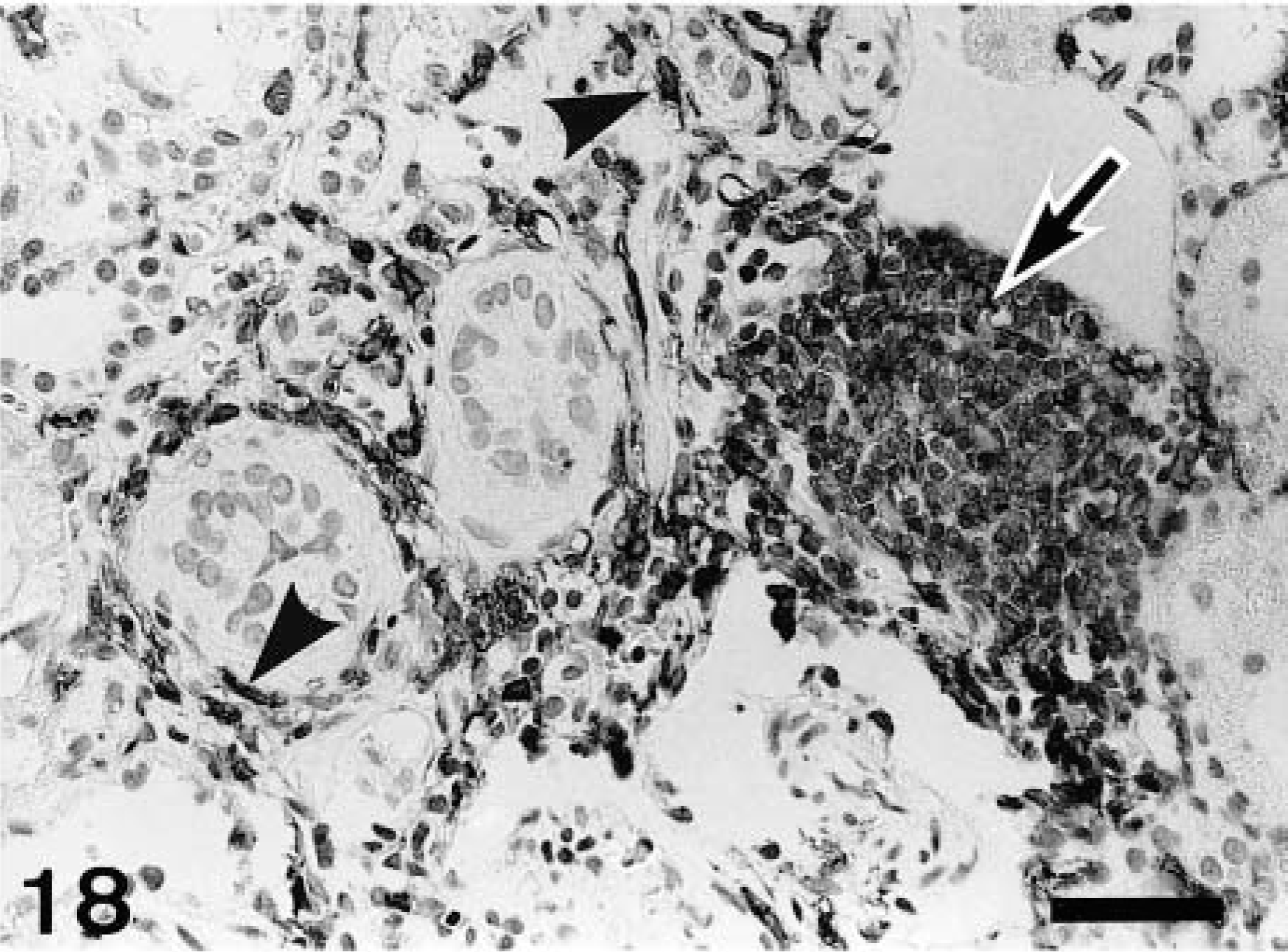
Kidney; CDDP-treated rat at PFI week 19. OX6-immunopositive cells (arrowheads) are seen around the atrophied renal tubules; they are oval or spindle-shaped. In addition, cells forming a lymphoid follicle give a positive reaction to OX6 (arrow), indicating B cells. Immunohistochemistry, counterstained with hematoxylin. Bar = 40 μm.
TGF-β1 immunoreactivity and apoptotic cells demonstrable by the TUNEL method
A faint immunoreaction to TGF-β1 was seen on the renal proximal tubules of control rats. Therefore, the reactivity in the CDDP group was semiquantitatively evaluated in contrast to that of controls. At PFI weeks 1, 3, and 5, a greater reactivity to TGF-β1 was seen in epithelial cells rimming the affected tubules in the corticomedullary junction, and occasional macrophages infiltrating the lumina of the dilated renal tubules revealed a positive reaction to TGF-β1 (Fig. 19). At PFI weeks 7–19, no marked difference was found in TGF-β1 immunoreaction on renal tubules between the control and the CDDP group, and macrophages present in the fibrotic areas did not react to TGF-β1.
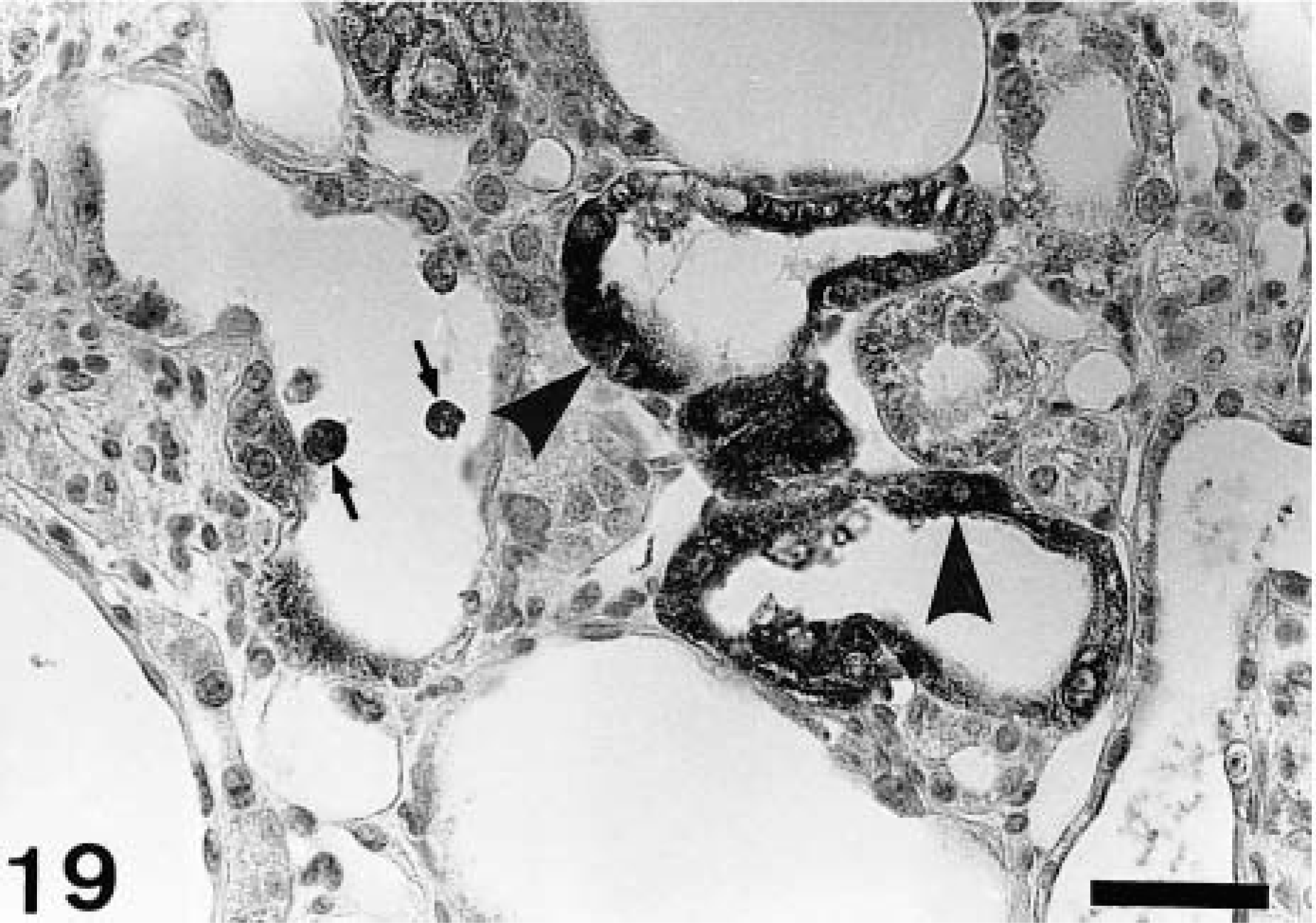
Kidney; CDDP-treated rat at PFI week 3. A greater immunoreactivity to TGF-β1 is seen in epithelial cells rimming the affected tubules in the corticomedullary junction (arrowheads), and some macrophages in the lumina of the dilated renal tubules show a positive reaction to TGF-β1 (arrows). Immunohistochemistry, counterstained with hematoxylin. Bar = 30 μm.
During the 19-week recovery period, cells undergoing apoptosis were seen infrequently in the fibrotic areas or dilated renal tubules in the affected corticomedullary junction (Fig. 20). The number was not more than five cells per 30 areas at each PFI week. Some of the positive cells seen in the dilated renal tubules appeared to be macrophages engulfing apoptotic bodies in the cytoplasm (Fig. 20). No cells reacting with the TUNEL method were present in controls.
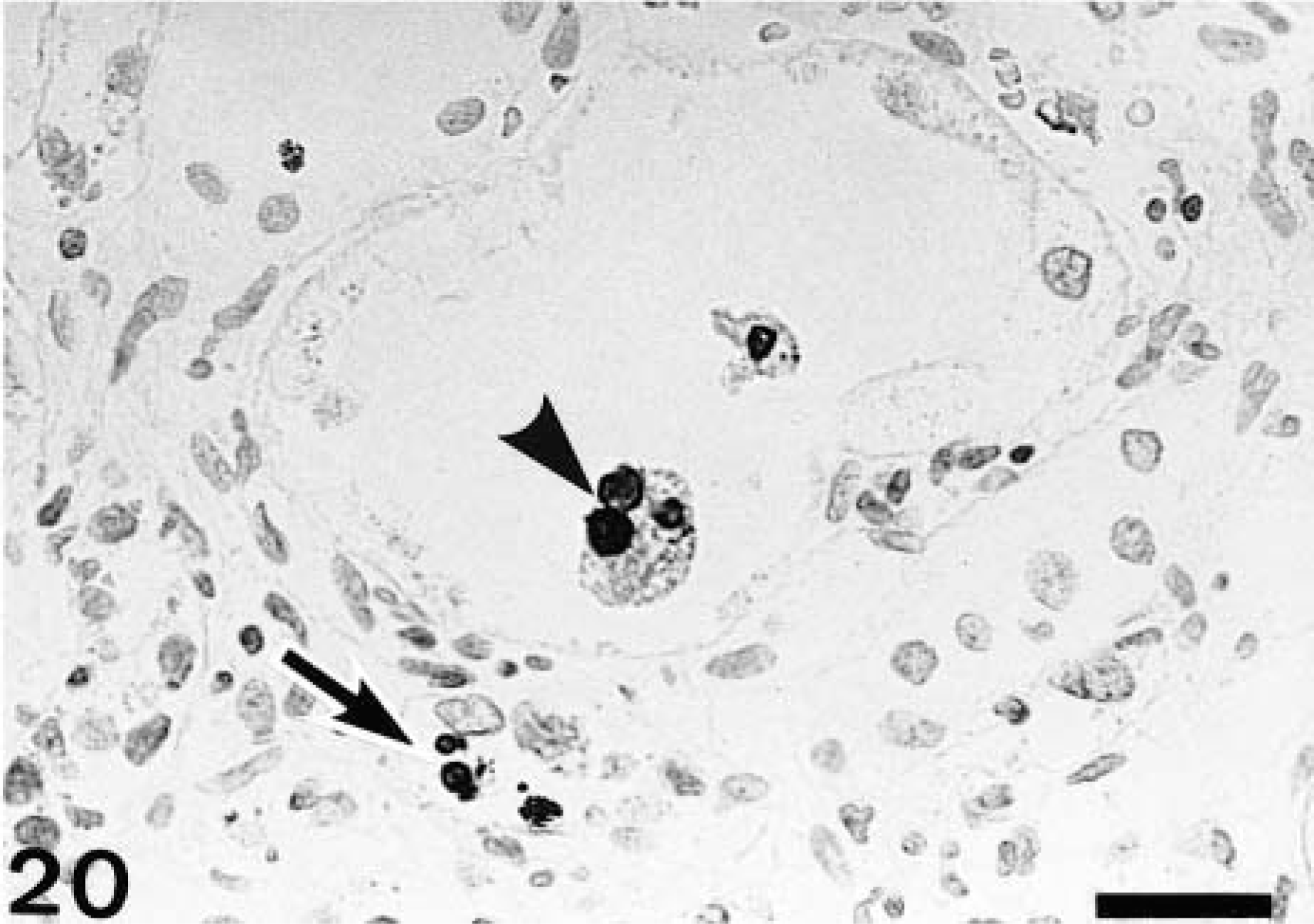
Kidney; CDDP-treated rat at PFI week 17. A few interstitial cells undergoing apoptosis, demonstrable by the TUNEL method, are seen (arrow), and a macrophage-like cell engulfing apoptotic bodies in the cytoplasm also is present in the dilated lumina of renal tubule (arrowhead). TUNEL method, counterstained with hematoxylin. Bar = 20 μm.
Discussion
CDDP has a toxic effect on the lower straight part (P3 segment) of the proximal tubules.21,46 Thus, CDDP-induced lesions are characterized by necrosis or degeneration or both of renal tubular epithelial cells in the corticomedullary junction and subsequent dilatation of the affected tubules.21,46 The present study confirmed our previous report42 of progressive renal interstitial fibrosis in the corticomedullary junction at the early stages in this model. However, more interestingly, all renal damages were repaired during the long-term observation period of 19 weeks after the cessation of CDDP injection by a reduction in fibrotic tissues and by replacing regenerated renal tubules. The healing was also indicated by the recoveries of body weight and values of BUN and creatinine at PFI week 19. Thus, the kinetics of myofibroblastic cells and macrophage populations appearing in progressive renal interstitial fibrosis and subsequent healing process could be analyzed.
Myofibroblastic cells, which have features intermediate between those of fibroblasts and smooth muscles, act as fibrogenic cells in hepatic,17,18 pulmonary,6 and myocardial fibrosis.26 In experimental rat renal fibrosis, similar myofibroblastic cells have been identified as a major cellular element.1,11,14,15,36,38,42,45,46 In the present study, many myofibroblastic cells were present from PFI weeks 1 to 7, within areas of increased fibrosis. BrdU-immunopositive interstitial cells seen in such areas at PFI weeks 1–7 were considered to be myofibroblastic cells, because proliferation of myofibroblastic cells has been demonstrated in isoproterenol-induced rat myocardial fibrosis and rat renal interstitial fibrosis after ureteral obstruction.26,45 Although macrophages may be another candidate for the proliferating cells, macrophages infiltrating injured areas have been considered to be short-lived, nondividing terminal cells.31,37 Recently, it has been reported that in addition to preexisting interstitial fibroblasts, renal epithelial cells may transdifferentiate into myofibroblastic cells in renal interstitial fibrosis.15,29 Further study of the nature and derivation of myofibroblastic cells seen in the present rat model would be interesting.
Severe depositions (3+) of ECM components such as collagen types I and III, and fibronectin, which are produced mainly by myofibroblastic cells,3,15,33 were seen in the fibrotic areas by PFI weeks 7–11. In addition to abnormal deposition of these interstitial ECM components, basement membrane–associated ECM components such as laminin and collagen type IV were markedly accumulated around the dilated or atrophied renal tubules. The basement membrane–related ECM components might be produced by regenerating renal epithelial cells.11,27 As has been reported in renal fibrosis,15,19,20,27,38,40 abnormal accumulation of ECM components was confirmed in the current rat model.
TGF-β1 is regarded as the most important soluble factor responsible for the induction of myofibroblastic cells.6,11,30,38 A greater immunoreactivity for TGF-β1 was seen in affected renal epithelial cells and some macrophages at PFI weeks 1, 3, and 5. In ureteral obstruction– or adriamycin-induced renal interstitial fibrosis of rats, TGF-β1 immunoreaction also was localized in renal epithelial cells as well as in a limited number of macrophages.38,40,45 TGF-β1 produced by renal epithelial cells and some exudate macrophages might participate in the development of myofibroblastic cells in the early stages of renal interstitial fibrosis.15,40,45
Apoptosis is an important phenomenon in remodeling of injured tissues.9,32,34 Although the number of TUNEL-positive cells was very small (fewer than five cells per 30 areas), cells were undergoing apoptosis in the present fibrotic areas. Apoptotic processes are extremely rapid means of cell deletion; the development of morphologic changes, uptake by phagocytosis, and degradation of the ingested apoptotic cells beyond histologic recognition are completed in 30 minutes to 3 hours.5,39 Thus, simultaneous detection of many apoptotic cells might be difficult.39 Myofibroblastic cells show apoptosis in skin wound healing and evolution of granulation tissues into a scar in which a striking decrease in cellularity occurs.9,32,34 Infiltrating macrophages can engulf apoptotic bodies and then macrophages themselves disappear via apoptosis after ending their functions.9,22,32,34 In the present study, TUNEL-positive interstitial cells and apoptotic body–ingesting cells in the lumina of renal tubule might be regarded as degraded myofibroblastic cells and macrophages with phagocytic activity, respectively. Thus, TUNEL-positive cells seen in the present study might imply a decreased number of myofibroblastic cells and inflammatory clearance by macrophages.
ED2 has been widely used to detect resident macrophages in rat lesions.17,18,26,35 Studies on cholestatic and carbon tetrachloride–induced acute liver injury with rats have demonstrated that ED2-positive activated Kupffer cells showed the kinetics similar to those of ED1-positive exudate macrophages, and that the expansion of these macrophage populations correlated well to the increased number of myofibroblastic cells.17,18 Likewise, in the present study, many ED1-positive cells were present at PFI weeks 3–11, the peak number occurred at PFI week 7, and the number of ED2-positive cells was maximum at PFI week 3 (Figs. 12, 13). These findings indicated that both ED1-positive exudate macrophages and ED2-positive resident macrophages contributed largely to the early stages of post–tissue injury renal fibrosis. In hepatic fibrosis, the roles of Kupffer cells are similar to those of exudate macrophages, particularly in terms of production of fibrogenic factors and phagocytosis.17,18,23,25 However, interestingly, ED2-positive macrophages have not been seen in ureteral obstruction–induced renal fibrosis and in interstitial fibrosis after acute renal tubular injury induced by a single injection of CDDP in rats.45,46 Furthermore, in isoproterenol-induced rat myocardial fibrosis, an increment of ED2-positive macrophages was different from that of ED1-positive macrophages in that the former appeared in the late stages consisting exclusively of scar tissues.26 Kupffer cells originally exist in the liver,8 and resident macrophages form a regular network of the interstitial cells throughout the normal cardiac tissues.26,35 In contrast, no cells reacting with ED2 were present in the interstitium of normal kidney tissues. The derivation and roles of ED2-positive macrophages seen in the present renal fibrosis should be investigated further.
OX6 is a monoclonal antibody against rat MHC class II antigen, and OX6 recognizes activated macrophages and dendritic cells as antigen-presenting cells.35,49,50 Activated macrophages originate from exudate or resident macrophages.41 The dendritic cells include interdigitating follicular cells in the lymphoid tissues, interstitial dendritic cells in the connective tissues of nonlymphoid organs, and Langerhans cells of the epidermis.31,35,37 The participation of OX6-positive cells has not been reported in rat renal interstitial fibrosis. In the present study, a number of OX6-positive cells had already appeared at PFI week 1, and the number peaked at PFI week 9. At PFI week 11, the number was decreased, but remained elevated until PFI week 19. These findings indicated that OX6-positive cells might sequentially participate not only in the development of renal interstitial fibrosis at the early stages but also in the remodeling processes at the late stages. MHC class II–expressing cells present antigens to unprimed T cells, and in turn Th1/Th2 lymphocyte polarization will be determined; cytokines produced mainly by Th2 lymphocytes stimulate B cell proliferation, and Th1-derived cytokines activate macrophage populations.31,35,37 During the 19-week recovery period, lymphocytes were distributed in the fibrotic areas, and lymphoid follicles were often formed in the late stages. Lymphocytes constituting the lymphoid follicles are B cells, because they reacted to OX6; B cells also present antigens to helper T cells.41 In rat myocardial fibrosis, interstitial dendritic cells participated in the activation of lymphocytes.49,50 The kidney-derived antigens and subtypes of lymphocytes appearing in relation to OX6-positive cells remain to be investigated. In addition, a subset of B cells has been reported to give rise to dendritic morphology and functions,4,24 although dendritic cells are generally considered to develop from bone marrow myeloid lineage precursors.31,37 Further study of the derivation of OX6-positive cells, in particular the association between OX6-positive interstitial cells and B cells, would be interesting.
In conclusion, the present study showed for the first time that different macrophage populations might contribute to the pathogenesis of chronically developing rat renal interstitial fibrosis. The participation of diverse macrophage populations has not been reported in rat renal fibrosis after acute damage induced by chemicals such as adriamycin,38 puromycin aminonucleoside,10,13 cyclosporine,48 and mercuric chloride.36 Macrophages exhibit heterogeneous functions, such as phagocytosis, antigen presentation, and inhibition or killing of pathogens, as well as production of fibrogenic factors, depending on microenvironmental conditions induced by cell–cell or cell–matrix interactions.37,44,47 The severity of fibrotic lesions has been regarded as a crucial determinant in progressive renal injury and its prognosis in humans.28,29 Therefore, the present rat renal interstitial fibrosis, which was characterized by myofibroblastic cell development in the progressive stages and subsequent disappearance of myofibroblastic cells in a healing process, would provide a valuable model to investigate the pathogenesis relating to the functions of macrophage populations.
Footnotes
Acknowledgements
This study was supported in part by grant-in-aid (No. 12660287) for Scientific Research C, the Ministry of Education, Science, Sports and Culture, Japan.
